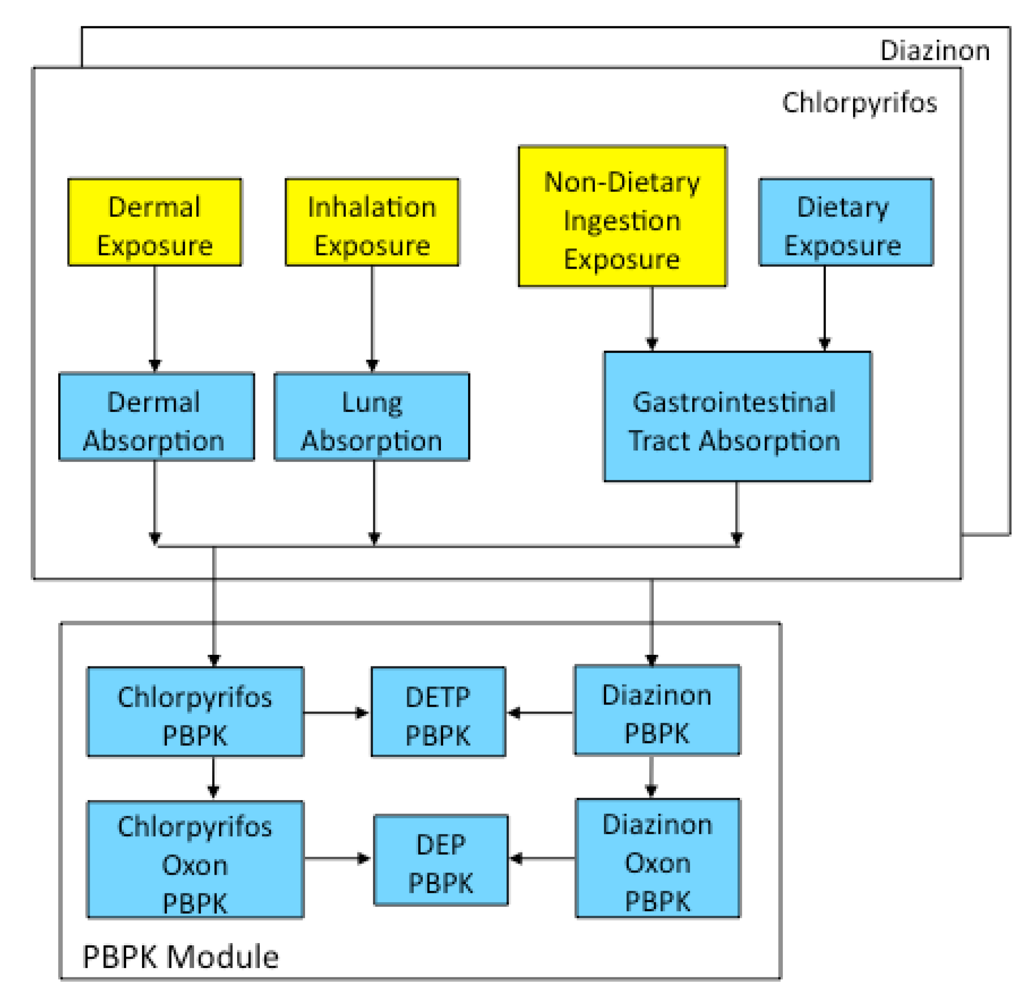
3. Results and Discussion
Our previous experiences and data collection efforts in this farmworker community [
3,
22] provide us with an opportunity to evaluate CACHED, and in particular the “microactivity” approach and child-specific PBPK modules. In
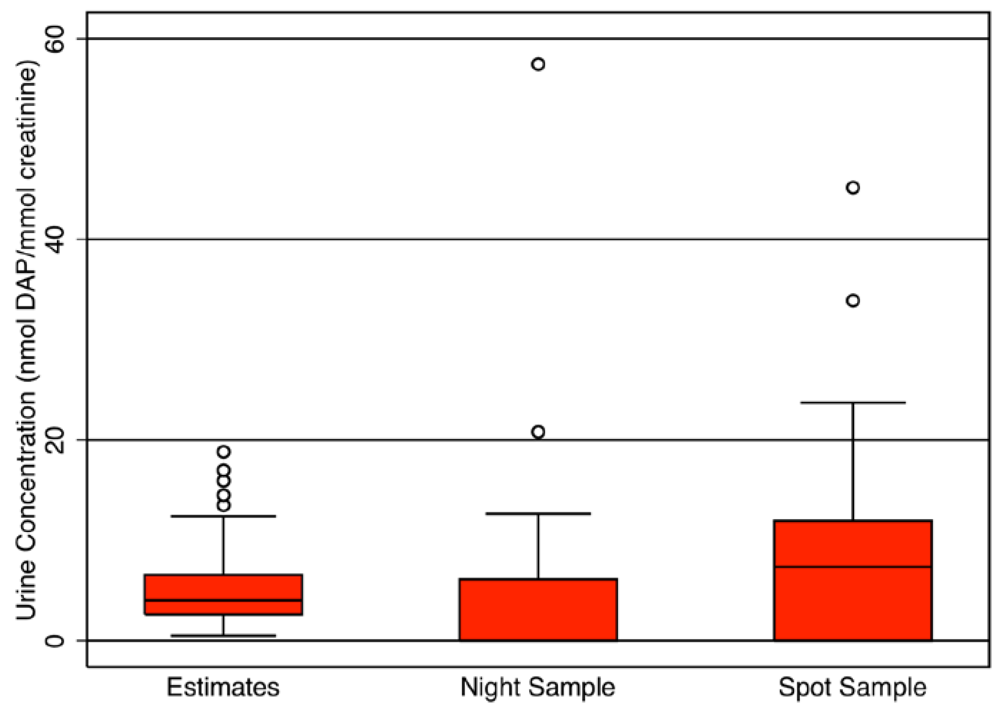
Figure 3, the estimated DAP urine concentrations (
n = 115,000, median = 4.1 nmol DAP/mmol creatinine) simulated from videotaped activity patterns for the farmworker children population is compared with the measured urine concentrations obtained from overnight and spot urine samples (
n = 20). No significant differences were found using the Wilcoxon rank sum test between the median modeled values and the measured values from the overnight (median = 0.7 nmol DAP/mmol creatinine,
p = 0.08) and spot (median = 7.2 nmol DAP/mmol creatinine,
p = 0.31) samples. The median DAP urine concentration estimates for the 11 children that participated in both videotaping and biomarker sampling were positively correlated with the measured values from their overnight (
ρ = 0.69,
p = 0.02) and spot urine samples (
ρ = 0.21,
p-value = 0.56). Interestingly, for these 11 children, there is a non-significant positive correlation between their overnight and spot urine samples (
ρ = 0.41,
p-value = 0.24).
Figure 3.
Comparison of DAP urine concentration estimated by CACHED for the children that had their activities videotaped [
22] with the DAP concentration measured in the overnight and spot urine samples of farmworker children [
3].
Figure 3.
Comparison of DAP urine concentration estimated by CACHED for the children that had their activities videotaped [
22] with the DAP concentration measured in the overnight and spot urine samples of farmworker children [
3].
The CACHED modeling framework was developed to represent the physical processes of exposure and dose through the incorporation of micro-activity time series, exposure mechanisms, and PBPK components. Utilizing careful assumptions for exposure factors and age-specific physiological parameters, CACHED simulations completed with activity patterns and environmental concentrations collected from the same farmworker children population, resulted in realistic estimates of pesticide metabolite concentration in the children’s urine (
Figure 3). These simulations also provide a rigorous and successful evaluation of the “microactivity” exposure assessment approach. Young children’s mouthing contacts with hands and non-dietary objects are very frequent (42 events/h), and of short duration (2 s) [
22], and realistic representations of these events are necessary to obtain accurate non-dietary ingestion exposure estimates, and to evaluate exposure route contribution. Given that children are not merely miniature adults, functions were created for the PBPK module of CACHED to adjust input parameters based on the demographic characteristics of the individual being simulated. While other PBPK models have been created for children [
38,
52,
53,
54,
55], unlike the one developed for CACHED, they have not been rigorously or successfully evaluated with biomonitoring data from children [
56]. Although we were able to use our model to successfully estimate pesticide metabolite urine concentration for this population of farmworker children, further investigations should be completed to evaluate the model with other populations of children simultaneously exposed to chlorpyrifos and diazinon.
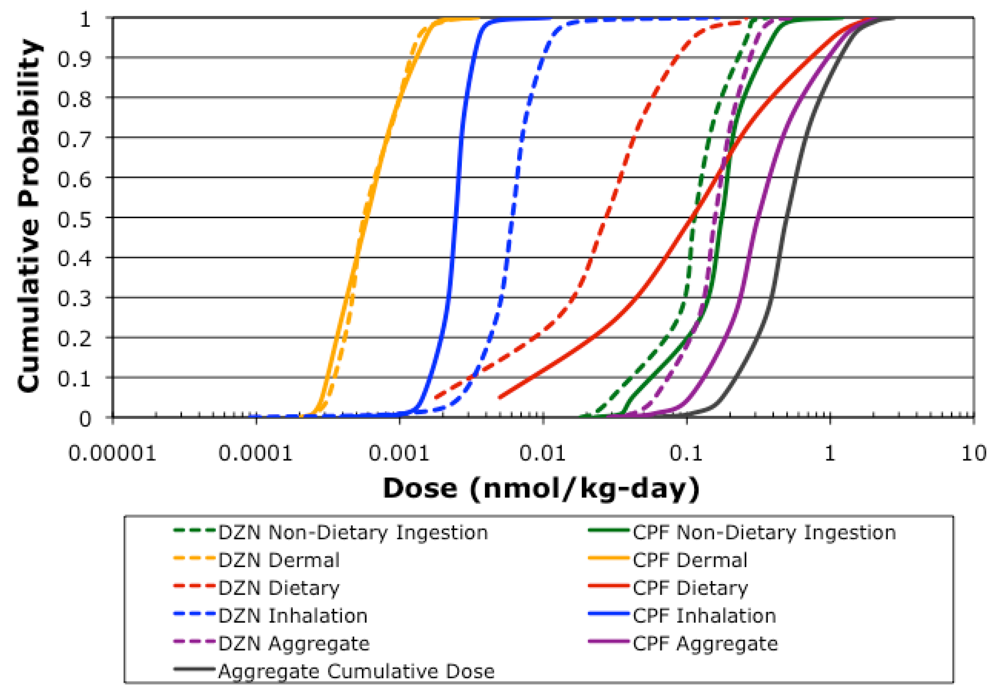
The aggregate cumulative dose estimates for the simulated farmworker children population (
n = 115,000) are depicted in
Figure 4, as well as the dose estimates for each route for each pesticide. Chlorpyrifos had a median aggregate dose of 0.294 nmol/kg-day, and contributed substantially more to the cumulative dose than diazinon, which had a median aggregate dose of 0.148 nmol/kg-day. Dietary exposure contributed the most to the higher aggregate dose estimates for chlorpyrifos (
Table 6), however below the 65th percentile, non-dietary ingestion exposure contributed substantially. Non-dietary ingestion exposure was the primary exposure route for diazinon. Neither inhalation nor dermal exposure contributed substantially to aggregate exposure for chlorpyrifos or diazinon. Even though chlorpyrifos contributed more to cumulative dose in general, there were simulations where diazinon contributed the most to cumulative dose, as evident by the range in
Table 6. This demonstrates how important it is to account for exposure to multiple pesticides in determining food tolerances under the FQPA, in particular for diazinon. The non-dietary ingestion exposure route contributed the most to aggregate dose (
Table 6), also demonstrating how important it is to account for exposure from routes other than dietary ingestion. Dietary ingestion was the second most significant route for aggregate dose. However, as evident by the range of proportional contributions, there were simulations when dietary exposure and even inhalation exposure were the most significant routes. The variability observed in the route and pesticide contributions to dose simulated by CACHED (
Table 6) confirms the importance of accounting for aggregate and cumulative exposure in establishing pesticide residue tolerances in food under the FQPA.
Figure 4.
Population dose distributions for each route, pesticide, and for cumulative and aggregate simulations (n = 115,000).
Figure 4.
Population dose distributions for each route, pesticide, and for cumulative and aggregate simulations (n = 115,000).
Table 6.
Dose estimations (nmol/kg-day) for the farmworker children population (n = 115,000) by pesticide and route.
Table 6.
Dose estimations (nmol/kg-day) for the farmworker children population (n = 115,000) by pesticide and route.
| Pesticide | Route | Range | Mean | Median |
|---|
| chlorpyrifos | dermal | 0.000–0.003 | (0.0–1.5%) | 0.001 | (0.3%) | 0.001 | (0.2%) |
| | inhalation | 0.000–0.011 | (0.0–5.3%) | 0.002 | (0.9%) | 0.002 | (0.8%) |
| | non-dietary ingestion | 0.023–1.206 | (1.5–84.5%) | 0.189 | (56.6%) | 0.175 | (59.2%) |
| | dietary | 0.000–2.120 | (0.0–94.4%) | 0.252 | (42.2%) | 0.109 | (39.4%) |
| | aggregate a | 0.031–2.43 | (28.2–96.8%) | 0.444 | (67.5%) | 0.314 | (65.7%) |
| diazinon | dermal | 0.000–0.003 | (0.0–1.4%) | 0.001 | (0.5%) | 0.001 | (0.4%) |
| | inhalation | 0.000–0.166 | (0.0–42.4%) | 0.006 | (4.8%) | 0.006 | (4.0%) |
| | non-dietary ingestion | 0.018–0.365 | (1.2–60.1%) | 0.123 | (72.1%) | 0.113 | (76.2%) |
| | dietary | 0.000–0.260 | (0.0–56.0%) | 0.039 | (22.6%) | 0.314 | (18.6%) |
| | aggregate a | 0.028–0.551 | (3.2–71.8%) | 0.169 | (32.5%) | 0.158 | (34.3%) |
| cumulative | dermal | 0.000–0.005 | (0.0–2.9%) | 0.001 | (0.3%) | 0.001 | (0.3%) |
| | inhalation | 0.001–0.170 | (0.1–43.5%) | 0.009 | (2.1%) | 0.009 | (1.7%) |
| | non-dietary ingestion | 0.051–1.48 | (3.0–99.1%) | 0.312 | (59.5%) | 0.292 | (62.4%) |
| | dietary | 0.000–2.20 | (0.0–96.5%) | 0.290 | (38.1%) | 0.150 | (34.7%) |
| | aggregate | 0.061–2.78 | | 0.613 | | 0.496 | |
Our results indicate that the absorbed dose from dermal exposure is two orders of magnitude lower than that from either ingestion route. Because bare hands have the greatest contact frequency we only estimated dermal absorption from the hands. However, this may have underestimated the contribution of dermal absorption. As part of the environmental sampling completed in the farmworker households, the children wore union suits and socks that were later analyzed for pesticide loading [
3]. Our dermal exposure estimates for the hand were comparable to the pesticide loading on the socks [
14]. We did not estimate dermal absorption for the feet because the children for the most part were wearing shoes and socks. However even had we included dermal absorption from the feet our absorbed dermal dose would approximately double and still be two orders of magnitude lower than other routes. The pesticide loading on the union suits was an order of magnitude lower than on the socks. For example, the mean chlorpyrifos loading was 0.01 ng/cm
2 and 0.11 ng/cm
2 for the union suit and socks, respectively. Given that the children were clothed and the lower pesticide loading on other parts of the body, it is not likely that dermal absorption from these other body parts would have contributed substantially to aggregate dose. Future investigations should be completed that examine relative dermal exposure from different body parts in relation to contact frequency, duration, surface area and clothing.
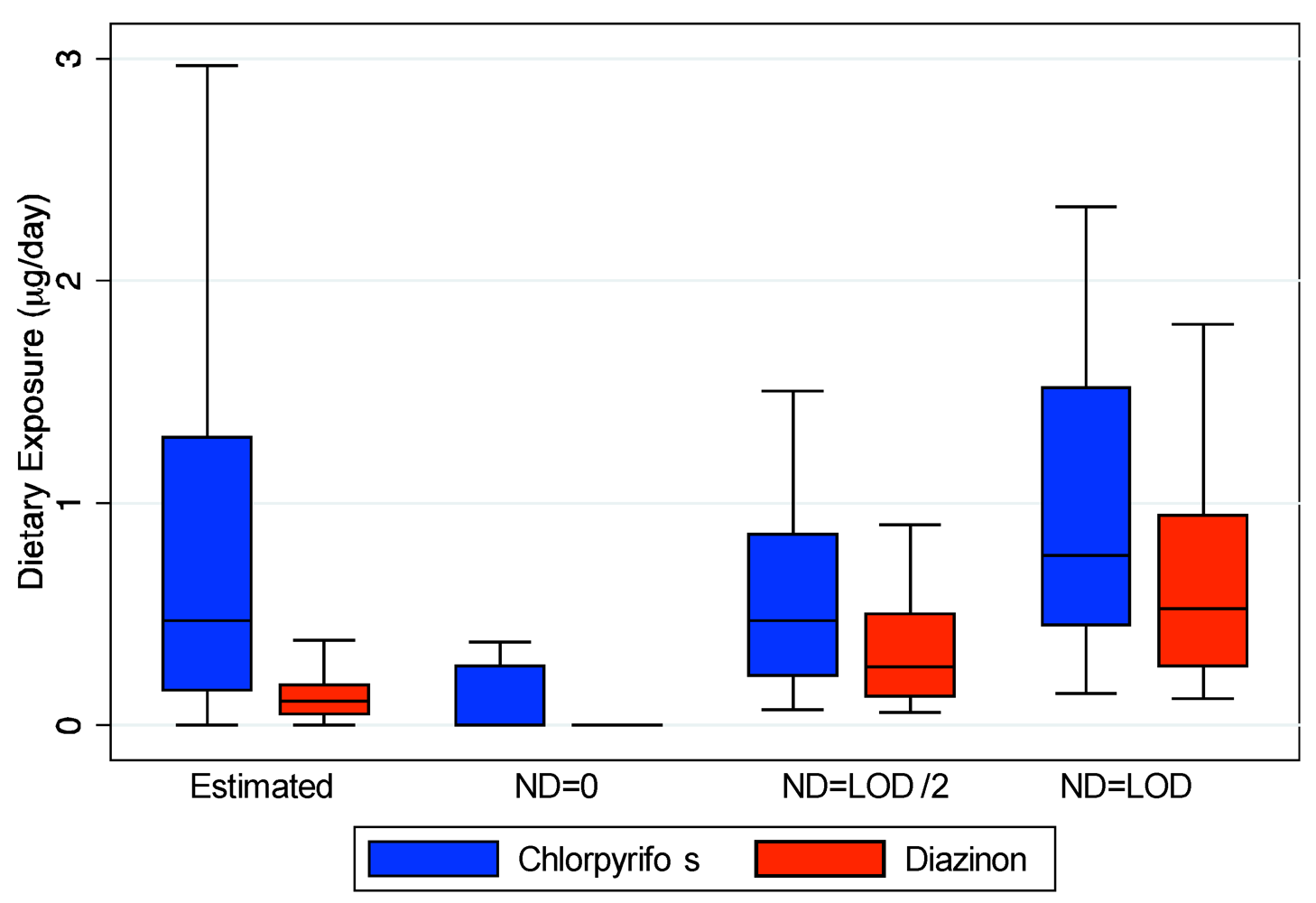
Our estimation of dietary exposure utilizing food diaries and pesticide residue values from national databases does not account for any contribution of ingestion exposure from handling the food by the child or other family members. In addition, the food may have higher pesticide residues depending upon how it is stored in these homes that may have increased residential pesticide contamination due to proximity to agricultural fields or through direct contact with contaminant surfaces [
57]. Although few pesticides were detected in the duplicate diet measurements, these measurements do account for potential contamination during food storage and preparation. As our estimated exposures are similar to the duplicate diet measurements (
Figure 2), these additional potential sources of dietary exposure may not be substantial. However, due to their young age, the children in our study ate mostly with their hands. Simulations from a different study indicate that children’s handling of food can account for 20–80% of their dietary intake [
58]. A major limitation of our current study is that we do not account for these additional sources of dietary exposure, thus underestimating the contribution of this route to aggregate dose. Although these exposures do contribute to overall dietary exposure, they should be quantified separately so that food tolerances developed under the FQPA take into account these additional exposures unique to children. It is also not currently clear how food handling may affect other routes of exposures. For example, it may decrease dermal exposure as a removal mechanism which in turn may decrease their non-dietary ingestion exposure.
Non-dietary ingestion exposure contributes most to aggregate cumulative dose and is a function of the children’s hand-to-mouth and object-to-mouth frequency. Hand-to-mouth frequency is highest during eating events and thus there is substantial non-dietary exposure while a child is handling food as well. Given that the mouthing frequencies quantified in these children [
22] exceed US EPA’s recommendations for use in risk assessments [
49], risk estimates based on their guidelines might underestimate the potential risk of residential pesticide exposure in children.
There was much higher within-child variability than between-child variability for absorbed cumulative dose for cumulative aggregate, inhalation and dietary exposure (
Table 7). However, there was much greater between-child variability compared to within-child variability for cumulative absorbed dose from non-dietary ingestion and dermal exposure. These findings are consistent with our analyses of exposures in this population [
14]. Given that we used the same pesticide concentration and exposure factor distributions for each child, and the within-child variability is so small, the variability between children for dermal and non-dietary ingestion dose is most likely attributed to their individual activity patterns even after accounting for differences in their physiology. There is over an order of magnitude difference in the non-dietary ingestion dose for the child with lowest exposure and the child with the highest exposure, highlighting that some children may be at substantially higher risk for residential pesticide exposure. In setting appropriate food tolerances under the FQPA, it is important that they are protective of at-risk children exhibiting unique activity patterns. Considering the recent associations with neurodevelopment and attention deficit/hyperactivity disorder and organophosphate pesticide exposure [
59,
60,
61], it would be important to examine in the future if these children’s increased exposure is as a function of increased activity during the first few years of life.
Table 7.
Within-child and between-child variance calculated for the 5000 simulations for each of the 23 children according to Rappaport [
46].
Table 7.
Within-child and between-child variance calculated for the 5000 simulations for each of the 23 children according to Rappaport [46].
| Pesticide | Route | Within-Child Variance | Between-Child Variance |
|---|
| Chlorpyrifos | Dermal | 0.01 | 0.26 |
| | Inhalation | 0.04 | 0.04 |
| | Non-dietary ingestion | 0.03 | 0.39 |
| | Dietary | 8.35 | 0.98 |
| | Aggregate | 0.43 | 0.13 |
| Diazinon | Dermal | 0.01 | 0.23 |
| | Inhalation | 0.18 | 0.06 |
| | Non-dietary ingestion | 0.02 | 0.37 |
| | Dietary | 5.12 | 0.51 |
| | Aggregate | 0.07 | 0.16 |
| Cumulative | Dermal | 0.01 | 0.24 |
| | Inhalation | 0.09 | 0.04 |
| | Non-dietary ingestion | 0.02 | 0.38 |
| | Dietary | 5.05 | 0.79 |
| | Aggregate | 0.27 | 0.12 |
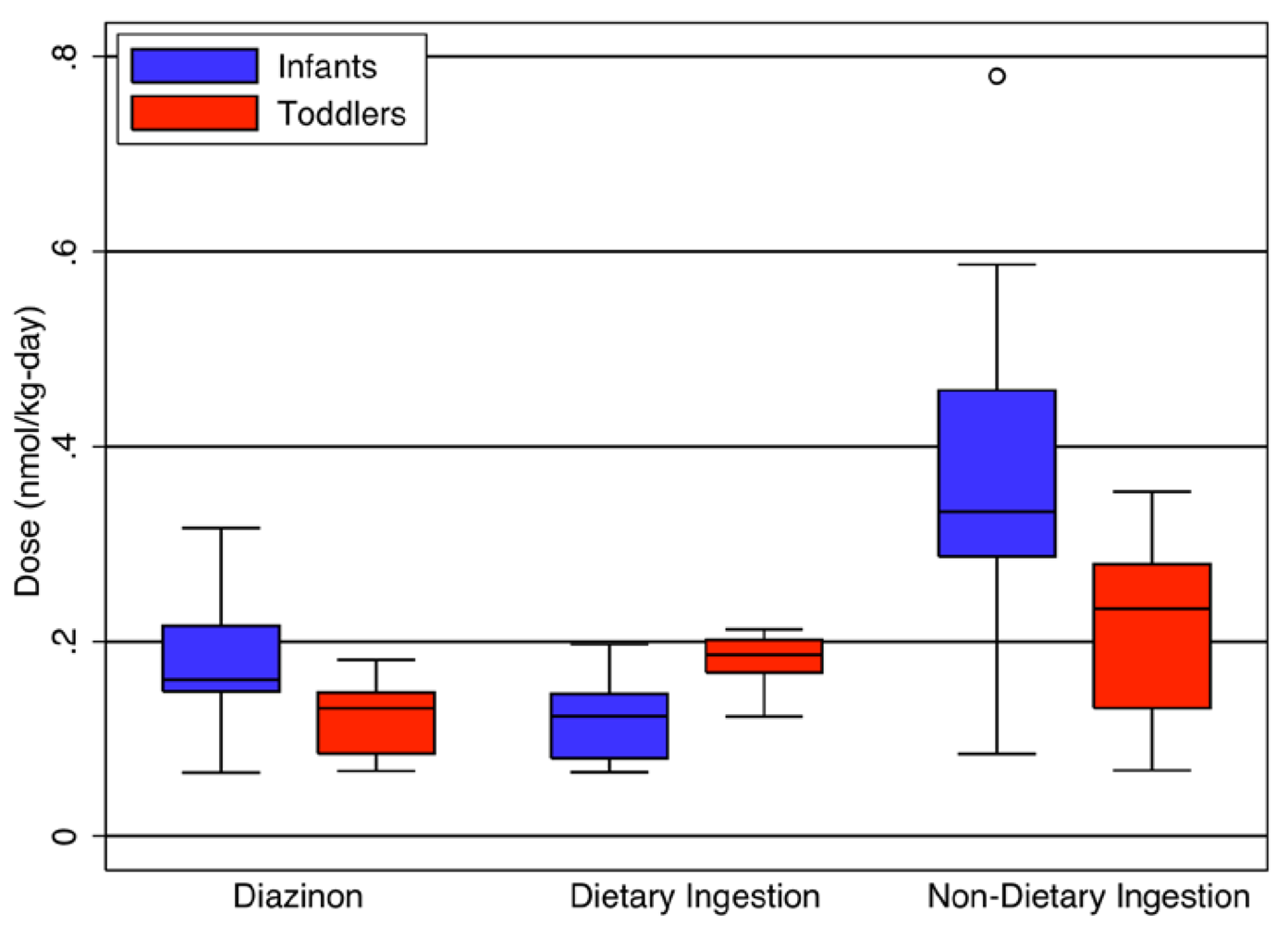
As in our analyses of our exposure simulations, no significant differences were observed in absorbed dose for any route as a function of gender for either chlorpyrifos or diazinon [
14]. However, several significant differences in absorbed dose were identified between infants (6–13 months) and toddlers (20–26 months) (
Figure 5). Toddlers had a higher dietary dose on a per body weight basis than infants (
p = 0.02). Conversely, infants had a higher non-dietary ingestion (
p = 0.008) and aggregate diazinon dose than toddlers (
p = 0.02). Both of these findings have particular importance for setting appropriate pesticide tolerances for food under the FQPA. Toddlers may be the most at-risk group for direct ingestion of pesticides from food, and special consideration should be given to their increased food consumption rates. Infants, however, are of particular concern because they receive a substantially larger dose from routes other than food ingestion, highlighting the importance of accounting for these routes of exposure in determining allowable pesticide levels on food.
Figure 5.
Significant differences (p < 0.05) between infants and toddlers in aggregate diazinon and cumulative dietary and non-dietary ingestion dose (nmol/kg-day).
Figure 5.
Significant differences (p < 0.05) between infants and toddlers in aggregate diazinon and cumulative dietary and non-dietary ingestion dose (nmol/kg-day).
The computed RIs and ARI for the farmworker children population according to the methods used by US EPA for aggregate exposure to chlorpyrifos and diazinon [
47,
48] and cumulative aggregate exposure to organophosphate pesticides [
49] are presented in
Table 8. According to these metrics, only less than 4% of the estimated non-dietary ingestion and aggregate diazinon doses pose a risk for this population. Conversely, while none of the children in the population (
n = 115,000) are potentially at risk from inhalation exposure to chlorpyrifos, approximately 4%, 57%, 83% and 98% are at risk from dermal, dietary, non-dietary ingestion and aggregate exposure to chlorpyrifos. As demonstrated in
Table 8, none of the exposure scenarios present a risk from cumulative exposure to chlorpyrifos and diazinon. However, the toxicological endpoint used for cumulative risk is the BMD
10, which is much higher than the NOAEL used for risk estimates of singular pesticides (
Table 4 and
Table 5). Given the very different results, it is not clear if the aggregate risk assessments using the NOAEL are too conservative or if the cumulative risk assessments using the BMD
10 are not protective enough. However, several adverse effects on neurodevelopment in children have already been associated with urinary DAP metabolite levels in the larger cohort of Latino children in our agricultural study community [
59,
60,
61]. This highlights the importance in determining more specific endpoints for risk analysis of pesticide exposure in children.
Table 8.
Route-specific and aggregate risk indices for farmworker children simulations (
n = 115,000) from chlorpyrifos, diazinon and cumulative exposure. An ARI or RI of less than one suggests a “risk of concern” [
50].
Table 8.
Route-specific and aggregate risk indices for farmworker children simulations (n = 115,000) from chlorpyrifos, diazinon and cumulative exposure. An ARI or RI of less than one suggests a “risk of concern” [50].
| Pesticide | Route | Range | Mean | Median |
|---|
| Chlorpyrifos a | Dermal | 0.5–10.3 | 4.6 | 4.3 |
| | Inhalation | 5.9–78.6 | 36.1 | 34.7 |
| | Non-dietary ingestion | 0.05–Inf | 4.2 | 0.7 |
| | Dietary | 0.04–2.4 | 0.7 | 0.5 |
| | Aggregate | 0.02–1.1 | 0.3 | 0.2 |
| Diazinon a | Dermal | 5280–42,100 | 19,200 | 19,500 |
| | Inhalation | 13.6–259 | 53.4 | 46.2 |
| | Non-dietary ingestion | 1.8–Inf | 1,070 | 23.0 |
| | Dietary | 0.5–29.0 | 7.4 | 5.8 |
| | Aggregate | 0.4–13.2 | 4.4 | 4.0 |
| Cumulative b | Dermal | 163,000–2,800,000 | 1,280,000 | 1,220,000 |
| | Inhalation | 9130–96,300 | 45,000 | 43,300 |
| | Non-dietary ingestion | 16.0–Inf | 2310 | 257 |
| | Dietary | 12.4–780 | 217 | 161 |
| | Aggregate | 7.7–480 | 105 | 91 |
While the modeled DAP urine concentration was quite similar to measured values collected from children in the same population (
Figure 3), the aggregate dose estimates are quite different than estimates from other studies (
Table 9). Other studies assumed values for necessary exposure factors, activity, and absorption fractions for each route to estimate aggregate dose from environmental concentrations, making it complicated to compare aggregate and route-specific dose estimates. O’Rourke and colleagues [
62] calculated “theoretical” absorbed daily dose assuming 100% clearance from the maximum DAP urine concentration measured in agricultural children 3–5 years old in Arizona. This resulted in aggregate dose estimates for chlorpyrifos and diazinon that were about an order of magnitude higher than the estimates from CACHED. Wilson and colleagues [
13] assumed that the contribution of dermal exposure is negligible, and their estimates for children 2–5 years old in North Carolina were lower than the estimates from CACHED, especially for diazinon. In a follow up study in the same population, estimated aggregate exposures for diazinon under-predicted urinary measurements by an order of magnitude [
63]. While these were not farmworker children, the environmental concentrations, with the exception of outdoor air, measured in their homes were comparable to the values in the farmworker children’s homes [
3]. The differences between these studies and the current study underscore how differences in the methods and assumptions used can result in very different estimates of absorbed dose and aggregate risk.
Similarly, Lu
et al. estimated aggregate chlorpyrifos dose for children in Washington using a linked exposure-PBPK model and from the TCP urine levels directly [
6]. They indicated that there model only reasonably predicted TCP metabolite levels for the two children who had chlorpyrifos quantified in their duplicate diet measurements. However, these children had comparable measured TCP levels in their urine with those that were assumed to have no dietary exposure. Assuming no-dietary exposure for children with duplicate diets may have underestimated their dietary exposures substantially (
Figure 2). Similarly they assumed no dermal exposure, only 20 hand-to-mouth contacts per day, and 100 mg/day of dust ingestion. We have previously demonstrated that these assumptions may vastly underestimate dermal and non-dietary ingestion exposure [
14], which our current analyses demonstrate are important components of aggregate exposure. Similarly, Lu
et al. [
6] did not adjust many of the PBPK parameters that are likely to be altered in young children such as partition coefficients, clearance rates and protein binding [
6]. In our model these are some of the most sensitive parameters [
24,
26]. It is likely that both inadequacies in aggregate exposure estimation and in developing their child-specific PBPK model, led to their models underestimation of TCP levels in urine.
A few studies have provided route-specific dose estimates for chlorpyrifos. Pang and colleagues [
11] estimated aggregate chlorpyrifos dose for 80 individuals over the age of 10 in Maryland. While their median estimate was lower than the CACHED estimates their reported range and mean values were comparable. Pang
et al. [
11] also report the dose for each route, and determined inhalation of indoor air to account for 85% of aggregate dose (
Table 9). They assumed 100% absorption of chlorpyrifos inhaled, which resulted in an inhalation dose much higher than the estimates from CACHED which were based on blood:air partition coefficients and child-specific ventilation rates. However, their estimated values for dose from the other routes are much lower than the current study, especially from ingestion for which they assumed 50% absorption. Morgan
et al. [
12] also report aggregate and route-specific chlorpyrifos dose estimates for children <1 to 5 years in North Carolina. While they did include dermal exposure, they used lower absorption fractions for inhalation and ingestion than Wilson
et al. [
13]. This resulted in estimates that were much lower than Wilson
et al. [
13] and the values in this current study (
Table 9). Their estimated values for each exposure route were lower than the CACHED estimates, especially for dietary and non-dietary ingestion. Morgan and colleagues [
12] also measured TCP in the children’s urine. Their aggregate dose estimates were unable to account for over 60% of the measured metabolite levels in urine. In contrast, the success of CACHED to simulate the DAP urine concentration of the farmworker children (
Figure 3) demonstrates the importance of utilizing a model that accounts for the physical process of exposure and dose, and incorporates micro-level activity patterns based on real children. Activity patterns from videotape were collected as part of the studies in Arizona and North Carolina [
12,
13,
62]. Thus, these studies could provide a unique opportunity to further evaluate the CACHED modeling framework and assess the relative contribution of exposure routes to aggregate risk in different children populations.
Table 9.
Comparison of route-specific and aggregate dose estimates from CACHED and other studies.
Table 9.
Comparison of route-specific and aggregate dose estimates from CACHED and other studies.
| Study | Units | Statistic | Dermal | Inhalation | Dietary | Non-Dietary | Aggregate |
|---|
| Current | ng/kg-day | Range | 0.1–1.2 | 0.1–3.9 | 0.0–743 | 8.0–423 | 10.9–853 |
| | | Mean | 0.3 | 0.9 | 88.2 | 66.2 | 157 |
| | | Median | 0.2 | 0.9 | 38.1 | 61.4 | 110 |
| | ng/day | Range | 0.8–13.3 | 1.5–42.5 | 0.0–8096 | 86.8–4609 | 118.4–9302 |
| | | Mean | 2.7 | 9.4 | 962 | 722 | 1,696 |
| | | Median | 2.2 | 9.6 | 402 | 656 | 1,128 |
| Morgan [12] | ng/kg-day | Range | 0.0–11.9 | 0.0–30.3 | 0.0–3.3 | 0.0–10,200 | 0.5–179 |
| | | Mean | 0.1 | 2.1 | 0.3 | 285 | 8.1 |
| | | Median | 0.0 | 0.8 | 0.1 | 0.0 | 3 |
| Pang [11] | ng/day | Range | 0.0–241 | 0.0–13,900 | 0.0–10,200 | 0.0–217 | 13.5–12,800 |
| | | Mean | 4.3 | 594 | 285 | 4.3 | 1,390 |
| | | Median | 0.0 | 103 | 0.0 | 0.0 | 112 |
| O’Rourke [62] | ng/kg-day | Range | | | | | 2,430–13,000 |
| | | Mean | | | | | 6006 |
| | | Median | | | | | 2590 |
| Wilson [13] | ng/kg/day | Range | | | | | 10.6–329 |
| | | Mean | | | | | 76.1 |
| | | Median | | | | | 30.0 |
| Wilson [63] | ng/kg/day | Range | | | | | 0.86–164 |
| | | Mean | | | | | |
| | | Median | | | | | 8.22 |
| Lu | ng/kg/day | Range | | | | | <1–2302 |
| (Predicted) [6] | | Mean | | | | | 180 |
| | | Median | | | | | 4 |
| Lu | ng/kg/day | Range | | | | | 40–1320 |
| (Measured) [6] | | Mean | | | | | 420 |
| | | Median | | | | | 330 |
Children’s aggregate and cumulative risk from exposure to multiple pesticides via multiple routes involves many complex mechanisms and processes. The CACHED modeling framework was developed in an attempt to describe these processes through mathematical calculations, with a particular emphasis on the complex mechanisms governing dermal and non-dietary ingestion exposure. Given the complexity of these exposure routes and the PBPK components, and the numerous assumptions made in developing the equations and selecting appropriate parameter values, it is remarkable that the estimates from CACHED were not significantly different from what was measured in the children. However, there are still several mechanisms of dermal and non-dietary exposure that should be explored in the future [
14]. For example, our modeling efforts did not account for non-dietary ingestion exposure from handling of food with contaminated hands or for dermal exposure to body parts other than the hands. For both the exposure and PBPK components there are still several parameters for which there is limited experimental data and assumptions had to be made. For dermal exposure more data is needed regarding the transfer of pesticides to the hands in residential settings, where the pesticides may be present in numerous phases (e.g., a residue or adhered to dust particles). There is also not much experimental data for two of the key parameters for non-dietary ingestion exposure: surface area of mouthing or saliva removal efficiency. Additional data on these parameters would also help refine estimates. The PBPK model is most sensitive to partition coefficients and the fraction of pesticide or metabolite bound to plasma proteins. There are currently very few experimental values for these parameters for chlorpyrifos and diazinon and virtually no experimental data for their metabolites. For our purposes these parameters were estimated and optimized during the initial PBPK model evaluation. Furthermore, there is also no experimental data for these parameters in children. Although the assumptions we have made in calculating these parameters for children seems to be adequate from the model evaluation, it would be helpful in the future to verify these assumptions with experimental measurements to refine future exposure models for children.