Collagen-Based Biomaterials for Tissue Engineering Applications
Abstract
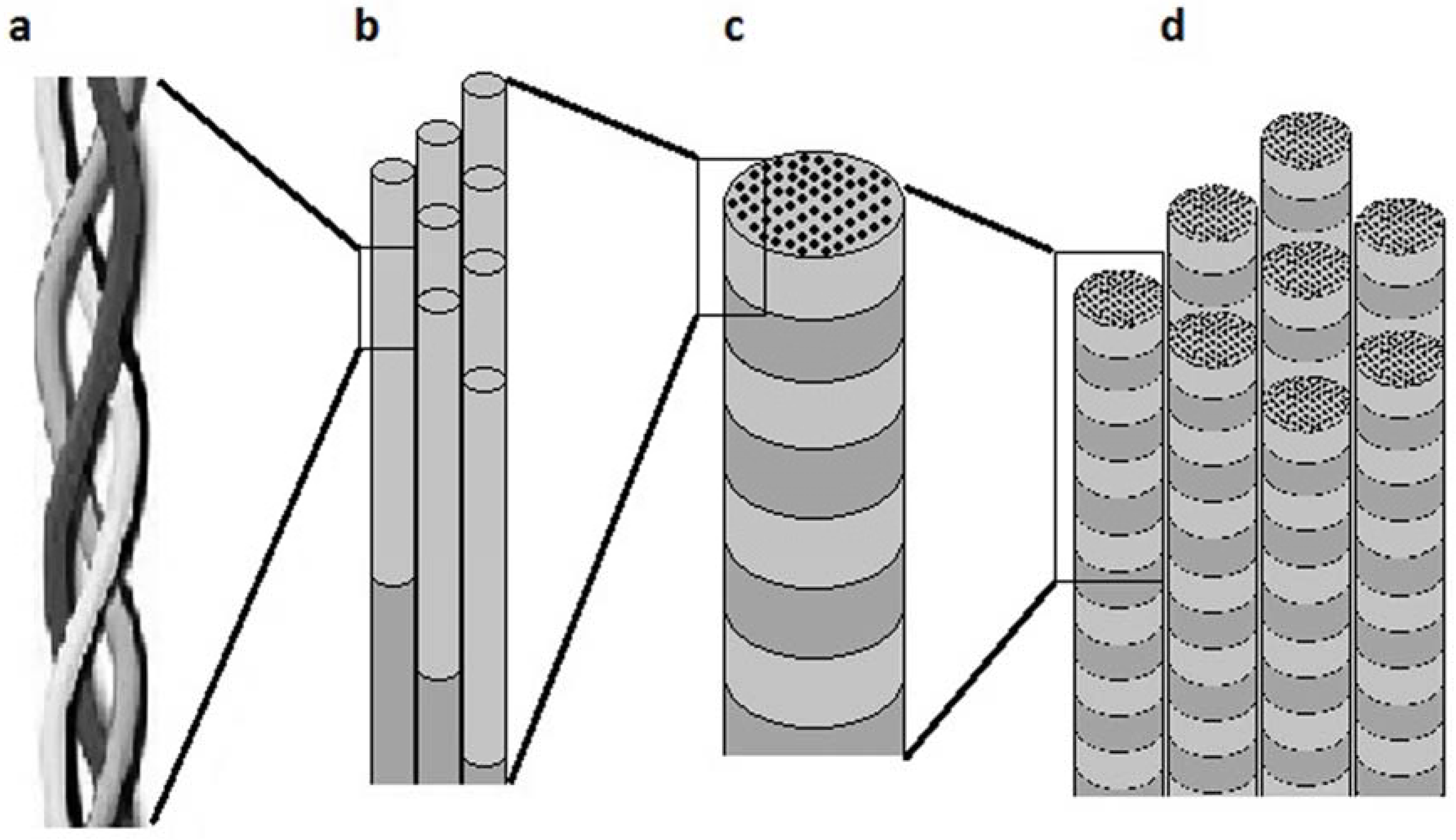
:1. Introduction
2. The Collagen Molecule
2.1. Distribution, biosynthesis and molecular structure
| Type | Molecular formula | Polymerized form | Tissue distribution | |
|---|---|---|---|---|
| Fibril-Forming (fibrillar) | I | [α1(I)]2α2(I) | fibril | bone, skin, tendons, ligaments, cornea (represent 90% of total collagen of the human body) |
| II | [α1(II)]3 | fibril | cartilage, intervertebrate disc, notochord, vitreous humor in the eye | |
| III | [α1(III)]3 | fibril | skin, blood vessels | |
| V | [α1(V)]2α2(V) and α1(V)α2(V)α3(V) | fibril (assemble with type I) | idem as type I | |
| XI | α1(XI)α2(XI)α3(XI) | fibril (assemble with type II) | idem as type II | |
| Fibril-associated | IX | α1(IX)α2(IX)α3(IX) | lateral association with type II fibril | cartilage |
| XII | [α1(XII)]3 | lateral association with type I fibril | tendons, ligaments | |
| Network-forming | IV | [α1(IV)]2α2(IV) | Sheet-like network | basal lamina |
| VII | [α1(VII)]3 | anchoring fibrils | beneath stratified squamous epithelia |
2.2. Immunogenicity and biocompatibility
2.3. Collagen origin and variability
2.4. Biodegradability and collagenases
2.5. Collagen and cellular interactions
3. Collagen-Based Biomaterials
3.1. Types of collagen-based biomaterials
3.2. Crosslinking methods and reinforcement with biopolymers combination
3.3. Sterilisation methods
4. Recent Advances in Collagen-Based Biomaterials
4.1. Experimental applications
4.2. Osteochondral defects
4.3. Vascular diseases
4.4. Skin and cornea
4.5. Urogenital system
4.6. Neural migration
4.7. Dermal filler, wound dressing and delivery systems
5. Conclusion
References and Notes
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K. Molecular Biology of The Cell; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- van der Rest, M.; Garrone, R. Collagen family of proteins. FASEB J. 1991, 5, 2814–2823. [Google Scholar]
- Prockop, D.J.; Kivirikko, K.I. Collagens: Molecular biology, diseases, and potentials for therapy. Annu Rev Biochem 1995, 64, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Crumpton, M. The molecular basis of antigenicity and immunogenicity. In Protein Antigens; Selan, M., Ed.; Academic Press: New York, NY, USA, 1974; pp. 1–78. [Google Scholar]
- Schmitt, F.O.; Levine, L.; Drake, M.P.; Rubin, A.L.; Pfahl, D.; Davison, P.F. The antigenicity of tropocollagen. Proc. Natl. Acad. Sci. USA 1964, 51, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Davison, P.F.; Levine, L.; Drake, M.P.; Rubin, A.; Bump, S. The serologic specificity of tropocollagen telopeptides. J. Exp. Med. 1967, 126, 331–346. [Google Scholar] [CrossRef] [PubMed]
- Steffen, C.; Timpl, R.; Wolff, I. Immunogenicity and specificity of collagen. V. Demonstration of three different antigenic determinants on calf collagen. Immunology 1968, 15, 135–144. [Google Scholar] [PubMed]
- Michaeli, D.; Martin, G.R.; Kettman, J.; Benjamini, E.; Leung, D.Y.; Blatt, B.A. Localization of antigenic determinants in the polypeptide chains of collagen. Science 1969, 166, 1522–1524. [Google Scholar] [CrossRef] [PubMed]
- Furthmayr, H.; Beil, W.; Timpl, R. Different antigenic determinants in the polypeptide chains of human collagen. FEBS Lett. 1971, 12, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Lindsley, H.; Mannik, M.; Bornstein, P. The distribution of antigenic determinants in rat skin collagen. J. Exp. Med. 1971, 133, 1309–1324. [Google Scholar] [CrossRef] [PubMed]
- Timpl, R.; Beil, W.; Furthmayr, H.; Meigel, W.; Pontz, B. Characterization of conformation independent antigenic determinants in the triple-helical part of calf and rat collagen. Immunology 1971, 21, 1017–1030. [Google Scholar] [PubMed]
- Furthmayr, H.; Timpl, R. Immunochemistry of collagens and procollagens. Int. Rev. Connect. Tissue Res. 1976, 7, 61–99. [Google Scholar] [PubMed]
- Weadock, K.S.; Miller, E.J.; Keuffel, E.L.; Dunn, M.G. Effect of physical crosslinking methods on collagen-fiber durability in proteolytic solutions. J. Biomed. Mater. Res. 1996, 32, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Harriger, M.D.; Supp, A.P.; Warden, G.D.; Boyce, S.T. Glutaraldehyde crosslinking of collagen substrates inhibits degradation in skin substitutes grafted to athymic mice. J. Biomed. Mater. Res. 1997, 35, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Charriere, G.; Bejot, M.; Schnitzler, L.; Ville, G.; Hartmann, D.J. Reactions to a bovine collagen implant. Clinical and immunologic study in 705 patients. J. Am. Acad. Dermatol. 1989, 21, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Eaglstein, W.H.; Alvarez, O.M.; Auletta, M.; Leffel, D.; Rogers, G.S.; Zitelli, J.A.; Norris, J.E.; Thomas, I.; Irondo, M.; Fewkes, J.; Hardin-Young, J.; Duff, R.G.; Sabolinski, M.L. Acute excisional wounds treated with a tissue-engineered skin (Apligraf). Dermatol. Surg. 1999, 25, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F.; Gilbert, T.W. Immune response to biologic scaffold materials. Semin. Immunol. 2008, 20, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.; Ogawa, M.; Portier, R.J.; Schexnayder, M.; Shirley, M.; Losso, J.N. Biochemical properties of alligator (Alligator mississippiensis) bone collagen. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2008, 151, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Rogers, G.J.; Roe, S.C.; Howlett, C.R.; Clayton, M.K.; Milthorpe, B.K.; Schindhelm, K. Nitrous acid pretreatment of tendon xenografts cross-linked with glutaraldehyde and sterilized with gamma irradiation. Biomaterials 1999, 20, 1003–1015. [Google Scholar] [CrossRef] [PubMed]
- Exposito, J.Y.; Cluzel, C.; Garrone, R.; Lethias, C. Evolution of collagens. Anat. Rec. 2002, 268, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Exposito, J.Y.; Le Guellec, D.; Lu, Q.; Garrone, R. Short chain collagens in sponges are encoded by a family of closely related genes. J. Biol. Chem. 1991, 266, 21923–21928. [Google Scholar] [PubMed]
- Sugiura, H.; Yunoki, S.; Kondo, E.; Ikoma, T.; Tanaka, J.; Yasuda, K. In vivo biological responses and bioresorption of tilapia scale collagen as a potential biomaterial. J Biomater Sci Polym. Ed. 2009, 20, 1353–1368. [Google Scholar] [CrossRef] [PubMed]
- Song, E.; Yeon Kim, S.; Chun, T.; Byun, H.J.; Lee, Y.M. Collagen scaffolds derived from a marine source and their biocompatibility. Biomaterials 2006, 27, 2951–2961. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.K.; Liu, D.C. Comparison of physical-chemical properties of type I collagen from different species. Food Chem. 2006, 99, 244–251. [Google Scholar] [CrossRef]
- Yang, C.; Hillas, P.J.; Baez, J.A.; Nokelainen, M.; Balan, J.; Tang, J.; Spiro, R.; Polarek, J.W. The application of recombinant human collagen in tissue engineering. BioDrugs 2004, 18, 103–119. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F. Xenogeneic extracellular matrix as a scaffold for tissue reconstruction. Transpl. Immunol. 2004, 12, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Goo, H.C.; Hwang, Y.S.; Choi, Y.R.; Cho, H.N.; Suh, H. Development of collagenase-resistant collagen and its interaction with adult human dermal fibroblasts. Biomaterials 2003, 24, 5099–5113. [Google Scholar] [CrossRef] [PubMed]
- Yannas, I.V.; Burke, J.F.; Orgill, D.P.; Skrabut, E.M. Wound tissue can utilize a polymeric template to synthesize a functional extension of skin. Science 1982, 215, 174–176. [Google Scholar] [CrossRef] [PubMed]
- Postlethwaite, A.E.; Seyer, J.M.; Kang, A.H. Chemotactic attraction of human fibroblasts to type I, II, and III collagens and collagen-derived peptides. Proc. Natl. Acad. Sci. USA 1978, 75, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Aimes, R.T.; Quigley, J.P. Matrix metalloproteinase-2 is an interstitial collagenase. Inhibitor-free enzyme catalyzes the cleavage of collagen fibrils and soluble native type I collagen generating the specific 3/4- and 1/4-length fragments. J. Biol. Chem. 1995, 270, 5872–5876. [Google Scholar] [CrossRef] [PubMed]
- Fields, G.B. A model for interstitial collagen catabolism by mammalian collagenases. J. Theor. Biol. 1991, 153, 585–602. [Google Scholar] [CrossRef] [PubMed]
- Freije, J.M.; Diez-Itza, I.; Balbin, M.; Sanchez, L.M.; Blasco, R.; Tolivia, J.; Lopez-Otin, C. Molecular cloning and expression of collagenase-3, a novel human matrix metalloproteinase produced by breast carcinomas. J. Biol. Chem. 1994, 269, 16766–16773. [Google Scholar] [PubMed]
- Knauper, V.; Lopez-Otin, C.; Smith, B.; Knight, G.; Murphy, G. Biochemical characterization of human collagenase-3. J. Biol. Chem. 1996, 271, 1544–1550. [Google Scholar] [CrossRef] [PubMed]
- Ohuchi, E.; Imai, K.; Fujii, Y.; Sato, H.; Seiki, M.; Okada, Y. Membrane type 1 matrix metalloproteinase digests interstitial collagens and other extracellular matrix macromolecules. J. Biol. Chem. 1997, 272, 2446–2451. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Kurkinen, M. Cloning and characterization of a novel matrix metalloproteinase (MMP), CMMP, from chicken embryo fibroblasts. CMMP, Xenopus XMMP, and human MMP19 have a conserved unique cysteine in the catalytic domain. J. Biol. Chem. 1998, 273, 17893–17900. [Google Scholar] [CrossRef] [PubMed]
- Allan, J.A.; Hembry, R.M.; Angal, S.; Reynolds, J.J.; Murphy, G. Binding of latent and high Mr active forms of stromelysin to collagen is mediated by the C-terminal domain. J. Cell Sci. 1991, 99, 789–795. [Google Scholar] [PubMed]
- Allan, J.A.; Docherty, A.J.; Barker, P.J.; Huskisson, N.S.; Reynolds, J.J.; Murphy, G. Binding of gelatinases A and B to type-I collagen and other matrix components. Biochem. J. 1995, 309, 299–306. [Google Scholar] [PubMed]
- Murphy, G.; Allan, J.A.; Willenbrock, F.; Cockett, M.I.; O'Connell, J.P.; Docherty, A.J. The role of the C-terminal domain in collagenase and stromelysin specificity. J. Biol. Chem. 1992, 267, 9612–9618. [Google Scholar] [PubMed]
- Lauer-Fields, J.L.; Fields, G.B. Triple-helical peptide analysis of collagenolytic protease activity. Biol. Chem. 2002, 383, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Ruoslahti, E.; Hayman, E.G.; Pierschbacher, M.D. Extracellular matrices and cell adhesion. Arteriosclerosis 1985, 5, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Smethurst, P.A.; Onley, D.J.; Jarvis, G.E.; O'Connor, M.N.; Knight, C.G.; Herr, A.B.; Ouwehand, W.H.; Farndale, R.W. Structural basis for the platelet-collagen interaction: The smallest motif within collagen that recognizes and activates platelet Glycoprotein VI contains two glycine-proline-hydroxyproline triplets. J. Biol. Chem. 2007, 282, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Knight, C.G.; Morton, L.F.; Onley, D.J.; Peachey, A.R.; Messent, A.J.; Smethurst, P.A.; Tuckwell, D.S.; Farndale, R.W.; Barnes, M.J. Identification in collagen type I of an integrin alpha2 beta1-binding site containing an essential GER sequence. J. Biol. Chem. 1998, 273, 33287–33294. [Google Scholar] [CrossRef] [PubMed]
- Knight, C.G.; Morton, L.F.; Peachey, A.R.; Tuckwell, D.S.; Farndale, R.W.; Barnes, M.J. The collagen-binding A-domains of integrins alpha(1)beta(1) and alpha(2)beta(1) recognize the same specific amino acid sequence, GFOGER, in native (triple-helical) collagens. J. Biol. Chem. 2000, 275, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Konitsiotis, A.D.; Raynal, N.; Bihan, D.; Hohenester, E.; Farndale, R.W.; Leitinger, B. Characterization of high affinity binding motifs for the discoidin domain receptor DDR2 in collagen. J. Biol. Chem. 2008, 283, 6861–6868. [Google Scholar] [CrossRef] [PubMed]
- Gullberg, D.; Gehlsen, K.R.; Turner, D.C.; Ahlen, K.; Zijenah, L.S.; Barnes, M.J.; Rubin, K. Analysis of alpha 1 beta 1, alpha 2 beta 1 and alpha 3 beta 1 integrins in cell--collagen interactions: Identification of conformation dependent alpha 1 beta 1 binding sites in collagen type I. EMBO J. 1992, 11, 3865–3873. [Google Scholar] [PubMed]
- Ruoslahti, E.; Vaheri, A. Novel human serum protein from fibroblast plasma membrane. Nature 1974, 248, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Pierschbacher, M.D.; Ruoslahti, E. Cell attachment activity of fibronectin can be duplicated by small synthetic fragments of the molecule. Nature 1984, 309, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, L.R.; Schonherr, E.; Waddington, R.; Niland, S.; Seidler, D.G.; Aeschlimann, D.; Eble, J.A. Decorin regulates endothelial cell motility on collagen I through activation of insulin-like growth factor I receptor and modulation of alpha2beta1 integrin activity. J. Biol. Chem. 2008, 283, 17406–17415. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, T.W.; Sellaro, T.L.; Badylak, S.F. Decellularization of tissues and organs. Biomaterials 2006, 27, 3675–3683. [Google Scholar] [PubMed]
- Boyce, S.T.; Christianson, D.J.; Hansbrough, J.F. Structure of a collagen-GAG dermal skin substitute optimized for cultured human epidermal keratinocytes. J. Biomed. Mater. Res. 1988, 22, 939–957. [Google Scholar] [CrossRef] [PubMed]
- Ellis, D.L.; Yannas, I.V. Recent advances in tissue synthesis in vivo by use of collagen-glycosaminoglycan copolymers. Biomaterials 1996, 17, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Marsilio, E.; Goldstein, R.H.; Yannas, I.V.; Spector, M. Formation of lung alveolar-like structures in collagen-glycosaminoglycan scaffolds in vitro. Tissue Eng. 2005, 11, 1436–1448. [Google Scholar] [CrossRef] [PubMed]
- Aprahamian, M.; Lambert, A.; Balboni, G.; Lefebvre, F.; Schmitthaeusler, R.; Damge, C.; Rabaud, M. A new reconstituted connective tissue matrix: Preparation, biochemical, structural and mechanical studies. J. Biomed. Mater. Res. 1987, 21, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Buijtenhuijs, P.; Buttafoco, L.; Poot, A.A.; Daamen, W.F.; van Kuppevelt, T.H.; Dijkstra, P.J.; de Vos, R.A.; Sterk, L.M.; Geelkerken, B.R.; Feijen, J.; Vermes, I. Tissue engineering of blood vessels: Characterization of smooth-muscle cells for culturing on collagen-and-elastin-based scaffolds. Biotechnol. Appl. Biochem. 2004, 39, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Garcia, Y.; Hemantkumar, N.; Collighan, R.; Griffin, M.; Rodriguez-Cabello, J.C.; Pandit, A. In vitro characterization of a collagen scaffold enzymatically cross-linked with a tailored elastin-like polymer. Tissue Eng. A 2009, 15, 887–899. [Google Scholar] [CrossRef]
- Damour, O.; Gueugniaud, P.Y.; Berthin-Maghit, M.; Rousselle, P.; Berthod, F.; Sahuc, F.; Collombel, C. A dermal substrate made of collagen--GAG--chitosan for deep burn coverage: First clinical uses. Clin. Mater. 1994, 15, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Shahabeddin, L.; Berthod, F.; Damour, O.; Collombel, C. Characterization of skin reconstructed on a chitosan-cross-linked collagen-glycosaminoglycan matrix. Skin Pharmacol. 1990, 3, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Black, L.; Santacana-Laffitte, G.; Patrick, C.W., Jr. Preparation and assessment of glutaraldehyde-crosslinked collagen-chitosan hydrogels for adipose tissue engineering. J. Biomed. Mater. Res. A 2007, 81, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Steven, F.S.; Tristram, G.R. The presence of non-protein nitrogen in acetic acid-soluble calf-skin collagen. Biochem. J. 1962, 83, 240–244. [Google Scholar] [PubMed]
- Steven, F.S.; Jackson, D.S. Purification and amino acid composition of monomeric and polymeric collagens. Biochem. J. 1967, 104, 534–536. [Google Scholar] [PubMed]
- Eastoe, J.E. The amino acid composition of mammalian collagen and gelatin. Biochem. J. 1955, 61, 589–600. [Google Scholar] [PubMed]
- Gross, J.; Highberger, J.H.; Schmitt, F.O. Extraction of collagen from connective tissue by neutral salt solutions. Proc. Natl. Acad. Sci. USA 1955, 41, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Grant, N.H.; Alburn, H.E. Collagen solubilization by mammalian proteinases. Arch. Biochem. Biophys. 1960, 89, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Steven, F.S. The nishihara technique for the solubilization of collagen. Application to the preparation of soluble collagens from normal and rheumatoid connective tissue. Ann. Rheum. Dis. 1964, 23, 300–301. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.P.; Davison, P.F.; Bump, S.; Schmitt, F.O. Action of proteolytic enzymes on tropocollagen and insoluble collagen. Biochemistry 1966, 5, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Rubin, A.L.; Pfahl, D.; Speakman, P.T.; Davison, P.F.; Schmitt, F.O. Tropocollagen: Significance of protease-induced alterations. Science 1963, 139, 37–39. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.J.; Rhodes, R.K. Preparation and characterization of the different types of collagen. Methods Enzymol. 1982, 82, 33–64. [Google Scholar] [PubMed]
- Miller, E.J. Structural studies on cartilage collagen employing limited cleavage and solubilization with pepsin. Biochemistry 1972, 11, 4903–4909. [Google Scholar] [CrossRef] [PubMed]
- Weadock, K.S.; Miller, E.J.; Bellincampi, L.D.; Zawadsky, J.P.; Dunn, M.G. Physical crosslinking of collagen fibers: Comparison of ultraviolet irradiation and dehydrothermal treatment. J. Biomed. Mater. Res. 1995, 29, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Ohan, M.P.; Weadock, K.S.; Dunn, M.G. Synergistic effects of glucose and ultraviolet irradiation on the physical properties of collagen. J. Biomed. Mater. Res. 2002, 60, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Chvapil, M.; Owen, J.A.; Clark, D.S. Effect of collagen crosslinking on the rate of resorption of implanted collagen tubing in rabbits. J. Biomed. Mater. Res. 1977, 11, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Yannas, I.V.; Burke, J.F. Design of an artificial skin. I. Basic design principles. J. Biomed. Mater. Res. 1980, 14, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Osborne, C.S.; Barbenel, J.C.; Smith, D.; Savakis, M.; Grant, M.H. Investigation into the tensile properties of collagen/chondroitin-6-sulphate gels: The effect of crosslinking agents and diamines. Med. Biol. Eng. Comput. 1998, 36, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Powell, H.M.; Boyce, S.T. EDC cross-linking improves skin substitute strength and stability. Biomaterials 2006, 27, 5821–5827. [Google Scholar] [CrossRef] [PubMed]
- Powell, H.M.; Boyce, S.T. Wound closure with EDC cross-linked cultured skin substitutes grafted to athymic mice. Biomaterials 2007, 28, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Sheardown, H. Crosslinking of collagen with dendrimers. J. Biomed. Mater. Res.A 2005, 75, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Castaneda, L.; Valle, J.; Yang, N.; Pluskat, S.; Slowinska, K. Collagen cross-linking with Au nanoparticles. Biomacromolecules 2008, 9, 3383–3388. [Google Scholar] [CrossRef] [PubMed]
- Zeeman, R.; Dijkstra, P.J.; van Wachem, P.B.; van Luyn, M.J.; Hendriks, M.; Cahalan, P.T.; Feijen, J. Successive epoxy and carbodiimide cross-linking of dermal sheep collagen. Biomaterials 1999, 20, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Everaerts, F.; Torrianni, M.; Hendriks, M.; Feijen, J. Biomechanical properties of carbodiimide crosslinked collagen: Influence of the formation of ester crosslinks. J. Biomed. Mater. Res. A 2008, 85, 547–555. [Google Scholar] [CrossRef] [PubMed]
- van Wachem, P.B.; van Luyn, M.J.; Olde Damink, L.H.; Dijkstra, P.J.; Feijen, J.; Nieuwenhuis, P. Biocompatibility and tissue regenerating capacity of crosslinked dermal sheep collagen. J. Biomed. Mater. Res. 1994, 28, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Zeugolis, D.I.; Paul, G.R.; Attenburrow, G. Cross-linking of extruded collagen fibers-a biomimetic three-dimensional scaffold for tissue engineering applications. J. Biomed. Mater. Res. A 2009, 89, 895–908. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.W.; Huang, R.N.; Huang, L.L.; Tsai, C.C. In vitro evaluation of cytotoxicity of a naturally occurring cross-linking reagent for biological tissue fixation. J. Biomater. Sci. Polym. Ed. 1999, 10, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Sundararaghavan, H.G.; Monteiro, G.A.; Lapin, N.A.; Chabal, Y.J.; Miksan, J.R.; Shreiber, D.I. Genipin-induced changes in collagen gels: Correlation of mechanical properties to fluorescence. J. Biomed. Mater. Res.A 2008, 87, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Speer, D.P.; Chvapil, M.; Eskelson, C.D.; Ulreich, J. Biological effects of residual glutaraldehyde in glutaraldehyde-tanned collagen biomaterials. J. Biomed. Mater. Res. 1980, 14, 753–764. [Google Scholar] [CrossRef] [PubMed]
- van Luyn, M.J.; van Wachem, P.B.; Olde Damink, L.H.; Dijkstra, P.J.; Feijen, J.; Nieuwenhuis, P. Secondary cytotoxicity of cross-linked dermal sheep collagens during repeated exposure to human fibroblasts. Biomaterials 1992, 13, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Berthod, F.; Saintigny, G.; Chretien, F.; Hayek, D.; Collombel, C.; Damour, O. Optimization of thickness, pore size and mechanical properties of a biomaterial designed for deep burn coverage. Clin. Mater. 1994, 15, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Berthod, F.; Hayek, D.; Damour, O.; Collombel, C. Collagen synthesis by fibroblasts cultured within a collagen sponge. Biomaterials 1993, 14, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Yung, C.W.; Wu, L.Q.; Tullman, J.A.; Payne, G.F.; Bentley, W.E.; Barbari, T.A. Transglutaminase crosslinked gelatin as a tissue engineering scaffold. J. Biomed. Mater. Res. A 2007, 83, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Khew, S.T.; Yang, Q.J.; Tong, Y.W. Enzymatically crosslinked collagen-mimetic dendrimers that promote integrin-targeted cell adhesion. Biomaterials 2008, 29, 3034–3045. [Google Scholar] [CrossRef] [PubMed]
- Yeo, T.K.; Brown, L.; Dvorak, H.F. Alterations in proteoglycan synthesis common to healing wounds and tumors. Am. J. Pathol. 1991, 138, 1437–1450. [Google Scholar] [PubMed]
- Teti, A. Regulation of cellular functions by extracellular matrix. J. Am. Soc. Nephrol. 1992, 2, 83–87. [Google Scholar]
- Huang-Lee, L.L.; Wu, J.H.; Nimni, M.E. Effects of hyaluronan on collagen fibrillar matrix contraction by fibroblasts. J. Biomed. Mater. Res. 1994, 28, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.; Yung, L.Y. Enhanced biological stability of collagen with incorporation of PAMAM dendrimer. J. Biomed. Mater. Res. A 2009, 91, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Caissie, R.; Gingras, M.; Champigny, M.F.; Berthod, F. In vivo enhancement of sensory perception recovery in a tissue-engineered skin enriched with laminin. Biomaterials 2006, 27, 2988–2993. [Google Scholar] [CrossRef] [PubMed]
- Antonicelli, F.; Bellon, G.; Lorimier, S.; Hornebeck, W. Role of the elastin receptor complex (S-Gal/Cath-A/Neu-1) in skin repair and regeneration. Wound Repair Regen. 2009, 17, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Suh, H.; Lee, J.E. Behavior of fibroblasts on a porous hyaluronic acid incorporated collagen matrix. Yonsei Med. J. 2002, 43, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Roe, S.C.; Milthorpe, B.K.; True, K.; Rogers, G.J.; Schindhelm, K. The effect of gamma irradiation on a xenograft tendon bioprosthesis. Clin. Mater. 1992, 9, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Cheung, D.T.; Perelman, N.; Tong, D.; Nimni, M.E. The effect of gamma-irradiation on collagen molecules, isolated alpha-chains, and crosslinked native fibers. J. Biomed. Mater. Res. 1990, 24, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Mollers, S.; Heschel, I.; Damink, L.H.; Schugner, F.; Deumens, R.; Muller, B.; Bozkurt, A.; Nava, J.G.; Noth, J.; Brook, G.A. Cytocompatibility of a novel, longitudinally microstructured collagen scaffold intended for nerve tissue repair. Tissue Eng. A 2009, 15, 461–472. [Google Scholar] [CrossRef]
- Ohan, M.P.; Dunn, M.G. Glucose stabilizes collagen sterilized with gamma irradiation. J. Biomed. Mater. Res. A 2003, 67, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Noah, E.M.; Chen, J.; Jiao, X.; Heschel, I.; Pallua, N. Impact of sterilization on the porous design and cell behavior in collagen sponges prepared for tissue engineering. Biomaterials 2002, 23, 2855–2861. [Google Scholar] [CrossRef] [PubMed]
- Friess, W.; Schlapp, M. Sterilization of gentamicin containing collagen/PLGA microparticle composites. Eur. J. Pharm. Biopharm. 2006, 63, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Grimes, M.; Pembroke, J.T.; McGloughlin, T. The effect of choice of sterilisation method on the biocompatibility and biodegradability of SIS (small intestinal submucosa). Biomed. Mater. Eng. 2005, 15, 65–71. [Google Scholar] [PubMed]
- Wilshaw, S.P.; Kearney, J.N.; Fisher, J.; Ingham, E. Production of an acellular amniotic membrane matrix for use in tissue engineering. Tissue Eng. 2006, 12, 2117–2129. [Google Scholar] [CrossRef] [PubMed]
- Doillon, C.J.; Drouin, R.; Cote, M.F.; Dallaire, N.; Pageau, J.F.; Laroche, G. Chemical inactivators as sterilization agents for bovine collagen materials. J. Biomed. Mater. Res. 1997, 37, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Gao, C.; Mao, Z.; Zhou, J.; Shen, J.; Hu, X.; Han, C. Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering. Biomaterials 2003, 24, 4833–4841. [Google Scholar] [CrossRef] [PubMed]
- Gingras, M.; Paradis, I.; Berthod, F. Nerve regeneration in a collagen-chitosan tissue-engineered skin transplanted on nude mice. Biomaterials 2003, 24, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Abel, M.; Ruth, P.; Wilhelms, T.; Schulze, D.; Norgauer, J.; Hipler, U.C. Effect of the sterilization method on the performance of collagen type I on chronic wound parameters in vitro. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 90, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Molnar, P.; Gregory, C.; Das, M.; Boland, T.; Hickman, J.J. Electrophysiological characterization of embryonic hippocampal neurons cultured in a 3D collagen hydrogel. Biomaterials 2009, 30, 4377–4383. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Fitzgerald, W.; Liu, Q.Y.; O'Shaughnessy, T.J.; Maric, D.; Lin, H.J.; Alkon, D.L.; Barker, J.L. CNS stem and progenitor cell differentiation into functional neuronal circuits in three-dimensional collagen gels. Exp. Neurol. 2004, 190, 276–288. [Google Scholar] [CrossRef] [PubMed]
- O'Shaughnessy, T.J.; Lin, H.J.; Ma, W. Functional synapse formation among rat cortical neurons grown on three-dimensional collagen gels. Neurosci. Lett. 2003, 340, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Gingras, M.; Beaulieu, M.M.; Gagnon, V.; Durham, H.D.; Berthod, F. In vitro study of axonal migration and myelination of motor neurons in a three-dimensional tissue-engineered model. Glia 2008, 56, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.M.; Jung, T.H.; Choi, J.H.; Yoon do, J.; Jeong, H.J.; Lee, E.J.; Kim, J. Collagen-based co-culture for invasive study on cancer cells-fibroblasts interaction. Biochem. Biophys. Res. Commun. 2006, 346, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Sabeh, F.; Shimizu-Hirota, R.; Weiss, S.J. Protease-dependent versus -independent cancer cell invasion programs: Three-dimensional amoeboid movement revisited. J. Cell Biol. 2009, 185, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Toda, S.; Narisawa, Y.; Sugihara, H. Subcutaneous adipocytes promote the differentiation of squamous cell carcinoma cell line (DJM-1) in collagen gel matrix culture. J. Invest. Dermatol. 2001, 117, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Shanmugasundaram, N.; Ravichandran, P.; Reddy, P.N.; Ramamurty, N.; Pal, S.; Rao, K.P. Collagen-chitosan polymeric scaffolds for the in vitro culture of human epidermoid carcinoma cells. Biomaterials 2001, 22, 1943–1951. [Google Scholar] [CrossRef] [PubMed]
- Stachowiak, A.N.; Irvine, D.J. Inverse opal hydrogel-collagen composite scaffolds as a supportive microenvironment for immune cell migration. J. Biomed. Mater. Res. A 2008, 85, 815–828. [Google Scholar] [CrossRef] [PubMed]
- Wolf, K.; Muller, R.; Borgmann, S.; Brocker, E.B.; Friedl, P. Amoeboid shape change and contact guidance: T-lymphocyte crawling through fibrillar collagen is independent of matrix remodeling by MMPs and other proteases. Blood 2003, 102, 3262–3269. [Google Scholar] [CrossRef] [PubMed]
- Spencer, N.J.; Cotanche, D.A.; Klapperich, C.M. Peptide- and collagen-based hydrogel substrates for in vitro culture of chick cochleae. Biomaterials 2008, 29, 1028–1042. [Google Scholar] [CrossRef] [PubMed]
- Cortial, D.; Gouttenoire, J.; Rousseau, C.F.; Ronziere, M.C.; Piccardi, N.; Msika, P.; Herbage, D.; Mallein-Gerin, F.; Freyria, A.M. Activation by IL-1 of bovine articular chondrocytes in culture within a 3D collagen-based scaffold. An in vitro model to address the effect of compounds with therapeutic potential in osteoarthritis. Osteoarthritis Cartilage 2006, 14, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Harley, B.A.; Lynn, A.K.; Wissner-Gross, Z.; Bonfield, W.; Yannas, I.V.; Gibson, L.J. Design of a multiphase osteochondral scaffold. II. Fabrication of a mineralized collagen-glycosaminoglycan scaffold. J. Biomed. Mater. Res. A 2010, 92, 1066–1077. [Google Scholar] [PubMed]
- Du, C.; Cui, F.Z.; Zhang, W.; Feng, Q.L.; Zhu, X.D.; de Groot, K. Formation of calcium phosphate/collagen composites through mineralization of collagen matrix. J. Biomed. Mater. Res. 2000, 50, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Ciardelli, G.; Gentile, P.; Chiono, V.; Mattioli-Belmonte, M.; Vozzi, G.; Barbani, N.; Giusti, P. Enzymatically crosslinked porous composite matrices for bone tissue regeneration. J. Biomed. Mater. Res. A 2009, 92, 137–151. [Google Scholar]
- Dubey, D.K.; Tomar, V. Role of the nanoscale interfacial arrangement in mechanical strength of tropocollagen-hydroxyapatite-based hard biomaterials. Acta Biomater. 2009, 5, 2704–2716. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Ngiam, M.; Chan, C.K.; Ramakrishna, S. Fabrication of nano-hydroxyapatite/collagen/osteonectin composites for bone graft applications. Biomed. Mater. 2009, 4, 25019. [Google Scholar] [CrossRef]
- Tamimi, F.; Kumarasami, B.; Doillon, C.; Gbureck, U.; Le Nihouannen, D.; Cabarcos, E.L.; Barralet, J.E. Brushite-collagen composites for bone regeneration. Acta Biomater. 2008, 4, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Jayaraman, M.; Subramanian, M.V. Preparation and characterization of two new composites: Collagen-brushite and collagen octa-calcium phosphate. Med. Sci. Monit. 2002, 8, BR481–487. [Google Scholar] [PubMed]
- Tebb, T.A.; Tsai, S.W.; Glattauer, V.; White, J.F.; Ramshaw, J.A.; Werkmeister, J.A. Development of porous collagen beads for chondrocyte culture. Cytotechnology 2006, 52, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Glattauer, V.; White, J.F.; Tsai, W.B.; Tsai, C.C.; Tebb, T.A.; Danon, S.J.; Werkmeister, J.A.; Ramshaw, J.A. Preparation of resorbable collagen-based beads for direct use in tissue engineering and cell therapy applications. J. Biomed. Mater. Res. A 2010, 92, 1301–1309. [Google Scholar] [PubMed]
- Gastel, J.A.; Muirhead, W.R.; Lifrak, J.T.; Fadale, P.D.; Hulstyn, M.J.; Labrador, D.P. Meniscal tissue regeneration using a collagenous biomaterial derived from porcine small intestine submucosa. Arthroscopy 2001, 17, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.L.; Fox, D.B.; Malaviya, P.; Tomlinson, J.L.; Farr, J.; Kuroki, K.; Cook, C.R. Evaluation of small intestinal submucosa grafts for meniscal regeneration in a clinically relevant posterior meniscectomy model in dogs. J. Knee Surg. 2006, 19, 159–167. [Google Scholar] [PubMed]
- De Franceschi, L.; Grigolo, B.; Roseti, L.; Facchini, A.; Fini, M.; Giavaresi, G.; Tschon, M.; Giardino, R. Transplantation of chondrocytes seeded on collagen-based scaffold in cartilage defects in rabbits. J. Biomed. Mater. Res. A 2005, 75, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Freyria, A.M.; Ronziere, M.C.; Cortial, D.; Galois, L.; Hartmann, D.; Herbage, D.; Mallein-Gerin, F. Comparative phenotypic analysis of articular chondrocytes cultured within type I or type II collagen scaffolds. Tissue Eng. A 2009, 15, 1233–1245. [Google Scholar] [CrossRef]
- Schneider, R.K.; Puellen, A.; Kramann, R.; Raupach, K.; Bornemann, J.; Knuechel, R.; Perez-Bouza, A.; Neuss, S. The osteogenic differentiation of adult bone marrow and perinatal umbilical mesenchymal stem cells and matrix remodelling in three-dimensional collagen scaffolds. Biomaterials 2010, 31, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.M.; Haugh, M.G.; O'Brien, F.J. The effect of mean pore size on cell attachment, proliferation and migration in collagen-glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials 2010, 31, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, T.; Deie, M.; Shinomiya, R.; Yasunaga, Y.; Yanada, S.; Ochi, M. Transplantation of meniscus regenerated by tissue engineering with a scaffold derived from a rat meniscus and mesenchymal stromal cells derived from rat bone marrow. Artif. Organs 2008, 32, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, T.W.; Ingram, J.; Katta, J.; Knight, R.; Korossis, S.; Fisher, J.; Ingham, E. Development and characterization of an acellular porcine medial meniscus for use in tissue engineering. Tissue Eng. A 2008, 14, 505–518. [Google Scholar] [CrossRef]
- Tedder, M.E.; Liao, J.; Weed, B.; Stabler, C.; Zhang, H.; Simionescu, A.; Simionescu, D.T. Stabilized collagen scaffolds for heart valve tissue engineering. Tissue Eng. A 2009, 15, 1257–1268. [Google Scholar] [CrossRef]
- Eitan, Y.; Sarig, U.; Dahan, N.; Machluf, M. Acellular cardiac extracellular matrix as a scaffold for tissue engineering: In-vitro cell support, remodeling and biocompatibility. Tissue Eng. C Methods 2010, in press. [Google Scholar]
- Somers, P.; Cuvelier, C.A.; Somer, F.D.; Cornelissen, M.; Cox, E.; Verloo, M.; Chiers, K.; van Nooten, G. Gamma radiation alters the ultrastructure in tissue-engineered heart valve scaffolds. Tissue Eng. A 2009, 15, 3597–3604. [Google Scholar] [CrossRef]
- Konuma, T.; Devaney, E.J.; Bove, E.L.; Gelehrter, S.; Hirsch, J.C.; Tavakkol, Z.; Ohye, R.G. Performance of CryoValve SG decellularized pulmonary allografts compared with standard cryopreserved allografts. Ann. Thorac. Surg. 2009, 88, 849–854; discussion 554–845. [Google Scholar] [CrossRef] [PubMed]
- O'Brien, M.F.; Goldstein, S.; Walsh, S.; Black, K.S.; Elkins, R.; Clarke, D. The SynerGraft valve: A new acellular (nonglutaraldehyde-fixed) tissue heart valve for autologous recellularization first experimental studies before clinical implantation. Semin. Thorac. Cardiovasc. Surg. 1999, 11, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Bechtel, J.F.; Muller-Steinhardt, M.; Schmidtke, C.; Brunswik, A.; Stierle, U.; Sievers, H.H. Evaluation of the decellularized pulmonary valve homograft (SynerGraft). J. Heart Valve Dis. 2003, 12, 734–739; discussion 739–740. [Google Scholar] [PubMed]
- Ott, H.C.; Matthiesen, T.S.; Goh, S.K.; Black, L.D.; Kren, S.M.; Netoff, T.I.; Taylor, D.A. Perfusion-decellularized matrix: Using nature's platform to engineer a bioartificial heart. Nat. Med. 2008, 14, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Teebken, O.E.; Puschmann, C.; Breitenbach, I.; Rohde, B.; Burgwitz, K.; Haverich, A. Preclinical development of tissue-engineered vein valves and venous substitutes using re-endothelialised human vein matrix. Eur. J. Vasc. Endovasc. Surg. 2009, 37, 92–102. [Google Scholar] [CrossRef] [PubMed]
- L'Heureux, N.; Paquet, S.; Labbe, R.; Germain, L.; Auger, F.A. A completely biological tissue-engineered human blood vessel. FASEB J. 1998, 12, 47–56. [Google Scholar] [PubMed]
- L'Heureux, N.; Stoclet, J.C.; Auger, F.A.; Lagaud, G.J.; Germain, L.; Andriantsitohaina, R. A human tissue-engineered vascular media: A new model for pharmacological studies of contractile responses. FASEB J. 2001, 15, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Laflamme, K.; Roberge, C.J.; Labonte, J.; Pouliot, S.; D'Orleans-Juste, P.; Auger, F.A.; Germain, L. Tissue-engineered human vascular media with a functional endothelin system. Circulation 2005, 111, 459–464. [Google Scholar] [CrossRef] [PubMed]
- McAllister, T.N.; Maruszewski, M.; Garrido, S.A.; Wystrychowski, W.; Dusserre, N.; Marini, A.; Zagalski, K.; Fiorillo, A.; Avila, H.; Manglano, X.; Antonelli, J.; Kocher, A.; Zembala, M.; Cierpka, L.; de la Fuente, L.M.; L'Heureux, N. Effectiveness of haemodialysis access with an autologous tissue-engineered vascular graft: A multicentre cohort study. Lancet 2009, 373, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- L'Heureux, N.; McAllister, T.N.; de la Fuente, L.M. Tissue-engineered blood vessel for adult arterial revascularization. N. Engl. J. Med. 2007, 357, 1451–1453. [Google Scholar] [CrossRef] [PubMed]
- Doillon, C.J.; Silver, F.H. Collagen-based wound dressing: Effects of hyaluronic acid and fibronectin on wound healing. Biomaterials 1986, 7, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.J. Biological dressings in burns-A review. Ann. Plastic. Surg. 1980, 4, 133–137. [Google Scholar] [CrossRef]
- Regnier, M.; Staquet, M.J.; Schmitt, D.; Schmidt, R. Integration of Langerhans cells into a pigmented reconstructed human epidermis. J. Invest. Dermatol. 1997, 109, 510–512. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, P.L.; Hudon, V.; Berthod, F.; Germain, L.; Auger, F.A. Inosculation of tissue-engineered capillaries with the host's vasculature in a reconstructed skin transplanted on mice. Am. J. Transplant. 2005, 5, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Bechetoille, N.; Dezutter-Dambuyant, C.; Damour, O.; Andre, V.; Orly, I.; Perrier, E. Effects of solar ultraviolet radiation on engineered human skin equivalent containing both Langerhans cells and dermal dendritic cells. Tissue Eng. 2007, 13, 2667–2679. [Google Scholar] [CrossRef] [PubMed]
- Blais, M.; Grenier, M.; Berthod, F. Improvement of nerve regeneration in tissue-engineered skin enriched with schwann cells. J. Invest. Dermatol. 2009, 129, 2895–2900. [Google Scholar] [CrossRef] [PubMed]
- Trottier, V.; Marceau-Fortier, G.; Germain, L.; Vincent, C.; Fradette, J. IFATS collection: Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells 2008, 26, 2713–2723. [Google Scholar] [CrossRef] [PubMed]
- Jean, J.; Lapointe, M.; Soucy, J.; Pouliot, R. Development of an in vitro psoriatic skin model by tissue engineering. J. Dermatol. Sci. 2009, 53, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Corriveau, M.P.; Boufaied, I.; Lessard, J.; Chabaud, S.; Senecal, J.L.; Grodzicky, T.; Chartier, S.; Raymond, Y.; Moulin, V.J. The fibrotic phenotype of systemic sclerosis fibroblasts varies with disease duration and severity of skin involvement: Reconstitution of skin fibrosis development using a tissue engineering approach. J. Pathol. 2009, 217, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Edmonds, M. Apligraf in the treatment of neuropathic diabetic foot ulcers. Int. J. Low Extrem. Wounds 2009, 8, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Karr, J. Utilization of living bilayered cell therapy (Apligraf) for heel ulcers. Adv. Skin Wound Care 2008, 21, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Nie, C.; Yang, D.; Morris, S.F. Local delivery of adipose-derived stem cells via acellular dermal matrix as a scaffold: A new promising strategy to accelerate wound healing. Med. Hypotheses 2009, 72, 679–682. [Google Scholar] [CrossRef] [PubMed]
- Altman, A.M.; Chiu, E.S.; Bai, X.; Yan, Y.; Song, Y.H.; Newsome, R.E.; Alt, E.U. Human adipose-derived stem cells adhere to acellular dermal matrix. Aesthetic Plast. Surg. 2008, 32, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Altman, A.M.; Matthias, N.; Yan, Y.; Song, Y.H.; Bai, X.; Chiu, E.S.; Slakey, D.P.; Alt, E.U. Dermal matrix as a carrier for in vivo delivery of human adipose-derived stem cells. Biomaterials 2008, 29, 1431–1442. [Google Scholar] [CrossRef] [PubMed]
- Levis, H.; Daniels, J.T. New technologies in limbal epithelial stem cell transplantation. Curr. Opin. Biotechnol. 2009, 20, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Schwab, I.R. Cultured corneal epithelia for ocular surface disease. Trans. Am. Ophthalmol. Soc. 1999, 97, 891–986. [Google Scholar] [PubMed]
- Zakaria, N.; Koppen, C.; Van Tendeloo, V.; Berneman, Z.; Hopkinson, A.; Tassignon, M.J. Standardized limbal epithelial stem cell graft generation and transplantation. Tissue Eng. C Methods 2010, in presss. [Google Scholar]
- Shortt, A.J.; Secker, G.A.; Lomas, R.J.; Wilshaw, S.P.; Kearney, J.N.; Tuft, S.J.; Daniels, J.T. The effect of amniotic membrane preparation method on its ability to serve as a substrate for the ex-vivo expansion of limbal epithelial cells. Biomaterials 2009, 30, 1056–1065. [Google Scholar] [CrossRef] [PubMed]
- Dravida, S.; Gaddipati, S.; Griffith, M.; Merrett, K.; Lakshmi Madhira, S.; Sangwan, V.S.; Vemuganti, G.K. A biomimetic scaffold for culturing limbal stem cells: A promising alternative for clinical transplantation. J. Tissue Eng. Regen. Med. 2008, 2, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Grueterich, M.; Espana, E.M.; Tseng, S.C. Ex vivo expansion of limbal epithelial stem cells: Amniotic membrane serving as a stem cell niche. Surv. Ophthalmol. 2003, 48, 631–646. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.; Jackson, W.B.; Lagali, N.; Merrett, K.; Li, F.; Fagerholm, P. Artificial corneas: A regenerative medicine approach. Eye (Lond) 2009, 23, 1985–1989. [Google Scholar] [CrossRef]
- Liu, Y.; Griffith, M.; Watsky, M.A.; Forrester, J.V.; Kuffova, L.; Grant, D.; Merrett, K.; Carlsson, D.J. Properties of porcine and recombinant human collagen matrices for optically clear tissue engineering applications. Biomacromolecules 2006, 7, 1819–1828. [Google Scholar] [CrossRef] [PubMed]
- Lagali, N.; Griffith, M.; Fagerholm, P.; Merrett, K.; Huynh, M.; Munger, R. Innervation of tissue-engineered recombinant human collagen-based corneal substitutes: A comparative in vivo confocal microscopy study. Invest. Ophthalmol. Vis. Sci. 2008, 49, 3895–3902. [Google Scholar] [CrossRef] [PubMed]
- Carrier, P.; Deschambeault, A.; Talbot, M.; Giasson, C.J.; Auger, F.A.; Guerin, S.L.; Germain, L. Characterization of wound reepithelialization using a new human tissue-engineered corneal wound healing model. Invest. Ophthalmol. Vis. Sci. 2008, 49, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Rafat, M.; Matsuura, T.; Li, F.; Griffith, M. Surface modification of collagen-based artificial cornea for reduced endothelialization. J. Biomed. Mater. Res. A 2009, 88, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Shekarriz, B.; Upadhyay, J.; Demirbilek, S.; Barthold, J.S.; Gonzalez, R. Surgical complications of bladder augmentation: Comparison between various enterocystoplasties in 133 patients. Urology 2000, 55, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Mingin, G.C.; Stock, J.A.; Hanna, M.K. Gastrocystoplasty: Long-term complications in 22 patients. J. Urol. 1999, 162, 1122–1125. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bharadwaj, S.; Lee, S.J.; Atala, A.; Zhang, Y. Optimization of a natural collagen scaffold to aid cell-matrix penetration for urologic tissue engineering. Biomaterials 2009, 30, 3865–3873. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.S.; Zhang, S.L.; Geng, H.; Pan, J.; Chen, F. Ex vivo functional evaluation of isolated strips in BAMG tissue-engineered bladders. Int. J. Artif. Organs 2009, 32, 159–165. [Google Scholar] [PubMed]
- Parshotam Kumar, G.; Barker, A.; Ahmed, S.; Gerath, J.; Orford, J. Urinary bladder auto augmentation using INTEGRA((R)) and SURGISIS ((R)): An experimental model. Pediatr. Surg. Int. 2010, 26, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Akbal, C.; Lee, S.D.; Packer, S.C.; Davis, M.M.; Rink, R.C.; Kaefer, M. Bladder augmentation with acellular dermal biomatrix in a diseased animal model. J. Urol. 2006, 176, 1706–1711. [Google Scholar] [CrossRef] [PubMed]
- Fiala, R.; Vidlar, A.; Vrtal, R.; Belej, K.; Student, V. Porcine small intestinal submucosa graft for repair of anterior urethral strictures. Eur. Urol. 2007, 51, 1702–1708; discussion 1708. [Google Scholar] [CrossRef] [PubMed]
- Farahat, Y.A.; Elbahnasy, A.M.; El-Gamal, O.M.; Ramadan, A.R.; El-Abd, S.A.; Taha, M.R. Endoscopic urethroplasty using small intestinal submucosal patch in cases of recurrent urethral stricture: A preliminary study. J. Endourol. 2009, 23, 2001–2005. [Google Scholar] [CrossRef] [PubMed]
- el-Kassaby, A.; AbouShwareb, T.; Atala, A. Randomized comparative study between buccal mucosal and acellular bladder matrix grafts in complex anterior urethral strictures. J. Urol. 2008, 179, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- Bouhout, S.; Perron, E.; Gauvin, R.; Bernard, G.; Ouellet, G.; Cattan, V.; Bolduc, S. In vitro Reconstruction of an autologous, watertight and resistant vesical equivalent. Tissue Eng. A 2010. [Google Scholar] [CrossRef]
- Atala, A.; Bauer, S.B.; Soker, S.; Yoo, J.J.; Retik, A.B. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 2006, 367, 1241–1246. [Google Scholar] [CrossRef] [PubMed]
- Magnan, M.; Berthod, F.; Champigny, M.F.; Soucy, F.; Bolduc, S. In vitro reconstruction of a tissue-engineered endothelialized bladder from a single porcine biopsy. J. Pediatr. Urol. 2006, 2, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Oberson, C.; Boubaker, A.; Ramseyer, P.; Meyrat, B.J.; Frey, P. Endoscopic and surgical treatment of vesico-ureteral reflux in children. Comparative long-term follow-up. Swiss Med. Wkly. 2007, 137, 471–475. [Google Scholar] [PubMed]
- Isom-Batz, G.; Zimmern, P.E. Collagen injection for female urinary incontinence after urethral or periurethral surgery. J. Urol. 2009, 181, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Koduri, S.; Goldberg, R.P.; Kwon, C.; Dobrez, D.G.; Sand, P.K. Factors influencing the long-term success of periurethral collagen therapy in the office. Int. Urogynecol. J. Pelvic. Floor. Dysfunct. 2006, 17, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, L.J.; Yannas, I.V.; Hsu, H.P.; Strichartz, G.; Spector, M. Collagen-GAG substrate enhances the quality of nerve regeneration through collagen tubes up to level of autograft. Exp. Neurol. 1998, 154, 315–329. [Google Scholar] [CrossRef] [PubMed]
- Archibald, S.J.; Krarup, C.; Shefner, J.; Li, S.T.; Madison, R.D. A collagen-based nerve guide conduit for peripheral nerve repair: An electrophysiological study of nerve regeneration in rodents and nonhuman primates. J. Comp. Neurol. 1991, 306, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Colin, W.; Donoff, R.B. Nerve regeneration through collagen tubes. J. Dent. Res. 1984, 63, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Hudson, T.W.; Zawko, S.; Deister, C.; Lundy, S.; Hu, C.Y.; Lee, K.; Schmidt, C.E. Optimized acellular nerve graft is immunologically tolerated and supports regeneration. Tissue Eng. 2004, 10, 1641–1651. [Google Scholar] [CrossRef] [PubMed]
- Madaghiele, M.; Sannino, A.; Yannas, I.V.; Spector, M. Collagen-based matrices with axially oriented pores. J. Biomed. Mater. Res. A 2008, 85, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, A.; Brook, G.A.; Moellers, S.; Lassner, F.; Sellhaus, B.; Weis, J.; Woeltje, M.; Tank, J.; Beckmann, C.; Fuchs, P.; Damink, L.O.; Schugner, F.; Heschel, I.; Pallua, N. In vitro assessment of axonal growth using dorsal root ganglia explants in a novel three-dimensional collagen matrix. Tissue Eng. 2007, 13, 2971–2979. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Lin, H.; Chen, B.; Zhao, W.; Zhao, Y.; Xiao, Z.; Dai, J. Collagen scaffolds loaded with collagen-binding NGF-beta accelerate ulcer healing. J. Biomed. Mater. Res. A 2009, 92A, 887–895. [Google Scholar]
- Sun, W.; Lin, H.; Chen, B.; Zhao, W.; Zhao, Y.; Dai, J. Promotion of peripheral nerve growth by collagen scaffolds loaded with collagen-targeting human nerve growth factor-beta. J. Biomed. Mater. Res. A 2007, 83, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, C.; Pluderi, M.; Colleoni, F.; Belicchi, M.; Meregalli, M.; Farini, A.; Parolini, D.; Draghi, L.; Fruguglietti, M.E.; Gavina, M.; Porretti, L.; Cattaneo, A.; Battistelli, M.; Prelle, A.; Moggio, M.; Borsa, S.; Bello, L.; Spagnoli, D.; Gaini, S.M.; Tanzi, M.C.; Bresolin, N.; Grimoldi, N.; Torrente, Y. Skin-derived stem cells transplanted into resorbable guides provide functional nerve regeneration after sciatic nerve resection. Glia 2007, 55, 425–438. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, A.; Deumens, R.; Beckmann, C.; Olde Damink, L.; Schugner, F.; Heschel, I.; Sellhaus, B.; Weis, J.; Jahnen-Dechent, W.; Brook, G.A.; Pallua, N. In vitro cell alignment obtained with a Schwann cell enriched microstructured nerve guide with longitudinal guidance channels. Biomaterials 2009, 30, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.W.; Syed, S.; Walsh, W.; Zochodne, D.W.; Midha, R. Collagen nerve conduits promote enhanced axonal regeneration, schwann cell association, and neovascularization compared to silicone conduits. Tissue Eng. A 2009, 15, 1975–1988. [Google Scholar] [CrossRef]
- Blais, M.; Grenier, M.; Berthod, F. Improvement of nerve regeneration in tissue-engineered skin enriched with schwann cells. J. Invest. Dermatol. 2009, 129, 2895–2900. [Google Scholar] [CrossRef] [PubMed]
- Bentkover, S.H. The biology of facial fillers. Facial Plast. Surg. 2009, 25, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Stosich, M.S.; Moioli, E.; Lee, C.H.; Fu, S.; Bastian, B.; Eisig, S.; Zemnick, C.; Ascherman, J.; Wu, J.; Rohde, C.; Ahn, J. Facial Reconstruction by Biosurgery: Cell Transplantation vs. Cell Homing. Tissue Eng. B Rev. 2010, in press. [Google Scholar]
- Gurney, T.A.; Kim, D.W. Applications of porcine dermal collagen (ENDURAGen) in facial plastic surgery. Facial Plast. Surg. Clin. North Am. 2007, 15, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Pons-Guiraud, A. Adverse reactions to injectable fillers. Ann. Dermatol. Venereol. 2008, 135, 171–174. [Google Scholar] [CrossRef]
- Ellis, D.A.; Segall, L. Review of non-FDA-approved fillers. Facial Plast. Surg. Clin. North Am. 2007, 15, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Adhirajan, N.; Shanmugasundaram, N.; Shanmuganathan, S.; Babu, M. Collagen-based wound dressing for doxycycline delivery: in-vivo evaluation in an infected excisional wound model in rats. J. Pharm. Pharmacol. 2009, 61, 1617–1623. [Google Scholar] [CrossRef] [PubMed]
- Adhirajan, N.; Shanmugasundaram, N.; Shanmuganathan, S.; Babu, M. Functionally modified gelatin microspheres impregnated collagen scaffold as novel wound dressing to attenuate the proteases and bacterial growth. Eur. J. Pharm. Sci. 2009, 36, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Sripriya, R.; Kumar, M.S.; Ahmed, M.R.; Sehgal, P.K. Collagen bilayer dressing with ciprofloxacin, an effective system for infected wound healing. J. Biomater. Sci. Polym. Ed. 2007, 18, 335–351. [Google Scholar] [CrossRef] [PubMed]
- Shanmugasundaram, N.; Sundaraseelan, J.; Uma, S.; Selvaraj, D.; Babu, M. Design and delivery of silver sulfadiazine from alginate microspheres-impregnated collagen scaffold. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 77, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Liyanage, S.H.; Purohit, G.S.; Frye, J.N.; Giordano, P. Anterior abdominal wall reconstruction with a Permacol implant. J. Plast. Reconstr. Aesthet. Surg. 2006, 59, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Bellows, C.F.; Jian, W.; McHale, M.K.; Cardenas, D.; West, J.L.; Lerner, S.P.; Amiel, G.E. Blood vessel matrix: A new alternative for abdominal wall reconstruction. Hernia 2008, 12, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Ansaloni, L.; Catena, F.; Gagliardi, S.; Gazzotti, F.; D'Alessandro, L.; Pinna, A.D. Hernia repair with porcine small-intestinal submucosa. Hernia 2007, 11, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, F.; Hokaiwado, N.; Honma, K.; Banas, A.; Ochiya, T. Local and systemic delivery of siRNAs for oligonucleotide therapy. Methods Mol. Biol. 2009, 487, 83–92. [Google Scholar] [PubMed]
- Takeshita, F.; Ochiya, T. Therapeutic potential of RNA interference against cancer. Cancer Sci. 2006, 97, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Sano, A.; Maeda, M.; Nagahara, S.; Ochiya, T.; Honma, K.; Itoh, H.; Miyata, T.; Fujioka, K. Atelocollagen for protein and gene delivery. Adv. Drug Deliv. Rev. 2003, 55, 1651–1677. [Google Scholar] [CrossRef] [PubMed]
- Mulder, G.; Tallis, A.J.; Marshall, V.T.; Mozingo, D.; Phillips, L.; Pierce, G.F.; Chandler, L.A.; Sosnowski, B.K. Treatment of nonhealing diabetic foot ulcers with a platelet-derived growth factor gene-activated matrix (GAM501): Results of a phase 1/2 trial. Wound Repair Regen. 2009, 17, 772–779. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863-1887. https://doi.org/10.3390/ma3031863
Parenteau-Bareil R, Gauvin R, Berthod F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials. 2010; 3(3):1863-1887. https://doi.org/10.3390/ma3031863
Chicago/Turabian StyleParenteau-Bareil, Rémi, Robert Gauvin, and François Berthod. 2010. "Collagen-Based Biomaterials for Tissue Engineering Applications" Materials 3, no. 3: 1863-1887. https://doi.org/10.3390/ma3031863




