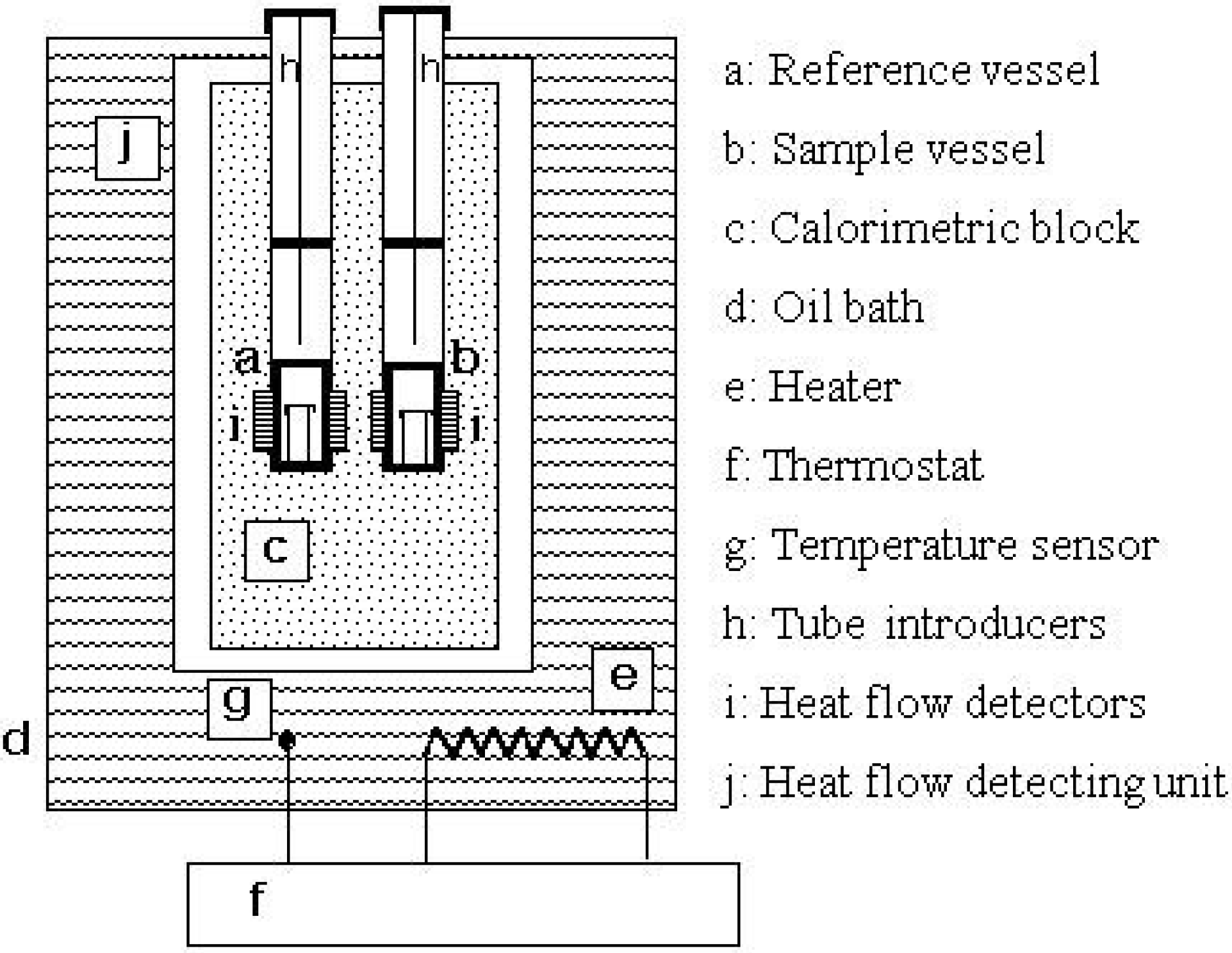
3. Results and Discussion
3.1. Calorimetry
Figure 4,
Figure 5,
Figure 6,
Figure 7,
Figure 8 and
Figure 9 show the C80 calorimetry results of the various fuel samples prepared without and with the addition of distilled water (equal to 20% of the sample). Distilled water was added to the samples in order to examine the effect of moisture content on heat generation.
Table 1 summarises the exothermic onset temperatures of each sample. The exothermic onset temperature was taken as the temperature at which the rate of heat generation increased to 0.005 mW.
Table 1.
Heat-generation onset temperatures of various biomass fuels, as determined by C80 calorimetry.
Table 1.
Heat-generation onset temperatures of various biomass fuels, as determined by C80 calorimetry.
| Sample | Heat generation onset temperature (°C) |
|---|
| Wood pellets (made from tree bark) | 59.3 |
| Wood pellets (made from tree bark) + distilled water 20% | 48.3 |
| Wood pellets (made from tree trunks) | 64.3 |
| Wood pellets (made from tree trunks) + distilled water 20% | 56.2 |
| Sludge fuel | 42.1 |
| Sludge fuel + distilled water 20% | 26.3 |
| Chicken dung | 28.5 |
| Chicken dung + distilled water 20% | 26.1 |
| Palm shell fuel | 90.7 |
| Palm shell fuel + distilled water 20% | 26.3 |
| Rice straw pellets | 52.3 |
| Rice straw pellets + distilled water 20% | 48.5 |
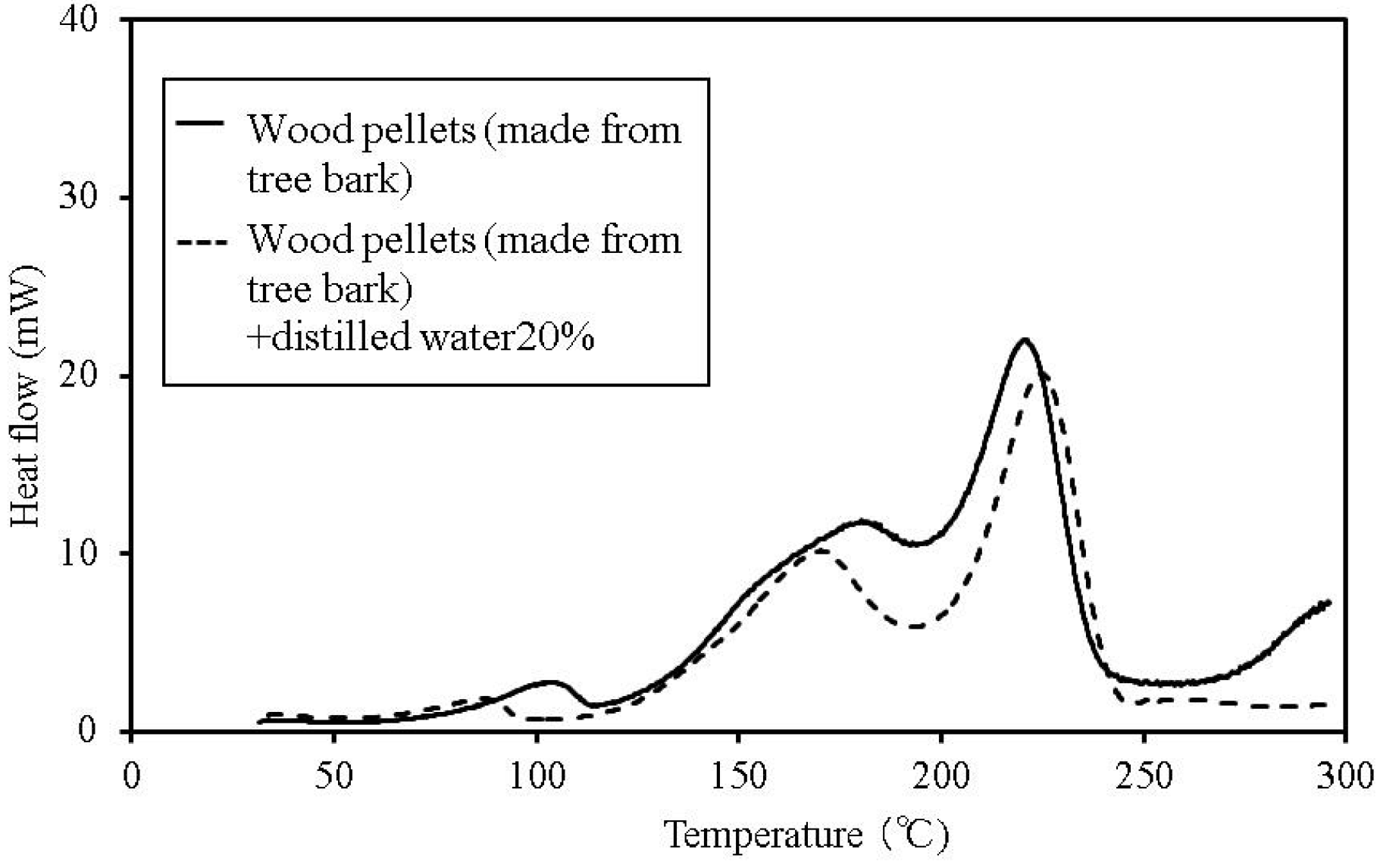
Figure 4.
C80 calorimetry curves for wood pellets (made from tree bark).
Figure 4.
C80 calorimetry curves for wood pellets (made from tree bark).
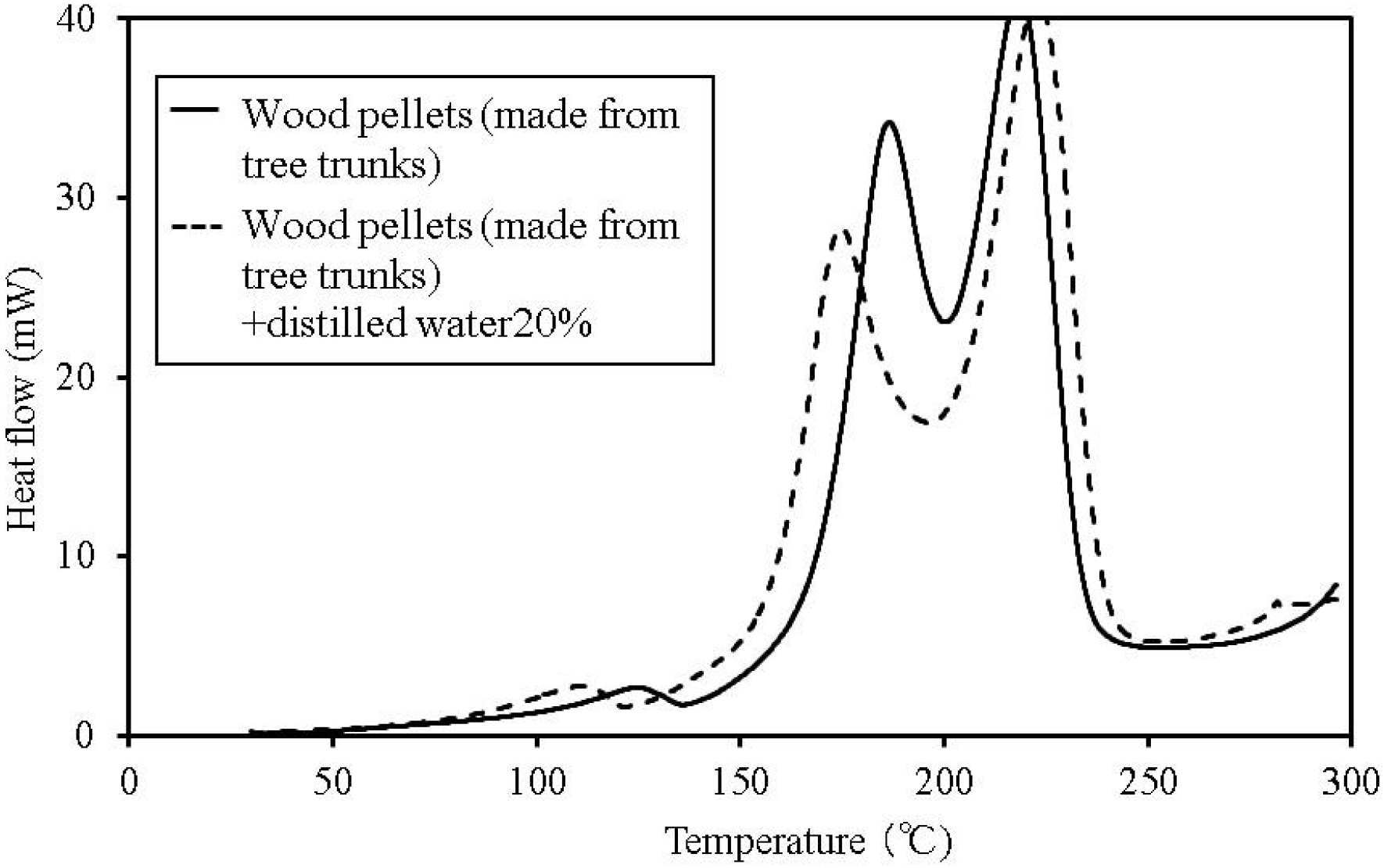
Figure 5.
C80 calorimetry curves for wood pellets (made from tree trunks).
Figure 5.
C80 calorimetry curves for wood pellets (made from tree trunks).
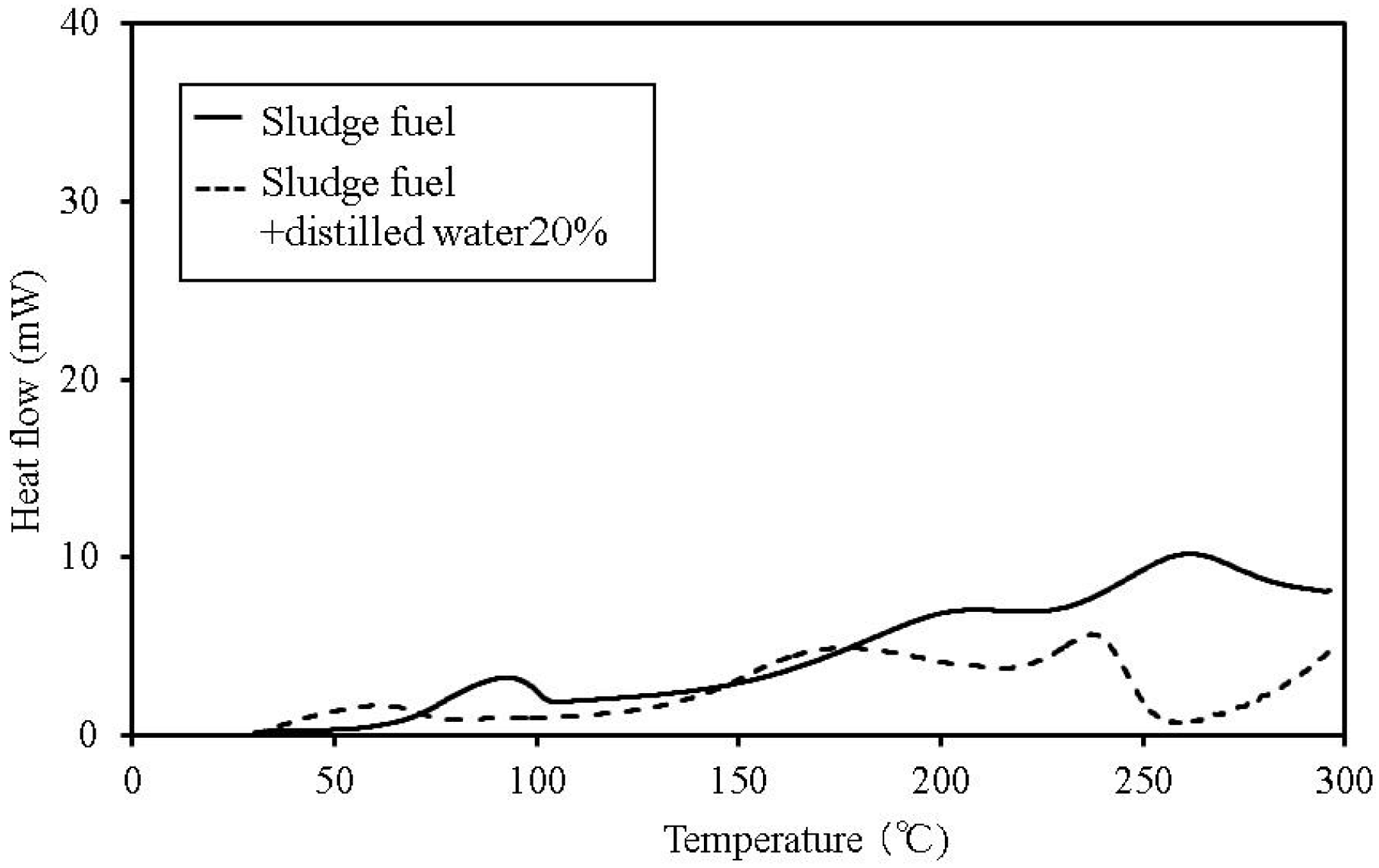
Figure 6.
C80 calorimetry curves for sludge fuel.
Figure 6.
C80 calorimetry curves for sludge fuel.
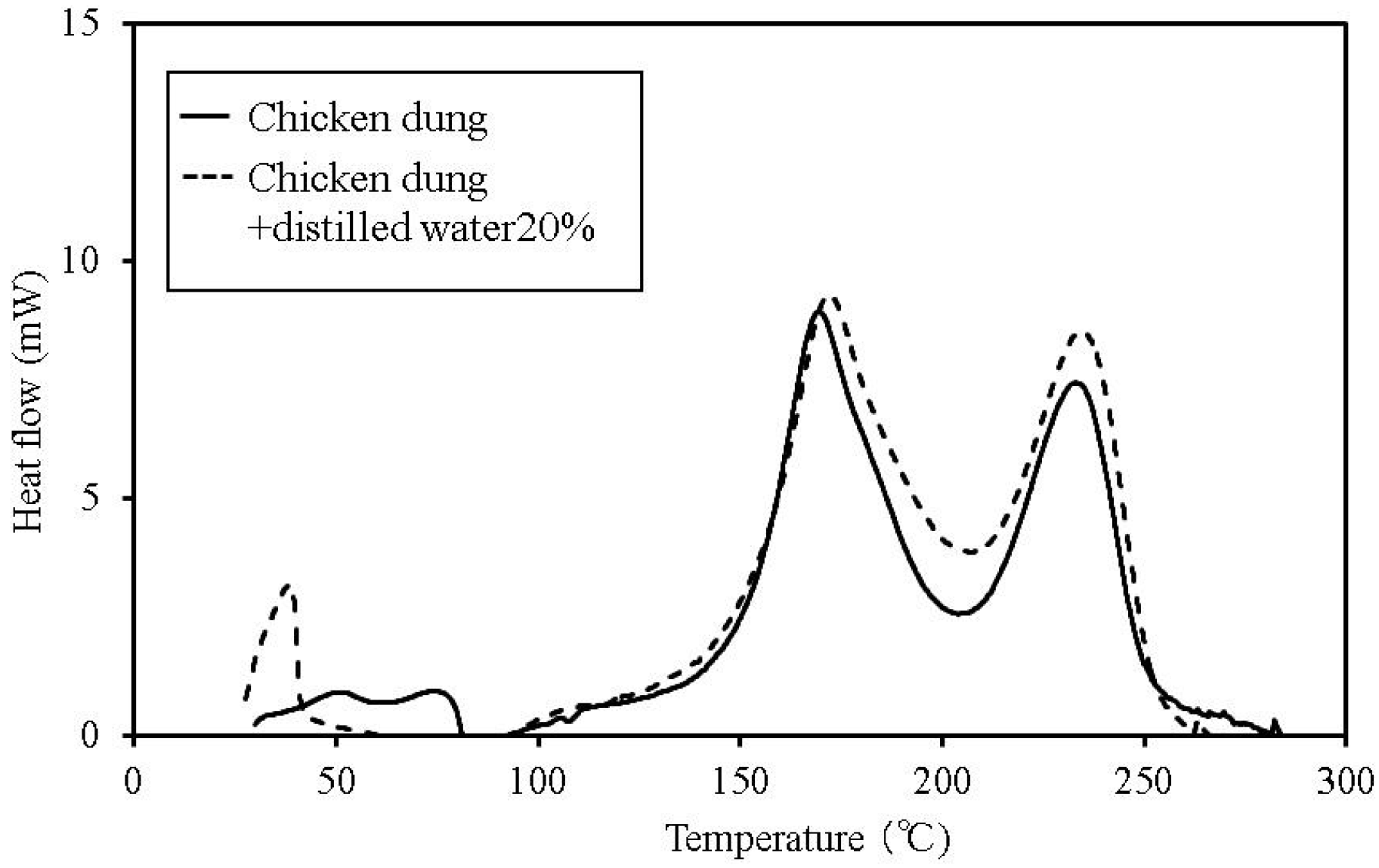
Figure 7.
C80 calorimetry curves for chicken dung.
Figure 7.
C80 calorimetry curves for chicken dung.
Figure 8.
C80 calorimetry curves for palm shell fuel.
Figure 8.
C80 calorimetry curves for palm shell fuel.
Figure 9.
C80 calorimetry curves for rice straw pellets.
Figure 9.
C80 calorimetry curves for rice straw pellets.
The temperature at which faint heat generation begins is lower in all of the samples with distilled water, compared to their counterparts without the added water. Heat generation over a temperature range of 80–100 °C is expected to be caused by the fatty acid esters contained in all the samples [
13]. For temperatures above this range, the heat generation is expected to be caused by decomposition [
14,
15]. The start of faint heat generation after adding moisture is expected to be caused by fermentation, and is observed in the sludge fuel and chicken dung almost immediately, even at temperatures of 30 °C or less.
The process of heat generation leading to spontaneous ignition of these items begins with a small amount of heat generated from fermentation as a result of microbial activity. Next, the fatty acid esters contained within the items begin to oxidise as the temperature gradually rises. Even if the microorganisms die, the temperature continues to rise and ultimately results in spontaneous ignition.
Based on the C80 calorimetry results, it is desirable to incorporate the following safety measures. First, if the internal temperature of the biomass fuel is between 30–50 °C, fermentation begins. Only faint heat is generated from fermentation and breaking down the pile. Conducting a heat dissipation treatment at this stage lowers the possibility of a further increase in temperature, which could lead to spontaneous ignition.
Second, if the internal temperature of the biomass fuel is between 50–80 °C, fermentation, continues and oxidation of the fatty acid esters produced by fermentation, occurs. Self-heating is already in progress at this stage and immediate evasive action is required. At this point, breaking down the pile increases the oxygen supply to the area of self-heating, rapidly increasing the temperature and the possibility of combustion. The amount of heat generated by these reactions is larger than that caused by fermentation. Venting inactive gas as needed is the recommended action. If water is sprayed onto the biomass fuel with the purpose of reducing its temperature, particular attention must be paid to preventing subsequent fermentation.
3.2. Highly Sensitive Isothermal Calorimeter
The TAM-III calorimeter can measure the change in activity of microorganisms over time.
Figure 10,
Figure 11,
Figure 12,
Figure 13,
Figure 14 and
Figure 15 show the TAM-III calorimetry curves of the various fuel samples prepared without and with the addition of distilled water (equal to 20% (w/w) of the sample), and
Table 2 summarizes the amount of heat generated. Heat generation has been considered for two periods: 0–24 h, and 24–72 h. In addition, heat generation was considered for a combination of both time periods. The TAM-III temperature was held at 50 °C for all tests, because microbial activity occurs most vigorously in this temperature range but becomes inactive at higher temperatures [
16,
17].
Table 2.
Heat generation of various biomass fuels, as determined by TAM calorimetry at 50 °C (untreated samples).
Table 2.
Heat generation of various biomass fuels, as determined by TAM calorimetry at 50 °C (untreated samples).
| Sample | Heat generation (J/g) 0–24 h | Heat generation (J/g) 24–72 h | Heat generation (J/g) 0–72 h |
|---|
| Wood pellets (made from tree bark) | 1.27 | 1.59 | 2.85 |
| Wood pellets (made from tree bark) + distilled water 20% | 7.95 | 10.36 | 18.31 |
| Wood pellets (made from tree trunks) | 1.74 | 1.96 | 3.71 |
| Wood pellets (made from tree trunks) + distilled water 20% | 10.11 | 2.86 | 12.97 |
| Sludge fuel | 17.74 | 11.14 * | 28.88 ** |
| Sludge fuel + distilled water 20% | 35.11 | 8.52 | 43.63 |
| Chicken dung | 3.67 | 5.68 | 9.35 |
| Chicken dung + distilled water 20% | 80.38 | 96.82 * | 177.2 ** |
| Palm shell fuel | 1.06 | 1.00 | 2.07 |
| Palm shell fuel + distilled water 20% | 8.96 | 6.14 | 15.10 |
| Rice straw pellets | 1.53 | 2.08 | 3.61 |
| Rice straw pellets + distilled water 20% | 8.24 | 6.58 | 14.82 |
Figure 10.
TAM calorimetry curves for wood pellets (made from tree bark).
Figure 10.
TAM calorimetry curves for wood pellets (made from tree bark).
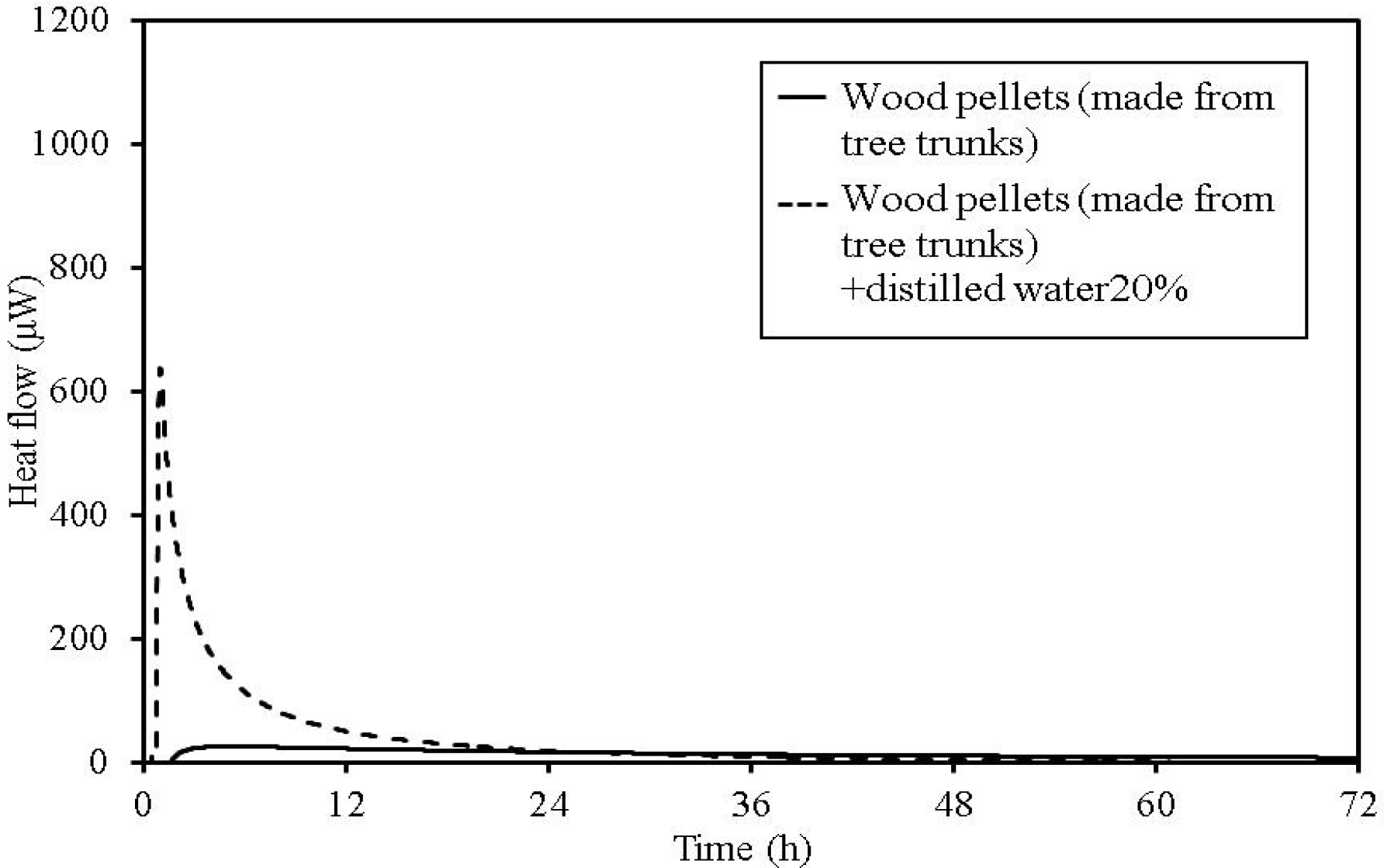
Figure 11.
TAM calorimetry curves for wood pellets (made from tree trunks).
Figure 11.
TAM calorimetry curves for wood pellets (made from tree trunks).
Figure 12.
TAM calorimetry curves for sludge fuel.
Figure 12.
TAM calorimetry curves for sludge fuel.
Figure 13.
TAM calorimetry curves for chicken dung.
Figure 13.
TAM calorimetry curves for chicken dung.
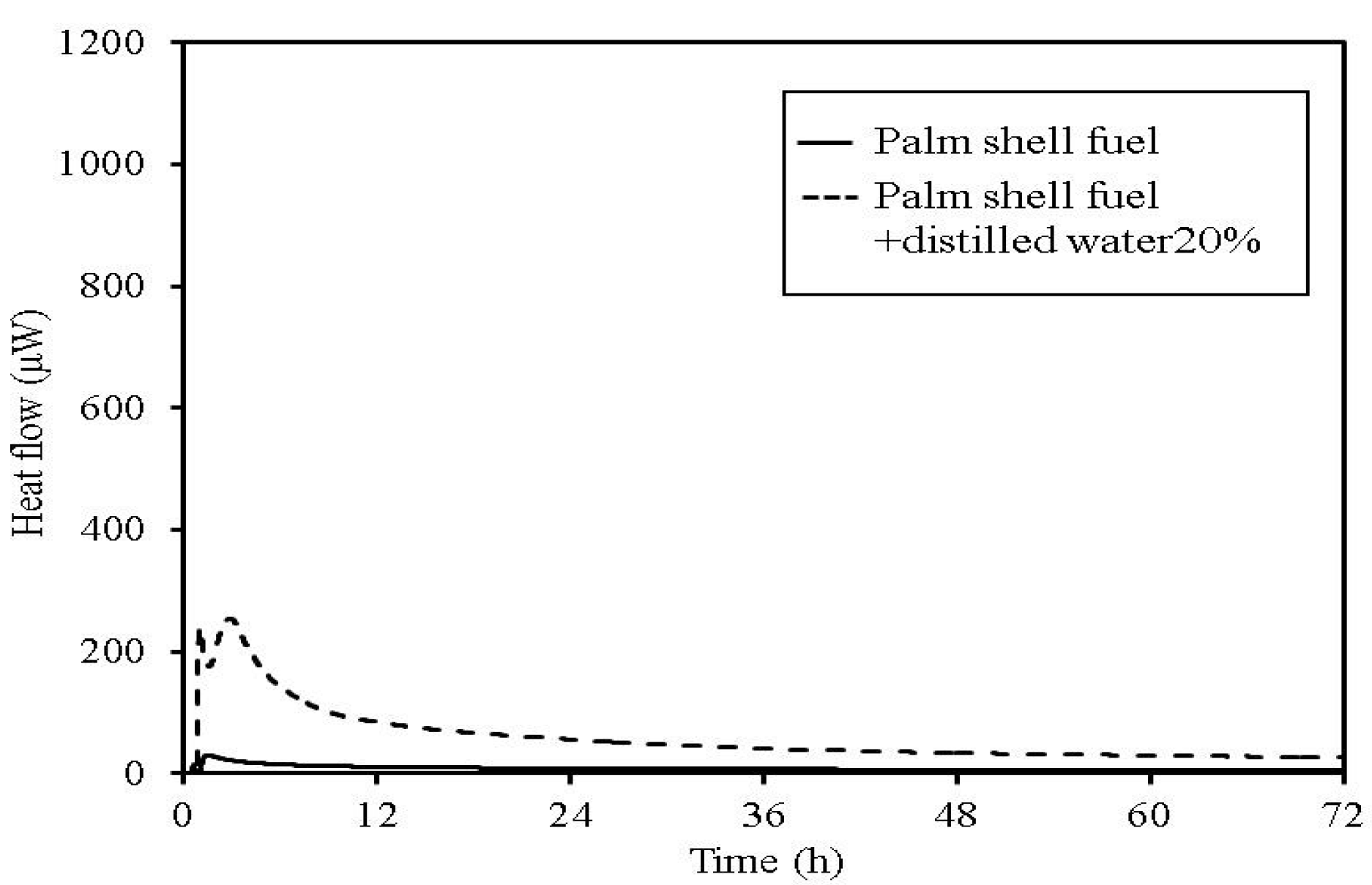
Figure 14.
TAM calorimetry curves for palm shell fuel.
Figure 14.
TAM calorimetry curves for palm shell fuel.
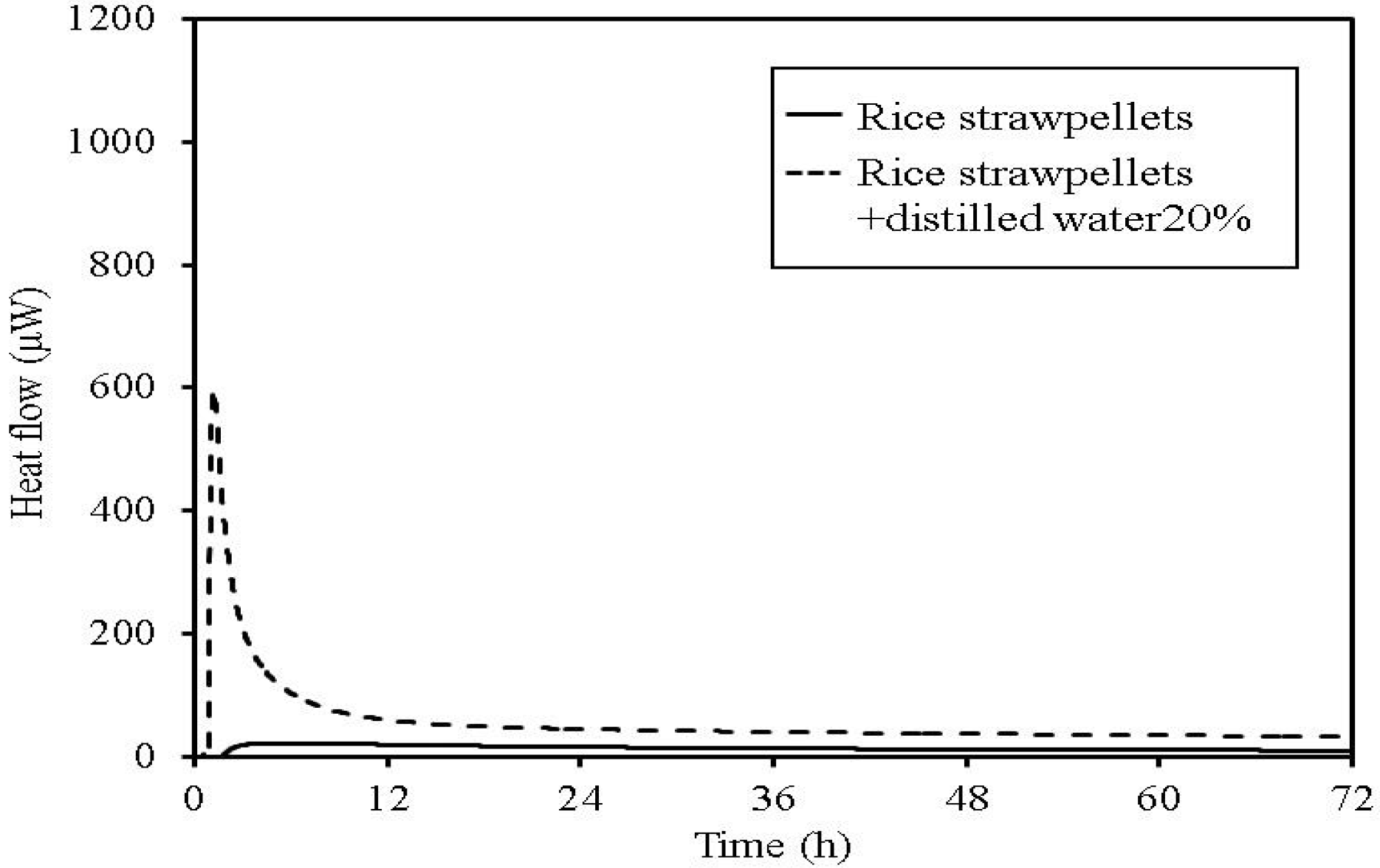
Figure 15.
TAM calorimetry curves for rice straw pellets.
Figure 15.
TAM calorimetry curves for rice straw pellets.
Considering the thermal behaviour of wood pellets (made from tree bark), once distilled water is added, heat generation begins immediately and increases again after 24 h. This is assumed to occur because microorganism activity increases over a period of time. Once oxygen in the sealed sample container is consumed by rapid fermentation, the reaction becomes inactive.
In wood pellets (made from tree trunks), similar to those made from tree bark, an increase in heat generation is observed when distilled water is added. However, the two-phase heat generation that occurs for the wood pellets made from bark, is not observed.
Furthermore, in sludge fuel, chicken dung and palm shell fuel, an increase in heat generation associated with the addition of distilled water is observed. The heat generated by chicken dung increases by approximately 19 times when distilled water is added. Sludge fuel and chicken dung are made from faecal matter, and are more likely to contain a large number of microorganisms, which are assumed to become active with the addition of distilled water.
3.3. Gas Chromatography
In regards to the risks related to the storage of biomass fuels, confirmation of the release of flammable gases such as H
2, CH
4, and CO is important. There have been cases in which waste in a waste disposer has fermented, filling the environment with flammable gases and causing an explosion [
18]. Additionally, there have been cases in which active fermentation has occurred in a storage location, releasing a large amount of CO
2, which resulted in the death of workers due to asphyxiation [
19].
The GC analysis results of the various fuel samples are shown in
Table 3. There is an increase in the amount of CO
2 generated in all samples when distilled water is added. H
2 generation is also observed, likely due to the high possibility of anaerobic fermentation, in addition to aerobic fermentation [
20]. CO and CH
4, however, are not detected according to the measurement conditions at the time.
Table 3.
GC results for untreated samples of various biomass fuels (25 °C).
Table 3.
GC results for untreated samples of various biomass fuels (25 °C).
| Sample | Storage period | Storage temperature | GC results % |
|---|
| O2 | N2 | H2 | CO | CH4 | CO2 |
|---|
| Wood pellets (made from tree bark) | 10 days | 25 °C | 19.55 | 76.97 | 0.0014 | - | - | 0.70 |
| Wood pellets (made from tree bark) + distilled water 20% | 1.37 | 68.87 | 0.014 | - | - | 21.67 |
| Wood pellets (made from tree trunks) | 20.67 | 77.23 | 0.002 | - | - | 0.08 |
| Wood pellets (made from tree trunks) + distilled water 20% | 17.88 | 78.53 | 0.0047 | - | - | 1.94 |
| Sludge fuel | 19.47 | 77.32 | - | - | - | 0.49 |
| Sludge fuel + distilled water 20% | 17.24 | 76.22 | - | - | - | 2.29 |
| Chicken dung | 9.53 | 70.12 | - | - | - | 14.20 |
| Chicken dung + distilled water 20% | 1.48 | 73.76 | 0.27 | - | - | 21.99 |
| Palm shell fuel | 20.46 | 76.13 | - | - | - | 0.06 |
| Palm shell fuel + distilled water 20% | 19,86 | 78.32 | - | - | - | 3.07 |
| Rice straw pellets | 20.57 | 76.39 | - | - | - | 0.04 |
| Rice straw pellets + distilled water 20% | 2.17 | 75.69 | 0.0031 | - | - | 16.93 |
In wood pellets (made from tree bark), a large increase in CO2 is observed with the addition of moisture. In chicken dung, high generation of CO2 is observed irrespective of the addition of distilled water. Considering its thermal behaviour, chicken dung is assumed to contain more moisture than the wood pellets, and therefore believed to be more conducive to fermentation. Other samples may also contain microorganisms, but these samples do not have enough moisture for microorganisms to become active, so fermentation is assumed difficult under normal conditions. However, when the amount of contained moisture is increased by some factor, fermentation is assumed to occur, leading to a temperature increase and spontaneous ignition.
Under normal conditions, even if flammable gas and CO2 are released, they are diffused in the air and their concentration decreases, so the risk from these gases is lower. However, when biomass fuel that is likely to ferment is transported in a tightly sealed container, or stored for a long period in a silo, caution is required to avoid ignition and explosion. Measuring the generated gas using a gas detection tube is recommended, in addition to temperature monitoring and regular confirmation of whether fermentation is occurring or flammable gases are being generated.
5. Risk Ranking of Biomass Fuels
A number of methods exist that can be used to estimate the risks involved with large piles of biomass fuels, based on experimental results. For example, the formulae established by Frank-Kamenetskii [
22] and Semenov [
23] can be used to estimate the critical ignition temperature, and therefore the risk of natural ignition presented by piled materials. Although such methods allow for the assumption of a critical ignition temperature relative to the height of the pile, it is not possible to clarify which of the articles contained in the pile present an increased risk of ignition.
It is important to identify materials that are most likely to generate heat and trigger a spontaneous ignition. To examine the feasibility of a new risk assessment method, we focussed on identifying materials that are likely to generate heat, based on the values obtained from our experiments. An attempt was made to evaluate the relative risk using the results for exothermic onset temperature (C80), amount of heat generated (TAM), and the amount of combustible gases generated (GC) as indices. This method was used to evaluate the simple progression of microbial fermentation. The “exothermic onset temperature” (Tonset) indicates how easily exothermal reactions occur, whereas the “amount of heat generated” (Q) indicates the scale of generated heat.
The temperature at which heat generation begins was determined using the C80 calorimetry results; a higher score represents a greater possibility of heat generation at a lower temperature, and therefore greater associated risk. The temperature at which heat generation begins was allocated a score according to the C80 calorimetry results (see
Table 3). Circled numbers show risk ranking.
The amount of heat generated immediately after the onset of TAM measurements at 50 °C, and the amount up to 72 h later was summed to give the total heat generation. The relative risk of heat generation in each sample was scored on the basis of the following standard, in which a higher score represents greater heat generation and a higher risk. The score for heat generation was based on the TAM results (see
Table 4).
Risk for flammable gases is derived directly from their concentration. Consider a mixture of flammable gases and air, in which the percentages of flammable gases are n, n′, n″…, and their limits are N, N′, N″..., respectively Le Chatelier [
24,
25] proposed the following rule: the flammability limit of the complex mixture occurs at percentages n, n′ and n′′, such that:
The rule can be justified according to Le Chatelier’s principle in the following way: it is more or less obvious that if two mixtures (G + air) and (G′ + air) combine to form a homogeneous mixture, then the latter will propagate a flame if both initial mixtures support propagation. It will not propagate a flame if both initial mixtures are either below the lower limit or above the upper limit. The result is a mixture in which G and G′ are present in percentages, n and n′, such that:
is at the propagation limit (lower or upper, depending on whether N and N′ are relative to the individual lower or upper limits). The argument can be easily extended to other gases, and leads to Equation (3).
The risk for the generation of flammable gases is therefore calculated by combining the concentrations of all evolved flammable gases such as H
2, CO and CH
4, as detected by the GC, and comparing these with their lower explosion limit (4.0% for H
2, 12.5% for CO and 5.0% for CH
4) [
26]. Moreover, the risk can be greater if a gas concentration exceed the its higher explosion limit although the risk is low. In order to compare the risk of flammable gas generation in each sample, the score was based on Equation (5). This calculation excludes the effect of CO
2:
The score for the generation of flammable gases was calculated according to the GC results (see
Table 3), giving:
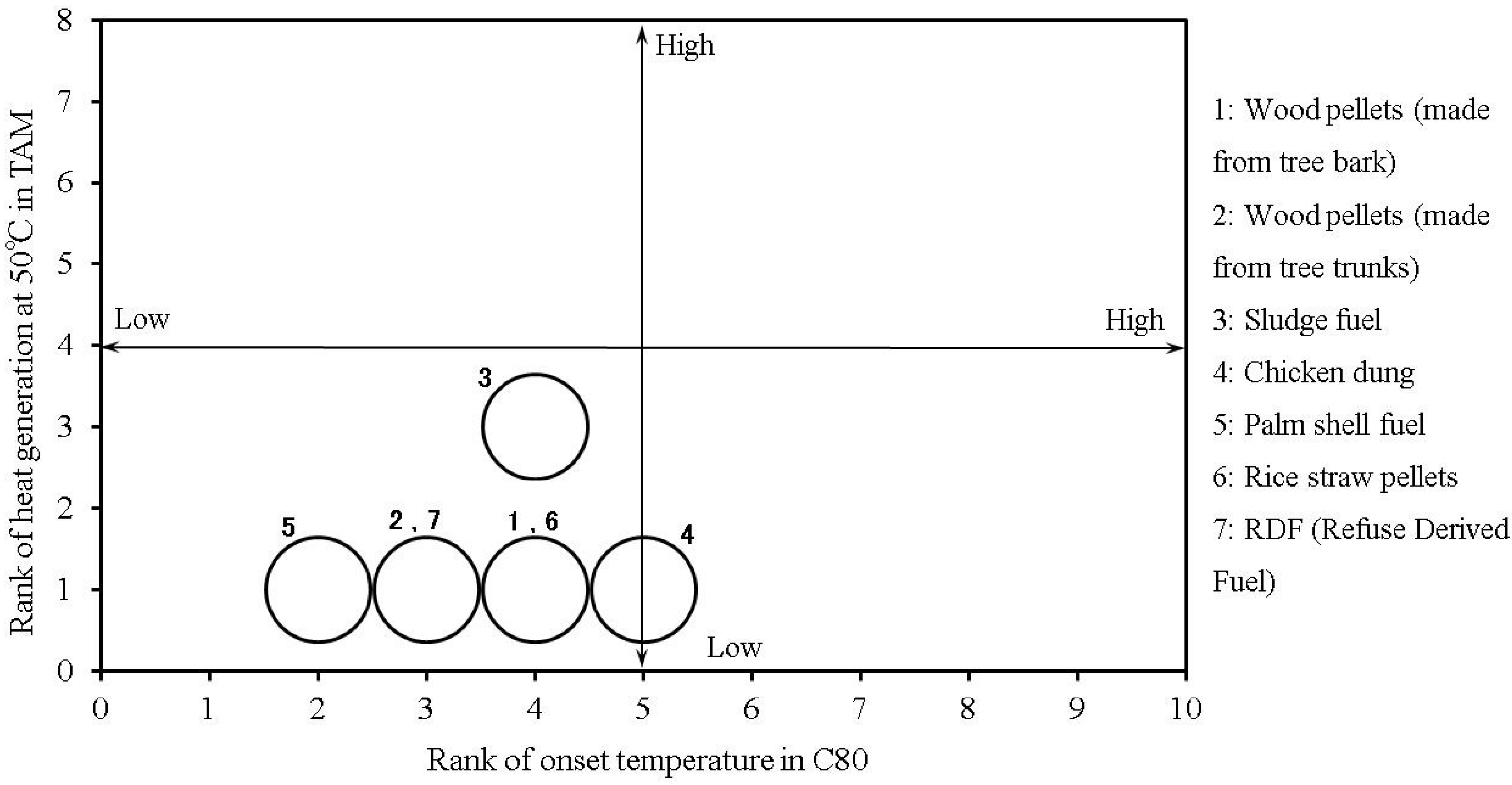
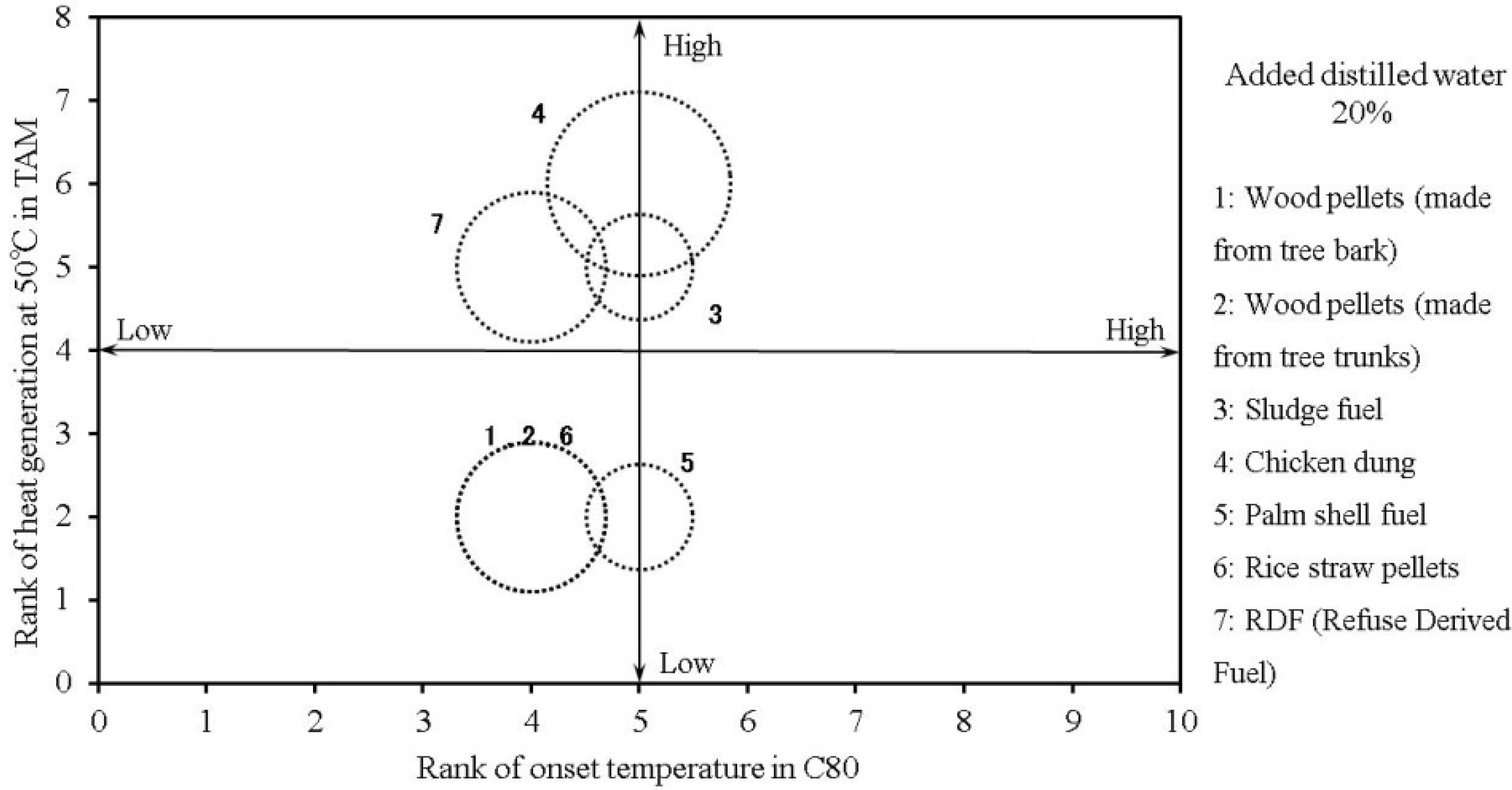
Table 4 (without added moisture) and Table 5 (with added moisture) show the relative risks associated with the biomass fuels, as evaluated by the methods discussed above.
Figure 17 and
Figure 18 show the results from
Table 4 and
Table 5, respectively. The size of the circle represents the “heat-generation amount risk” on the vertical axis, and “heat-generation starting temperature risk” on the horizontal axis. The figures show that the thermal risk increases towards the upper-right corner of the plots. The risk of generating combustible gas also increases as the size of the circle becomes larger. Measured RDF values were also used as a reference for the same thermal hazard evaluation method [
8].
Overall, chicken dung, sludge fuel and palm shell fuels may generate heat even when the temperature is below 30 °C. Moreover, the addition of water increases the amount of generated heat, further raising the risk of combustible gas generation. With the other samples, the addition of moisture lowers the temperature at which heat generation starts, but the amount of generated heat, and the risk of combustible gas generation are not as high as those for chicken dung, and sludge fuel.
Table 4.
Risk ranking of the tested samples.
Table 4.
Risk ranking of the tested samples.
| Sample name | Thermal risk | Flammable gas risk |
|---|
| Tonset (C80) | Q (TAM) | 25 ± 5 °C (GC) |
|---|
| Wood pellets (made from tree bark) | 4 | 1 | 1 |
| Wood pellets (made from tree trunks) | 3 | 1 | 1 |
| Sludge fuel | 4 | 3 | 1 |
| Chicken dung | 5 | 1 | 1 |
| Palm shell fuel | 2 | 1 | 1 |
| Rice straw pellets | 4 | 1 | 1 |
Table 5.
Risk ranking of the tested samples + distilled water 20%.
Table 5.
Risk ranking of the tested samples + distilled water 20%.
| Sample name | Thermal risk | Flammable gas risk |
|---|
| Tonset (C80) | Q (TAM) | 25 ± 5 °C (GC) |
|---|
| Wood pellets (made from tree bark) + distilled water 20% | 4 | 2 | 2 |
| Wood pellets (made from tree trunks) + distilled water 20% | 4 | 2 | 2 |
| Sludge fuel + distilled water 20% | 5 | 5 | 1 |
| Chicken dung + distilled water 20% | 5 | 6 | 3 |
| Palm shell fuel + distilled water 20% | 5 | 2 | 1 |
| Rice straw pellets + distilled water 20% | 4 | 2 | 2 |
Figure 17.
Comparison of risks for onset temperature, heat generation, and flammable gas generation for biomass fuels.
Figure 17.
Comparison of risks for onset temperature, heat generation, and flammable gas generation for biomass fuels.
The RDF values reveal that the addition of moisture increases each risk value in the same manner as with poultry manure, and bio-solid fuels. The results also indicate that adding moisture increases the potential for “thermal risk” or “flammable gas risk”. This method is assumed to be effective in confirming heat generation that can trigger spontaneous ignition, and in determining the associated risk. Sensitive thermal- and gas-analysis equipment is first used to determine whether minute levels of heat or flammable gas are generated, which may trigger spontaneous ignition.
Figure 18.
Comparison of risks for onset temperature, heat generation, and flammable gas generation for biomass fuels + distilled water 20%.
Figure 18.
Comparison of risks for onset temperature, heat generation, and flammable gas generation for biomass fuels + distilled water 20%.
Next, for samples with higher risk, the possibility of an increase in temperature by minute heat generation under more adiabatic conditions is considered. Moreover, for samples with higher ignition temperature, the sample quantity can be increased and the results studied more closely. This permits estimation of the ignition temperature at a large scale, utilising data obtained by a theoretical method such as the Frank-Kamenetskii [
22] method.
Based on the experimental results, the following conclusions were obtained:
(1) The temperature at which heat generation begins is low in all samples when distilled water is added before the measurement. The start of heat generation under these conditions is assumed to be caused by fermentation, which is observed in sludge fuel and chicken dung immediately after the measurements began. Sludge fuel and chicken dung are more likely to generate heat, even at temperatures of 30 °C or less.
(2) Fermentation begins when the internal temperature of the stored biomass fuel is between 30–50 °C. Only a small amount of heat is generated from fermentation and breaking down the pile. If the internal temperature of the stored biomass fuel is between 50–80 °C, fermentation and oxidation of the fatty acid esters produced by fermentation occurs. Self-heating is already in progress at this stage and immediate evasive is required.
(3) All samples generate a greater amount of heat as a consequence of increased moisture content. This micro-heat generation is the trigger of fires. Moreover, it is believed that moderate moisture content in an item enables better fermentation and generates heat more easily. Sludge fuel and chicken dung, which are both composed of faecal matter, are more likely to contain a large amount of microorganisms, which are assumed to become active with the addition of distilled water.
(4) There is an increase in the amount of CO2 generated when distilled water is added. Under normal conditions, even if flammable gases and CO2 are released, they are diffused in the air and their concentration decreases, so the risk from these gases is low. However, when biomass fuel that is likely to ferment is transported in a tightly sealed container, or stored for a long period in a silo, caution is required to avoid ignition and explosion.
(5) The risk of at least one of heat generation temperature, heat generation amount, or flammable gas generation increases due to the addition of distilled water. There is a possibility that chicken dung and sludge fuel may generate heat even at 30 °C or less, and that the amount of heat generated when distilled water is added also increases the risk of generating flammable gases. For other samples, the temperature at which heat generation begins is lowered by adding distilled water, but the amount of heat generated and the risk of flammable gas is not lowered to the same extent as for chicken dung and sludge fuel.