Morphological Effect of Pd Catalyst on Ethanol Electro-Oxidation Reaction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Catalysts
2.2. Materials Characterization
2.3. Electrochemical Measurements
3. Results and Discussion
3.1. Materials Characterization
3.1.1. XRD Analysis

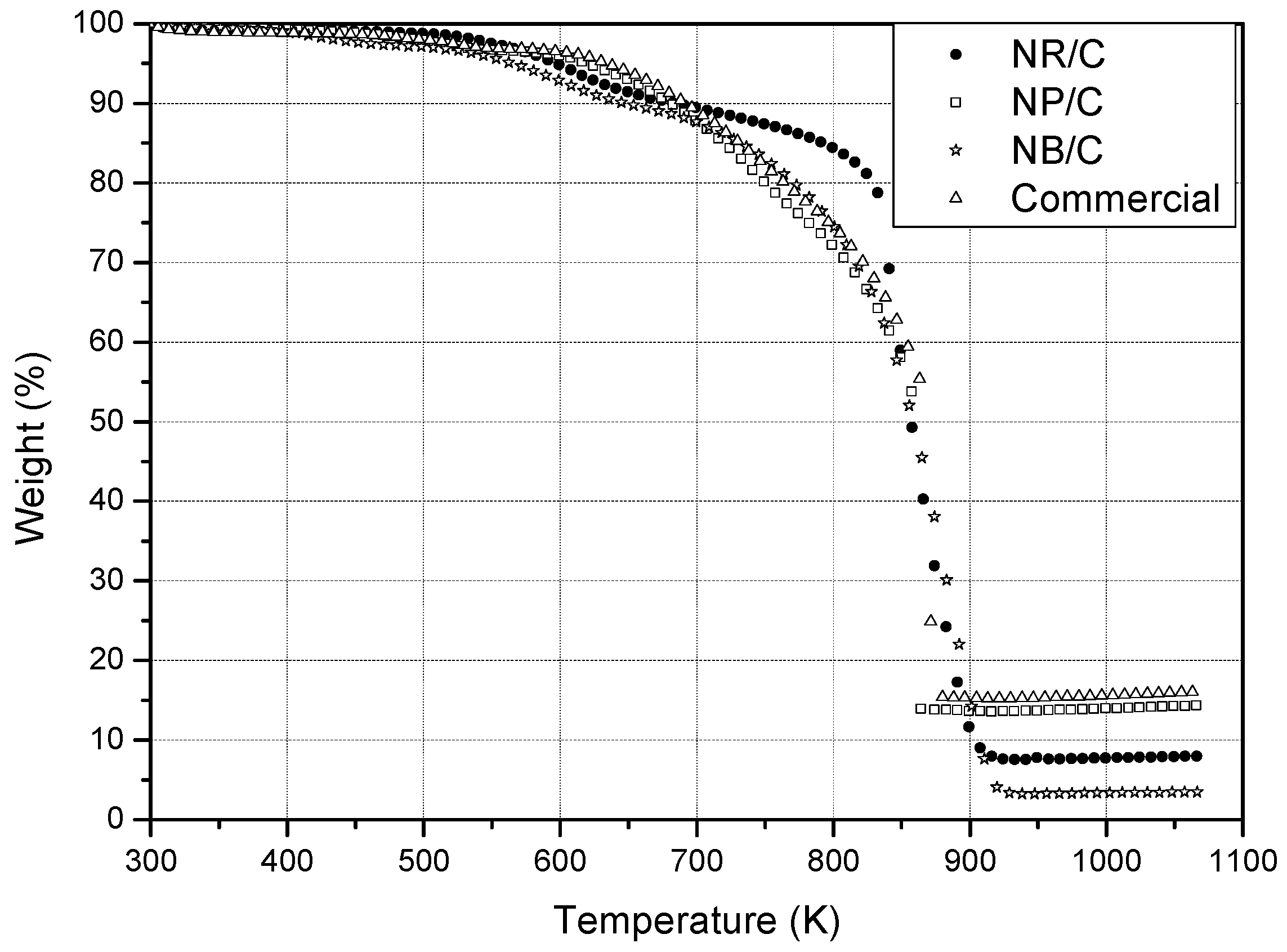
3.1.2. Thermogravimetric Analysis.
3.1.3. TEM Observations

3.2. Electrochemical Characterization
3.2.1. Cyclic Voltammetry in Absence of Ethanol

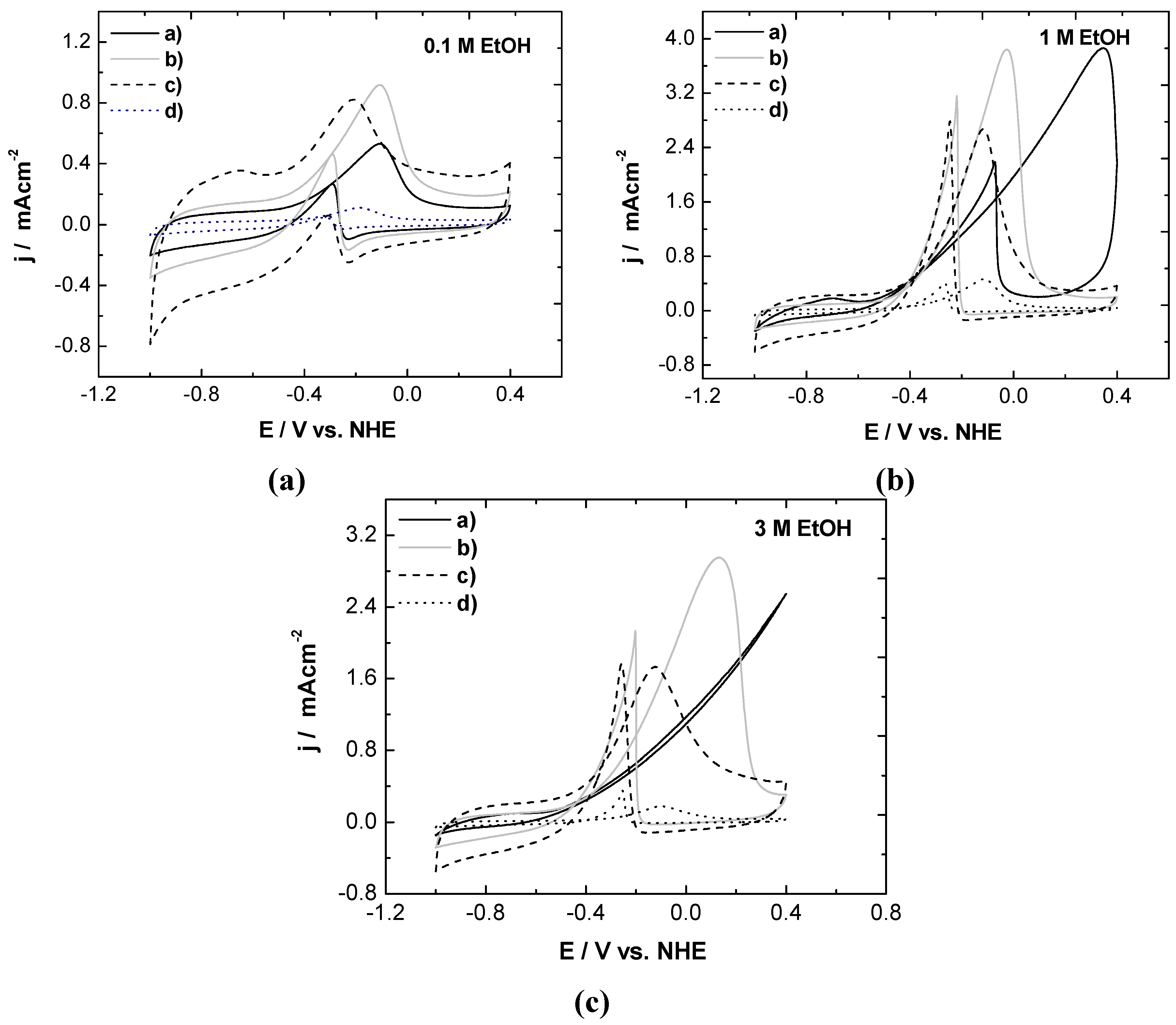
3.2.2. Cyclic Voltammetry in Presence of Ethanol
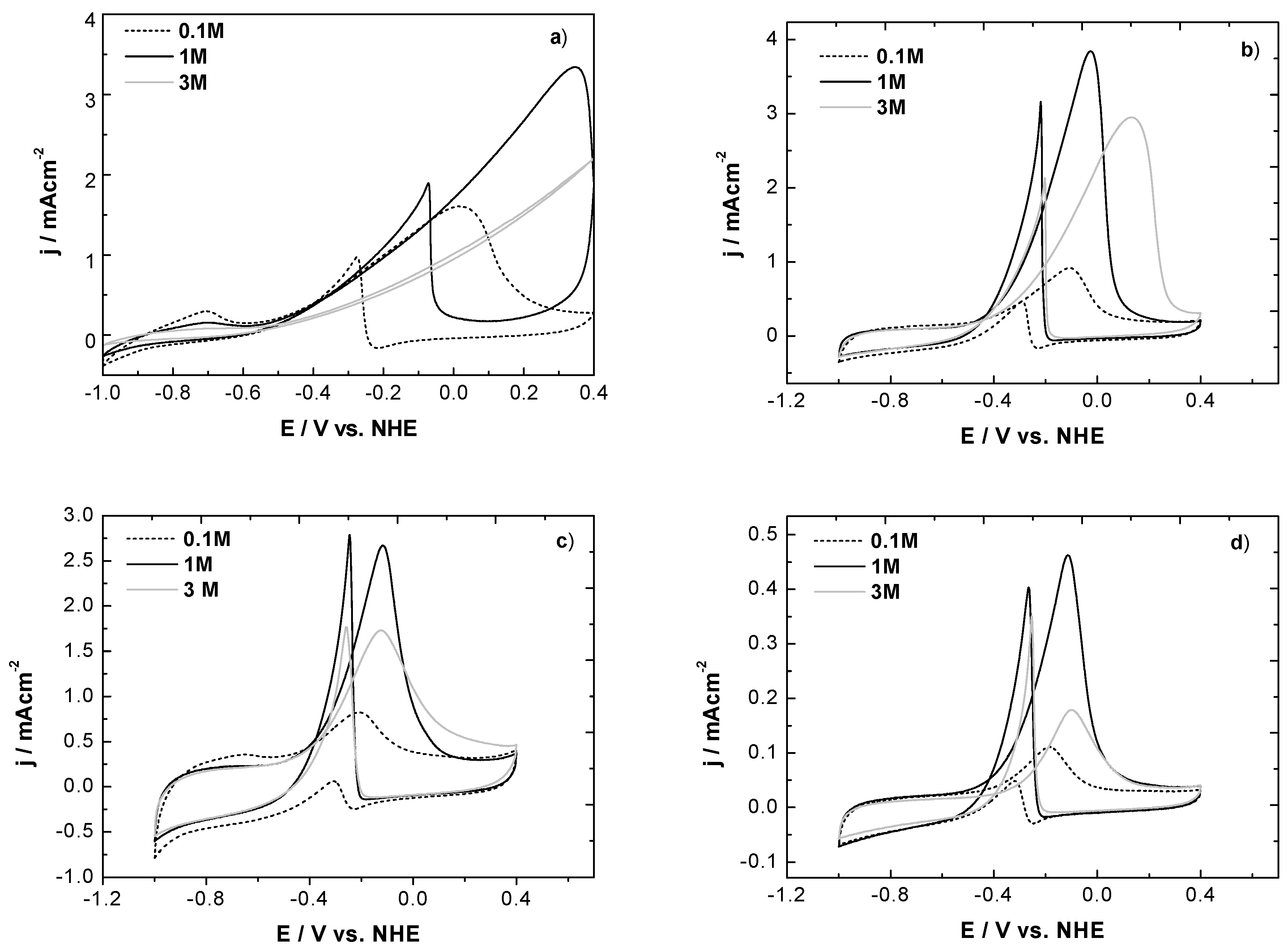
| Ethanol concentration | Ef/V | |||
|---|---|---|---|---|
| Commercial | NB/C | NR/C | NP/C | |
| 0.1 M | 0.016 | −0.210 | −0.108 | −0.183 |
| 1 M | 0.347 | −0.117 | −0.026 | −0.113 |
| 3 M | - | −0.122 | 0.132 | −0.099 |
4. Conclusions
Acknowledgments
References
- Rostrup-Nielsen, J.R. Fuels and energy for the future: the role of catalysis. Catal. Rev. 2004, 46, 247–270. [Google Scholar] [CrossRef]
- Demirbas, M.F.; Balat, M.; Balat, H. Biowastes-to-biofuels. Energy Convers. Manage. 2011, 52, 1815–1828. [Google Scholar] [CrossRef]
- Haryanto, A.; Fernando, S.; Murali, N.; Adhikari, S. Current status of hydrogen production techniques by steam reforming of ethanol: A review. Energy Fuel. 2005, 5, 2098–2106. [Google Scholar] [CrossRef]
- Ni, M.; Leung, D.Y.C.; Leung, M.K.H. A review on reforming bio-ethanol for hydrogen production. Int. J. Hydrogen Energy 2007, 32, 3238–3247. [Google Scholar] [CrossRef]
- Carrera, R.C.; Ramírez, R.F.; Alvarado, A.F.A.; Rosales, J.M.M.; García, T.V.; Esquivel, I.R.G. Steam reforming of ethanol over Ni/Al2O3−La2O3 catalysts synthesized by Sol−Gel. Ind. Eng. Chem. Res. 2011, 50, 2576–2584. [Google Scholar] [CrossRef]
- Song, S.Q.; Zhou, W.J.; Zhou, Z.H.; Jiang, L.H.; Sun, G.Q.; Xin, Q.; Leontidis, V.; Kontou, S.; Tsiakaras, P. Direct ethanol PEM fuel cells: The case of platinum based anodes. Int. J. Hydrogen Energy 2005, 30, 995–1001. [Google Scholar] [CrossRef]
- Lopes, T.; Antolini, E.; Gonzalez, E.R. Carbon supported Pt-Pd alloy as an ethanol tolerant oxygen reduction electrocatalyst for direct ethanol fuel cells. Int. J. Hydrogen Energy 2008, 33, 5563–5570. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Xin, L.; Sun, K.; Li, W.Z. PdNi electrocatalysts for efficient ethanol oxidation reaction in alkaline electrolyte. Int. J. Hydrogen Energy 2011, 36, 12686–12697. [Google Scholar] [CrossRef]
- Zhao, T.S.; Li, Y.S.; Shen, S.Y. Anion-exchange membrane direct ethanol fuel cells: Status and perspective. Front. Energy Power Eng. China 2010, 4, 443–458. [Google Scholar] [CrossRef]
- Lee, Y.W.; Oh, J.K.; Kim, H.S.; Lee, J.K.; Han, S.B.; Choi, W.J.; Park, K.W. Shape-controlled Pd nanostructure catalysts for highly efficient electrochemical power sources. J. Power Sources 2010, 195, 5896–5901. [Google Scholar] [CrossRef]
- Xiao, L.; Zhuang, L.; Liu, Y.; Lu, J.T.; Abruña, H.D. Activating Pd by morphology tailoring for oxygen reduction. J. Am. Chem. Soc. 2009, 2, 602–608. [Google Scholar] [CrossRef]
- Han, S.B.; Song, Y.J.; Lee, J.M.; Kim, J.Y.; Park, K.W. Platinum nanocube catalysts for methanol and ethanol electrooxidation. Electrochem. Commun. 2008, 10, 1044–1047. [Google Scholar] [CrossRef]
- Wang, E.D.; Xu, J.B.; Zhao, T.S. Density functional theory studies of the structure sensitivity of ethanol oxidation on palladium surfaces. J. Phys. Chem. C 2010, 114, 10489–10497. [Google Scholar] [CrossRef]
- Xiong, Y.J.; Cai, H.G.; Wiley, B.J.; Wang, J.G.; Kim, M.J.; Xia, Y.N. Synthesis and mechanistic study of palladium nanobars and nanorods. J. Am. Chem. Soc. 2007, 129, 3665–3675. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.Y.; Zhao, T.S.; Xu, J.B.; Li, Y.S. Synthesis of PdNi catalysts for the oxidation of ethanol in alkaline direct ethanol fuel cells. J. Power Sources 2010, 195, 1001–1006. [Google Scholar] [CrossRef]
- Do, J.S.; Chen, Y.T.; Lee, M.H. Effect of thermal annealing on the properties of Co rich core—Pt rich shell/C oxygen reduction electrocatalyst. J. Power Sources 2007, 172, 623–632. [Google Scholar] [CrossRef]
- Xiao, C.W.; Ding, H.; Shen, C.M.; Yang, T.Z.; Hui, C.; Gao, H.J. Shape-controlled synthesis of palladium nanorods and their magnetic properties. J. Phys. Chem. C 2009, 113, 13466–13469. [Google Scholar] [CrossRef]
- Liang, Z.X.; Zhao, T.S.; Xu, J.B.; Zhu, L.D. Mechanism study of the ethanol oxidation reaction on palladium in alkaline media. Electrochim. Acta 2009, 54, 2203–2208. [Google Scholar] [CrossRef]
- Nguyen, S.T.; Law, H.M.; Nguyen, H.T.; Kristian, N.; Wang, S.Y.; Chan, S.H.; Wang, X. Enhancement effect of Ag for Pd/C towards the ethanol electro-oxidation in alkaline media. App. Catal. B Environ. 2009, 91, 507–515. [Google Scholar] [CrossRef]
- Singh, R.N.; Singh, A.; Anindita, A. Electrocatalytic activity of binary and ternary composite films of Pd, MWCNT, and Ni for ethanol electro-oxidation in alkaline solutions. Carbon 2009, 47, 271–278. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cerritos, R.C.; Guerra-Balcázar, M.; Ramírez, R.F.; Ledesma-García, J.; Arriaga, L.G. Morphological Effect of Pd Catalyst on Ethanol Electro-Oxidation Reaction. Materials 2012, 5, 1686-1697. https://doi.org/10.3390/ma5091686
Cerritos RC, Guerra-Balcázar M, Ramírez RF, Ledesma-García J, Arriaga LG. Morphological Effect of Pd Catalyst on Ethanol Electro-Oxidation Reaction. Materials. 2012; 5(9):1686-1697. https://doi.org/10.3390/ma5091686
Chicago/Turabian StyleCerritos, Raúl Carrera, Minerva Guerra-Balcázar, Rosalba Fuentes Ramírez, Janet Ledesma-García, and Luis Gerardo Arriaga. 2012. "Morphological Effect of Pd Catalyst on Ethanol Electro-Oxidation Reaction" Materials 5, no. 9: 1686-1697. https://doi.org/10.3390/ma5091686




