Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds
Abstract
:Table of Content
- Introduction
- 1. Ring-Closing Enyne Metathesis
- 2. Ring-Closing Dienyne Metathesis
- 3. Cross Enyne Metathesis
- 4. Ring-Opening Enyne Metathesis
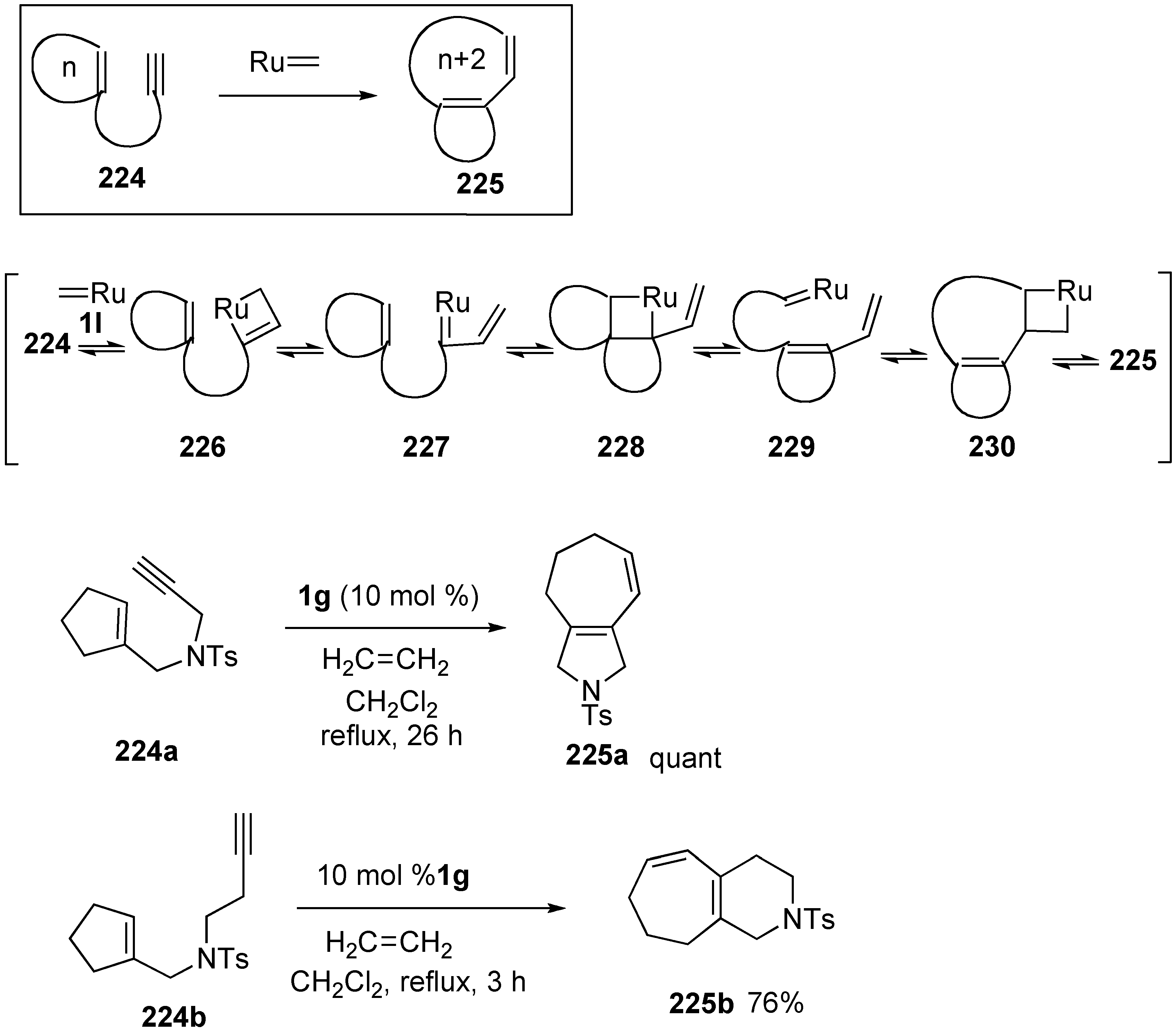
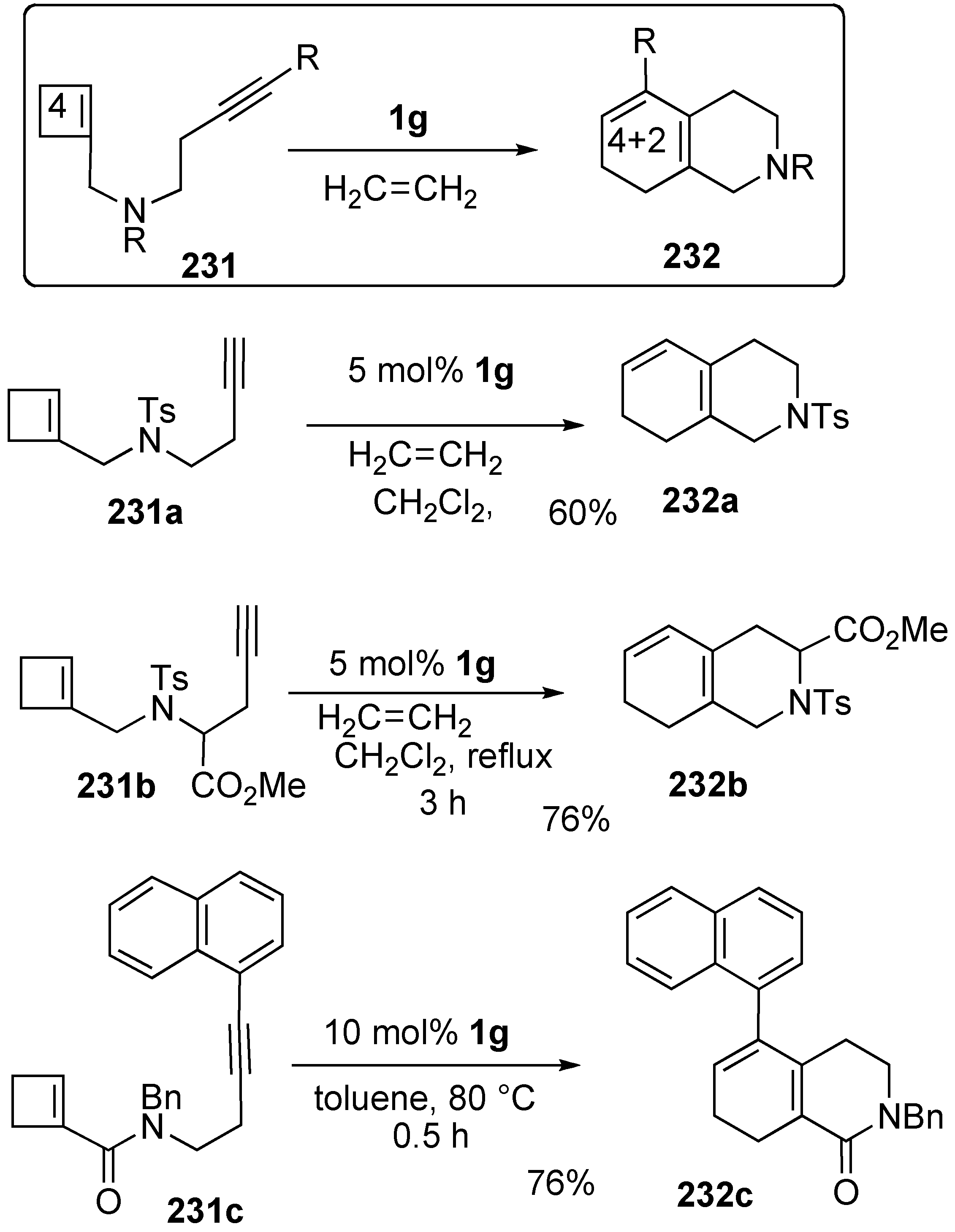
- 5. Skeletal Reorganization Using Transition Metals
- 6. Metallotropic Rearrangement
- Perspective
Introduction
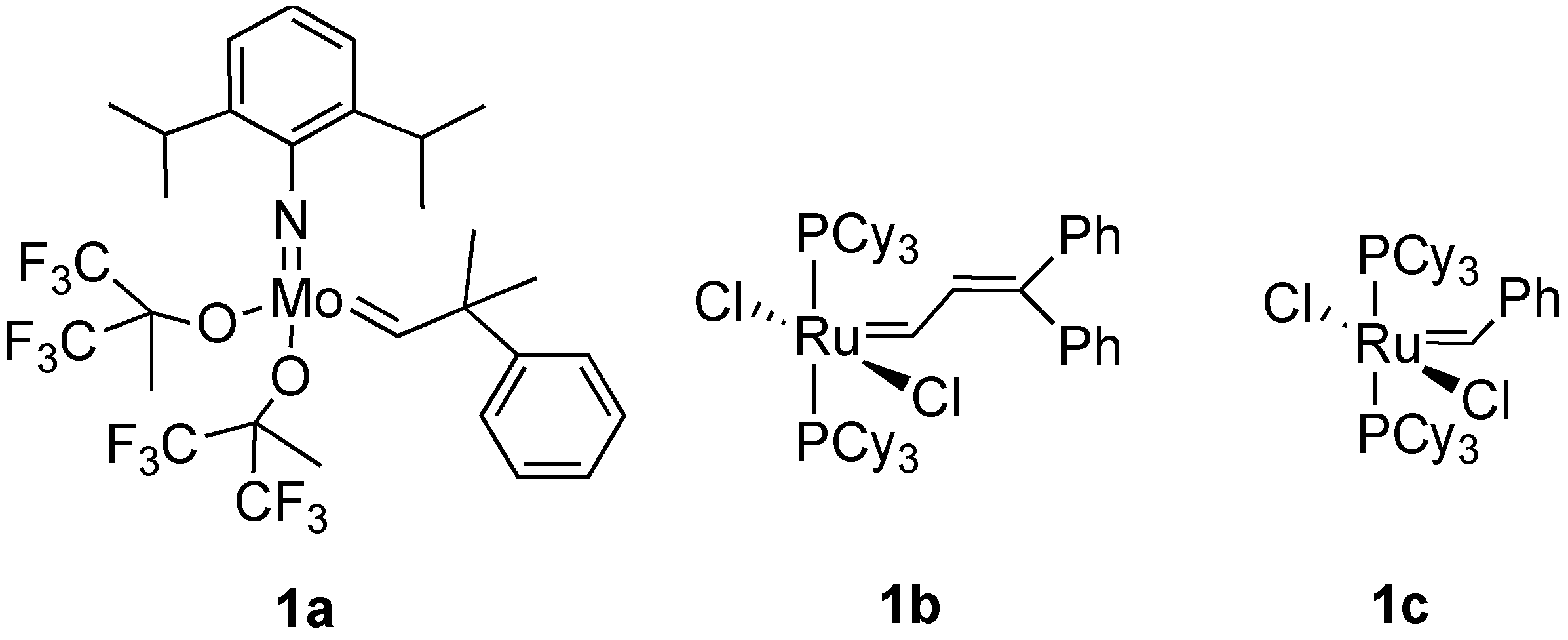
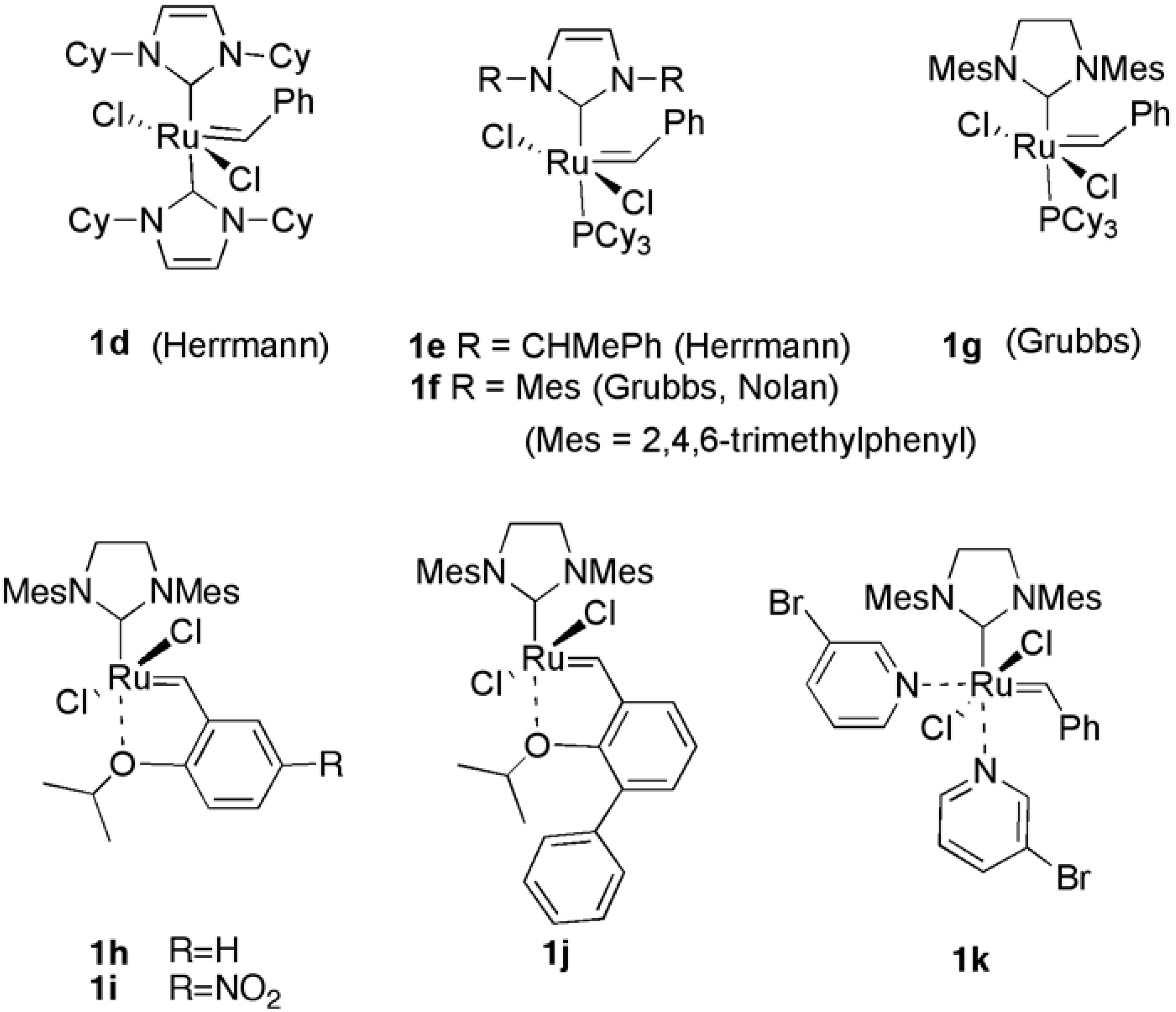
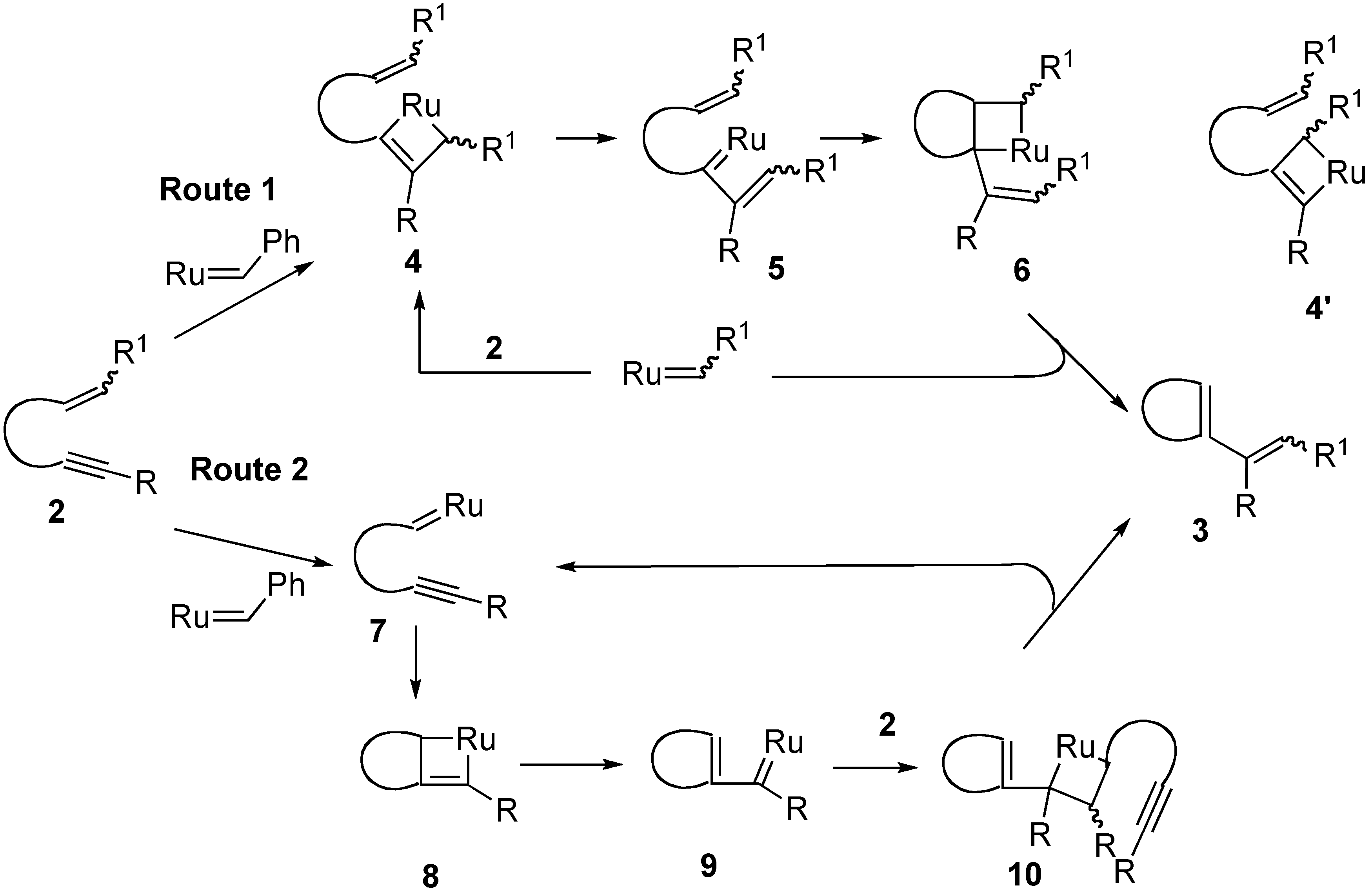
1. Ring-Closing Enyne Metathesis (RCM)
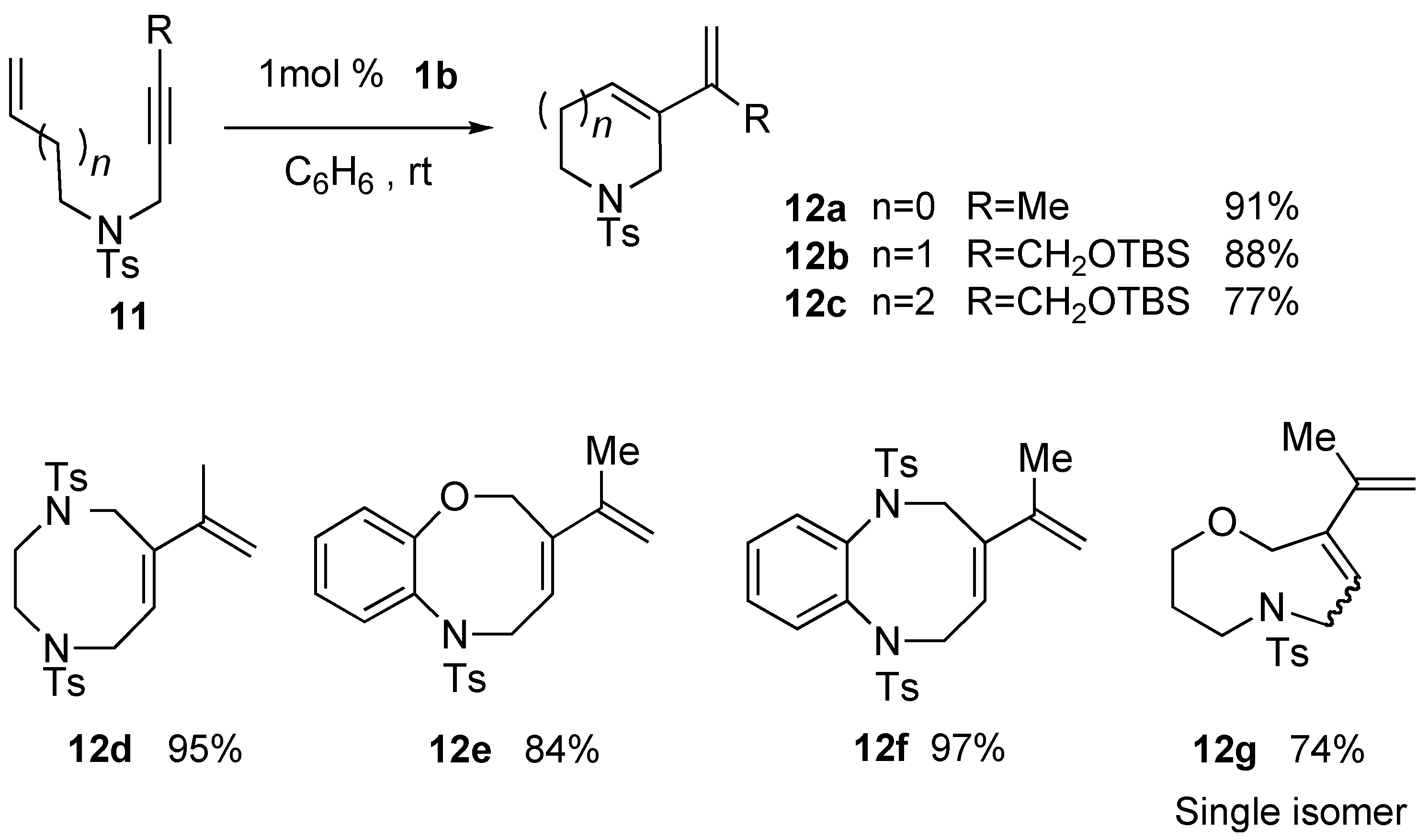
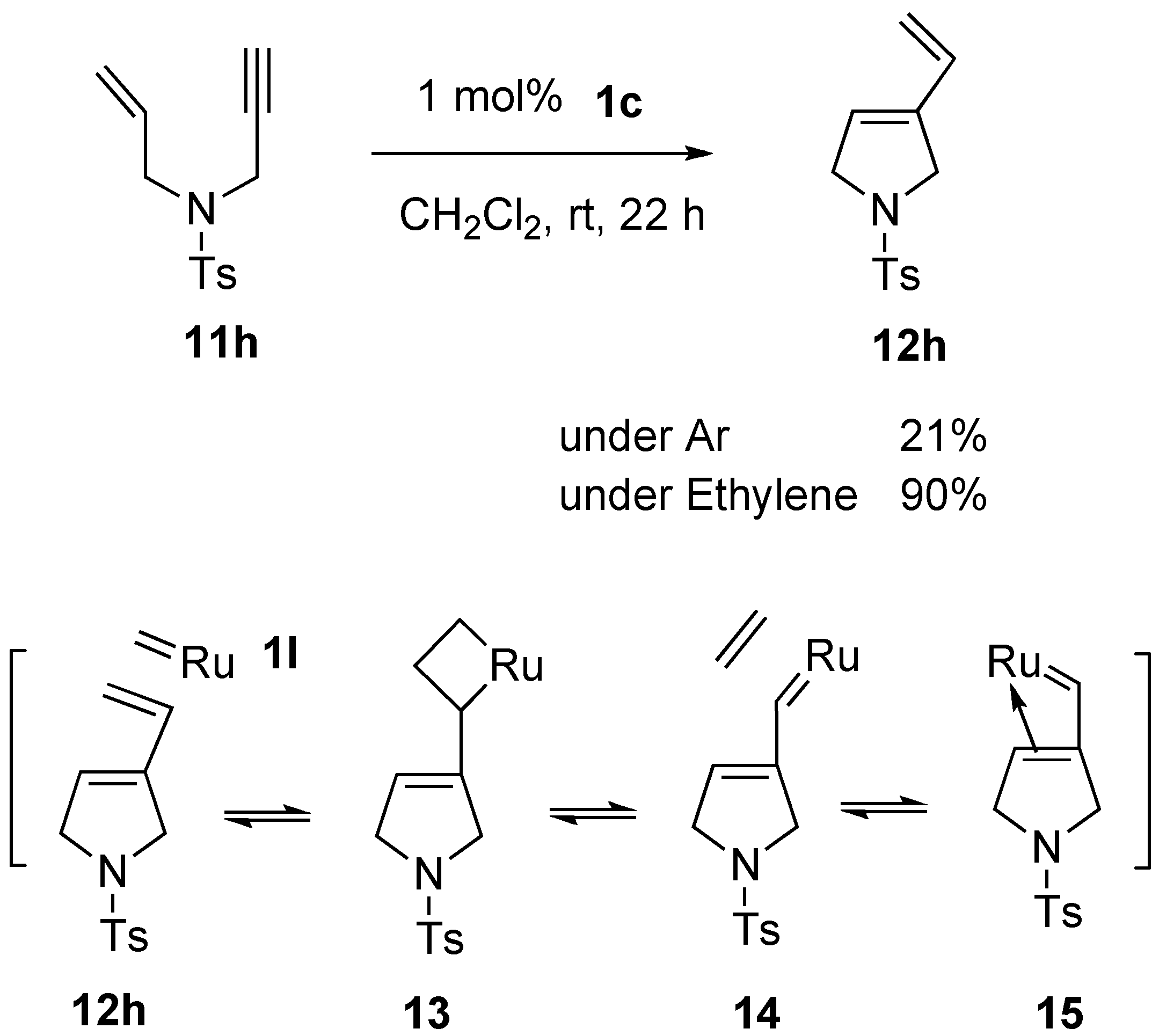
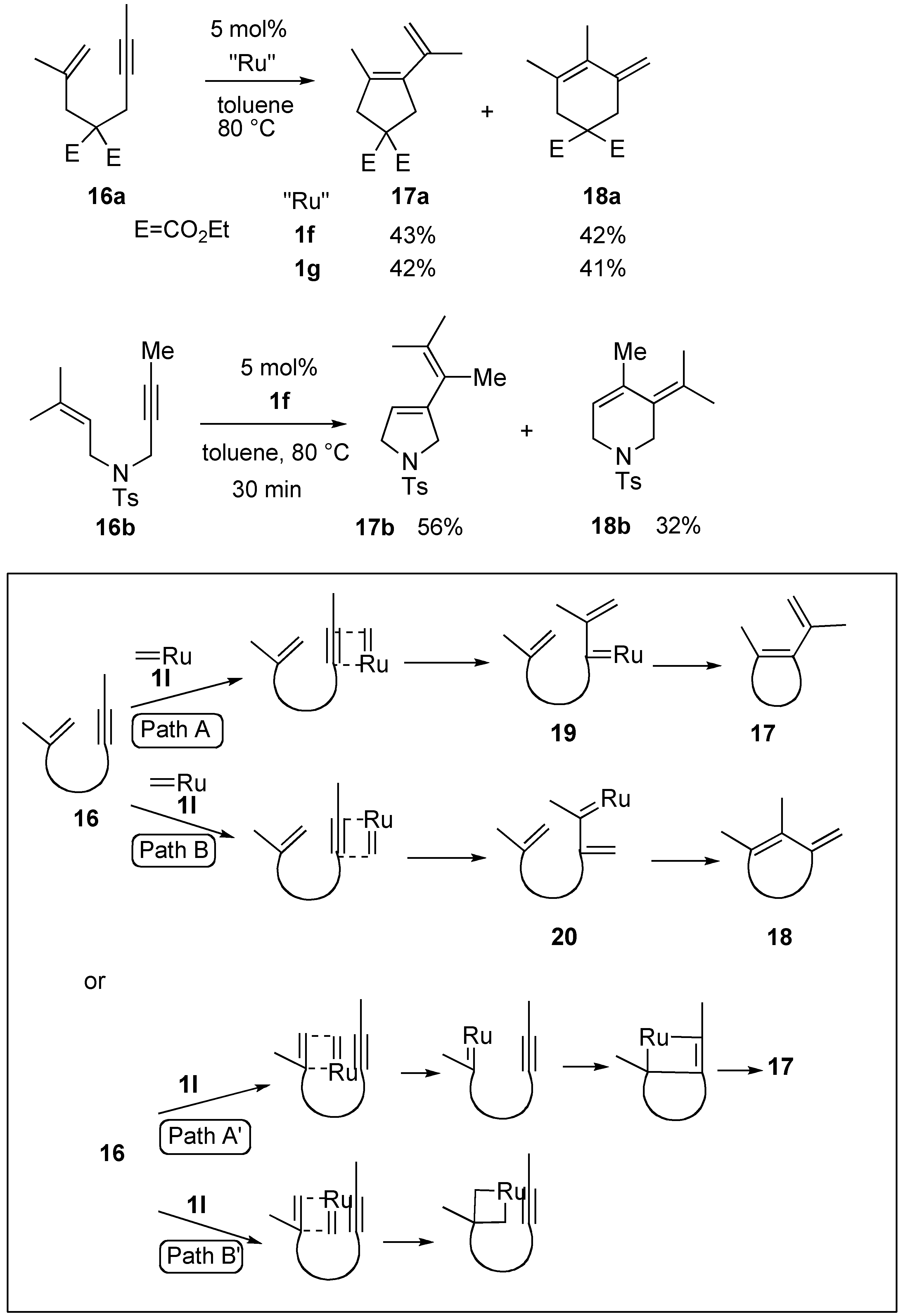
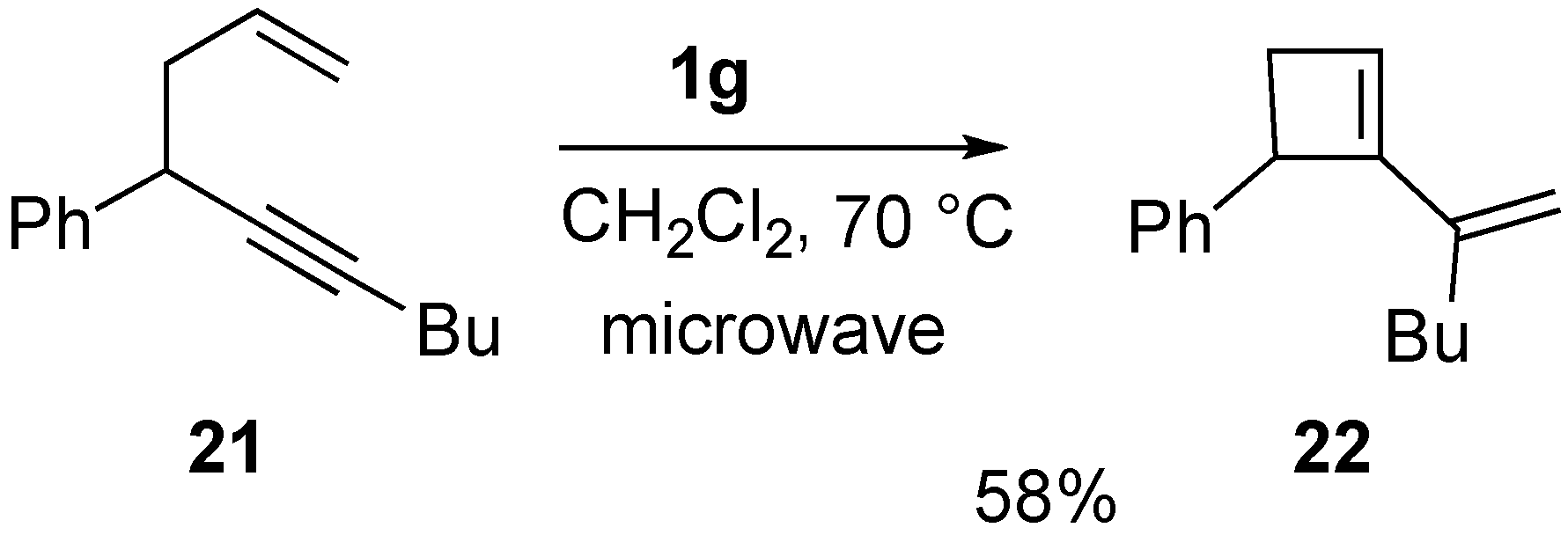
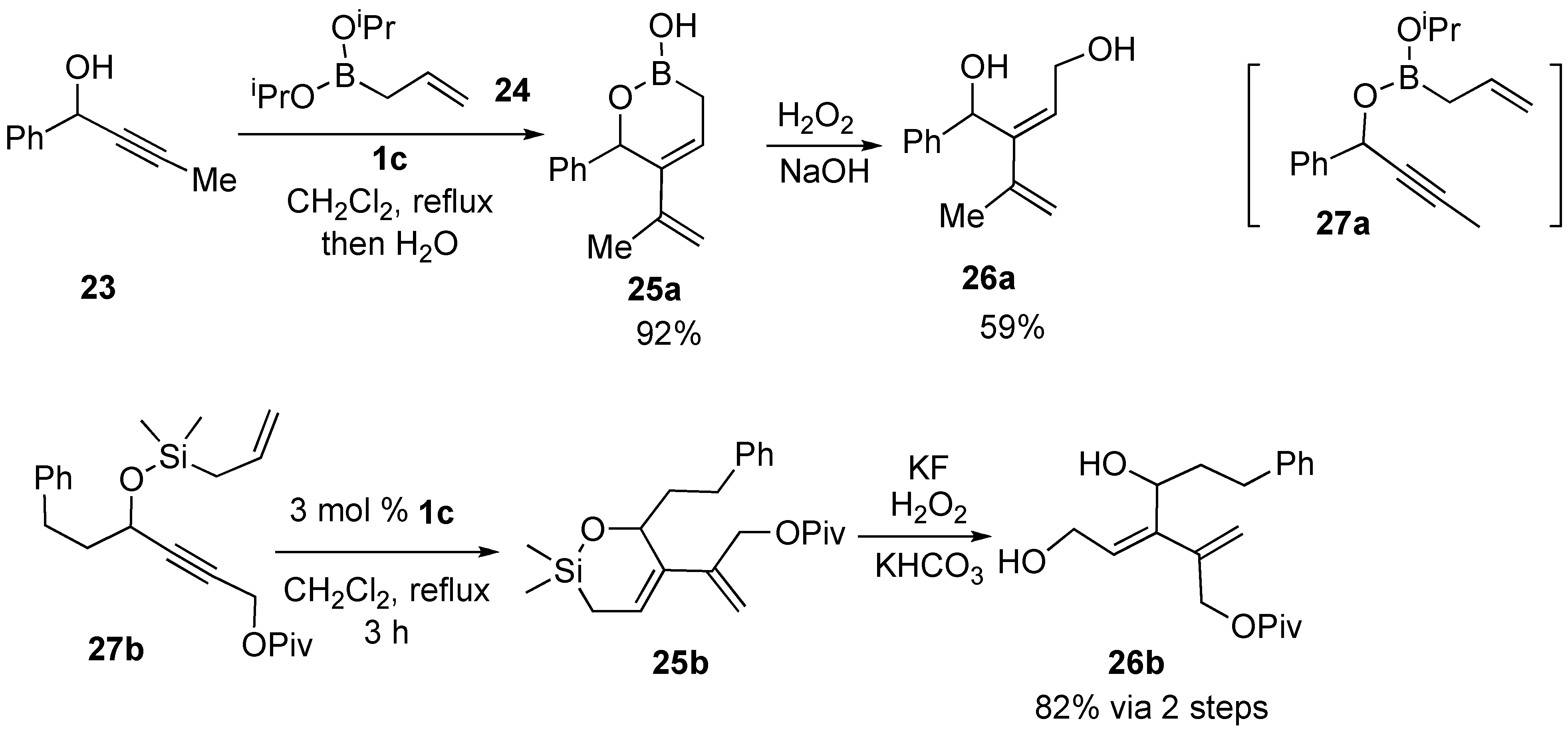
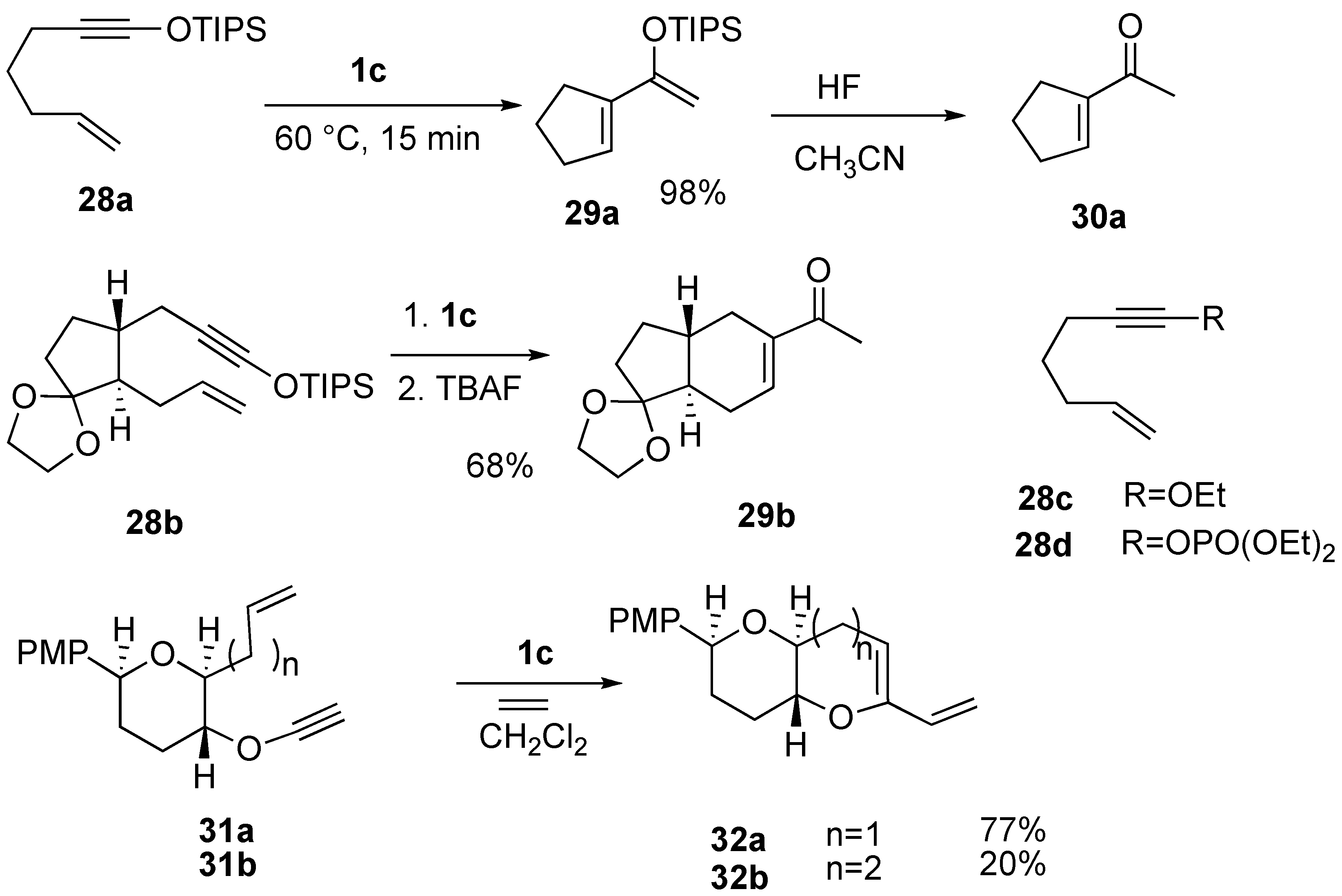
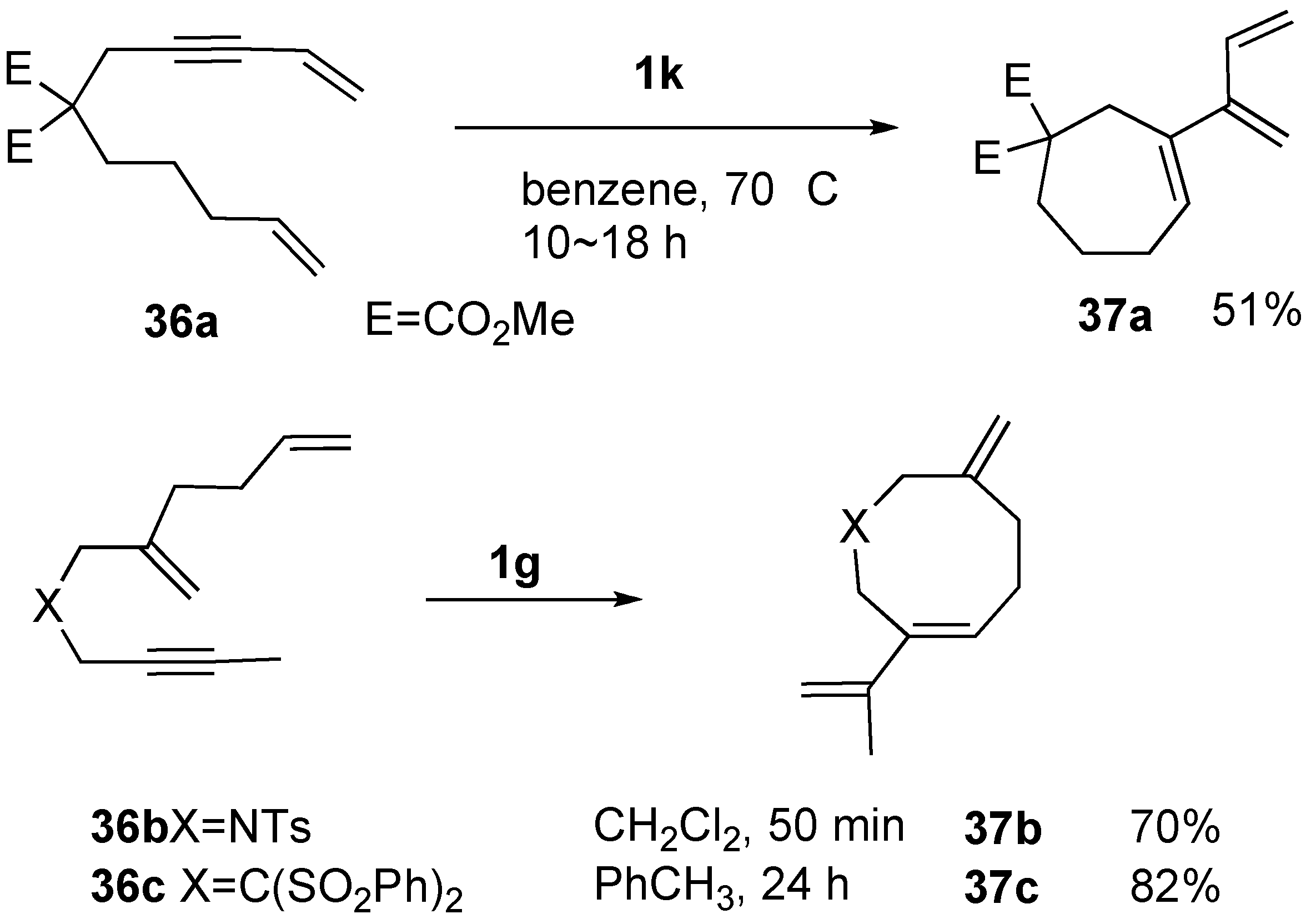
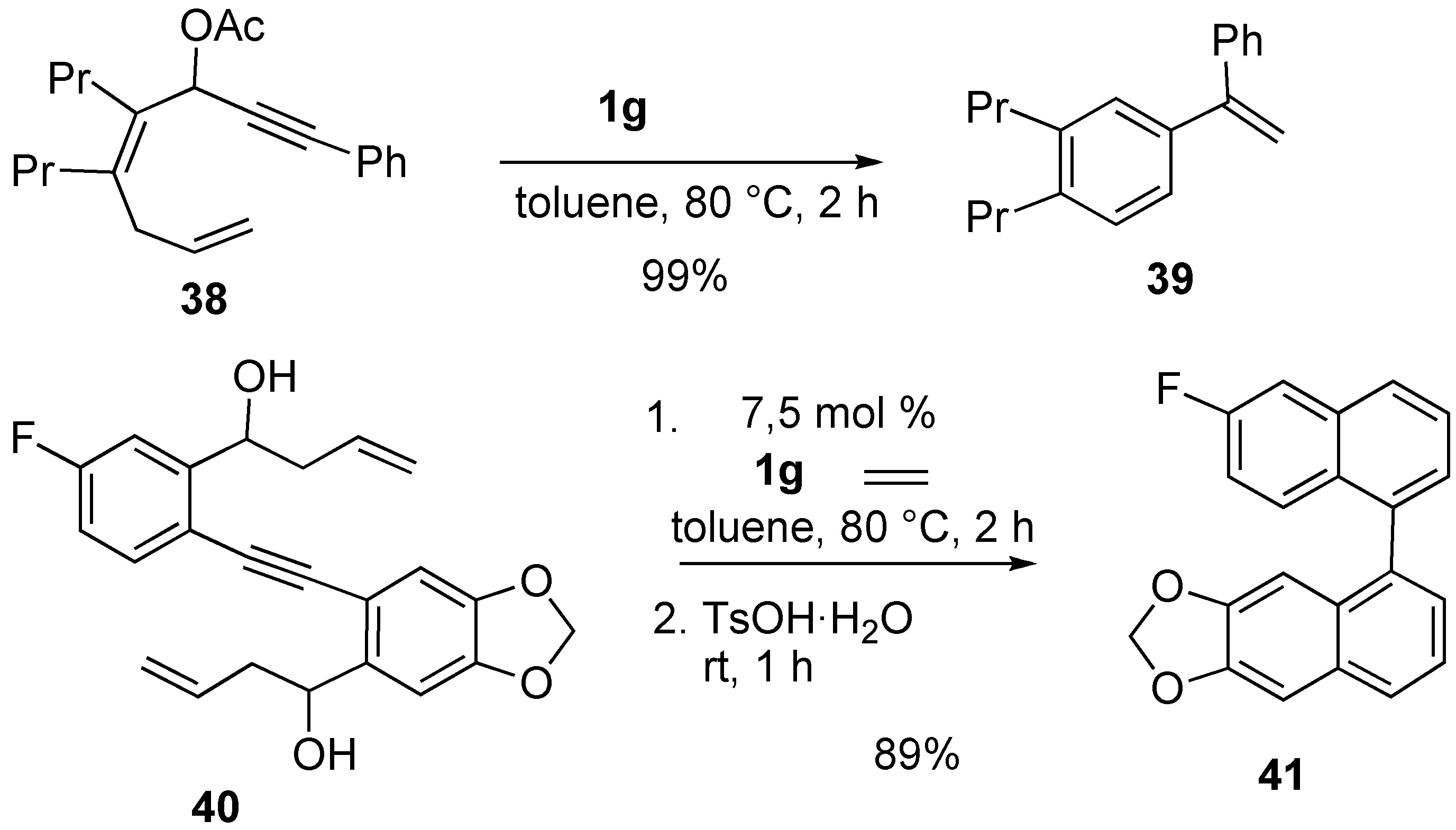
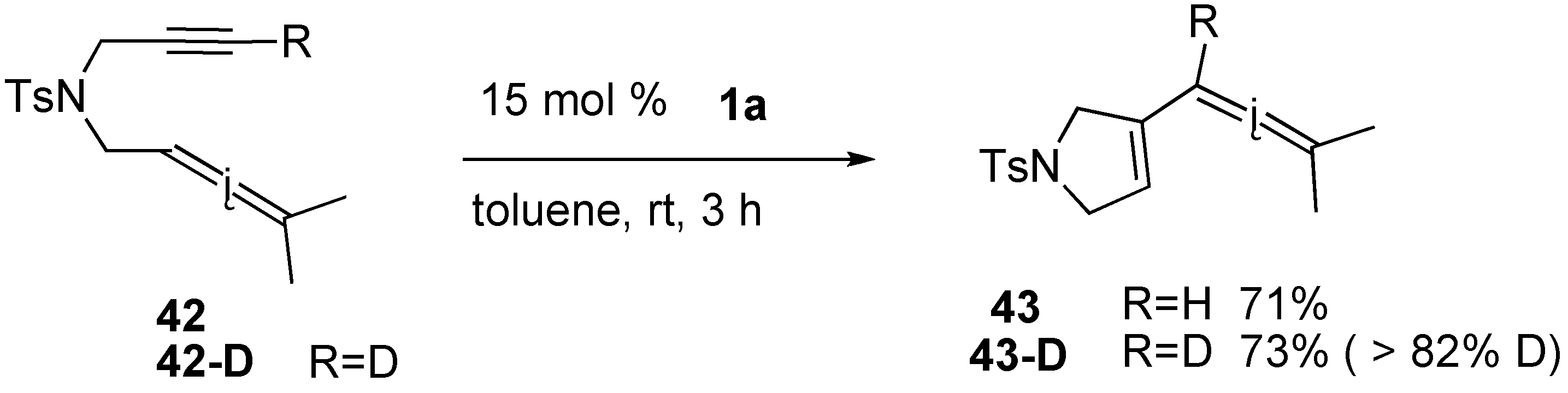
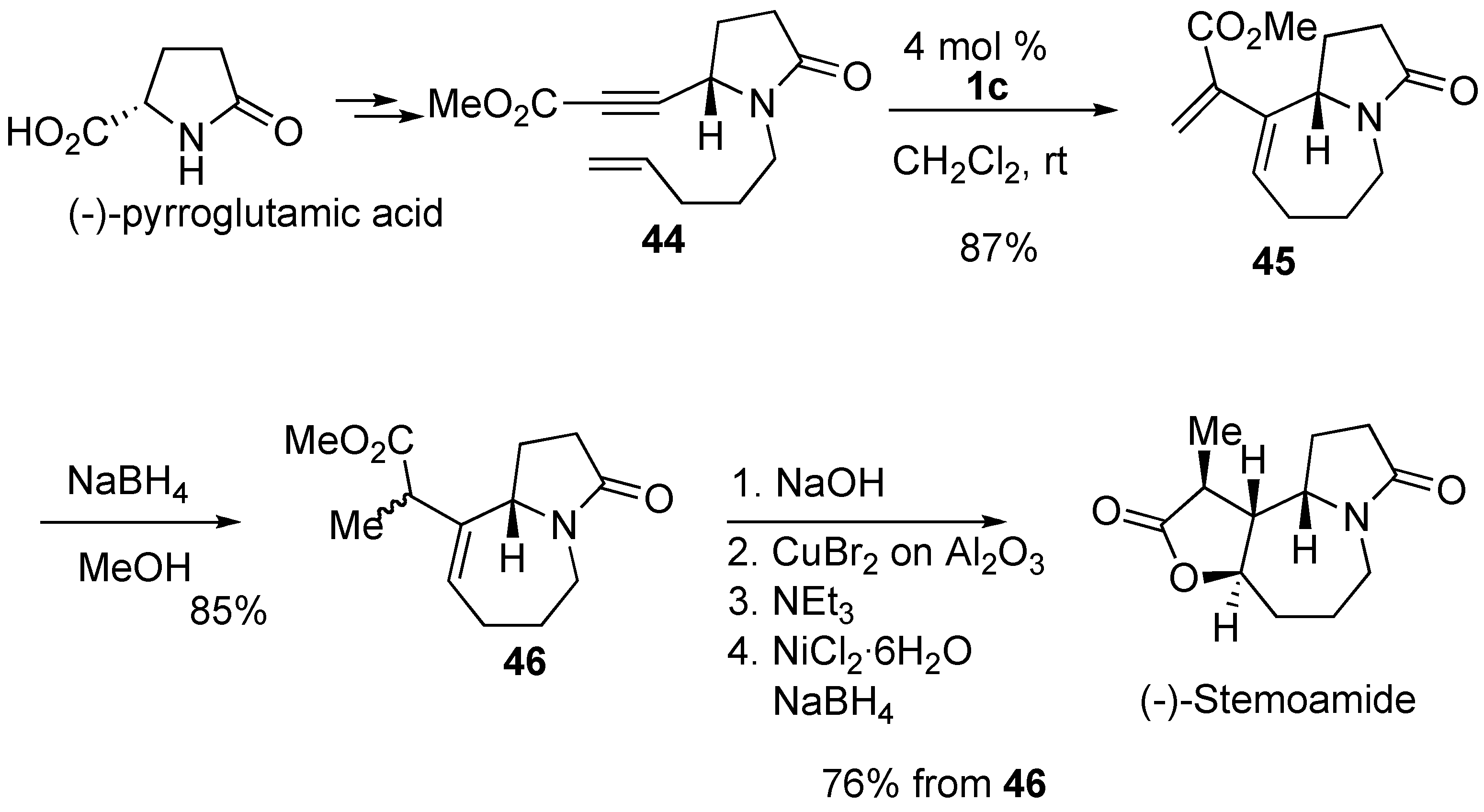
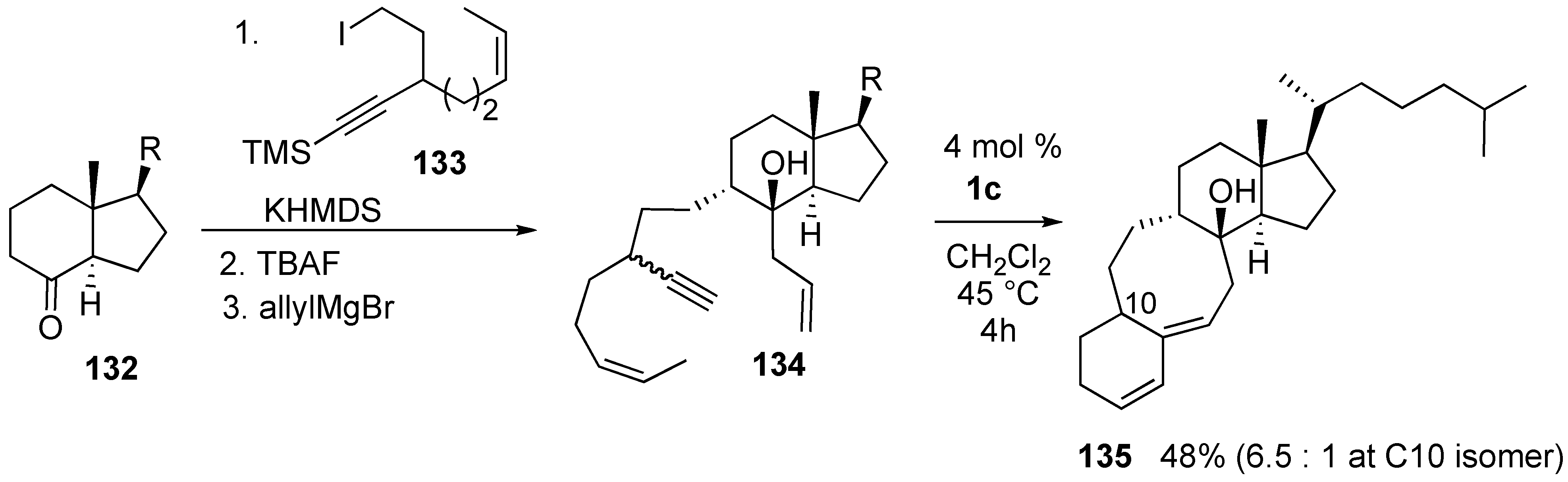
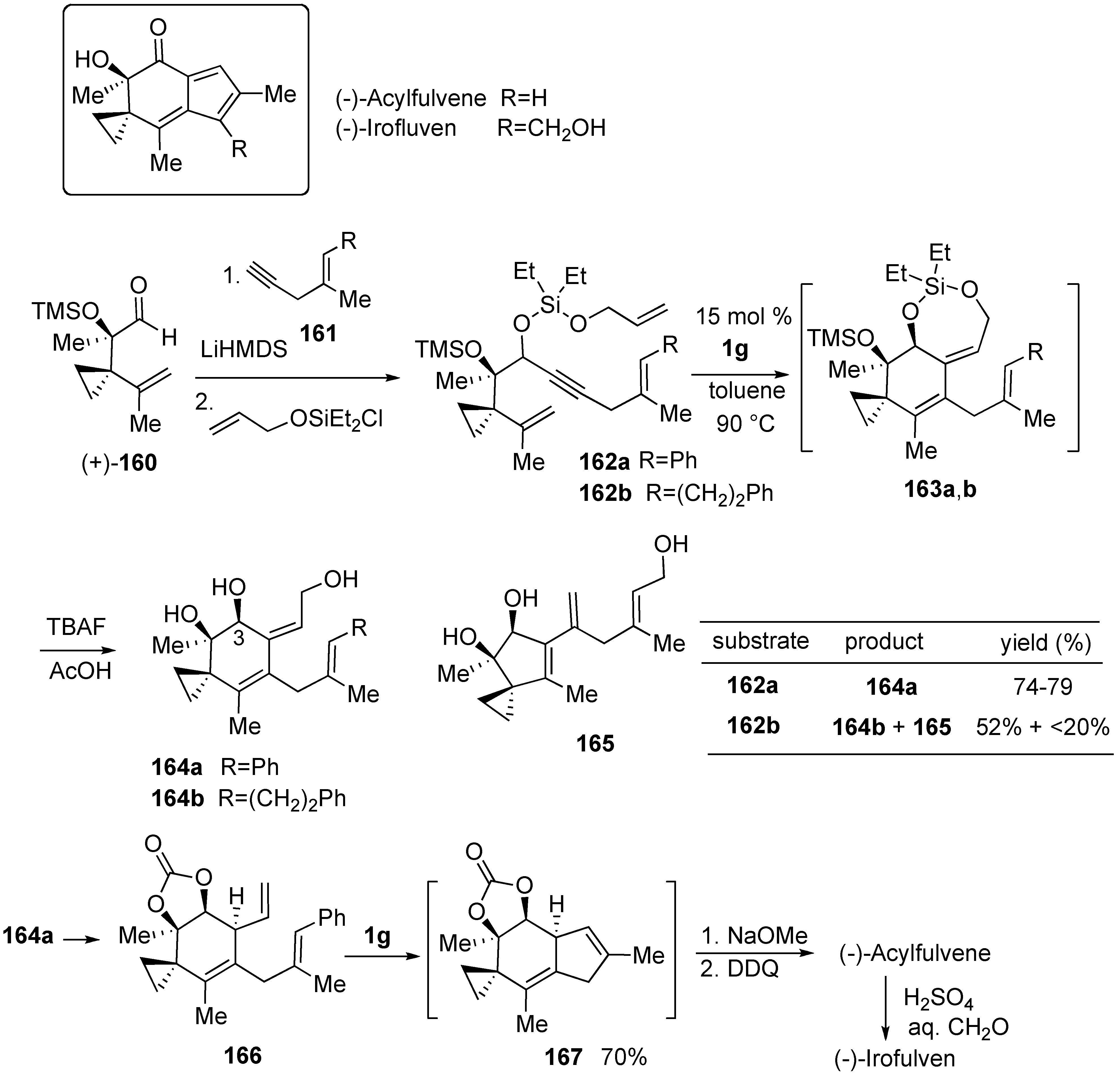
Syntheses of Natural Products and Related Compounds Using Ring-Closing Enyne Metathesis (RCM)
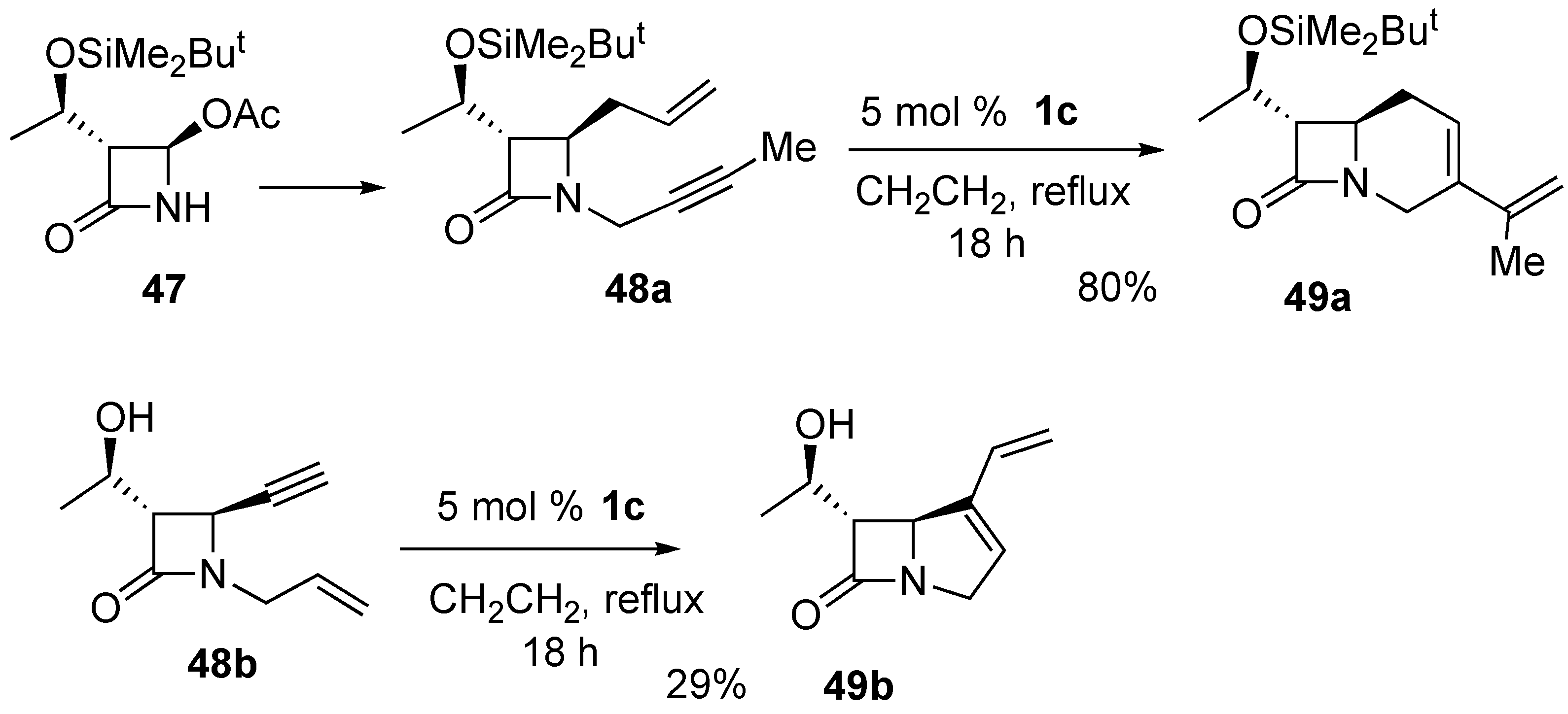
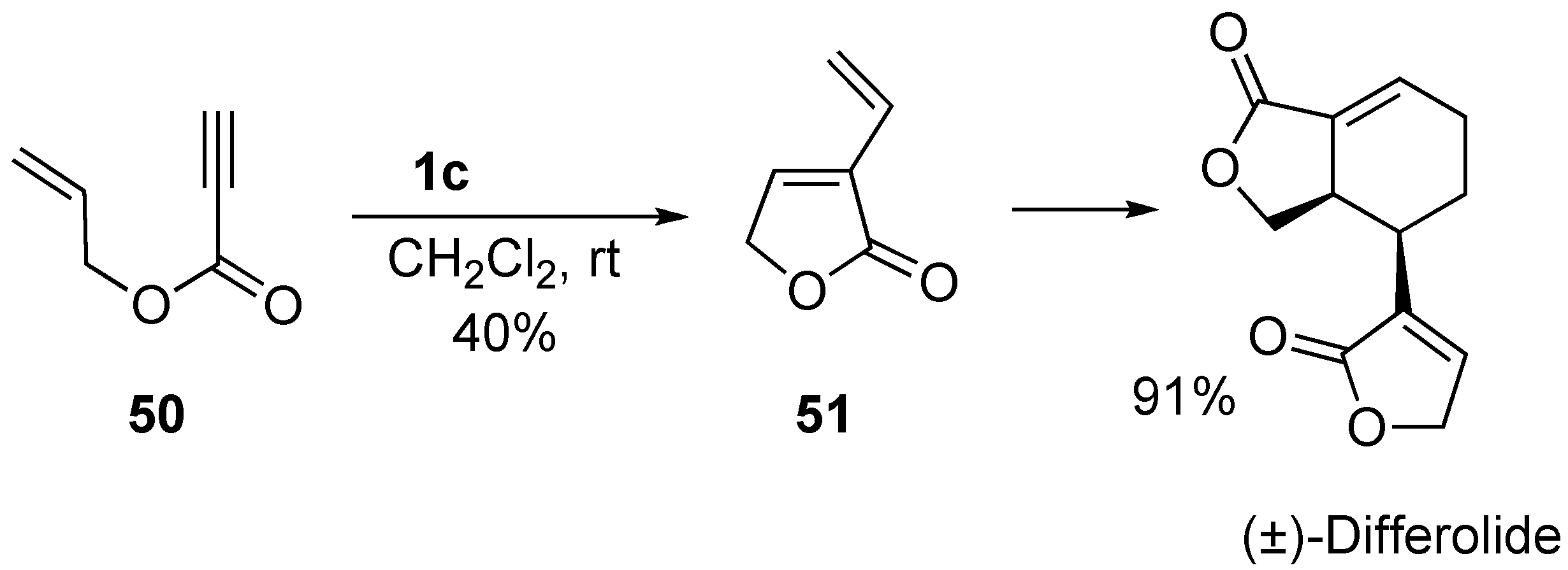

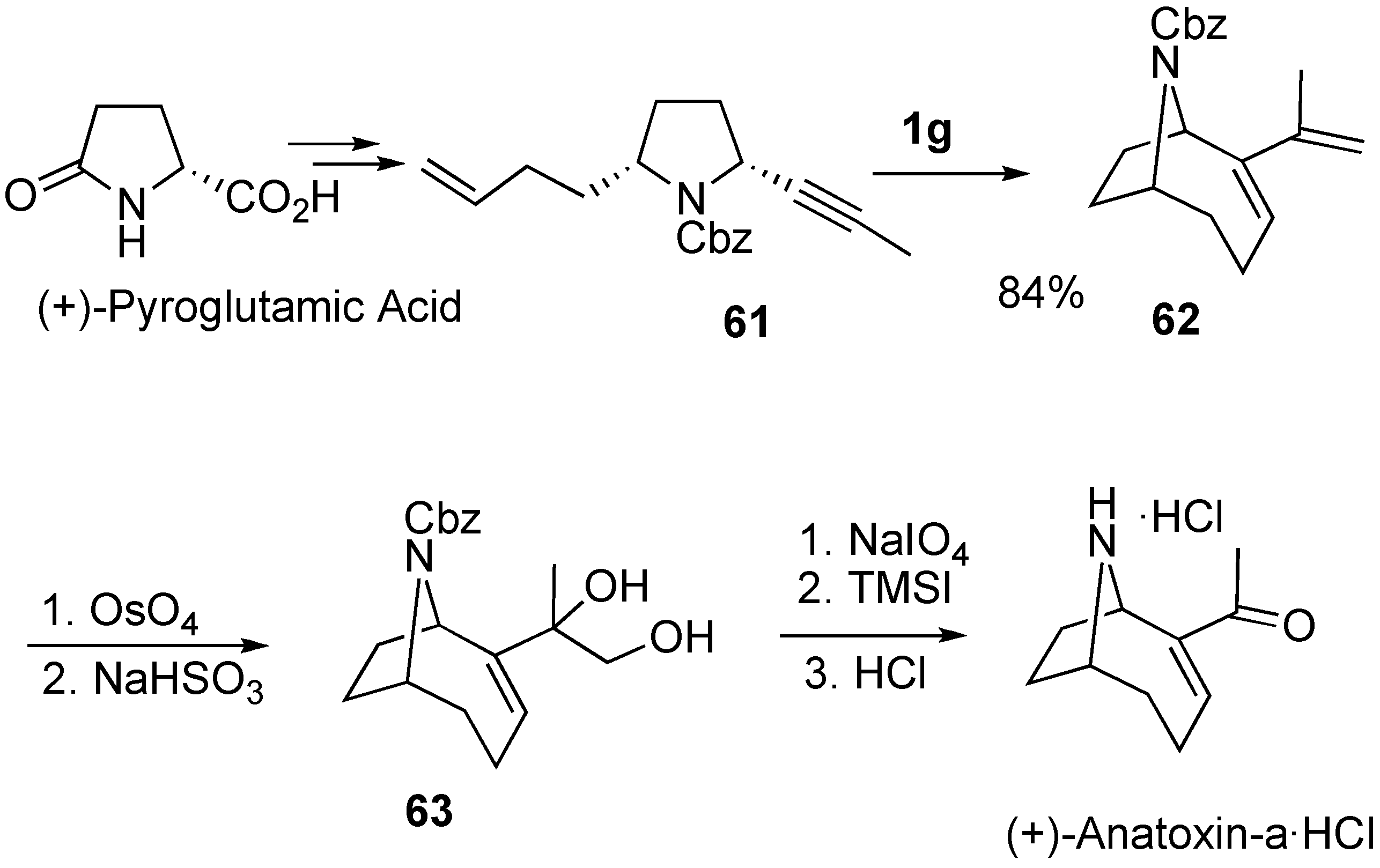
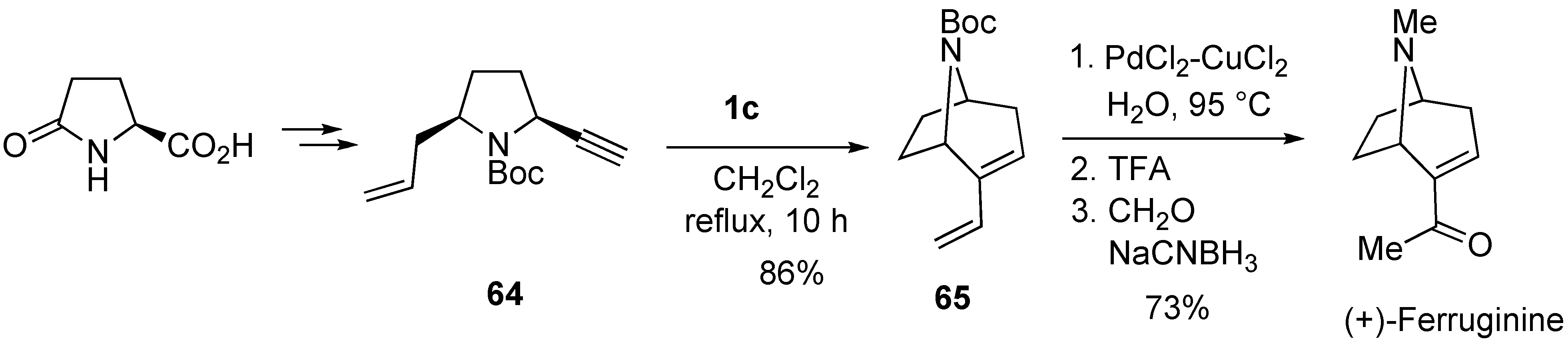

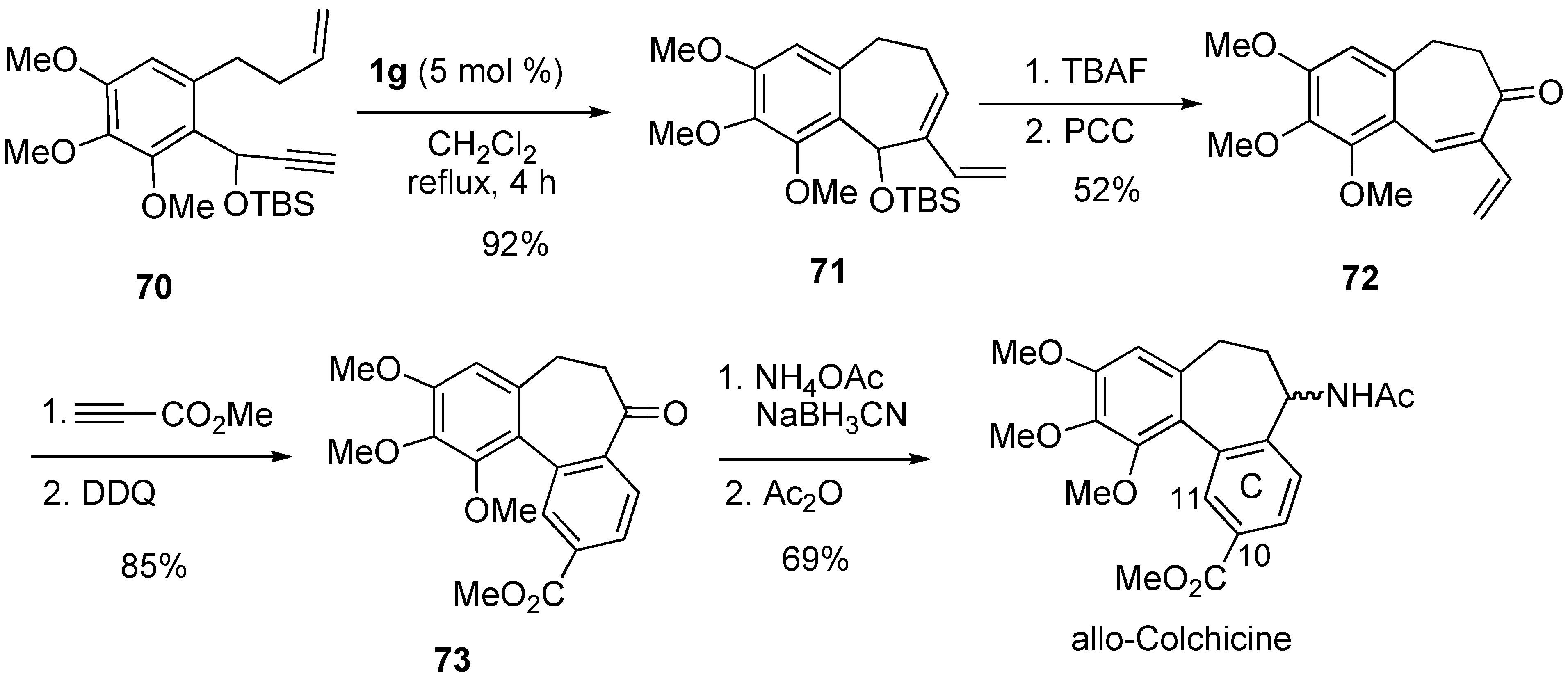

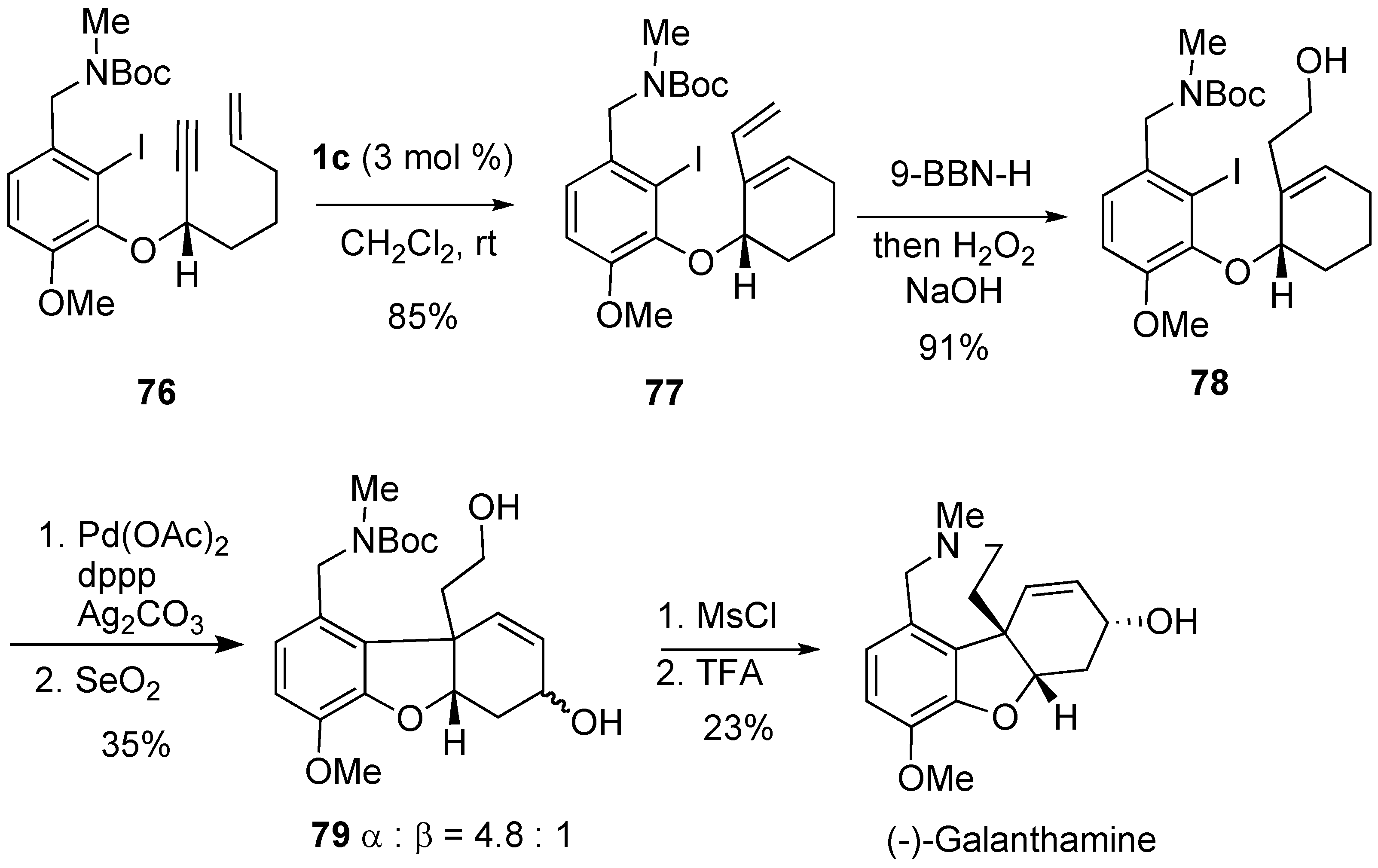
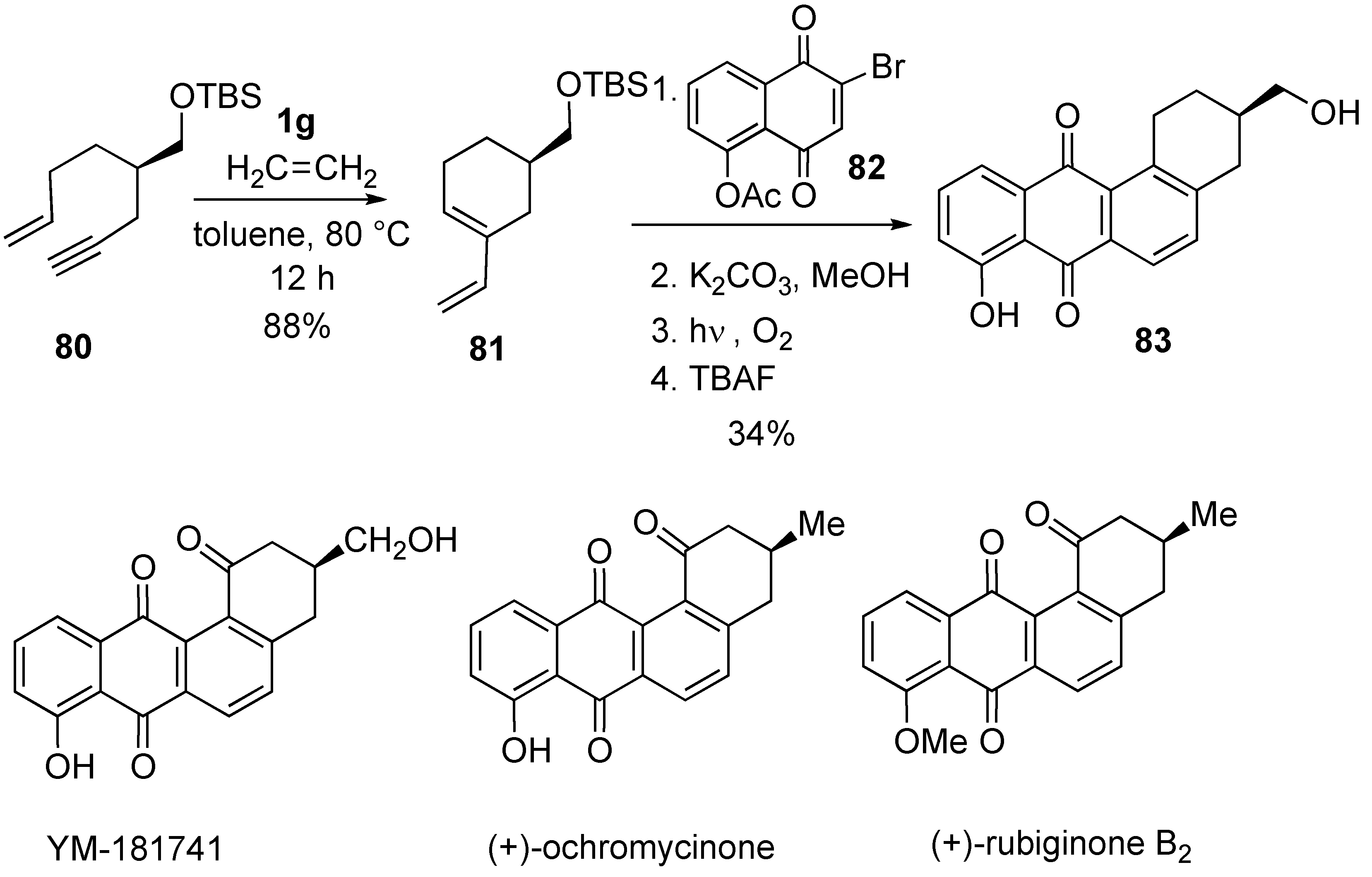
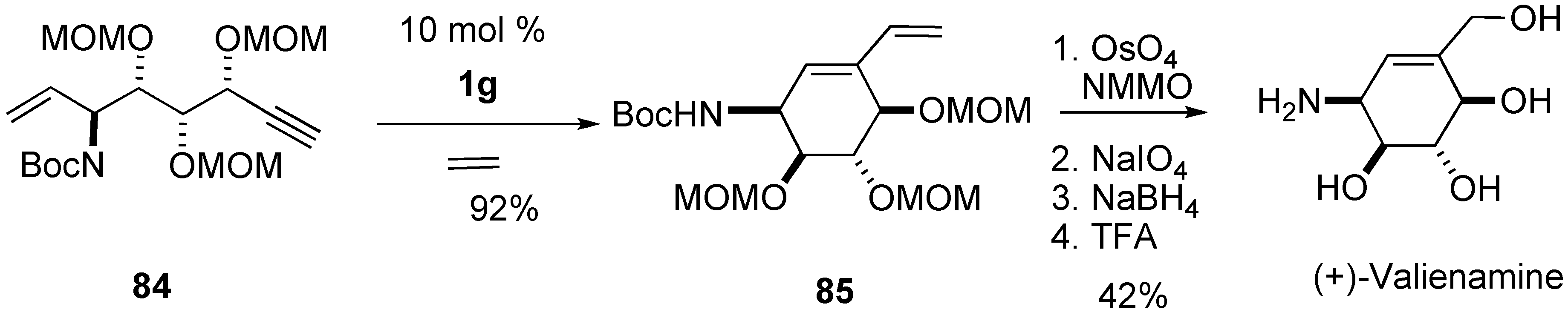
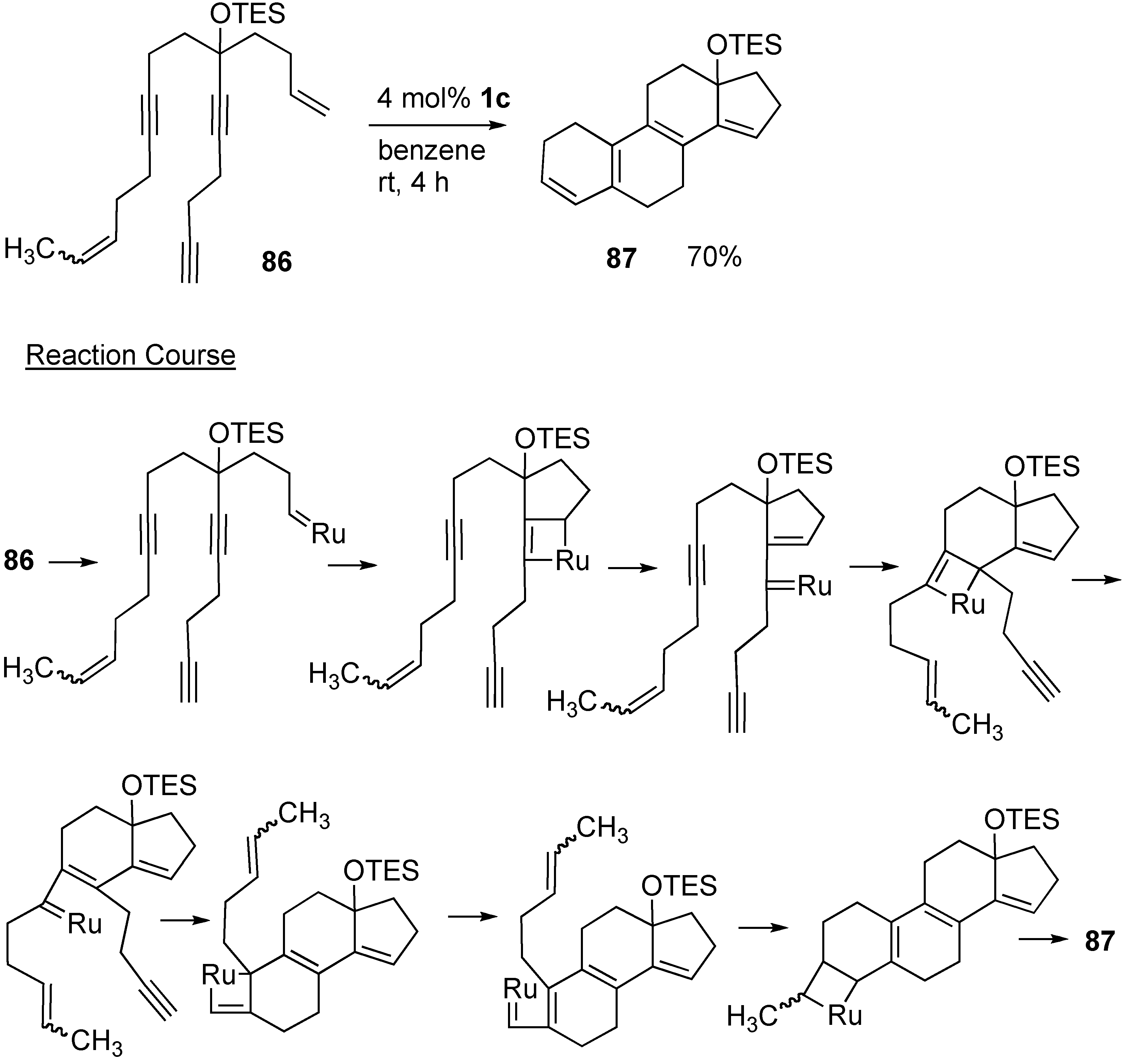
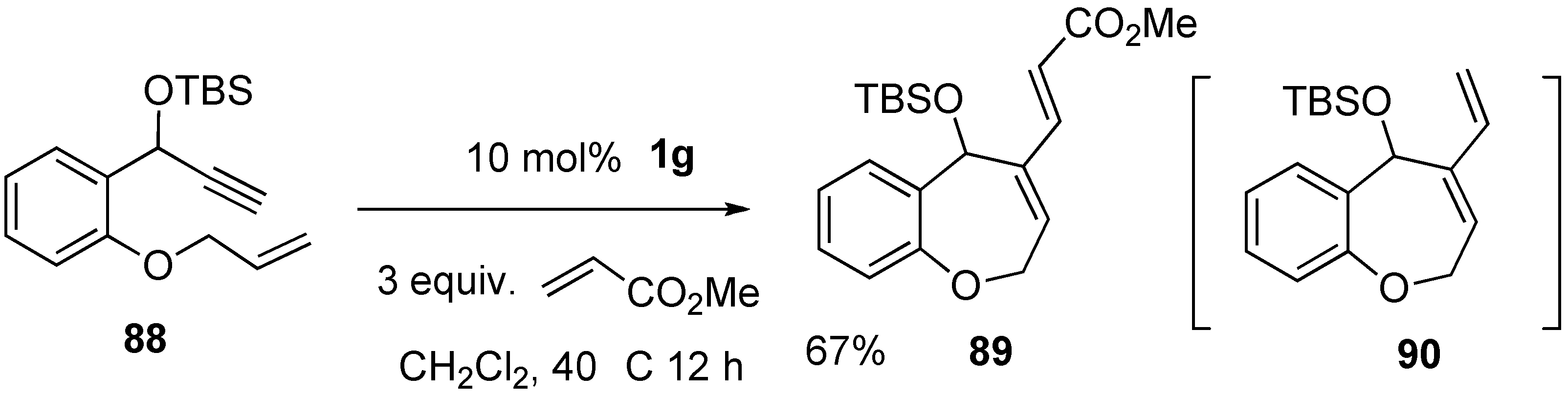
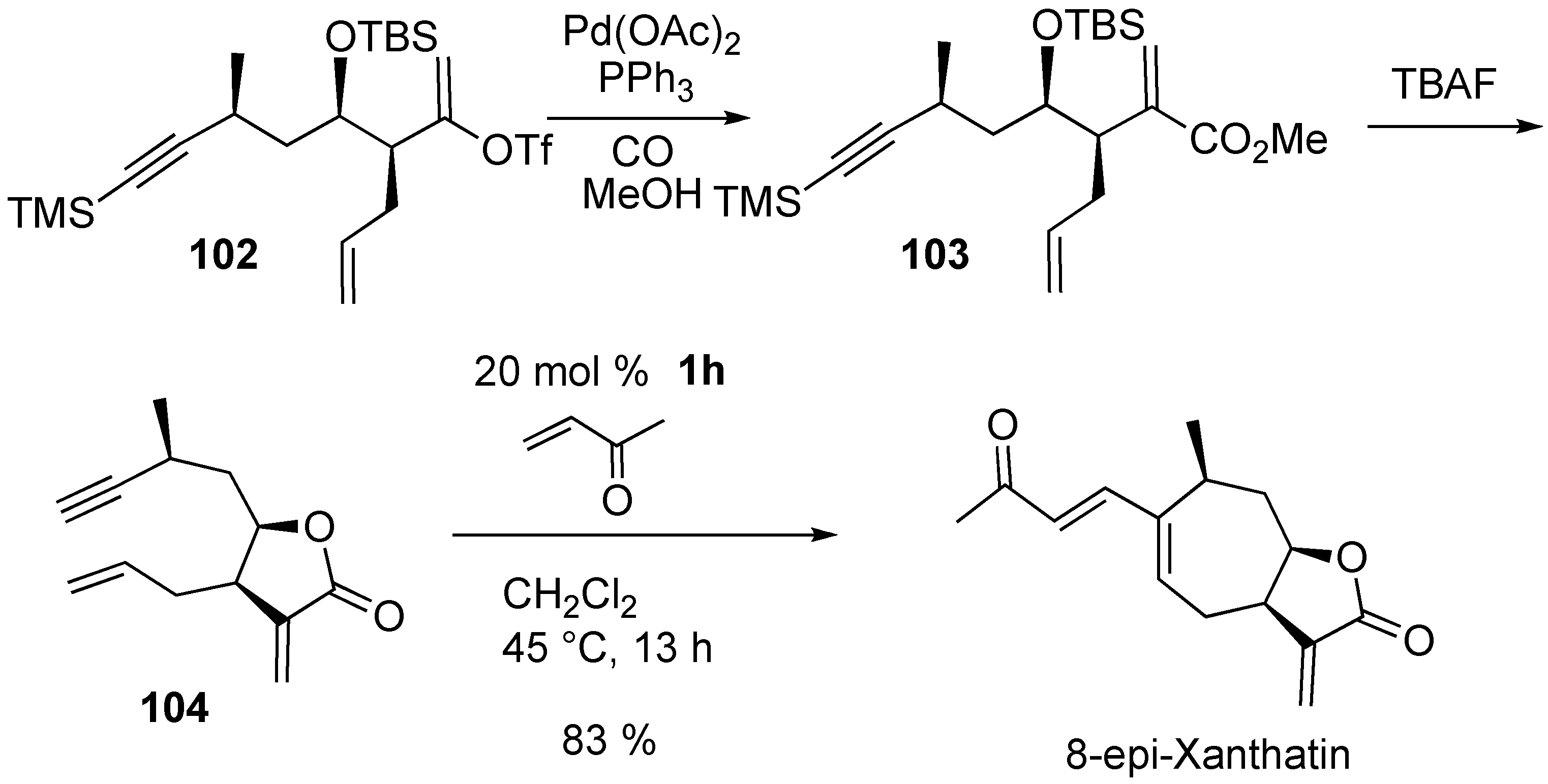

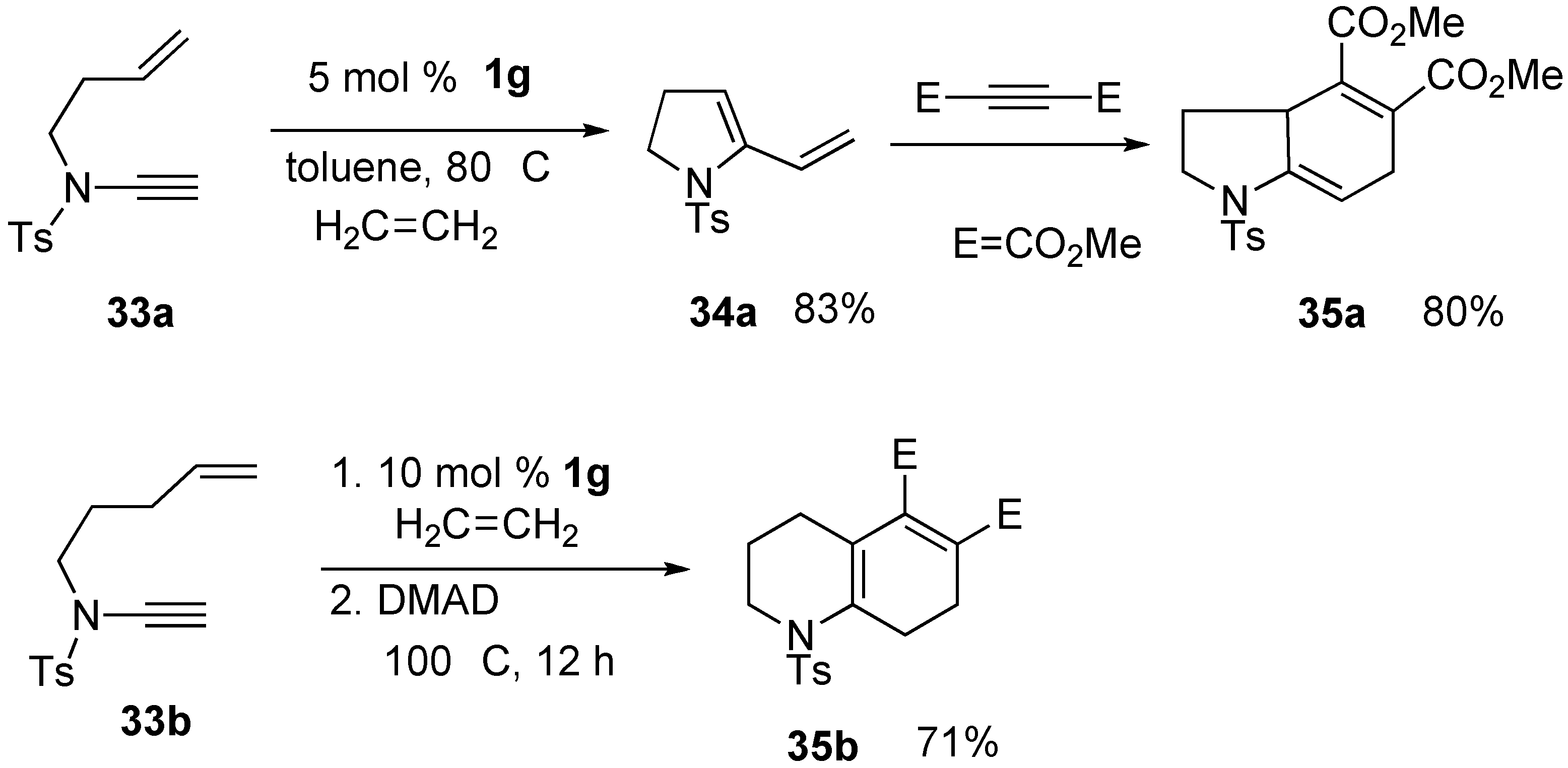
2. Ring-Closing Dienyne Metathesis
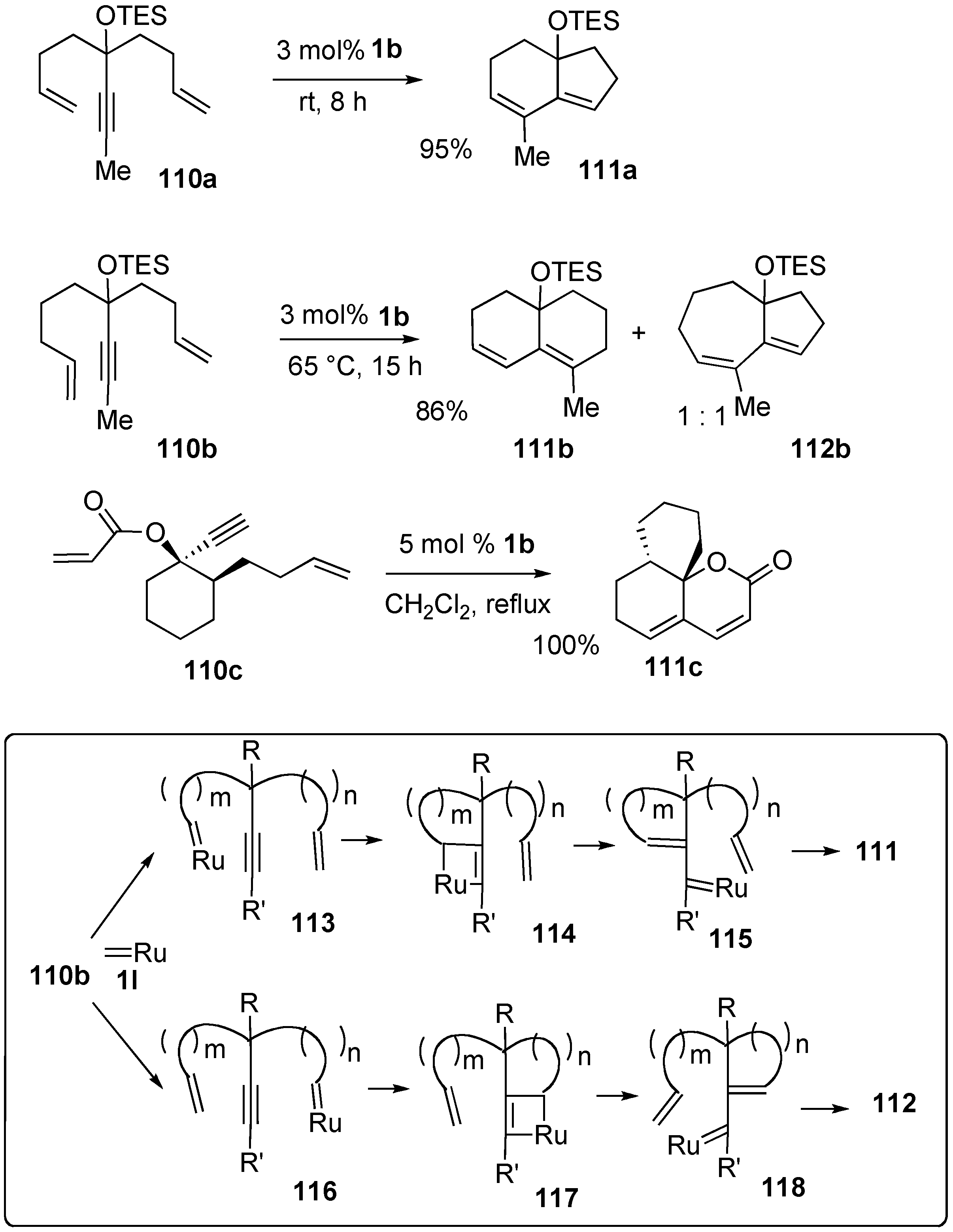


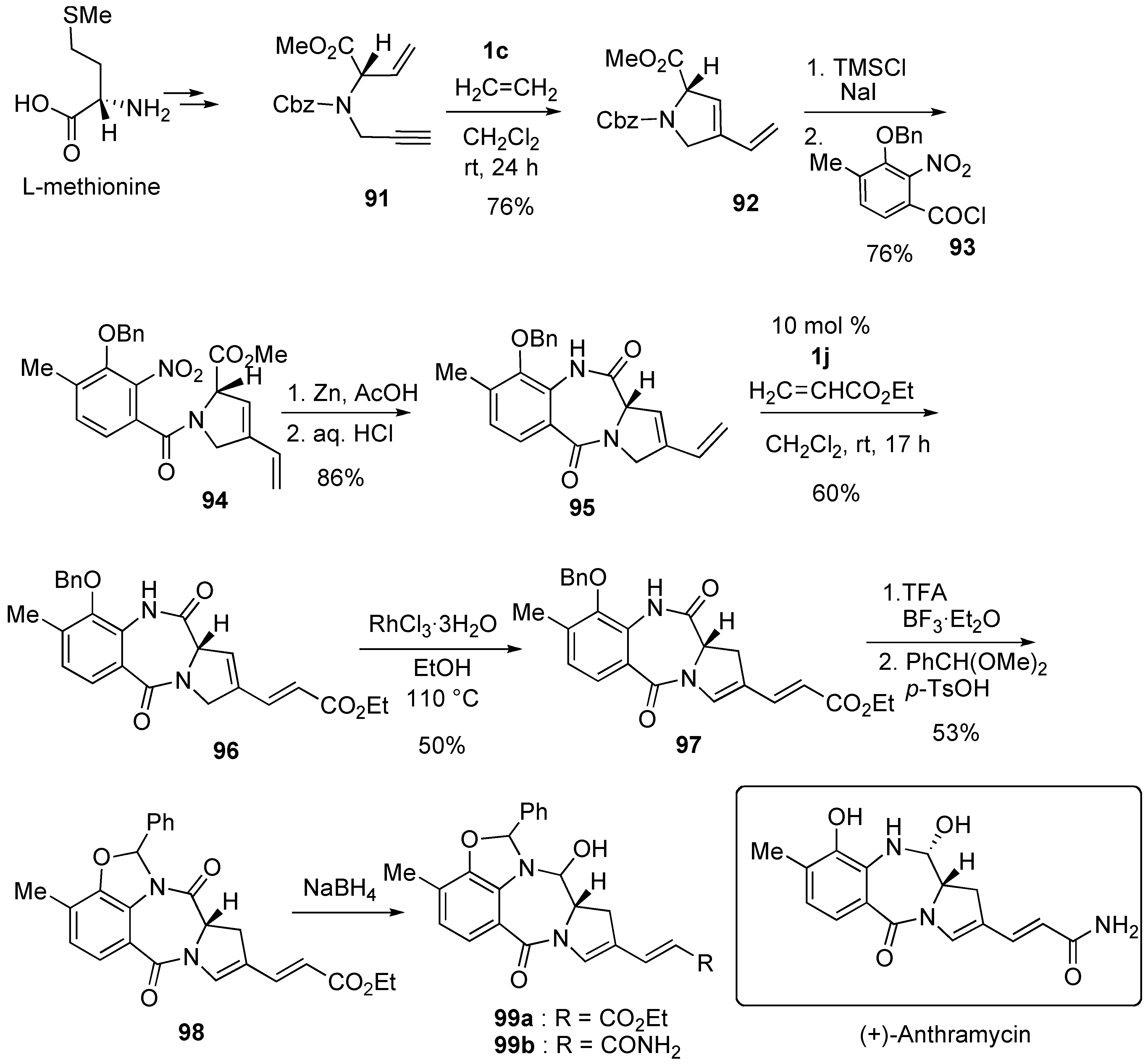
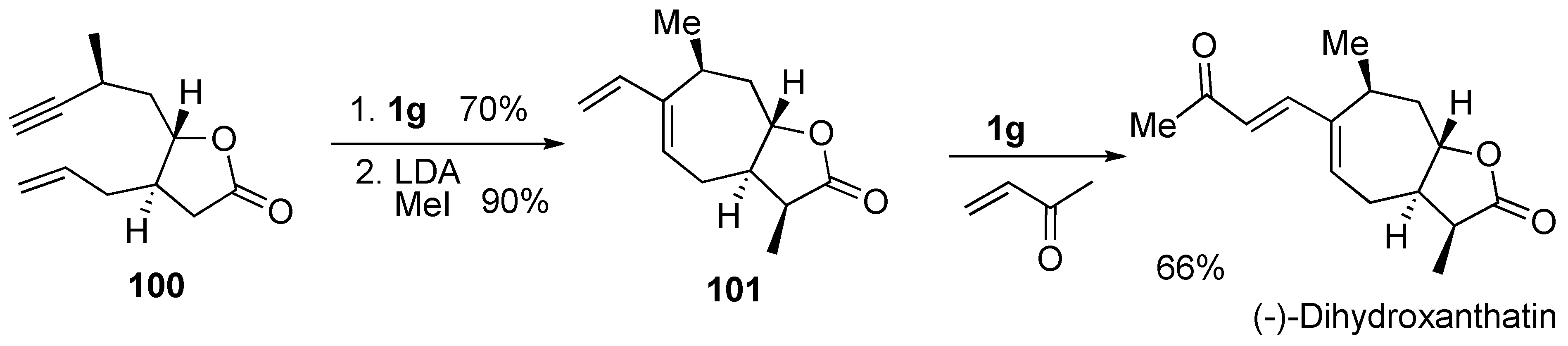
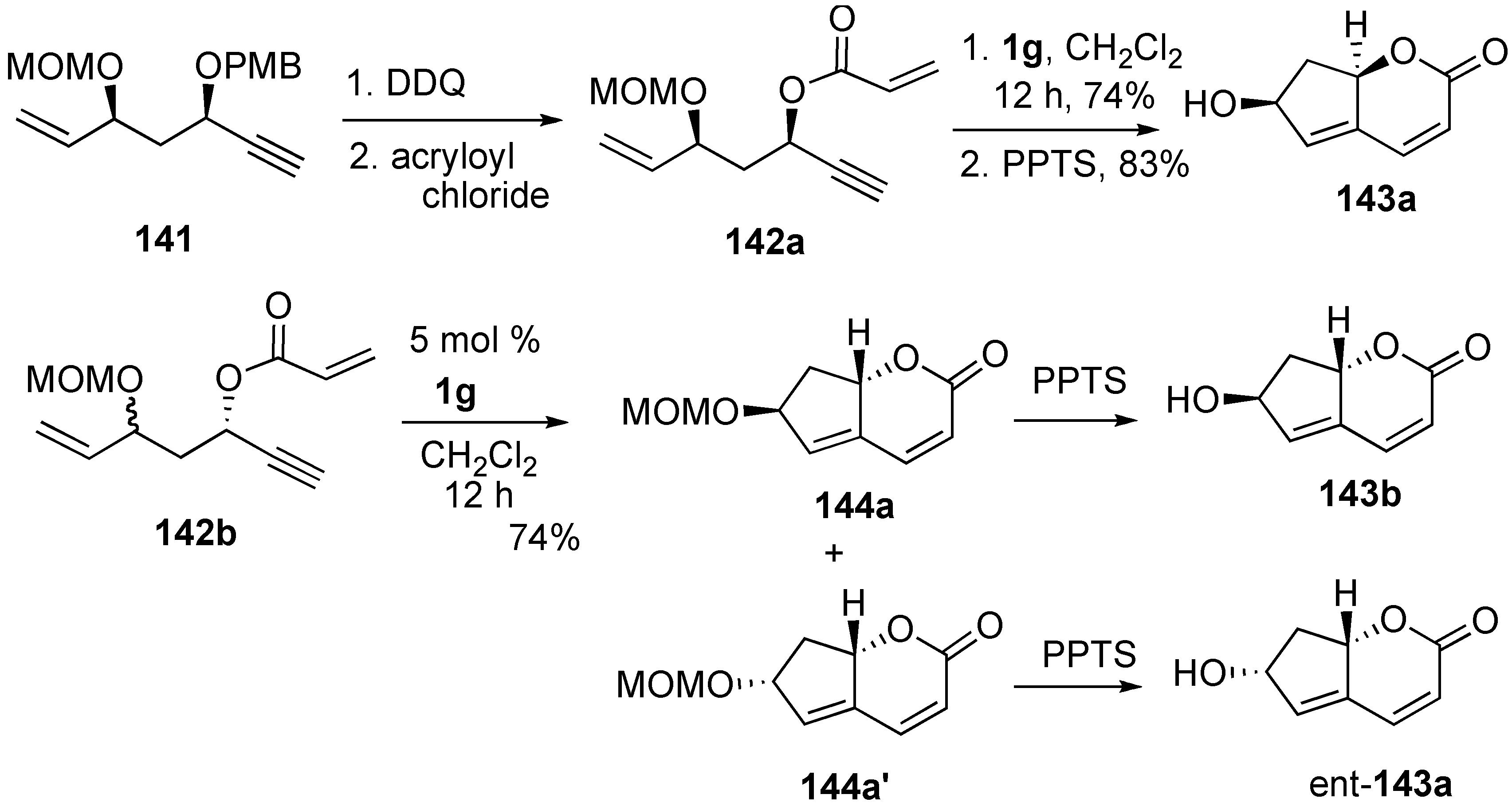
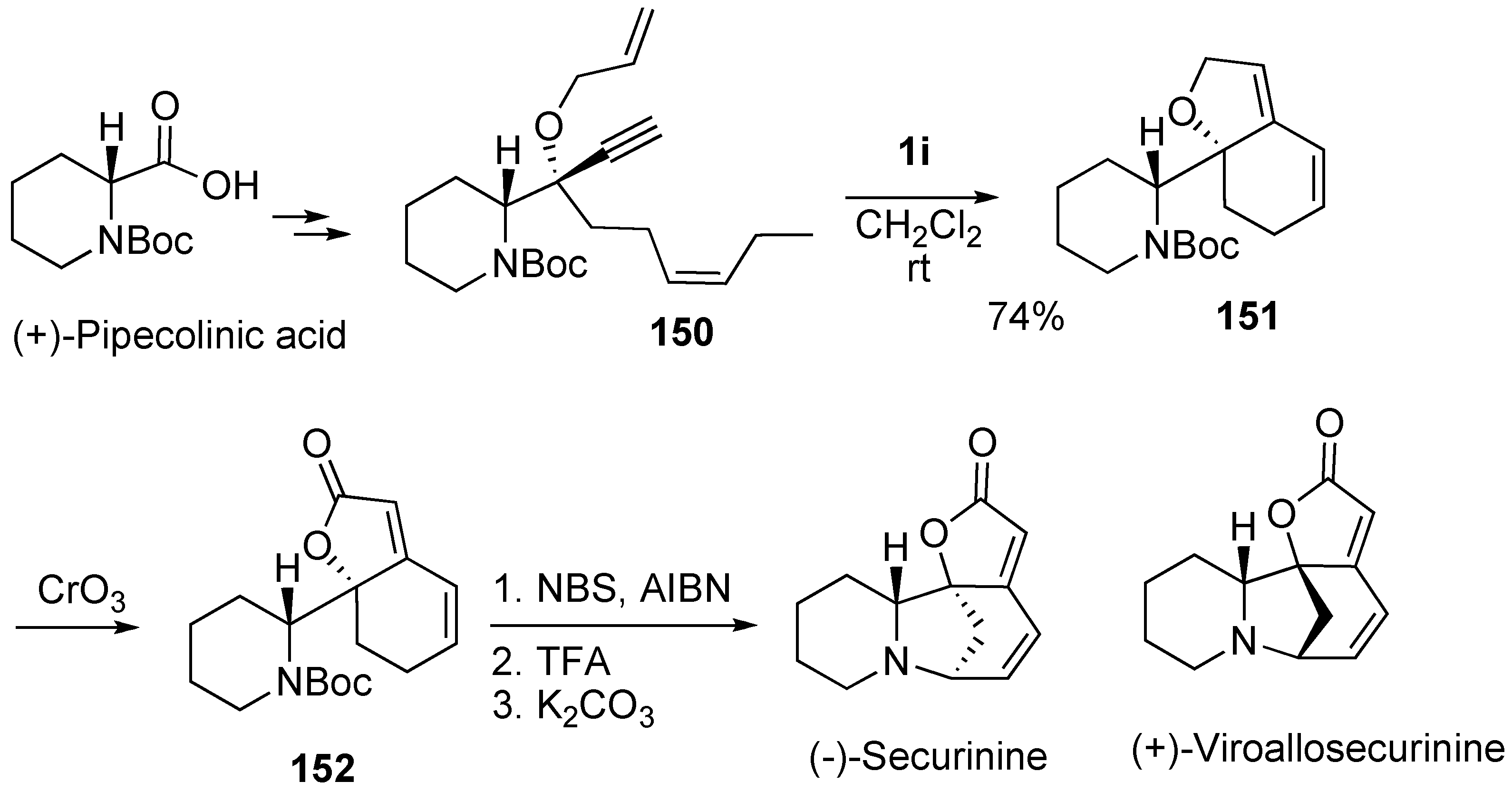
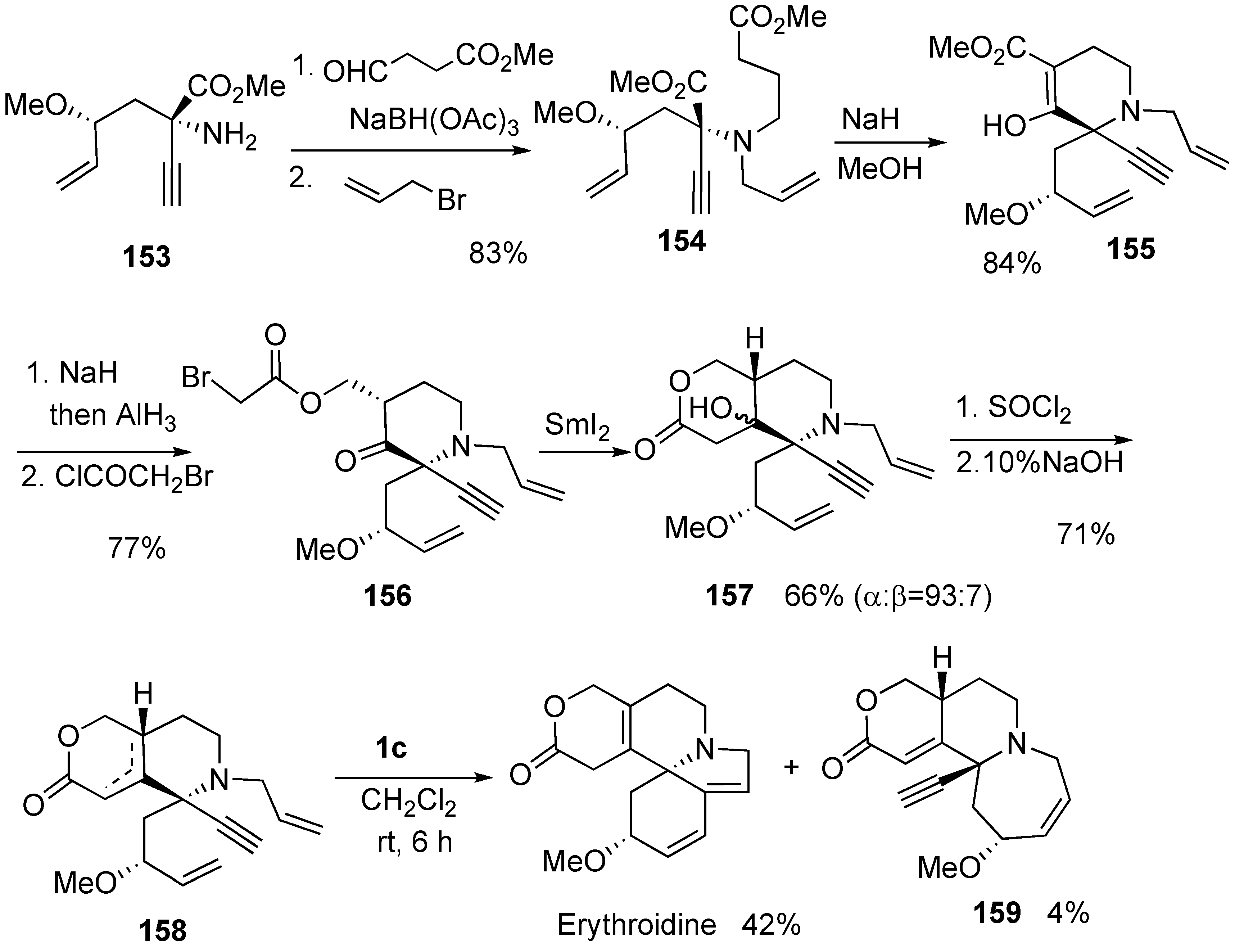
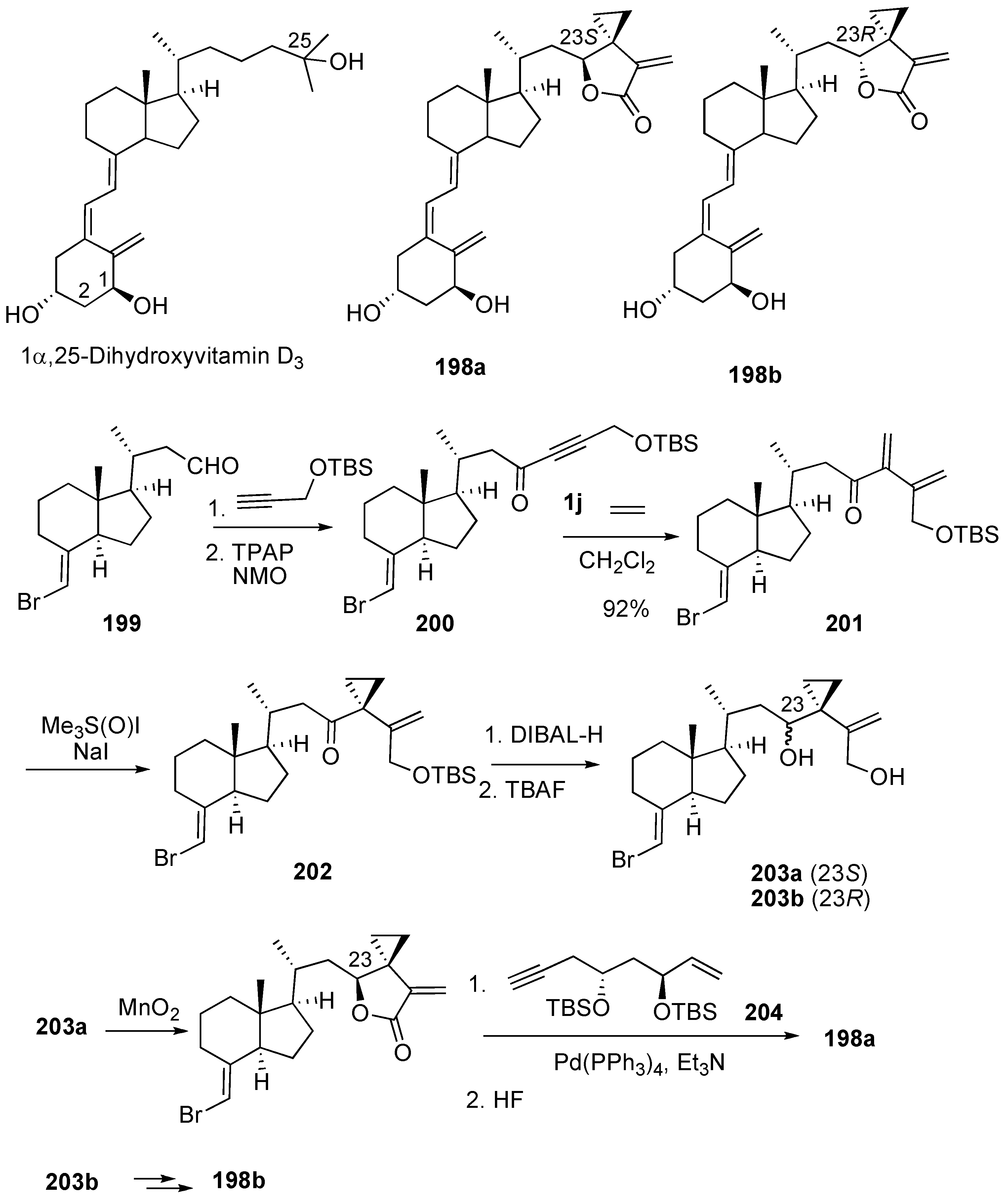
Syntheses of Natural Products and Related Compounds Using Dienyne Metathesis



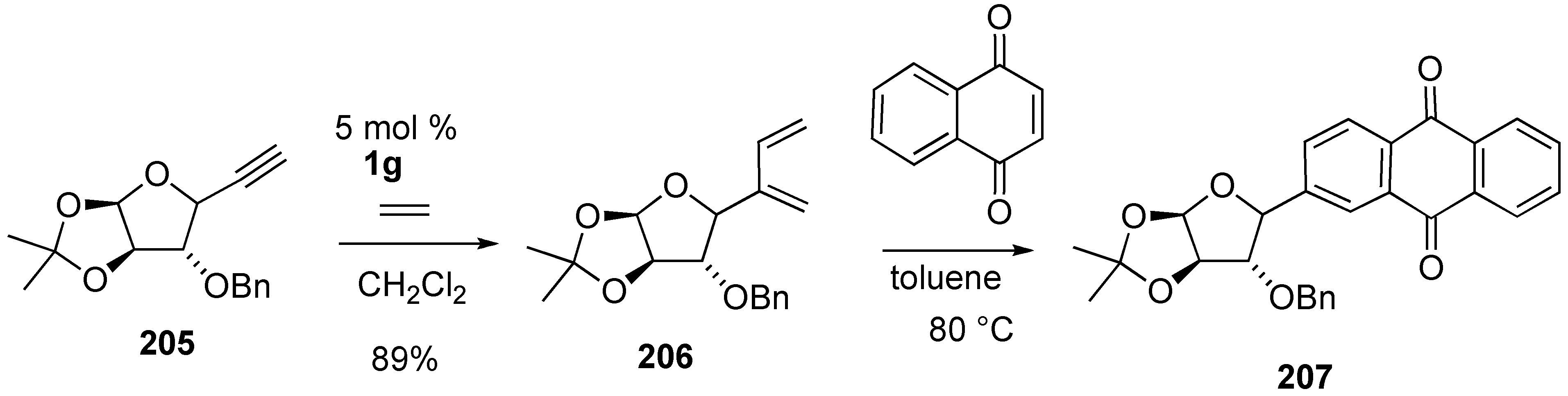
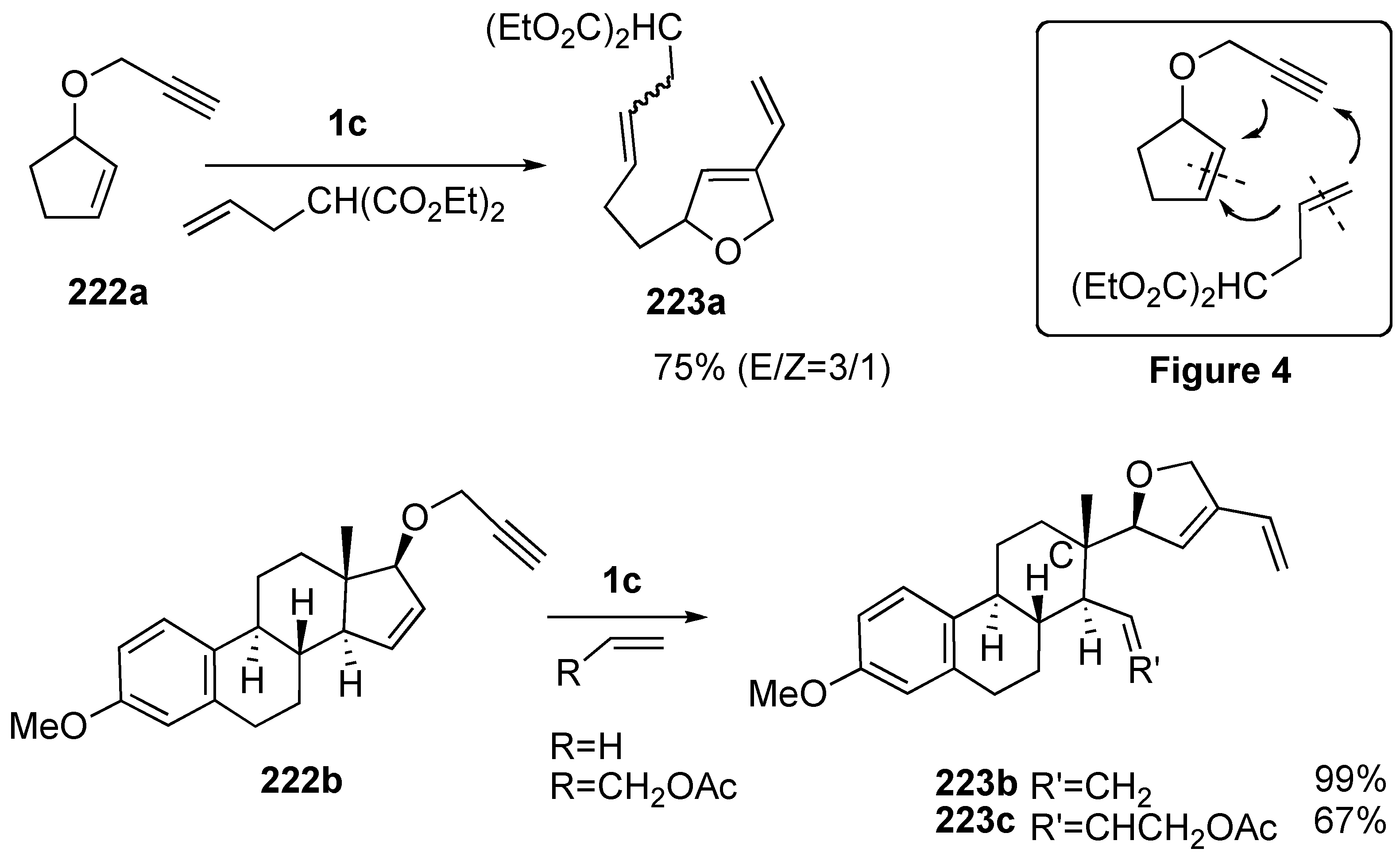
3. Cross Enyne Metathesis
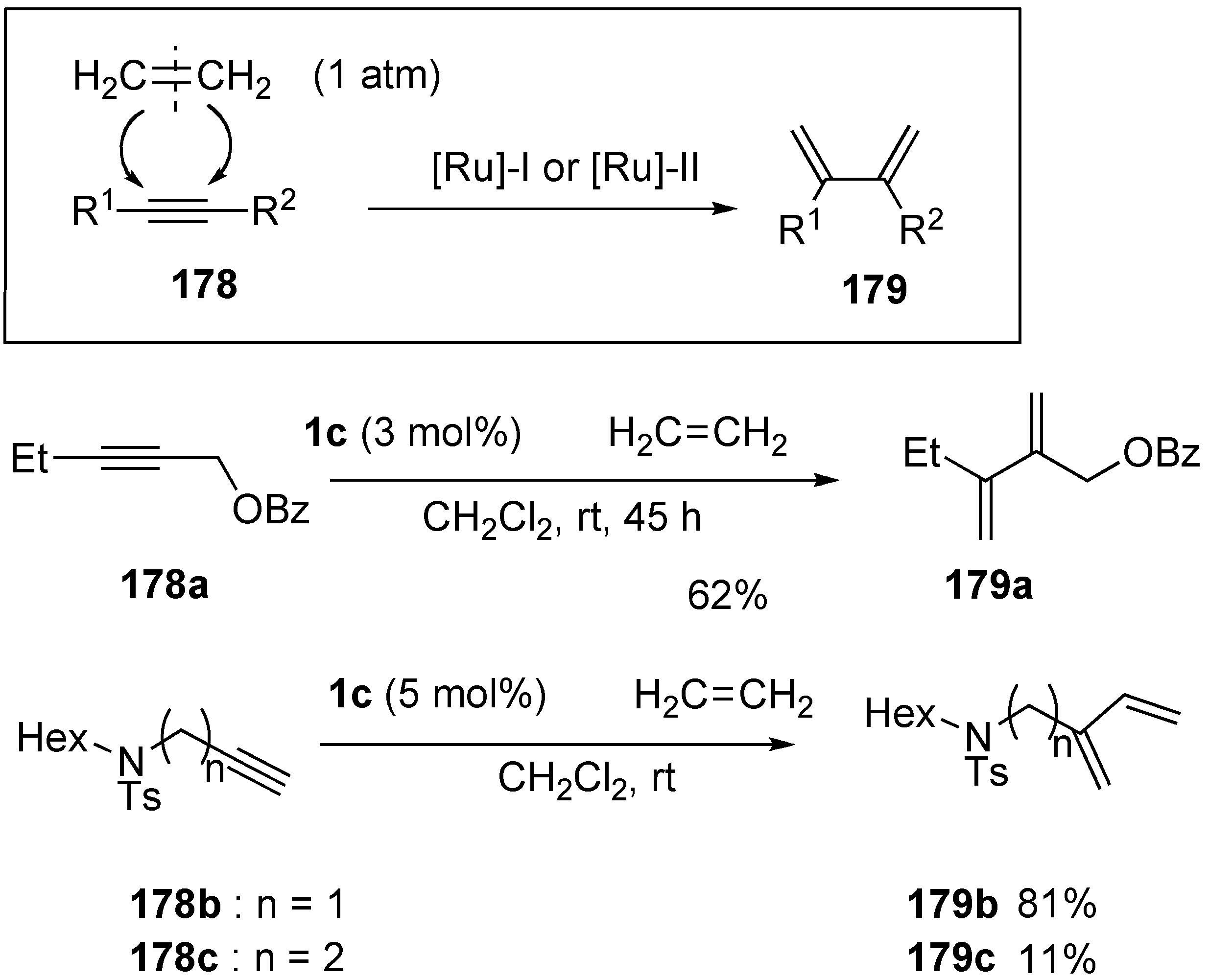

| entry | alkyne 178 | 1,3-diene 179 | time (h) | yield (%) |
|---|---|---|---|---|
| 1 |  |  | 0.5 | 88 |
| 2 |  |  | 0.5 | 71 |
| 3 |  |  | 0.5 | 85 |
| 4 |  |  | 16 | 87 |
| 5 |  |  | 16 | 43 |
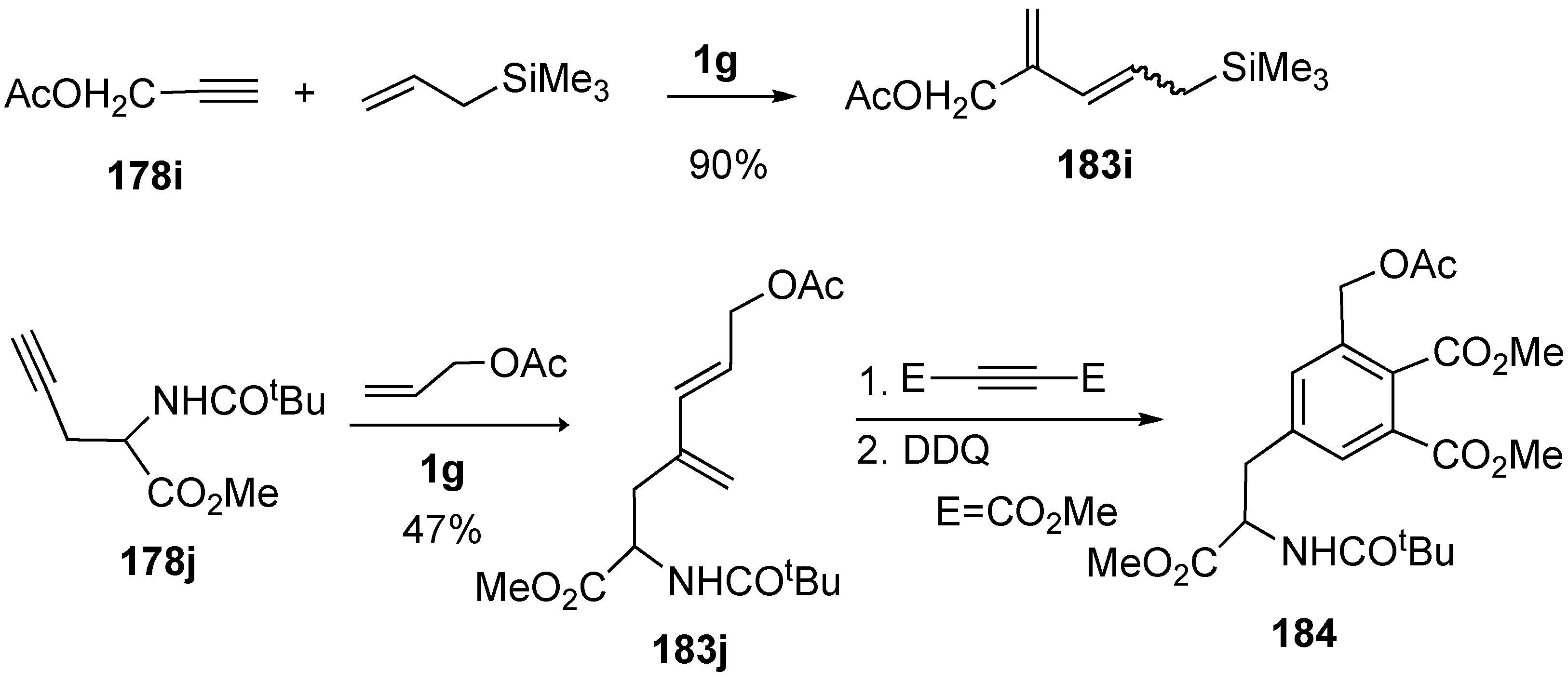
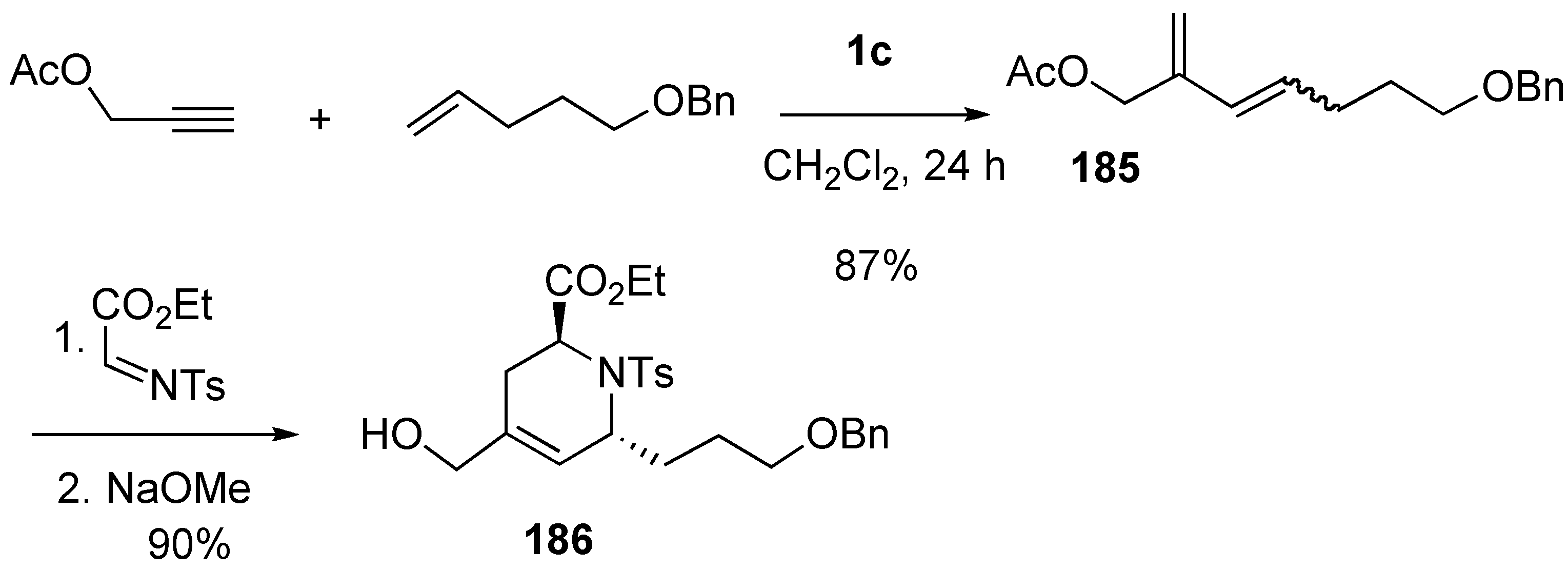

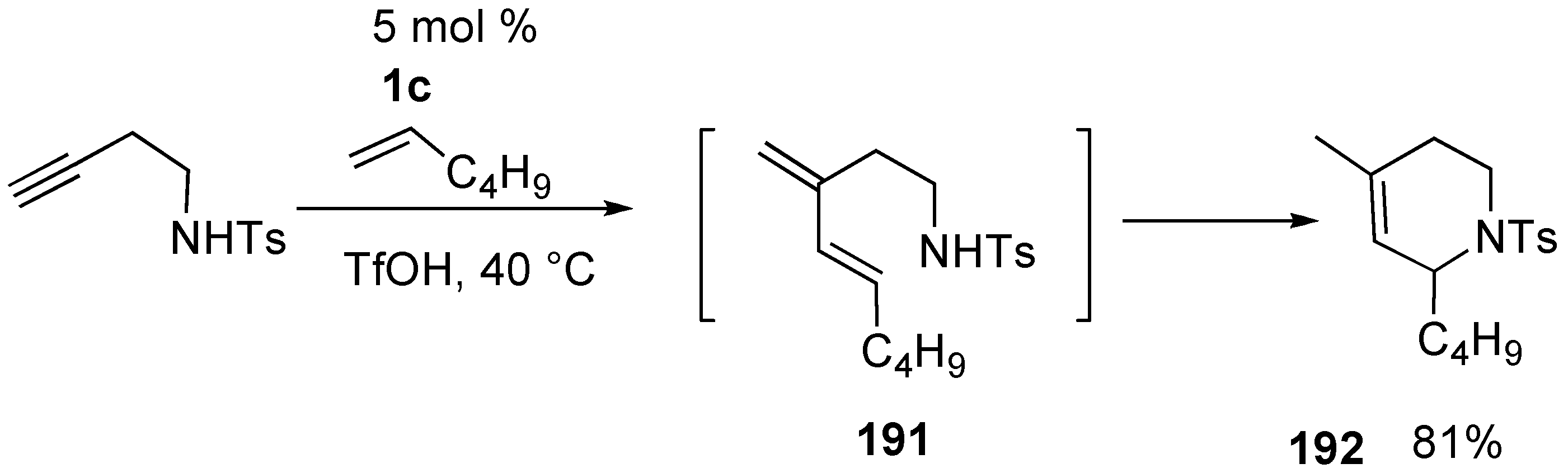
Synthesis of Natural Products and Related Compounds Using Cross Metathesis



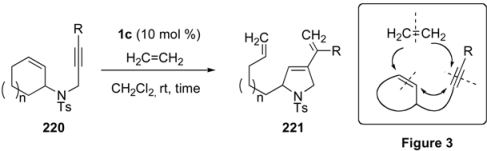
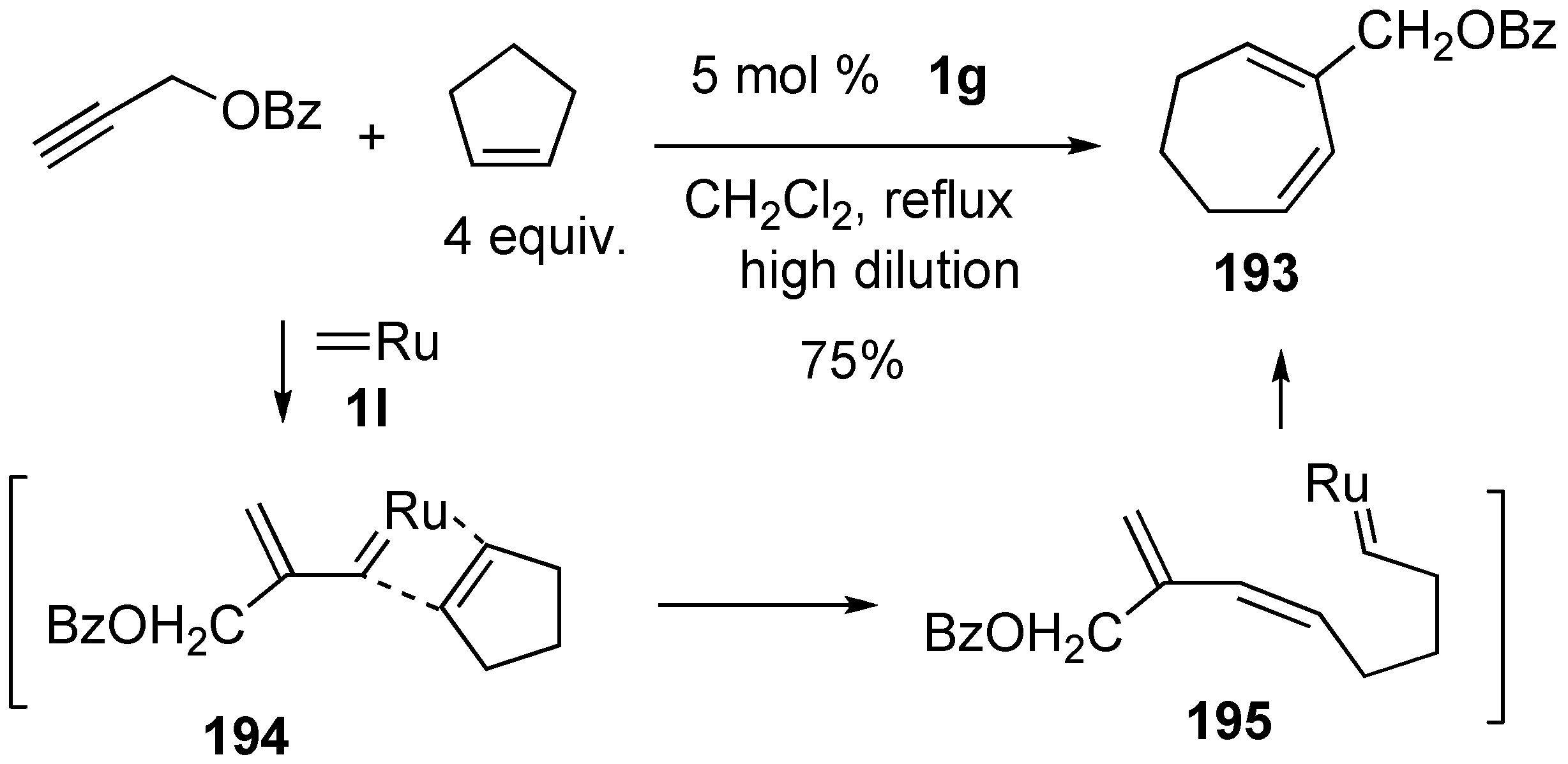
4. Ring-Opening Enyne Metathesis
| entry | R | ring size | N | time (h) | yield (%)a |
|---|---|---|---|---|---|
| 1 | 220a Me | 7 | 2 | 24 | 221a 56b |
| 2 | 220b H | 6 | 1 | 4 | 221b 78 |
| 3 | 220c H | 7 | 2 | 1 | 221c 70 |
| 4 | 220d H | 8 | 3 | 1 | 221d 75 |
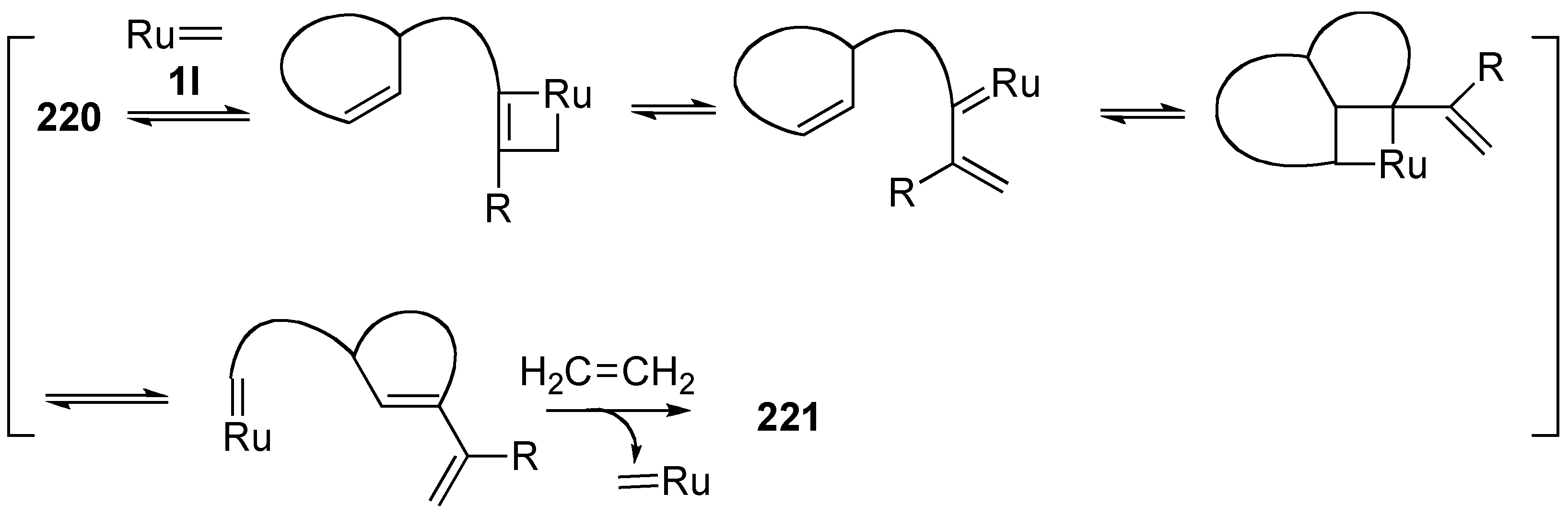


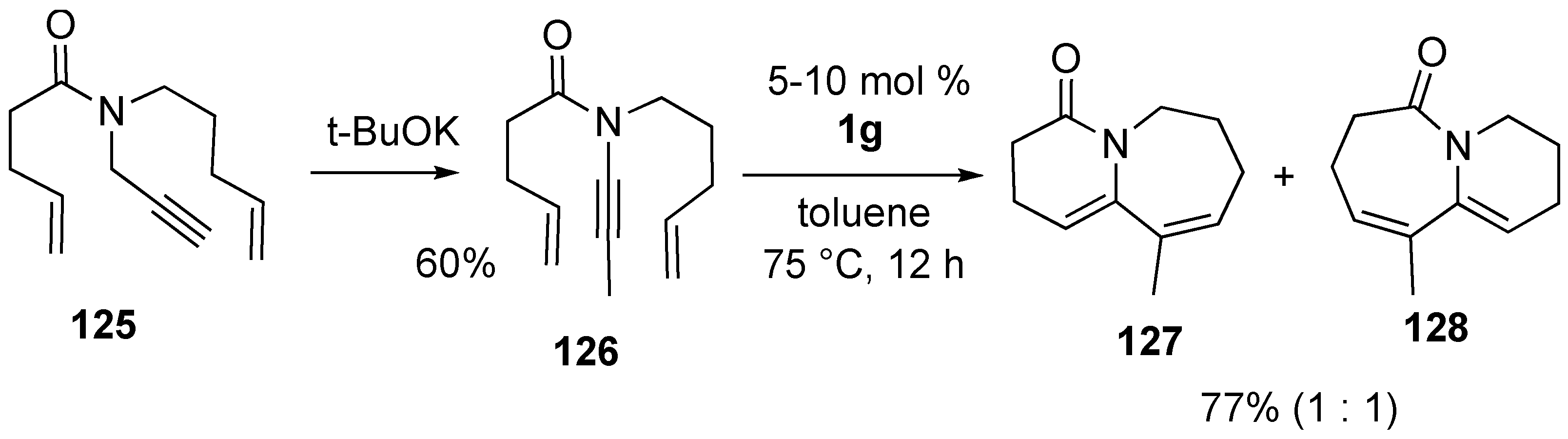
5. Skeletal Reorganization Using Transition Metals
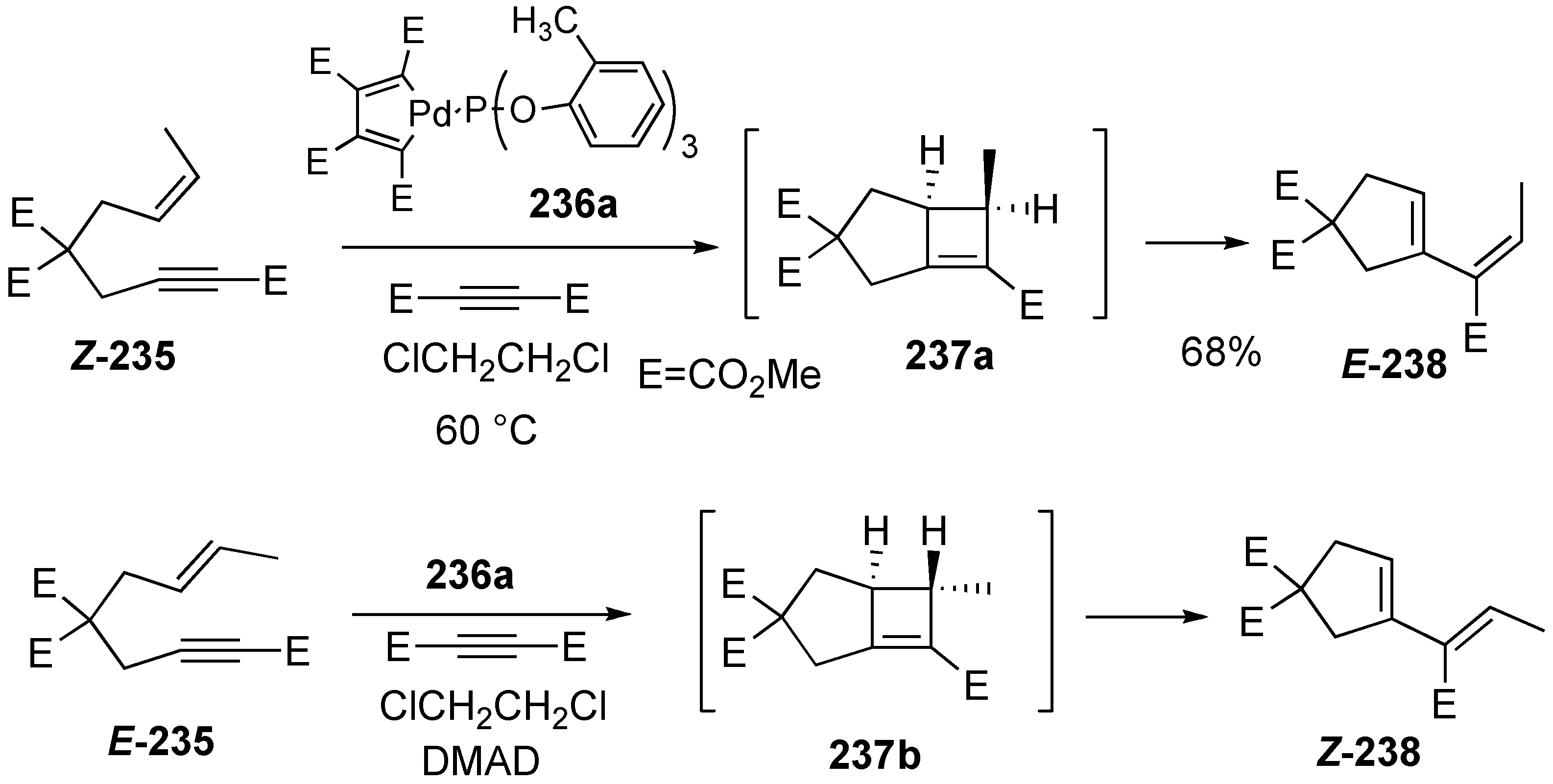
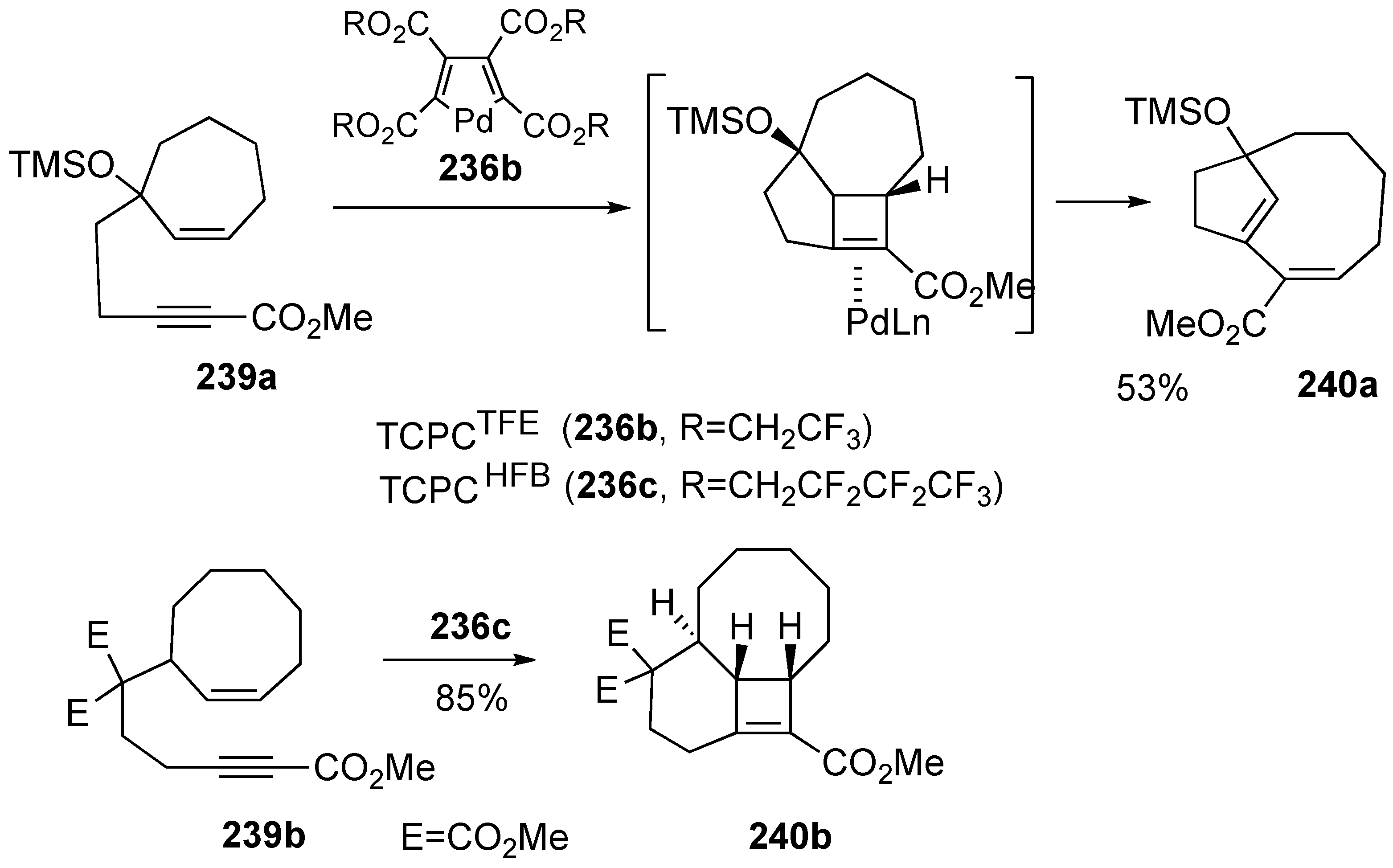

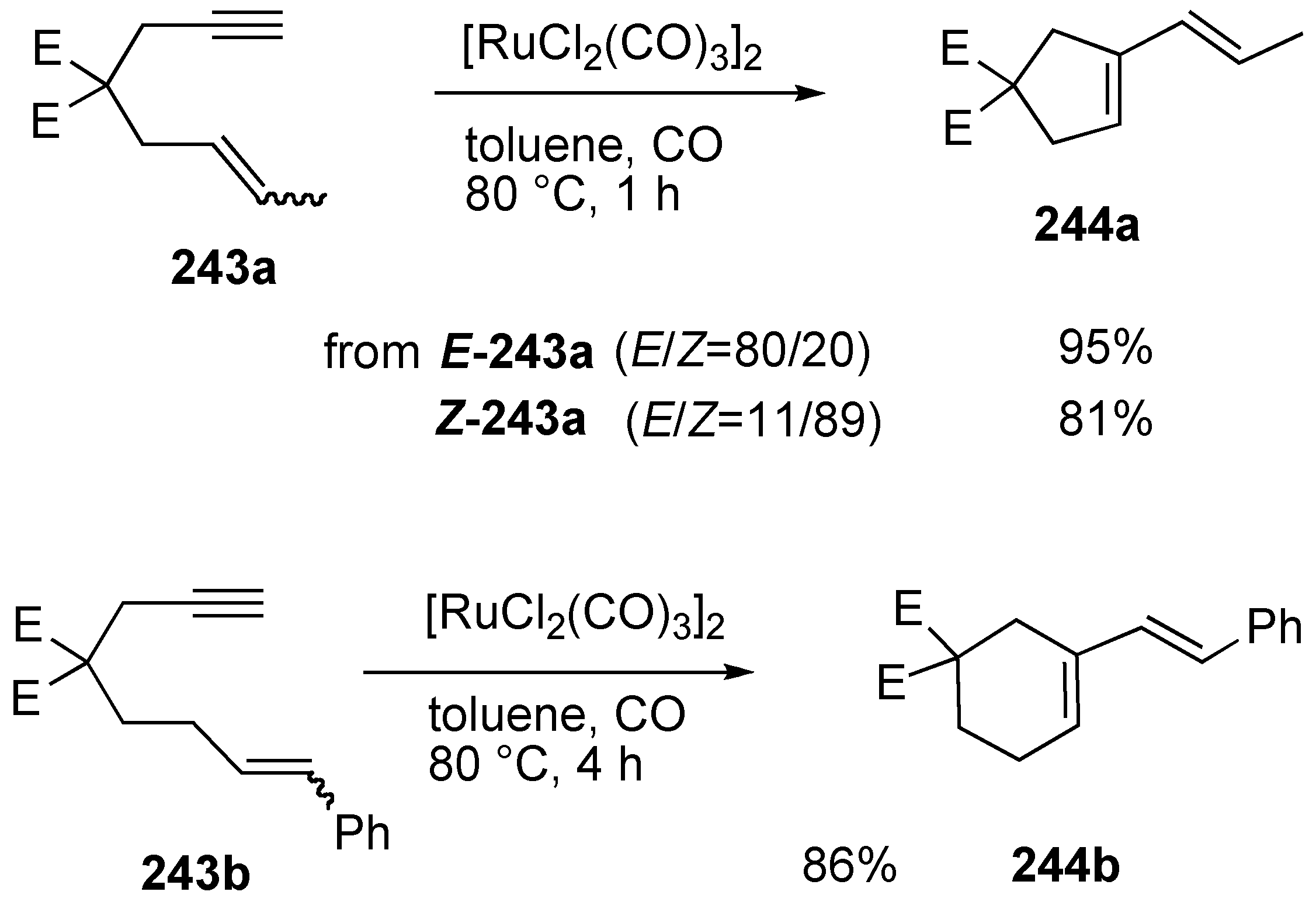
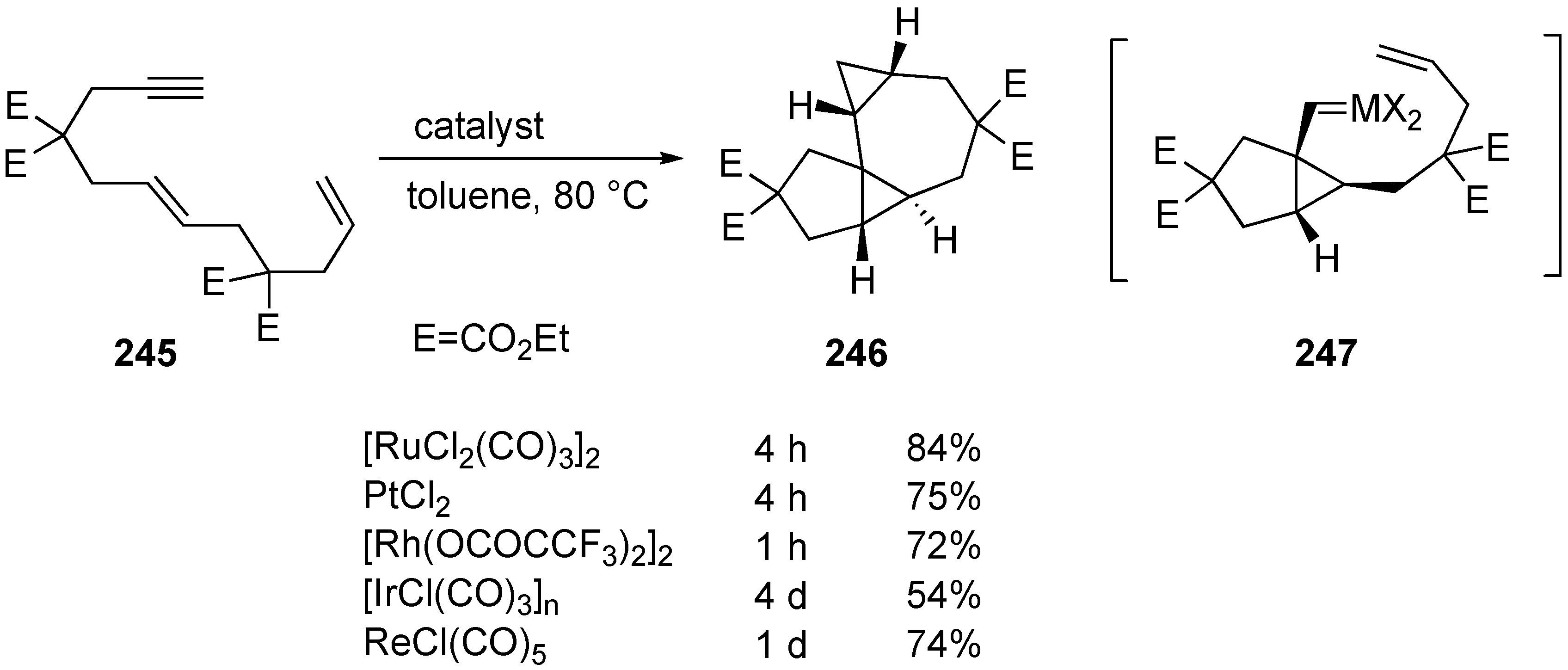

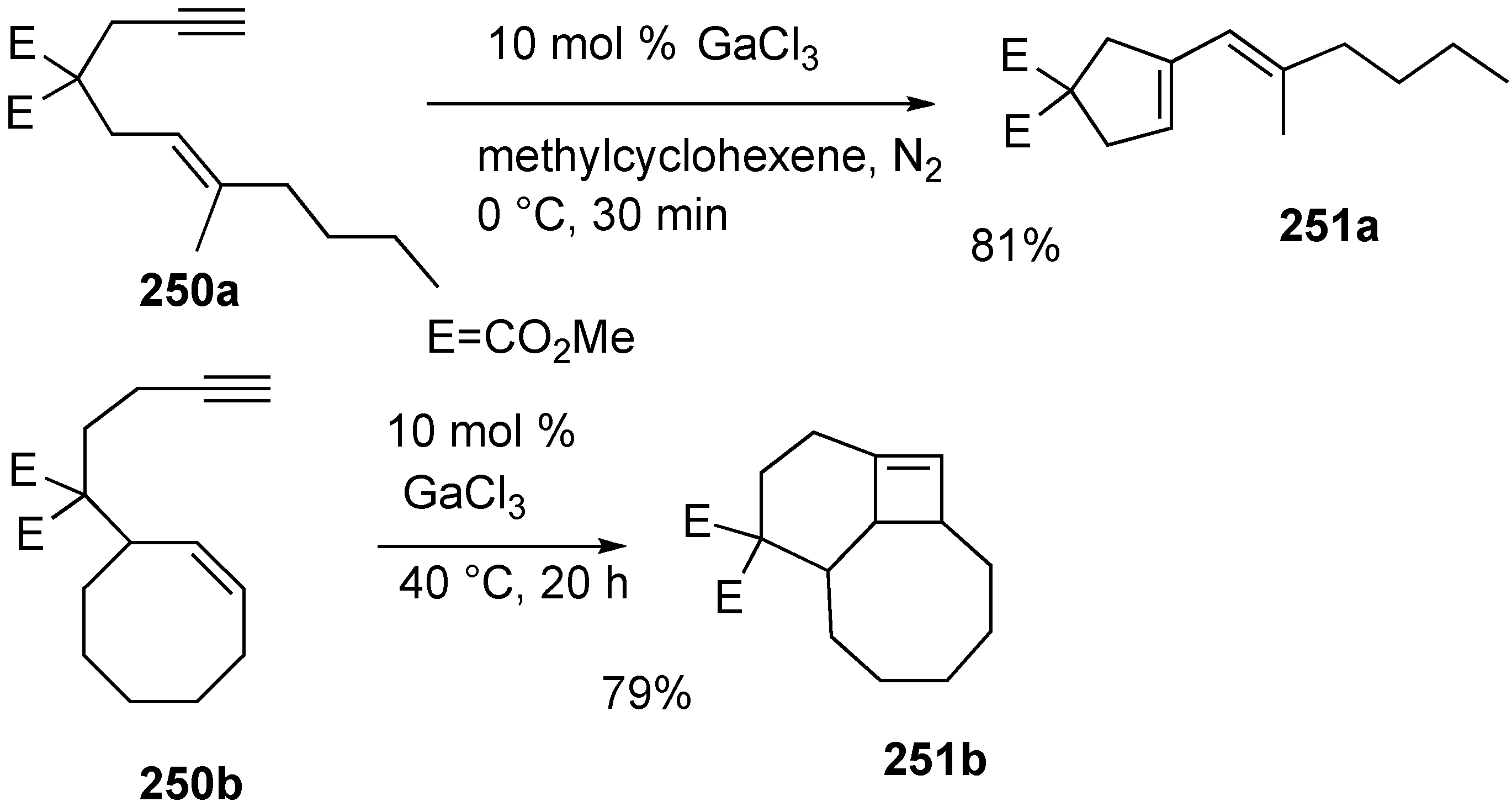
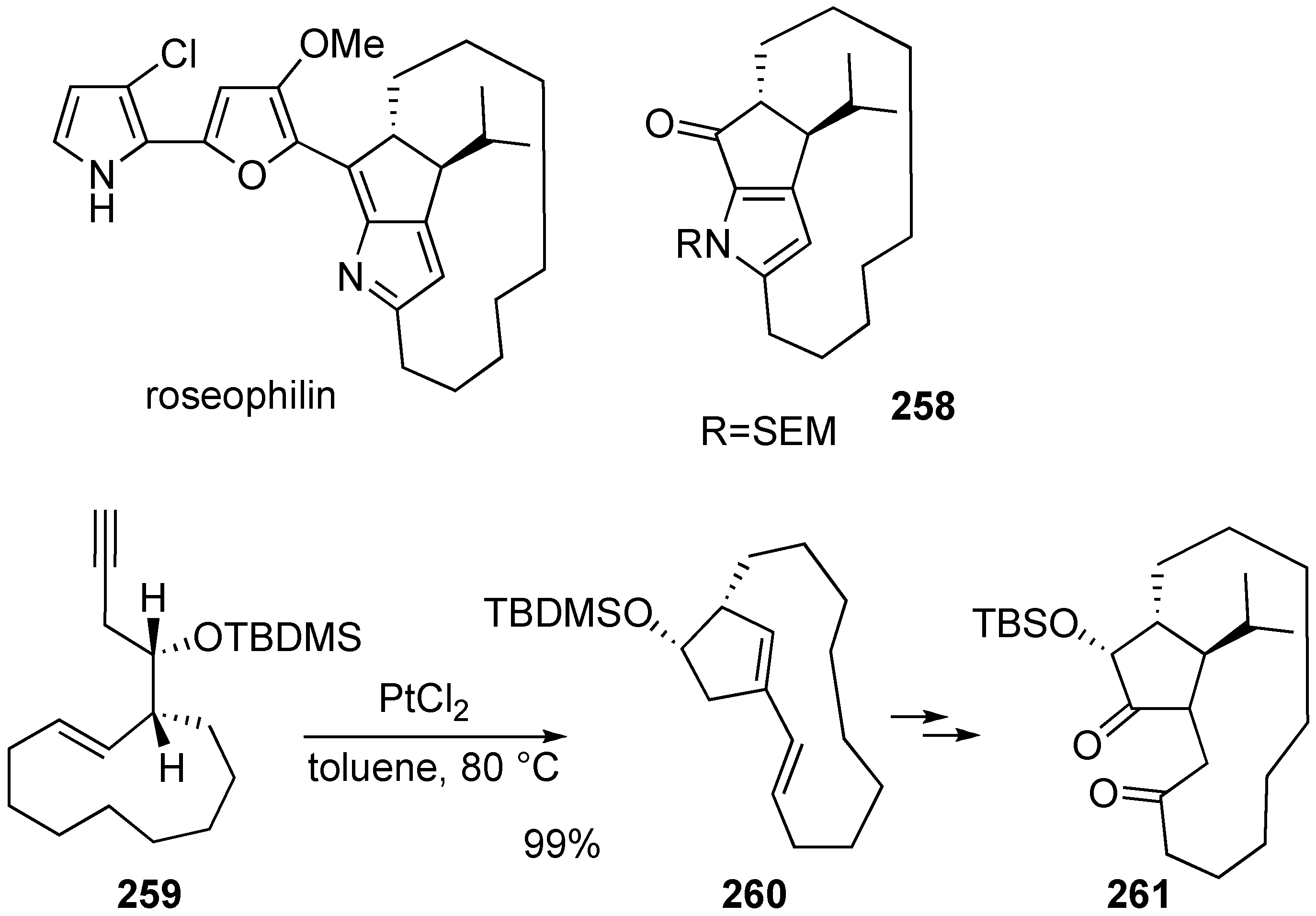
Synthesis of Natural products Using Skeletal Reorganization

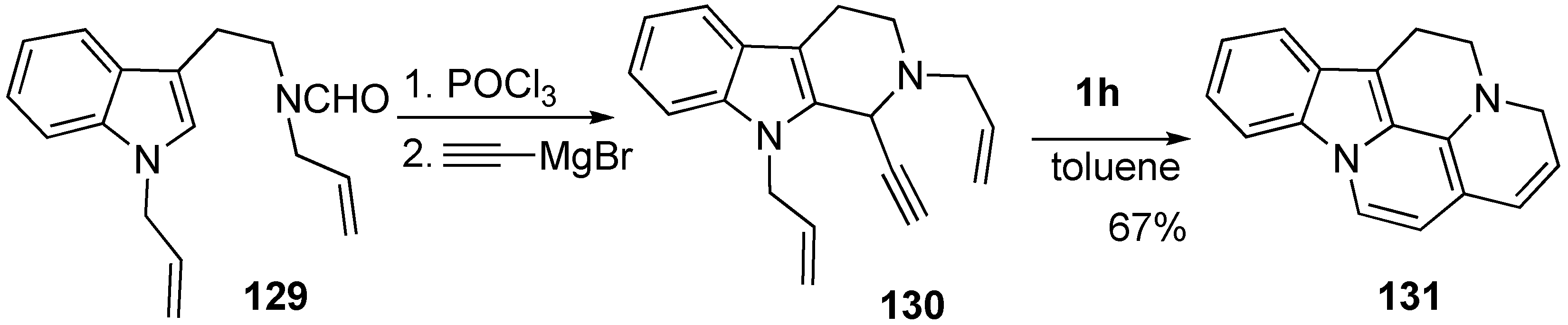
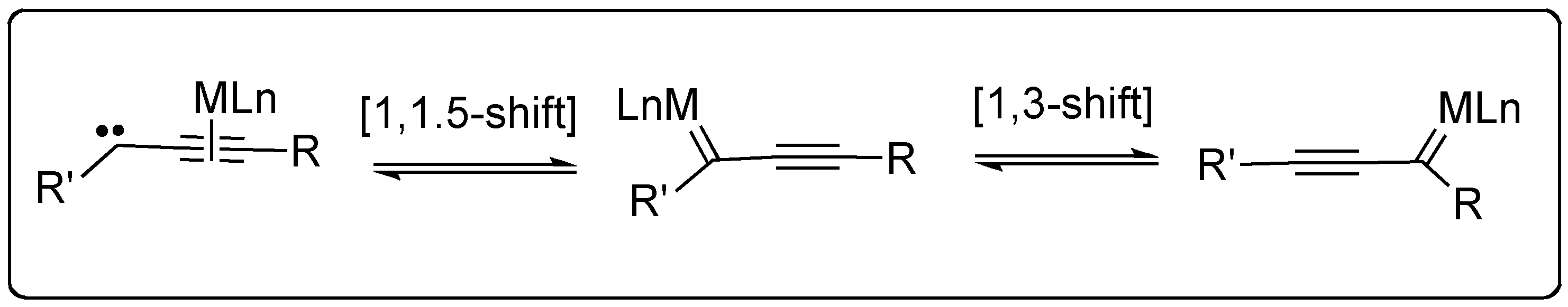
6. Metallotropic Rearrangement
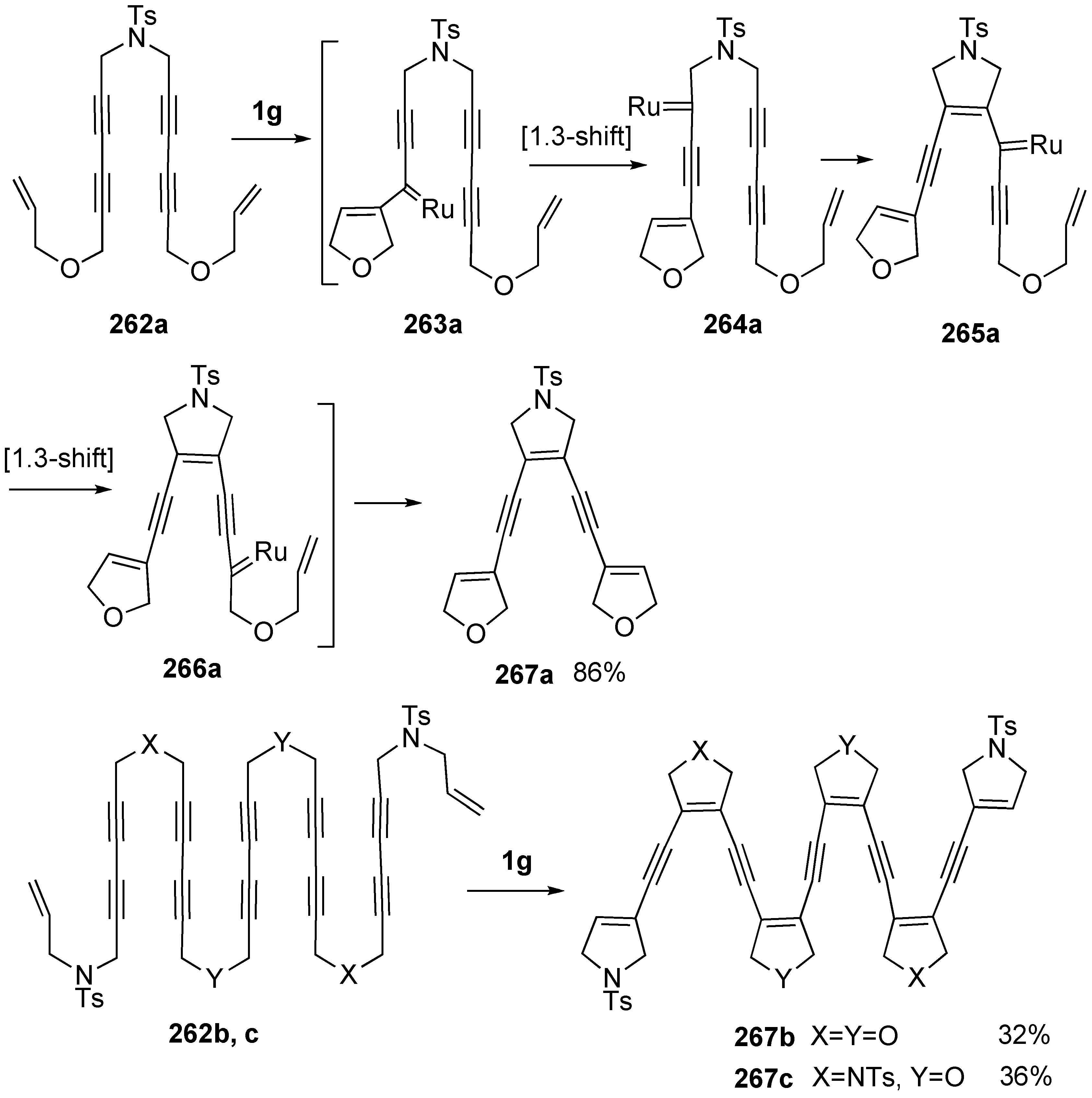
Synthesis of Natural Products Using Metallotropic Rearrangement
Perspective
References
- Schrock, R.R.; Murdzek, J.S.; Bazan, G.C.; Robbins, J.; DiMare, M.; O’Regan, M. Synthesis of molybdenum imido alkylidene complexes and some reactions involving acyclic olefins. J. Am. Chem. Soc. 1990, 112, 3875–3886. [Google Scholar]
- Nguyen, S.-B.T.; Johnson, L.K.; Grubbs, R.H.; Ziller, J.W. Ring-opening metathesis polymerization (ROMP) of norbornene by a group VIII carbene complex in protic media. J. Am. Chem. Soc. 1992, 114, 3974–3975. [Google Scholar]
- Fu, G.C.; Grubbs, R.H. The application of catalytic ring-closing olefin metathesis to the synthesis of unsaturated oxygen heterocycles. J. Am. Chem. Soc. 1992, 114, 5426–5427. [Google Scholar]
- Fu, G.C.; Grubbs, R.H. The synthesis of nitrogen heterocycles via catalytic ring-closing metathesis of dienes. J. Am. Chem. Soc. 1992, 114, 7324–7325. [Google Scholar] [CrossRef]
- Fu, G.C.; Grubbs, R.H. Synthesis of cycloalkenes via alkylidene-mediated olefin metathesis and carbonyl olefination. J. Am. Chem. Soc. 1993, 115, 3800–3801. [Google Scholar] [CrossRef]
- Fu, G.C.; Nguyen, S.–B.T.; Grubbs, R.H. Catalytic ring-closing metathesis of functionalized dienes by a ruthenium carbene complex. J. Am. Chem. Soc. 1993, 115, 9856–9857. [Google Scholar]
- Schwab, P.; France, M.B.; Ziller, J.W.; Grubbs, R.H. A series of well-defined metathesis catalysts-synthesis of [RuCl2(CHR)(PR3)2] and its reactions. Angew. Chem. Int. Ed. Engl. 1995, 34, 2039–2041. [Google Scholar] [CrossRef]
- Weskamp, T.; Schattenmann, W.C.; Spiegler, M.; Herrmann, W.A. A novel class of ruthenium catalysts for olefin metathesis. Angew. Chem. Int. Ed. 1998, 37, 2490–2493. [Google Scholar] [CrossRef]
- Weskamp, T.; Kohl, F.J.; Hieringer, W.; Gleich, D.; Herrmann, W.A. highly active ruthenium catalysts for olefin metathesis: The synergy of N-heterocyclic carbenes and coordinatively labile ligands. Angew. Chem. Int. Ed. 1999, 38, 2416–2419. [Google Scholar] [CrossRef]
- Huang, J.; Stevens, E.D.; Nolan, S.P.; Peterson, J.L. Olefin metathesis-active ruthenium complexes bearing a nucleophilic carbene ligand. J. Am. Chem. Soc. 1999, 121, 2674–2678. [Google Scholar] [CrossRef]
- Huang, J.; Schanz, H.-J.; Stevens, E.D.; Nolan, S.P. Influence of sterically demanding carbene ligation on catalytic behavior and thermal stability of ruthenium olefin metathesis catalysts. Organometallics 1999, 18, 5375–5380. [Google Scholar] [CrossRef]
- Scholl, M.; Ding, S.; Lee, C.W.; Grubbs, R.H. Synthesis and activity of a new generation of ruthenium-based olefin metathesis catalysts coordinated with 1,3-dimesityl-4,5-dihydroimidazol-2-ylidene ligands. Org. Lett. 1999, 1, 953–956. [Google Scholar] [CrossRef] [PubMed]
- Scholl, M.; Trnka, T.M.; Morgan, J.P.; Grubbs, R.H. Increased ring closing metathesis activity of ruthenium-based olefin metathesis catalysts coordinated with imidazolin-2-ylidene ligands. Tetrahedron Lett. 1999, 40, 2247–2250. [Google Scholar] [CrossRef]
- Chaterjee, A.K.; Morgan, J.P.; Scholl, M.; Grubbs, R.H. Synthesis of functionalized olefins by cross and ring-closing metatheses. J. Am. Chem. Soc. 2000, 122, 3783–3784. [Google Scholar] [CrossRef]
- Choi, T.-L.; Lee, C.W.; Chatterjee, A.K.; Grubbs, R.H. Olefin metathesis involving ruthenium enoic carbene complexes. J. Am. Chem. Soc. 2001, 123, 10417–10418. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.K.; Grubbs, R.H. Formal vinyl CH activation and allylic oxidation by olefin metathesis. Angew. Chem. Int. Ed. 2002, 41, 3171–3174. [Google Scholar] [CrossRef]
- For 1h: Garber, S.B.; Kingsbury, J.S.; Gray, B.L.; Hoveyda, A.H. Efficient and recyclable monomeric and dendritic Ru-based metathesis catalysts. J. Am. Chem. Soc. 2000, 122, 8168–8179. [Google Scholar] [CrossRef]
- For 1i: Michrowska, A.; Bujok, R.; Harutyunyan, S.; Sashuk, V.; Dolgonos, G.; Grela, K. Nitro-substituted Hoveyda-Grubbs ruthenium carbenes: Enhancement of catalyst activity through electronic activation. J. Am. Chem. Soc. 2004, 126, 9318–9325. [Google Scholar] [CrossRef] [PubMed]
- For 1i: Michrowska, A.; Mennecke, K.; Kunz, U.; Kirschning, A.; Grela, K. A new concept for the noncovalent binding of a ruthenium-based olefin metathesis catalyst to polymeric phases: Preparation of a catalyst on raschig rings. J. Am. Chem. Soc. 2006, 128, 13261–13267. [Google Scholar] [CrossRef] [PubMed]
- For 1j: Wakamatsu, H.; Blechert, S. A new highly efficient ruthenium metathesis catalyst. Angew. Chem., Int. Ed. 2002, 41, 2403–2405. [Google Scholar] [CrossRef]
- For 1j: Zaja, M.; Connon, S.J.; Dunne, A.M.; Rivard, M.; Buschmann, N.; Jiricek, J.; Blechert, S. Ruthenium olefin metathesis catalysts with modified styrene ethers: Influence of steric and electronic effects. Tetrahedron 2003, 59, 6545–6558. [Google Scholar] [CrossRef]
- For 1k: Love, J.A.; Morgan, J.P.; Trnka, T.M.; Grubbs, R.H. A practical and highly active ruthenium-based catalyst that effects the cross metathesis of acrylonitrile. Angew. Chem. Int. Ed. 2002, 41, 4035–4037. [Google Scholar] [CrossRef]
- Mori, M. Enyne metathesis. Top. Organomet. Chem. 1998, 1, 133–154. [Google Scholar]
- Poulsen, C.S.; Madsen, R. Enyne metathesis catalyzed by ruthenium carbene complexes. Synthesis 2003, 1–18. [Google Scholar]
- Mori, M. Handbook of Metathesis; Grubbs, R.H., Ed.; WILEY-VCH: Weinheim, Germany, 2003; Volume 2, pp. 176–204. [Google Scholar]
- Giessert, A.J.; Diver, S.T. Enyne metathesis (enyne bond reorganization). Chem. Rev. 2004, 104, 1317–1382. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Kitamura, T. Ene-yne and alkyne metathesis. In Comprehensive Organometallic Chemistry III, Transition Metal Organometallics in Organic Synthesis; Hiyama, T., Ed.; Elsevier: London, UK, 2005; pp. 271–310. [Google Scholar]
- Katz, T.J.; Sivavec, T.M. Metal-catalyzed rearrangement of alkene-alkynes and the stereochemistry of metallacyclobutene ring opening. J. Am. Chem. Soc. 1985, 107, 737–738. [Google Scholar] [CrossRef]
- Sivavec, T.M.; Katz, T.J.; Chiang, M.Y.; Yang, G.X.-Q. A metal pentadienyl prepared by reacting a metal carbyne with an enyne. Organometallics 1989, 8, 1620–1625. [Google Scholar] [CrossRef]
- Katz, T.J.; Yang, G.X.-Q. Reactions of enynes with adsorbed metal-carbenes. Tetrahedron Lett. 1991, 32, 5895–5898. [Google Scholar] [CrossRef]
- Watanuki, S.; Ochifuji, N.; Mori, M. Chromium-catalyzed intramolecular enyne metathesis. Organometallics 1994, 13, 4129–4130. [Google Scholar] [CrossRef]
- Watanuki, S.; Ochifuji, N.; Mori, M. Chromium-catalyzed intramolecular enyne metathesis. Organometallics 1995, 14, 5062–5067. [Google Scholar] [CrossRef]
- Mori, M.; Watanuki, S. New synthesis of pyrrolidine derivatives via the chromacyclobutanes generated from enynes and Fischer carbene complexes. J. Chem. Soc. Chem. Commun. 1992, 1082–1084. [Google Scholar]
- Watanuki, S.; Mori, M. New synthesis of piperidine derivatives via the chromacyclobutane. Heterocycles 1993, 35, 679–682. [Google Scholar] [CrossRef]
- Kinoshita, A.; Mori, M. Ruthenium catalyzed enyne metathesis. Synlett 1994, 1020–1022. [Google Scholar] [CrossRef]
- Kinoshita, A.; Sakakibara, N.; Mori, M. Novel 1,3-diene synthesis from alkyne and ethylene by ruthenium-catalyzed enyne metathesis. Tetrahedron 1999, 55, 8155–8167. [Google Scholar] [CrossRef]
- Lippstreu, J.J.; Straub, B.F. Mechanism of enyne metathesis catalyzed by Grubbs ruthenium-carbene complexes: A DFT study. J. Am. Chem. Soc. 2005, 127, 7444–7457. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Sakakibara, N.; Kinoshita, A. Remarkable effect of ethylene gas in the intramolecular enyne metathesis of terminal alkynes. J. Org. Chem. 1998, 63, 6082–6083. [Google Scholar] [CrossRef]
- Kitamura, T.; Sato, Y.; Mori, M. Unexpected results of enyne metathesis using a ruthenium complex containing an N-heterocyclic carbene ligand. Chem. Commun. 2001, 1258–1259. [Google Scholar] [CrossRef]
- Kitamura, T.; Sato, Y.; Mori, M. Effects of Substituents on the multiple bonds on ring-closing metathesis of enynes. Adv. Synth. Catal. 2002, 344, 678–693. [Google Scholar] [CrossRef]
- Debleds, O.; Campagne, J.-M. 1,5-Enyne metathesis. J. Am. Chem. Soc. 2008, 130, 1562–1563. [Google Scholar] [CrossRef] [PubMed]
- Micalizio, G. C.; Schreiber, S.L. A boronic ester annulation strategy for diversity-oriented organic synthesis. Angew. Chem. Int. Ed. 2002, 41, 152–154. [Google Scholar] [CrossRef]
- Micalizio, G.C.; Schreiber, S.L. An alkynylboronic ester annulation: Development of synthetic methods for application to diversity-oriented organic synthesis. Angew. Chem. Int. Ed. 2002, 41, 3272–3276. [Google Scholar] [CrossRef]
- Yao, Q. Rapid assembly of structurally defined and highly functionalized conjugated dienes via tethered enyne metathesis. Org. Lett. 2001, 3, 2069–2072. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.L.; Maifeld, S.V.; Lee, D. Ruthenium-catalyzed silyl ether formation and enyne metathesis sequence: Synthesis of siloxacycles from terminal alkenyl alcohols and alkynylsilanes. Org. Lett. 2004, 6, 2773–2776. [Google Scholar]
- Schramm, M.P.; Reddy, D.S.; Kozmin, S.A. siloxyalkyne-alkene metathesis: Rapid access to highly functionalized enones. Angew. Chem. Int. Ed. 2001, 40, 4274–4277. [Google Scholar] [CrossRef]
- Clark, J.S.; Trevitt, G.P.; Boyall, D.; Stammen, B. Synthesis of alkenyl-substituted cyclic enol ethers by catalytic ring-closing metathesis of alkynyl ethers. Chem. Commun. 1998, 2629–2630. [Google Scholar] [CrossRef]
- Saito, N.; Sato, Y.; Mori, M. Synthesis of cyclic dienamide using ruthenium-catalyzed ring-closing metathesis of ene-ynamide. Org. Lett. 2002, 4, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Wakamatsu, H.; Saito, N.; Sato, Y.; Narita, R.; Sato, Y.; Fujita, R. Synthesis of cyclic dienamide using ruthenium-catalyzed ring-closing metathesis of ene–ynamide. Tetrahedron 2006, 62, 3872–3881. [Google Scholar] [CrossRef]
- Villar, H.; Frings, M.; Bolm, C. Ring closing enyne metathesis: A powerful tool for the synthesis of heterocycles,Chem. Soc. Rev. 2007, 36, 55–66. [Google Scholar] [CrossRef]
- Kotha, S.; Meshram, M.; Tiwari, A. Advanced approach to polycyclics by a synergistic combination of enyne metathesis and Diels-Alder reaction. Chem. Soc. Rev. 2009, 38, 2065–2092. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.; Kim, D.H.; Do, Y.; Chang, S. Conjugated enynes as a new type of substrates for olefin metathesis. Org. Lett. 2003, 5, 3041–3043. [Google Scholar] [CrossRef] [PubMed]
- Peppers, B.P.; Diver, S.T. Tandem cyclopropanation/ring-closing metathesis of dienynes. J. Am. Chem. Soc. 2004, 126, 9524–9525. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Shishikura, Y.; Takahashi, H.; Imamoto, T. Synthesis of styrenes using ruthenium-catalyzed ring-closing enyne metathesis. Org. Lett. 2008, 10, 2777–2780. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Yoshida, K.; Yanagisawa, A. Synthesis of carbocyclic aromatic compounds using ruthenium-catalyzed ring-closing enyne metathesis. J. Org. Chem. 2009, 74, 3632–3640. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Kadowaki, S.; Matsuda, T. Molybdenum-catalyzed ring-closing metathesis of allenynes. Org. Lett. 2005, 7, 3953–3956. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, A.; Mori, M. Total synthesis of (-)-stemoamide using ruthenium-catalyzed enyne metathesis reaction. J. Org. Chem. 1996, 61, 8356–8357. [Google Scholar] [CrossRef]
- Kinoshita, A.; Mori, M. Total synthesis of (-)-stemoamide using ruthenium-catalyzed enyne metathesis reaction. Heterocycles 1997, 46, 287–299. [Google Scholar] [CrossRef]
- Barrett, A.G.M.; Baugh, S.P.D.; Braddock, D.C.; Flack, K.; Gibson, V.C.; Giles, M.R.; Marshall, E.L.; Procopiou, P.A.; White, A.J.P.; Williams, D.J. Rapid entry into mono-, bi-, and tricyclic β-lactam arrays via alkene metathesis. J. Org. Chem. 1998, 63, 7893–7907. [Google Scholar] [CrossRef]
- Duboc, R.; Henaut, C.; Savignac, M.; Genet, J.-P.; Bhatnagar, N. Synthetic approach to tricyclic β-lactams using metathesis and Diels–Alder reactions. Tetrahedron Lett. 2001, 42, 2461–2464. [Google Scholar] [CrossRef]
- Hoye, T.R.; Donaldson, S.M.; Vos, T.J. An enyne metathesis/(4 + 2)- dimerization Route to (±)-differolide. Org. Lett. 1999, 1, 277–279. [Google Scholar] [CrossRef]
- Layton, M.E.; Morales, C.A.; Shair, M.D. Biomimetic synthesis of (-)-Longithorone A. J. Am. Chem. Soc. 2002, 124, 773–775. [Google Scholar] [CrossRef] [PubMed]
- Brenneman, J.B.; Martin, S.F. Application of intramolecular enyne metathesis to the synthesis of aza[4.2.1]bicyclics: Enantiospecific total synthesis of (+)-anatoxin-a. Org. Lett. 2004, 6, 1329–1331. [Google Scholar] [CrossRef] [PubMed]
- Brenneman, J.B.; Machauer, R.; Martin, S.F. Enantioselective synthesis of (+)-anatoxin-a via enyne metathesis. Tetrahedron 2004, 60, 7301–7314. [Google Scholar] [CrossRef]
- Mori, M.; Tomita, T.; Kita, Y.; Kitamura, T. Synthesis of (+)-anatoxin-a using enyne metathesis. Tetrahedron Lett. 2004, 45, 4397–4399. [Google Scholar] [CrossRef]
- Tomita, T.; Kita, Y.; Kitamura, T.; Sato, Y.; Mori, M. Synthesis of (+)-anatoxin-a using enyne metathesis. Tetrahedron 2006, 62, 10518–10527. [Google Scholar] [CrossRef]
- Aggarwal, V.K.; Astle, C.J.; Roger-Evans, M. A concise asymmetric route to the bridged bicyclic tropane alkaloid ferruginine using enyne ring-closing metathesis. Org. Lett. 2004, 6, 1469–1471. [Google Scholar] [CrossRef] [PubMed]
- Reddy, D.S.; Kozmin, S.A. Efficient and general approach to eremophilanes using siloxyalkyne-alkene metathesis. J. Org. Chem. 2004, 69, 4860–4862. [Google Scholar] [CrossRef] [PubMed]
- Boyer, F.-D.; Hanna, I. Synthesis of allocolchicines using sequential ring-closing enyne metathesis-Diels-Alder reactions. Org. Lett. 2007, 9, 715–718. [Google Scholar] [CrossRef] [PubMed]
- Bedel, O.; Francais, A.; Haudrechy, A. Diastereoselective access to the spirotetronate subunit of the quartromicins. Synlett 2005, 2313–2316. [Google Scholar]
- Satcharoen, V.; McLean, N.J.; Kemp, S.C.; Camp, N.P.; Brown, R.C.D. Stereocontrolled synthesis of (-)-galanthamine. Org. Lett. 2007, 9, 1867–1869. [Google Scholar] [CrossRef] [PubMed]
- Kaliappan, K.P.; Ravikumar, V. Angucyclinone Antibiotics: Total syntheses of YM-181741, (+)-ochromycinone, (+)-rubiginone B2, (-)-tetrangomycin, and MM-47755. J. Org. Chem. 2007, 72, 6116–6126. [Google Scholar] [CrossRef] [PubMed]
- Krishna, P.R.; Reddy, P.S. Stereoselective total synthesis of (+)-valienamine and (+)-4-epi-valienamine via a ring-closing enyne metathesis protocol. Synlett 2009, 209–212. [Google Scholar]
- Imahori, T.; Ojima, H.; Yoshimura, Y.; Takahata, H. Acceleration effect of an allylic hydroxy group on ring-closing metathesis of terminal alkynes: Scope, application, and mechanistic insights. Chem. Eur. J. 2008, 14, 10762–10771. [Google Scholar] [CrossRef] [PubMed]
- Zuercher, W.J.; Scholl, M.; Grubbs, R.H. Ruthenium-catalyzed polycyclization reactions. J. Org. Chem. 1998, 63, 4291–4298. [Google Scholar] [CrossRef]
- Royer, F.; Vilain, C.; Elkaiem, L.; Grimaud, L. Selective domino ring-closing metathesis-cross-metathesis reactions between enynes and electron-deficient alkenes. Org. Lett. 2003, 5, 2007–2009. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, T.; Sato, Y.; Mori, M. Synthetic study of (+)-anthramycin using ring-closing enyne metathesis and cross-metathesis. Tetrahedron 2004, 60, 9649–9657. [Google Scholar] [CrossRef]
- Evans, M.A.; Morken, J.P. Asymmetric synthesis of (-)-dihydroxanthatin by the stereoselective Oshima-Utimoto reaction. Org. Lett. 2005, 7, 3371–3373. [Google Scholar]
- Kummer, D.A.; Brenneman, J.B.; Martin, S.F. Application of a domino intramolecular enyne metathesis/cross metathesis reaction to the total synthesis of (+)-8-epi-xanthatin. Org. Lett. 2005, 7, 4621–4623. [Google Scholar] [CrossRef] [PubMed]
- Paquette, L.A.; Lai, K.W. Studies directed toward the total synthesis of lancifodilactone G: An expeditious route to the ABC subunit. Org. Lett. 2008, 10, 2111–2113. [Google Scholar]
- Kim, S.-H.; Bowden, N.; Grubbs, R.H. Catalytic ring closing metathesis of dienynes: Construction of fused bicyclic rings. J. Am. Chem. Soc. 1994, 116, 10801–10802. [Google Scholar] [CrossRef]
- Kim, S.-H.; Zuercher, W.J.; Bowden, N.; Grubbs, R.H. Catalytic ring closing metathesis of dienynes: Construction of fused bicyclic [n.m.0] rings. J. Org. Chem. 1996, 61, 1073–1081. [Google Scholar] [CrossRef]
- Renaud, J.; Graf, C.-D.; Oberer, L. Ruthenium-catalyzed enyne metathesis of acetylenic boronates: A concise route for the construction of cyclic 1,3-dienylboronic esters. Angew. Chem. Int. Ed. 2000, 39, 3101–3104. [Google Scholar] [CrossRef]
- Timmer, M.S.M.; Ovaa, H.; Filippov, D.V.; van der Marel, G.A.; van Boom, J.H. Synthesis of phosphorus mono- and bicycles by catalytic ring-closing metathesis. Tetrahedron Lett. 2001, 42, 8231–8233. [Google Scholar] [CrossRef]
- Huang, J.; Xiong, H.; Hsung, R.P.; Rameshkumar, C.; Mulder, J.A.; Grebe, T.P. The first successful base-promoted isomerization of propargyl amides to chiral ynamides. Applications in ring-closing metathesis of ene-ynamides and tandem RCM of diene-ynamides. Org. Lett. 2002, 4, 2417–2420. [Google Scholar]
- Gonzalez-Gomez, A.; Dominguez, G.; Perez-Castells, J. Enyne and dienyne metathesis reactions in β-carbolines. Tetrahedron Lett. 2005, 46, 7267–7270. [Google Scholar] [CrossRef]
- Codesido, E.M.; Castedo, L.; Granja, J.R. Access to [6.4.0]carbocyclic systems by tandem metathesis of dienynes. A step toward the synthesis of a PreD3-D3 transition state analogue. Org. Lett. 2001, 3, 1483–1486. [Google Scholar] [CrossRef] [PubMed]
- Boyer, F.-D.; Hanna, I.; Ricard, L. Formal synthesis of (±)-guanacastepene A: A tandem ring-closing metathesis approach. Org. Lett. 2004, 6, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Krishna, P.R.; Narsingam, M. Studies directed towards the stereoselective total synthesis of ilexlactone via a tandem ring-closing enyne metathesis protocol. Tetrahedron Lett. 2007, 48, 8721–8724. [Google Scholar] [CrossRef]
- Shimizu, K.; Takimoto, M.; Mori, M. Novel synthesis of heterocycles having a functionalized carbon center via nickel-mediated carboxylation: Total synthesis of erythrocarine. Org. Lett. 2003, 5, 2323–2325. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, H.; Esumi, T.; Ishihara, J.; Hatakeyama, S. Total synthesis of (±)-erythravine based on ring closing dienyne metathesis. Tetrahedron Lett. 2003, 44, 8047–8049. [Google Scholar] [CrossRef]
- Honda, T.; Namiki, H.; Kaneda, K.; Mizutani, H. First diastereoselective chiral synthesis of (-)-securinine. Org. Lett. 2004, 6, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Namiki, H.; Watanabe, M.; Mizutani, H. First total synthesis of (+)-viroallosecurinine. Tetrahedron Lett. 2004, 45, 5211–5213. [Google Scholar] [CrossRef]
- Fukumoto, H.; Takahashi, K.; Ishihara, J.; Hatakeyama, S. Total synthesis of (+)-β-erythroidine. Angew. Chem. Int. Ed. 2006, 45, 2731–2734. [Google Scholar] [CrossRef]
- Movassaghi, M.; Piizzi, G.; Siegel, D.S.; Piersanti, G. Enantioselective total synthesis of (-)-acylfulvene and (-)-irofulven. Angew. Chem. Int. Ed. 2006, 45, 5859–5863. [Google Scholar] [CrossRef]
- Mukherjee, S.; Lee, D. Application of tandem ring-closing enyne metathesis: Formal total synthesis of (-)-cochleamycin A. Org. Lett. 2009, 11, 2916–2919. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, A.; Sakakibara, N.; Mori, M. Novel 1,3-diene synthesis from alkyne and ethylene by ruthenium-catalyzed enyne metathesis. J. Am. Chem. Soc. 1997, 119, 12388–12389. [Google Scholar] [CrossRef]
- Kinoshita, A.; Sakakibara, N.; Mori, M. Novel 1,3-diene synthesis from alkyne and ethylene by ruthenium-catalyzed enyne metathesis. Tetrahedron 1999, 55, 8155–8167. [Google Scholar] [CrossRef]
- Mori, M.; Tonogaki, K.; Kinoshita, A. Synthesis of 1,3-diene from alkyne and ethylene : Acetic acid 2-methylene-3-phenethyl-but-3-enyl ester. Org. Synth. 2004, 81, 1–13. [Google Scholar]
- Tonogaki, K.; Mori, M. An improved 1,3-diene synthesis from alkyne and ethylene using cross-enyne metathesis. Tetrahedron Lett. 2002, 43, 2235–2238. [Google Scholar] [CrossRef]
- Smulik, J.A.; Diver, S.T. Terminal alkyne-ethylene cross-metathesis: Reaction of 1-substituted propargyl esters at elevated ethylene pressure. J. Org. Chem. 2000, 65, 1788–1792. [Google Scholar]
- Smulik, J.A.; Diver, S.T. Expanded scope in ethylene-alkyne cross-metathesis: Coordinating heteroatom functionality at the propargylic position. Org. Lett. 2000, 2, 2271–2274. [Google Scholar] [CrossRef] [PubMed]
- Smulik, J.A.; Giessert, A.J.; Diver, S.T. Ethylene metathesis of sulfur-containing alkynes. Tetrahedron Lett. 2002, 43, 209–211. [Google Scholar]
- Stragies, R.; Schuster, M.; Blechert, S. A crossed yne-ene metathesis showing atom economy. Angew. Chem. Int. Ed. 1997, 36, 2518–2520. [Google Scholar]
- Kotha, S.; Halder, S.; Brahmachary, E. Synthesis of highly functionalized phenylalanine derivatives via cross-enyne metathesis reactions. Tetrahedron 2002, 58, 9203–9208. [Google Scholar]
- Schürer, S.C.; Blechert, S. A versatile synthesis of substituted tetrahydropyridines. Tetrahedron Lett. 1999, 40, 1877–1880. [Google Scholar]
- Mix, S.; Blechert, S. Synthesis of cis-fused carbo-bicycles by domino enyne cross-metathesis/intramolecular Diels-Alder reaction. Org. Lett. 2005, 7, 2015–2018. [Google Scholar] [PubMed]
- Kalbarczyk, K.P.; Diver, S.T. Enyne metathesis/brønsted acid-promoted heterocyclization. J. Org. Chem. 2009, 74, 2193–2196. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.A.; Diver, S.T. Cycloheptadiene ring synthesis by tandem intermolecular enyne metathesis. Org. Lett. 2003, 5, 3463–3466. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Tonogaki, K.; Nishiguchi, N. Syntheses of anolignans A and B using ruthenium-catalyzed cross-enyne metathesis. J. Org. Chem. 2002, 67, 224–226. [Google Scholar] [CrossRef]
- Saito, N.; Masuda, M.; Saito, H.; Takenouchi, K.; Ishizuka, S.; Namekawa, J.; Takimoto-Kamimura, M.; Kittaka, A. synthesis of 24,24-ethanovitamin D3 lactones using ruthenium-catalyzed intermolecular enyne metathesis: Potent vitamin D receptor antagonists. Synthesis 2005, 2533–2543. [Google Scholar]
- Kaliappan, K.P.; Subrahmanyam, A.V. A new versatile strategy for C-aryl glycosides. Org. Lett. 2007, 9, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; An, H.J.; Shin, W.K.; Yu, W.; Woo, S.K.; Jung, S.K.; Lee, E. Stereoselective synthesis of (-)-amphidinolide E. E. Chem. Asian J. 2008, 3, 1523–1534. [Google Scholar] [CrossRef]
- Kim, C.H.; An, H.J.; Shin, W.K.; Yu, W.; Woo, S.K.; Jung, S.K.; Lee, E. Total synthesis of (-)-amphidinolide E. Angew. Chem. Int. Ed. 2006, 45, 8019–8021. [Google Scholar] [CrossRef]
- Ko, H.M.; Lee, C.W.; Kwon, H.K.; Chung, H.S.; Choi, S.Y.; Chung, Y.K.; Lee, E. Total synthesis of (-)-amphidinolide K. Angew. Chem. Int. Ed. 2009, 48, 2364–2366. [Google Scholar] [CrossRef]
- Fürstner, A.; Flügge, S.; Larionov, O.; Takahashi, Y.; Kubota, T.; Kobayashi, J. Total synthesis and biological evaluation of amphidinolide V and analogues. Chem. Eur. J. 2009, 15, 4011–4029. [Google Scholar] [CrossRef] [PubMed]
- Fürstner, A.; Larionov, O.; Flugge, S. What is amphidinolide V? Report on a likely conquest. Angew. Chem. Int. Ed. 2007, 46, 5545–5548. [Google Scholar] [CrossRef]
- Kitamura, T.; Mori, M. Ruthenium-catalyzed ring-opening and ring-closing enyne metathesis. Org. Lett. 2001, 3, 1161–1164. [Google Scholar] [CrossRef] [PubMed]
- Randl, S.; Lucas, N.; Connon, S.J.; Blechert, S. A mechanism switch in enyne metathesis reactions involving rearrangement: Influence of heteroatoms in the propargylic position. Adv. Synth. Catal. 2002, 344, 631–633. [Google Scholar] [CrossRef]
- Rückert, A.; Eisele, D.; Blechert, S. Domino metathesis of alkynyl substituted cycloolefins. Tetrahedron Lett. 2001, 42, 5245–5247. [Google Scholar] [CrossRef]
- Mori, M.; Kuzuba, Y.; Kitamura, T.; Sato, Y. Ruthenium-catalyzed ROM−RCM of cycloalkene-yne. Org. Lett. 2002, 4, 3855–3858. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, T.; Kuzuba, Y.; Sato, Y.; Wakamatsu, H.; Fujita, R.; Mori, M. ROM-RCM of cycloalkene-yne. Tetrahedron 2004, 60, 7375–7389. [Google Scholar] [CrossRef]
- Mori, M.; Wakamatsu, H.; Tonogaki, K.; Fujita, R.; Kitamura, T.; Sato, Y. Synthesis of isoquinoline derivatives using ROM-RCM of cyclobutene-yne. J. Org. Chem. 2005, 70, 1066–1069. [Google Scholar] [CrossRef] [PubMed]
- Arjona, O.; Csaky, A.G.; Murcia, M.C.; Plumet, J. Regioselective domino metathesis of 7-oxanorbornene derivatives as a new stereoselective entry into 2,6-dioxabicyclo[4.3.0]nonenes. Tetrahedron Lett. 2000, 41, 9777–9779. [Google Scholar] [CrossRef]
- Arjona, O.; Csaky, A.G.; Leon, V.; Medel, R.; Plumet, J. Control of product distribution in the domino metathesis reactions of N-alkynyl 2-azabicyclo[2.2.1]hept-5-en-3-ones. A convenient synthesis of functionalized γ-lactams and indolizidinones. Tetrahedron Lett. 2004, 45, 565–567. [Google Scholar] [CrossRef]
- Mori, M.; Wakamatsu, H.; Sato, Y.; Fujita, R. ROM-RCM of azabicycloheptene derivatives: Study of products distribution by the substituent on alkyne. J. Mol. Cat. A: Chem. 2006, 254, 64–67. [Google Scholar] [CrossRef]
- Banti, D.; North, M. Totally atom economical tandem-metathesis and Diels–Alder approach to polycyclic compounds. Tetrahedron Lett. 2002, 43, 1561–1564. [Google Scholar] [CrossRef]
- Banti, D.; North, M. Enyne Metathesis of Norbornene Derivatives: A facile approach to polycyclic heterocycles. Adv. Synth. Catal. 2002, 344, 694–704. [Google Scholar] [CrossRef]
- Trost, B.M.; Tanoury, G.J. An unusual mechanism of a palladium-catalyzed intramolecular carbametalation. A novel palladium-catalyzed rearrangement. J. Am. Chem. Soc. 1988, 110, 1636–1638. [Google Scholar] [CrossRef]
- Trost, B.M.; Trost, M.K. Intramolecular enyne metathesis reaction. Route to bridged bicycles with bridgehead olefins. J. Am. Chem. Soc. 1991, 113, 1850–1852. [Google Scholar] [CrossRef]
- Trost, B.M.; Trost, M.K. Mechanistic dichotomies in Pd-catalyzed enyne metathesis of cyclic olefins. Tetrahedron Lett. 1991, 32, 3647–3650. [Google Scholar] [CrossRef]
- Trost, B.M.; Yanai, M.; Hoogsteen, K. A palladium-catalyzed [2 + 2] cycloaddition. Mechanism of a Pd-catalyzed enyne metathesis. J. Am. Chem. Soc. 1993, 115, 5294–5295. [Google Scholar] [CrossRef]
- Trost, B.M.; Hashmi, A.S.K. A Cycloaddition approach to cyclopentenes via metalladienes as 4.pi. partners. J. Am. Chem. Soc. 1994, 116, 2183–2184. [Google Scholar] [CrossRef]
- Trost, B.M.; Hashmi, A.S.K. On the mechanism of the TCPCHFB-catalyzed metathesis of 1,6-enyne: Evidence for alkylidenepalladium intermediates. Angew. Chem. Int. Ed. 1993, 32, 1085–1087. [Google Scholar] [CrossRef]
- Trost, B.M.; Chang, V.K. An Approach to Botrydianes: On the steric demands of a metal catalyzed enyne metathesis. Synthesis 1993, 824–832. [Google Scholar]
- Chatani, N.; Furukawa, N.; Sakurai, H.; Murai, S. PtCl2-catalyzed conversion of 1,6- and 1,7-enynes to 1-vinylcycloalkenes. Anomalous bond connection in skeletal reorganization of enynes. Organometallics 1996, 15, 901–903. [Google Scholar]
- Chatani, N.; Morimoto, T.; Muto, T.; Murai, S. Highly selective skeletal reorganization of 1,6- and 1,7-enynes to 1-vinylcycloalkenes catalyzed by [RuCl2(CO)3]2. J. Am. Chem. Soc. 1994, 116, 6049–6050. [Google Scholar] [CrossRef]
- Chatani, N.; Kataoka, K.; Murai, S.; Furukawa, N.; Seki, Y. Construction of novel polycyclic ring systems by transition-metal-catalyzed cycloisomerization of ene-ene-ynes. Interception of a carbenoid intermediate in skeletal reorganization of enynes. J. Am. Chem. Soc. 1998, 120, 9104–9105. [Google Scholar]
- Ota, K.; Lee, S.I.; Tang, J.-M.; Takachi, M.; Nakai, H.; Morimoto, T.; Sakurai, H.; Kataoka, K.; Chatani, N. Rh(II)-catalyzed skeletal reorganization of 1,6- and 1,7-enynes through electrophilic activation of alkynes. J. Am. Chem. Soc. 2009, 131, 15203–15211. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.; Chatani, N. Rh(II)-catalyzed skeletal reorganization of enynes involving selective cleavage of C–C triple bonds. Chem. Commun. 2008, 2906. [Google Scholar] [CrossRef]
- Fürstner, A.; Stelzer, F.; Szillat, H. Platinum-catalyzed cycloisomerization reactions of enynes. J. Am. Chem. Soc. 2001, 123, 11863–11869. [Google Scholar] [CrossRef] [PubMed]
- Chatani, N.; Inoue, H.; Kotsuma, T.; Murai, S. Skeletal reorganization of enynes to 1-vinylcycloalkenes catalyzed by GaCl3. J. Am. Chem. Soc. 2002, 124, 10294–10295. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Núñez, E.; Claverie, C.K.; Bour, C.; Cárdenas, D.J.; Echavarren, A.M. cis-Selective single-cleavage skeletal rearrangement of 1,6-enynes reveals the multifaceted character of the intermediates in metal-catalyzed cycloisomerizations. Angew. Chem. Int. Ed. 2008, 47, 7892–7895. [Google Scholar] [CrossRef]
- Fürstner, A.; Szillat, H.; Gabor, B.; Mynott, R. Platinum- and acid-catalyzed Enyne metathesis reactions: Mechanistic studies and applications to the syntheses of streptorubin B and metacycloprodigiosin. J. Am. Chem. Soc. 1998, 120, 8305–8314. [Google Scholar] [CrossRef]
- Trost, B.M.; Doherty, G.A. An asymmetric synthesis of the tricyclic core and a formal total synthesis of roseophilin via an enyne metathesis. J. Am. Chem. Soc. 2000, 122, 3801–3810. [Google Scholar] [CrossRef]
- Padwa, A.; Austin, D.J.; Gareau, Y.; Kassir, J.M.; Xu, S.L. Rearrangement of alkynyl and vinyl carbenoids via the rhodium(II)-catalyzed cyclization reaction of .alpha.-diazo ketones. J. Am. Chem. Soc. 1993, 115, 2637–2647. [Google Scholar] [CrossRef]
- Casey, C.P.; Dzwiniel, T.L. Manganese trifluoroacetoxycarbene complexes are convenient intermediates in the synthesis of cyclic enediynes. Organometallics 2003, 22, 5285–5290. [Google Scholar] [CrossRef]
- Casey, C.P.; Kraft, S.; Powell, D.R. Formation of cis-enediyne complexes from rhenium alkynylcarbene complexes. J. Am. Chem. Soc. 2002, 124, 2584–2594. [Google Scholar] [CrossRef] [PubMed]
- Barluenga, J.; de la Rua, R.B.; de Saa, D.; Ballesteros, A.; Tomas, M. Formal alkyne insertion into alkoxycarbene complexes: Simple access to enantiopure group 6 alkynyl(alkoxy)carbene complexes. Angew. Chem. Int. Ed. 2005, 44, 4981–4983. [Google Scholar] [CrossRef]
- Kim, M.; Lee, D. Metathesis and metallotropy: A versatile combination for the synthesis of oligoenynes. J. Am. Chem. Soc. 2005, 127, 18024–18025. [Google Scholar] [CrossRef] [PubMed]
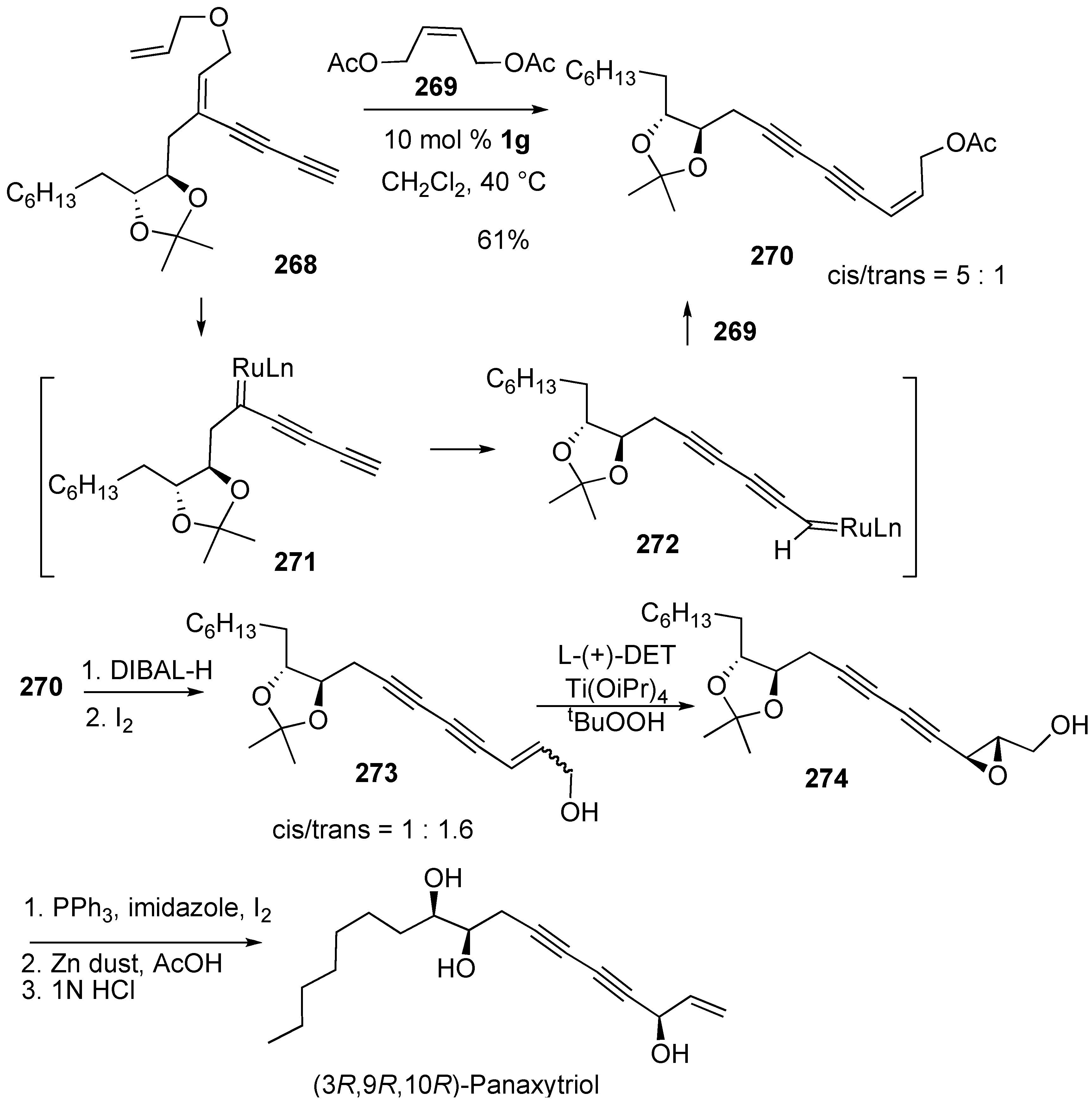
- Cho, E.J.; Lee, D. Total synthesis of (3R,9R,10R)-panaxytriol via tandem metathesis and metallotropic [1,3]-shift as a key step. Org. Lett. 2008, 10, 257–259. [Google Scholar] [CrossRef] [PubMed]
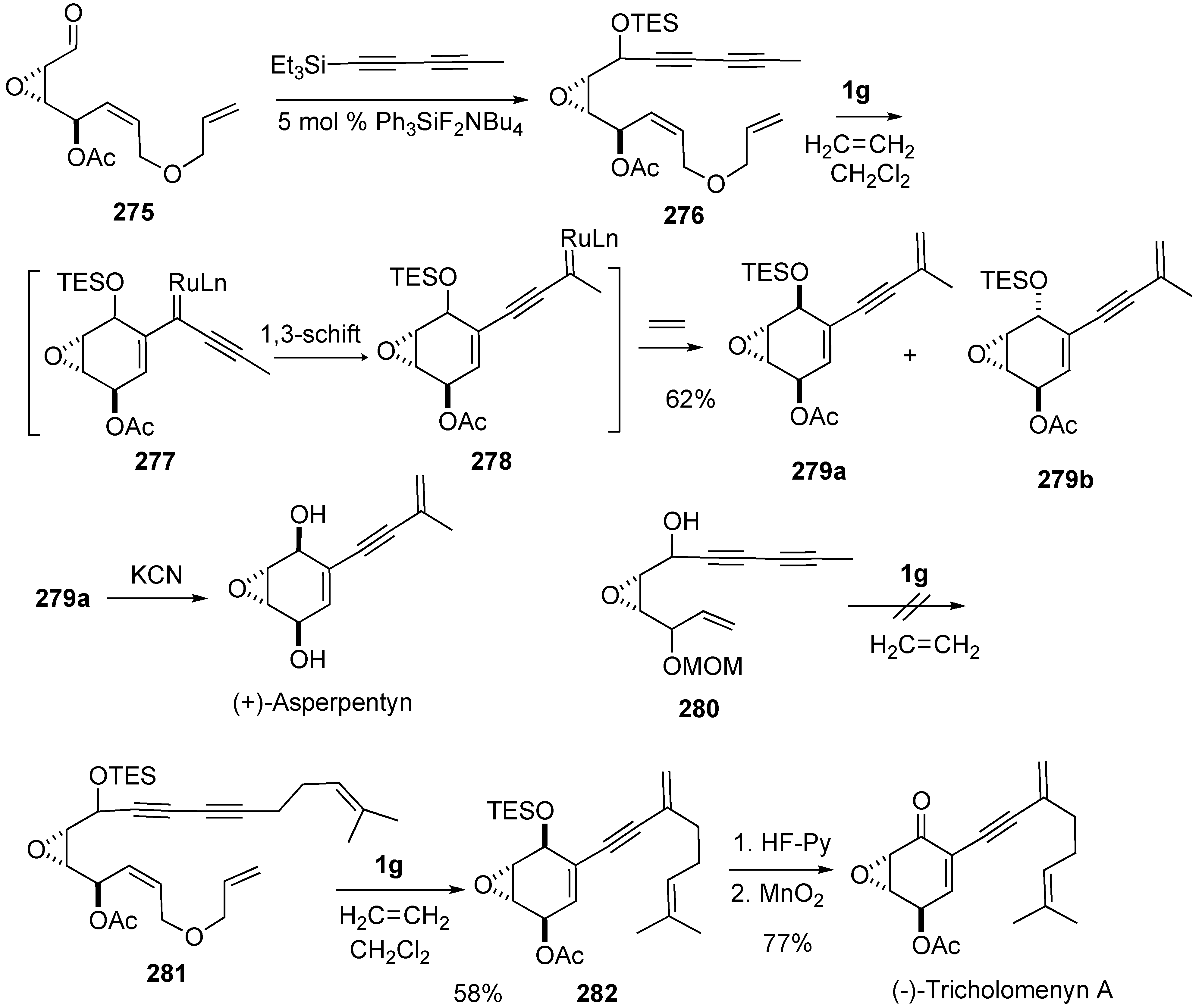
- Li, J.; Park, S.; Miller, R.L.; Lee, D. Tandem enyne metathesis-metallotropic [1,3]-shift for a concise total syntheses of (+)-asperpentyn, (-)-harveynone, and (-)-tricholomenyn A. Org. Lett. 2009, 11, 571–574. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mori, M. Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds. Materials 2010, 3, 2087-2140. https://doi.org/10.3390/ma3032087
Mori M. Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds. Materials. 2010; 3(3):2087-2140. https://doi.org/10.3390/ma3032087
Chicago/Turabian StyleMori, Miwako. 2010. "Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds" Materials 3, no. 3: 2087-2140. https://doi.org/10.3390/ma3032087