Parametric Studies of Titania-Supported Gold-Catalyzed Oxidation of Carbon Monoxide
Abstract
:1. Introduction
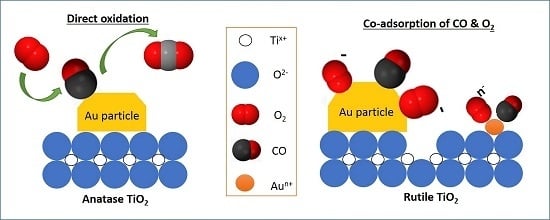
2. Results
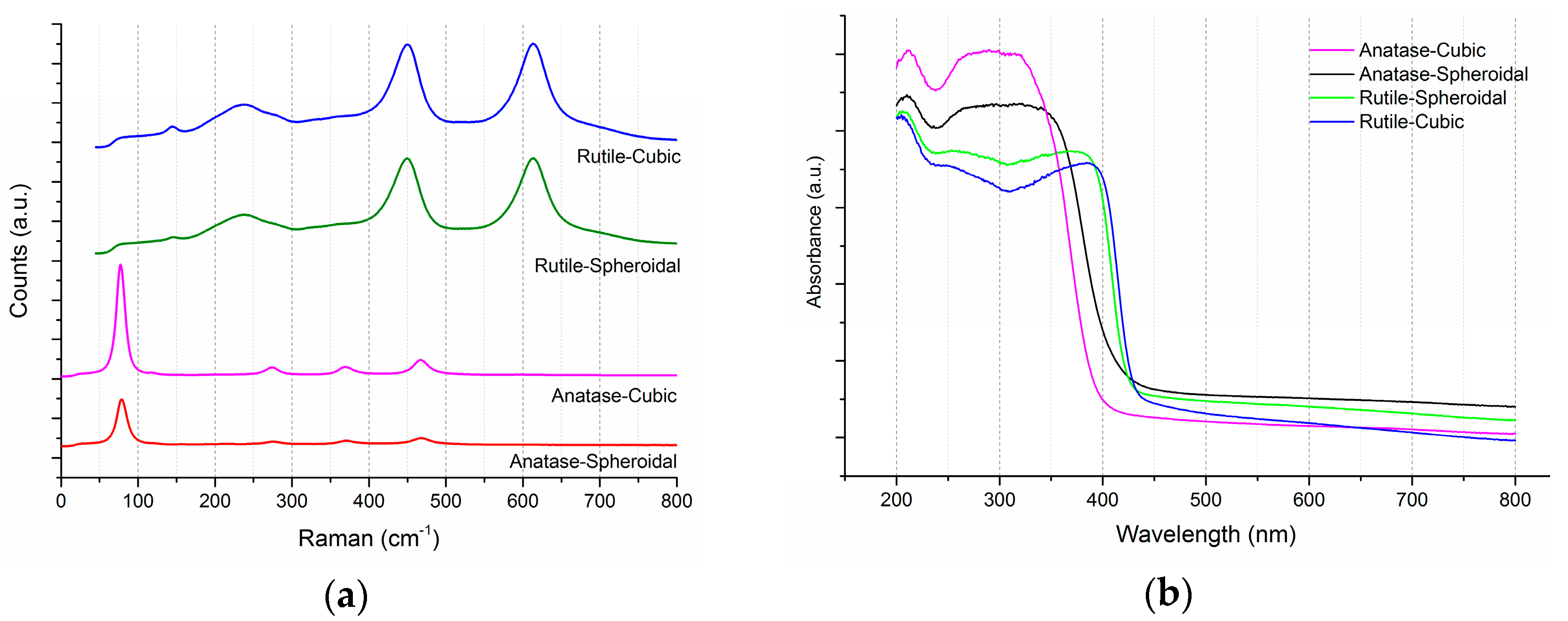
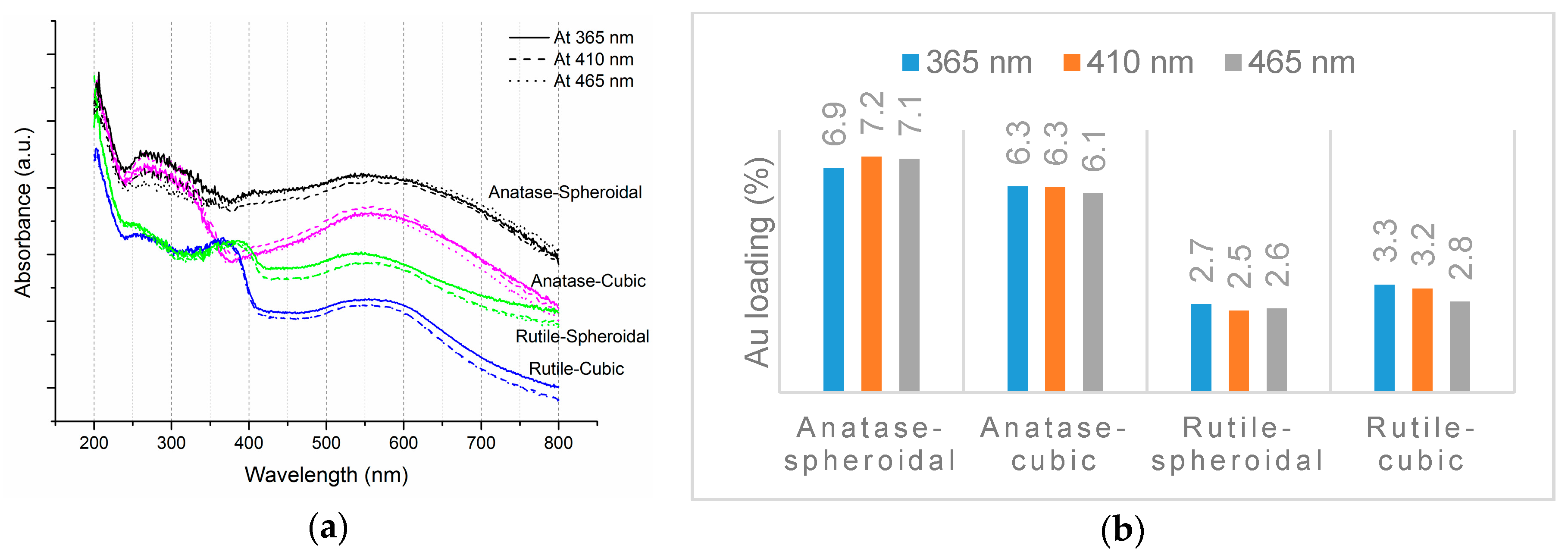
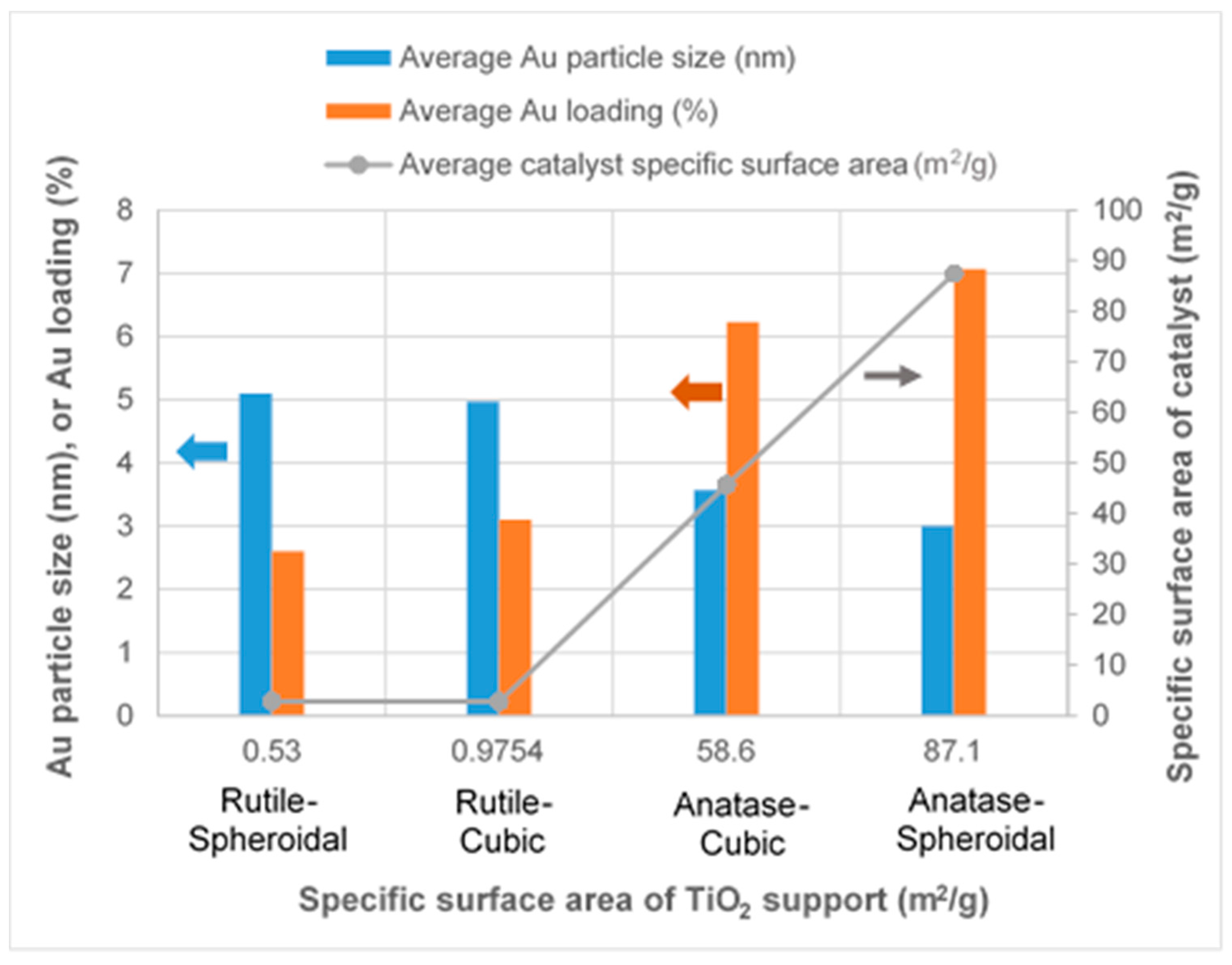
2.1. TiO2 Supports and Gold Deposition
2.2. CO Oxidation
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bone, W.A.; Andrew, G.W. Studies upon Catalytic Combustion. Part I. The Union of Carbon Monoxide and Oxygen in Contact with a Gold Surface. R. Soc. Lond. 1925, 109, 459–476. [Google Scholar] [CrossRef]
- Bond, G.C.; Sermon, P.A. Gold catalysts for olefin hydrogenation. Gold Bull. 1973, 6, 102–105. [Google Scholar] [CrossRef]
- Huber, H.; McIntosh, D.; Ozin, G. A metal atom model for the oxidation of carbon monoxide to carbon dioxide. The gold atom-carbon monoxide-dioxygen reaction and the gold atom-carbon dioxide reaction. Inorg. Chem. 1977, 16, 975–979. [Google Scholar] [CrossRef]
- McIntosh, D.; Ozin, G.A. Direct synthesis using gold atoms. Monodioxygen gold, Au(O2). Inorg. Chem. 1976, 15, 2869–2871. [Google Scholar] [CrossRef]
- Haruta, M.; Yamada, N.; Kobayashi, T.; Iijima, S. Gold catalysts prepared by coprecipitation for low-temperature oxidation of hydrogen and of carbon monoxide. J. Catal. 1989, 115, 301–309. [Google Scholar] [CrossRef]
- Haruta, M.; Tsubota, S.; Kobayashi, T.; Kageyama, H.; Genet, M.J.; Delmon, B. Low-Temperature Oxidation of CO over Gold Supported on TiO2, α-Fe2O3, and Co3O4. J. Catal. 1993, 144, 175–192. [Google Scholar] [CrossRef]
- Mellor, J.; Palazov, A.; Grigorova, B.; Greyling, J.; Reddy, K.; Letsoalo, M.; Marsh, J. The application of supported gold catalysts to automotive pollution abatement. Catal. Today 2002, 72, 145–156. [Google Scholar] [CrossRef]
- Petrov, L.A. Gold Based Catalyst for Exhaust Gas Purification. WO Patent 199851401 A1, 19 November 1998. [Google Scholar]
- Marecot, P.; Emmanuel, R. Rhone Poulenc Chimie. Low Temperature Oxidation of CO. French Patent 2771310 A1, 28 May 1999. [Google Scholar]
- Buelow, M.T.; Chin, S.W.; Hoke, J.B.; Leclerc, N.R.; Robinson, D.M. Catalyst Coatings for Pollution Control. Google Patents WO2015066272 A3, 30 October 2014. [Google Scholar]
- Boccuzzi, F.; Chiorino, A.; Manzoli, M. FTIR study of methanol decomposition on gold catalyst for fuel cells. J. Power Sources 2003, 118, 304–310. [Google Scholar] [CrossRef]
- Tumilty, J.A.J.; Mellor, J.; Grigorova, B. Gold Catalyst for Fuel Cell. Google Patents WO2000013791 A1, 16 March 2000. [Google Scholar]
- Tabata, T.; Terada, T.; Nagata, T.; Kataoka, M.; Takahashi, H.; Mizutani, N.; Horiuchi, Y. Fuel Cell and Supported Catalyst Used Therefor. Google Patents US 20100196802 A1, 5 August 2010. [Google Scholar]
- Hussain, A. A Computational Study of Catalysis by Gold in Applications of CO Oxidation. Ph.D. Thesis, Technische Universiteit Eindhoven, Eindhoven, The Netherlands, 2010. [Google Scholar]
- Chen, S.; Zhang, B.; Su, D.; Huang, W. Titania Morphology-Dependent Gold—Titania Interaction, Structure, and Catalytic Performance of Gold/Titania Catalysts. ChemCatChem 2015, 7, 3290–3298. [Google Scholar] [CrossRef]
- Cao, L.; Chen, D.; Li, W.; Caruso, R.A. Hierarchically Porous Titania Networks with Tunable Anatase: Rutile Ratios and Their Enhanced Photocatalytic Activities. ACS Appl. Mater. Interfaces 2014, 6, 13129–13137. [Google Scholar] [CrossRef] [PubMed]
- Chong, S.; Yang, T.C.-K. Illumination wavelength and time dependent nano gold photo-deposition and CO oxidation. Results Phys. 2017, 7, 1167–1174. [Google Scholar] [CrossRef]
- Stevanovic, A.; Ma, S.; Yates, J.T., Jr. Effect of Gold Nanoparticles on Photoexcited Charge Carriers in Powdered TiO2—Long Range Quenching of Photoluminescence. J. Phys. Chem. C 2014, 118, 21275–21280. [Google Scholar] [CrossRef]
- Landmann, M.; Rauls, E.; Schmidt, W. The electronic structure and optical response of rutile, anatase and brookite TiO2. J. Phys. Condens. Matter 2012, 24, 195503. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.Y.; Yun, H.J.; Yu, S.; Kim, W.; Kim, N.D.; Yi, J. Effect of TiO2 crystalline phase on CO oxidation over CuO catalysts supported on TiO2. J. Mol. Catal. A Chem. 2013, 368, 72–77. [Google Scholar] [CrossRef]
- Saavedra, J.; Powell, C.; Panthi, B.; Pursell, C.J.; Chandler, B.D. CO oxidation over Au/TiO2 catalyst: pretreatment effects, catalyst deactivation, and carbonates production. J. Catal. 2013, 307, 37–47. [Google Scholar] [CrossRef]
- Valden, M.; Lai, X.; Goodman, D.W. Onset of catalytic activity of gold clusters on titania with the appearance of nonmetallic properties. Science 1998, 281, 1647–1650. [Google Scholar] [CrossRef] [PubMed]
- Mavrikakis, M.; Stoltze, P.; Nørskov, J.K. Making gold less noble. Catal. Lett. 2000, 64, 101–106. [Google Scholar] [CrossRef]
- Overbury, S.; Schwartz, V.; Mullins, D.R.; Yan, W.; Dai, S. Evaluation of the Au size effect: CO oxidation catalyzed by Au/TiO2. J. Catal. 2006, 241, 56–65. [Google Scholar] [CrossRef]
- Lopez, N.; Nørskov, J.K. Catalytic CO oxidation by a gold nanoparticle: A density functional study. J. Am. Chem. Soc. 2002, 124, 11262–11263. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-P.; Hu, P.; Alavi, A. Catalytic role of gold in gold-based catalysts: A density functional theory study on the CO oxidation on gold. J. Am. Chem. Soc. 2002, 124, 14770–14779. [Google Scholar] [CrossRef] [PubMed]
- Lopez, N.; Janssens, T.V.W.; Clausen, B.S.; Xu, Y.; Mavrikakis, M.; Bligaard, T.; Nørskov, J.K. On the origin of the catalytic activity of gold nanoparticles for low-temperature CO oxidation. J. Catal. 2004, 223, 232–235. [Google Scholar] [CrossRef]
- Cho, A. Connecting the dots to custom catalysts. Science 2003, 299, 1684–1685. [Google Scholar] [CrossRef] [PubMed]
- Haruta, M. Size- and support-dependency in the catalysis of gold. Catal. Today 1997, 36, 153–166. [Google Scholar] [CrossRef]
- Park, E.D.; Lee, J.S. Effects of Pretreatment Conditions on CO Oxidation over Supported Au Catalysts. J. Catal. 1999, 186, 1–11. [Google Scholar] [CrossRef]
- Wang, G.-H.; Li, W.-C.; Jia, K.-M.; Spliethoff, B.; Schüth, F.; Lu, A.-H. Shape and size controlled α-Fe2O3 nanoparticles as supports for gold-catalysts: Synthesis and influence of support shape and size on catalytic performance. Appl. Catal. A 2009, 364, 42–47. [Google Scholar] [CrossRef]
- Li, S.; Zhu, H.; Qin, Z.; Wang, G.; Zhang, Y.; Wu, Z.; Li, Z.; Chen, G.; Dong, W.; Wu, Z.; et al. Morphologic effects of nano CeO2–TiO2 on the performance of Au/CeO2–TiO2 catalysts in low-temperature CO oxidation. Appl. Catal. B 2014, 144, 498–506. [Google Scholar] [CrossRef]
- Jovic, V.; Chen, W.-T.; Sun-Waterhouse, D.; Blackford, M.G.; Idriss, H.; Waterhouse, G.I. Effect of gold loading and TiO2 support composition on the activity of Au/TiO2 photocatalysts for H2 production from ethanol–water mixtures. J. Catal. 2013, 305, 307–317. [Google Scholar] [CrossRef]
- Yang, K.; Liu, J.; Si, R.; Chen, X.; Dai, W.; Fu, X. Comparative study of Au/TiO2 and Au/Al2O3 for oxidizing CO in the presence of H2 under visible light irradiation. J. Catal. 2014, 317, 229–239. [Google Scholar] [CrossRef]
- Ramirez-Garza, R.E.; Pawelec, B.; Zepeda, T.A.; Martinez-Hernandez, A. Total CO oxidation over Fe-containing Au/HMS catalysts: Effects of gold loading and catalyst pretreatment. Catal. Today 2011, 172, 95–102. [Google Scholar] [CrossRef]
- Zanella, R.; Giorgio, S.; Shin, C.-H.; Henry, C.R.; Louis, C. Characterization and reactivity in CO oxidation of gold nanoparticles supported on TiO2 prepared by deposition-precipitation with NaOH and urea. J. Catal. 2004, 222, 357–367. [Google Scholar] [CrossRef]
- Ho, K.; Yeung, K. Properties of TiO2 support and the performance of Au/TiO2 catalyst for CO oxidation reaction. Gold Bull. 2007, 40, 15–30. [Google Scholar] [CrossRef]
- Bond, G.C.; Thompson, D.T. Gold-catalysed oxidation of carbon monoxide. Gold Bull. 2000, 33, 41–50. [Google Scholar] [CrossRef]
- Falicov, L.; Somorjai, G. Correlation between catalytic activity and bonding and coordination number of atoms and molecules on transition metal surfaces: Theory and experimental evidence. Proc. Natl. Acad. Sci. USA 1985, 82, 2207–2211. [Google Scholar] [CrossRef] [PubMed]
- Bond, G.C.; Thompson, D.T. Catalysis by Gold. Catal. Rev. Sci. Eng. 1999, 41, 319–388. [Google Scholar] [CrossRef]
- Hodge, N.A.; Kiely, C.J.; Whyman, R.; Siddiqui, M.R.H.; Hutchings, G.J.; Pankhurst, Q.A.; Wagner, F.E.; Rajaram, R.R.; Golunski, S.E. Microstructural comparison of calcined and uncalcined gold/iron-oxide catalysts for low-temperature CO oxidation. Catal. Today 2002, 72, 133–144. [Google Scholar] [CrossRef]
- Guo, L.-W.; Du, P.-P.; Fu, X.-P.; Ma, C.; Zeng, J.; Si, R.; Huang, Y.-Y.; Jia, C.-J.; Zhang, Y.-W.; Yan, C.-H. Contributions of distinct gold species to catalytic reactivity for carbon monoxide oxidation. Nat. Commun. 2016, 7, 13481. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, M.; Lippits, M.; Nieuwenhuys, B. CO adsorption and oxidation on Au/TiO2. Catal. Lett. 1998, 56, 195–197. [Google Scholar] [CrossRef]
- Boccuzzi, F.; Chiorino, A.; Manzoli, M.; Lu, P.; Akita, T.; Ichikawa, S.; Haruta, M. Au/TiO2 nanosized samples: A catalytic, TEM, and FTIR study of the effect of calcination temperature on the CO oxidation. J. Catal. 2001, 202, 256–267. [Google Scholar] [CrossRef]
- Srinivas, G.; Wright, J.; Bai, C.S.; Cook, R. Au/metal oxides for low temperature CO oxidation. Stud. Surf. Sci. Catal. 1996, 101, 427–433. [Google Scholar]
- Green, I.X.; Tang, W.; Neurock, M.; Yates, J.T. Spectroscopic observation of dual catalytic sites during oxidation of CO on a Au/TiO2 catalyst. Science 2011, 333, 736–739. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bell, A.T. The mechanism of dimethyl carbonate synthesis on Cu-exchanged zeolite Y. J. Catal. 2008, 255, 153–161. [Google Scholar] [CrossRef]
- Boccuzzi, F.; Chiorino, A.; Tsubota, S.; Haruta, M. FTIR study of carbon monoxide oxidation and scrambling at room temperature over gold supported on ZnO and TiO2. 2. J. Phys. Chem. 1996, 100, 3625–3631. [Google Scholar] [CrossRef]
- Raphulu, M.; McPherson, J.; Van der Lingen, E.; Anderson, J.; Scurrell, M. Investigation of the active site and the mode of Au/TiO2 catalyst deactivation using Diffuse Reflectance Infrared Fourier Transform Spectroscopy (DRIFTS). Gold Bull. 2010, 43, 21–28. [Google Scholar] [CrossRef]
- Bollinger, M.A.; Vannice, M.A. A kinetic and DRIFTS study of low-temperature carbon monoxide oxidation over Au-TiO2 catalysts. Appl. Catal. B Environ. 1996, 8, 417–443. [Google Scholar] [CrossRef]


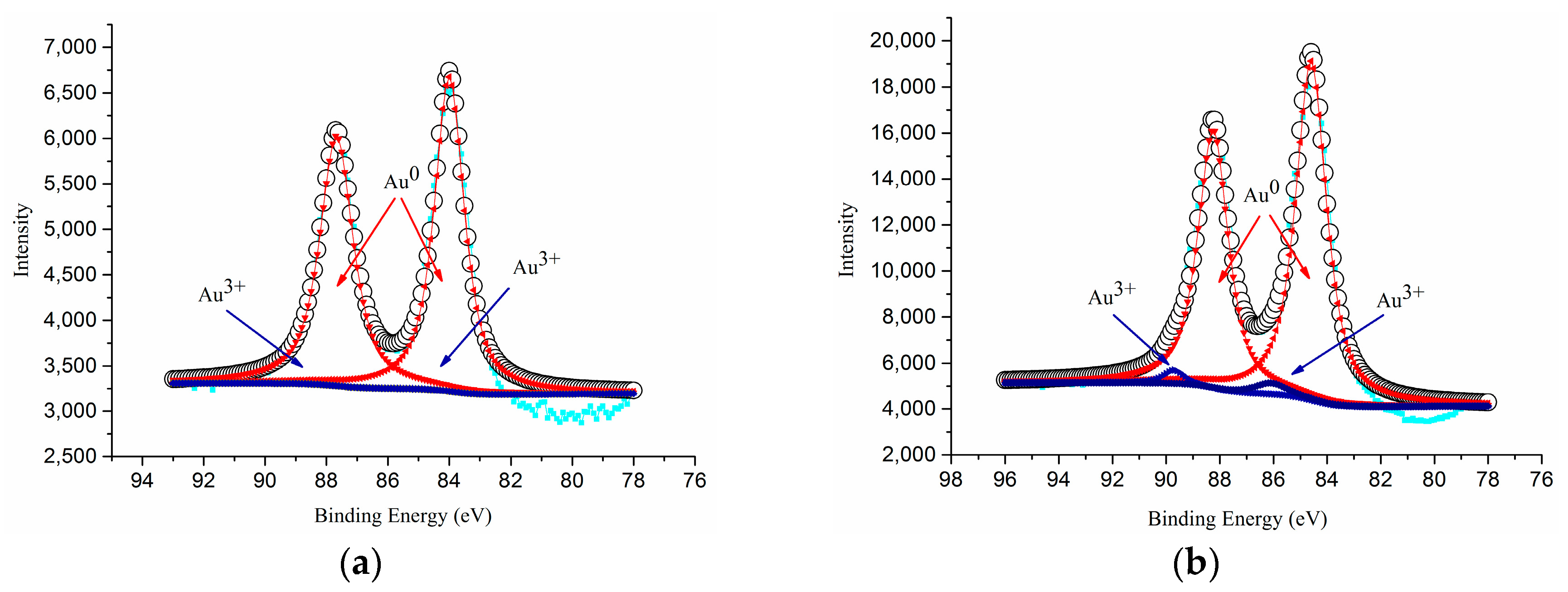
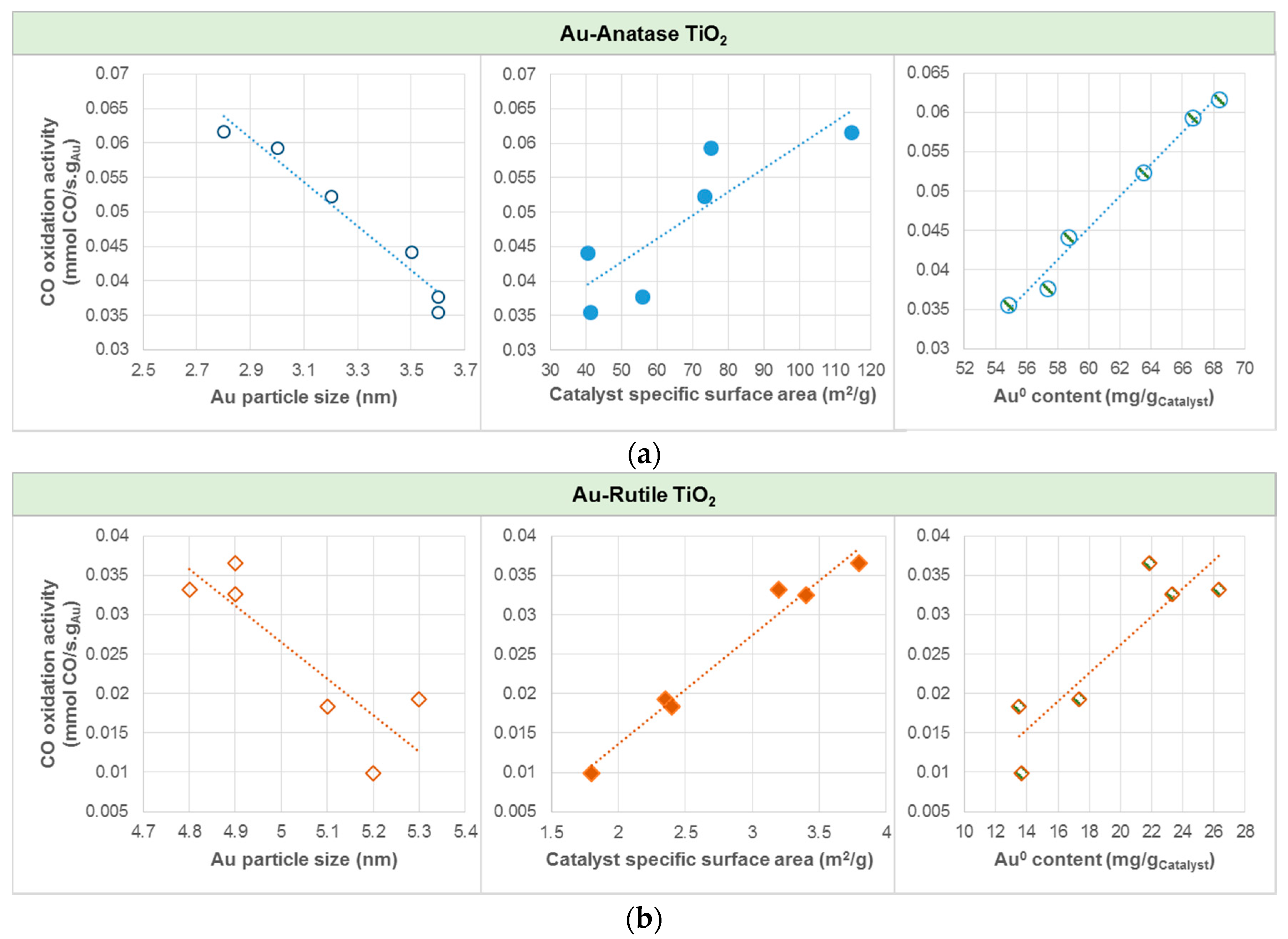
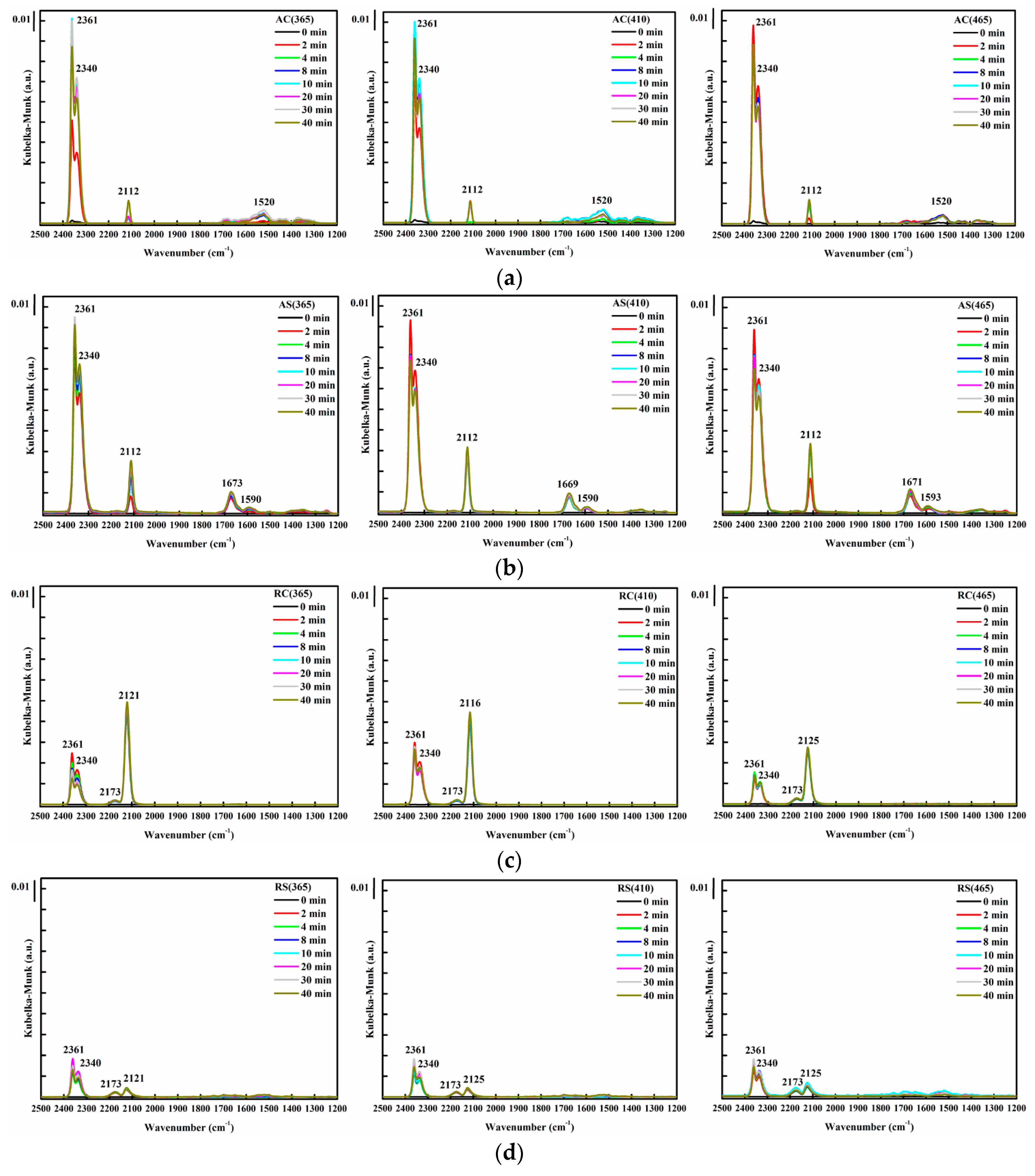
| TiO2 Support | Sample Notation | Au Loading (%) | Au Particle Size (nm) | Au0 (Atomic %) | Au3+ (Atomic %) | Au3+/Au0 Ratio | Specific Surface Area (m2/g) | Conversion (%) | CO Oxidation Activity (mmol CO/s.gAu) × 10−3 |
|---|---|---|---|---|---|---|---|---|---|
| Anatase–Spheroidal | - | - | - | - | - | - | 87.1 | - | - |
| AS(365) | 6.9 | 3.2 | 92 | 8 | 0.087 | 73.0 | 72.8 | 52.3 | |
| AS(410) | 7.2 | 2.8 | 95 | 5 | 0.052 | 114.4 | 89.5 | 61.7 | |
| AS(465) | 7.1 | 3.0 | 94 | 6 | 0.064 | 74.9 | 84.9 | 59.3 | |
| Anatase–Cubic | - | - | - | - | - | - | 58.6 | - | - |
| AC(365) | 6.3 | 3.5 | 93 | 7 | 0.075 | 40.3 | 56.1 | 44.2 | |
| AC(410) | 6.3 | 3.6 | 91 | 9 | 0.099 | 55.7 | 47.9 | 37.7 | |
| AC(465) | 6.1 | 3.6 | 90 | 10 | 0.111 | 41.0 | 43.7 | 35.5 | |
| Rutile–Spheroidal | - | - | - | - | - | - | 0.53 | - | - |
| RS(365) | 2.7 | 4.9 | 81 | 19 | 0.235 | 3.8 | 19.9 | 36.6 | |
| RS(410) | 2.5 | 5.3 | 69 | 31 | 0.449 | 2.4 | 9.7 | 19.2 | |
| RS(465) | 2.6 | 5.1 | 52 | 48 | 0.923 | 2.4 | 9.6 | 18.3 | |
| Rutile–Cubic | - | - | - | - | - | - | 0.9754 | - | - |
| RC(365) | 3.3 | 4.8 | 80 | 20 | 0.250 | 3.2 | 22.1 | 33.2 | |
| RC(410) | 3.2 | 4.9 | 73 | 27 | 0.370 | 3.4 | 21.0 | 32.6 | |
| RC(465) | 2.8 | 5.2 | 49 | 51 | 1.041 | 1.8 | 5.6 | 9.9 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chong, S.; Yang, T.C.-K. Parametric Studies of Titania-Supported Gold-Catalyzed Oxidation of Carbon Monoxide. Materials 2017, 10, 756. https://doi.org/10.3390/ma10070756
Chong S, Yang TC-K. Parametric Studies of Titania-Supported Gold-Catalyzed Oxidation of Carbon Monoxide. Materials. 2017; 10(7):756. https://doi.org/10.3390/ma10070756
Chicago/Turabian StyleChong, Siewhui, and Thomas Chung-Kuang Yang. 2017. "Parametric Studies of Titania-Supported Gold-Catalyzed Oxidation of Carbon Monoxide" Materials 10, no. 7: 756. https://doi.org/10.3390/ma10070756