Identification of Candidate Genes and Physiological Pathways Involved in Gonad Deformation in Whitefish (Coregonus spp.) from Lake Thun, Switzerland
Abstract
:1. Introduction
2. Experimental Section
2.1. Sampling
2.2. Experimental Design
2.3. Microarrays
2.4. cRNA Synthesis, Hybridizations, Imaging and Feature Extraction
2.5. Normalization and Data Filtering
2.6. Gene-by-Gene Analysis
2.7. Gene Ontology Analysis
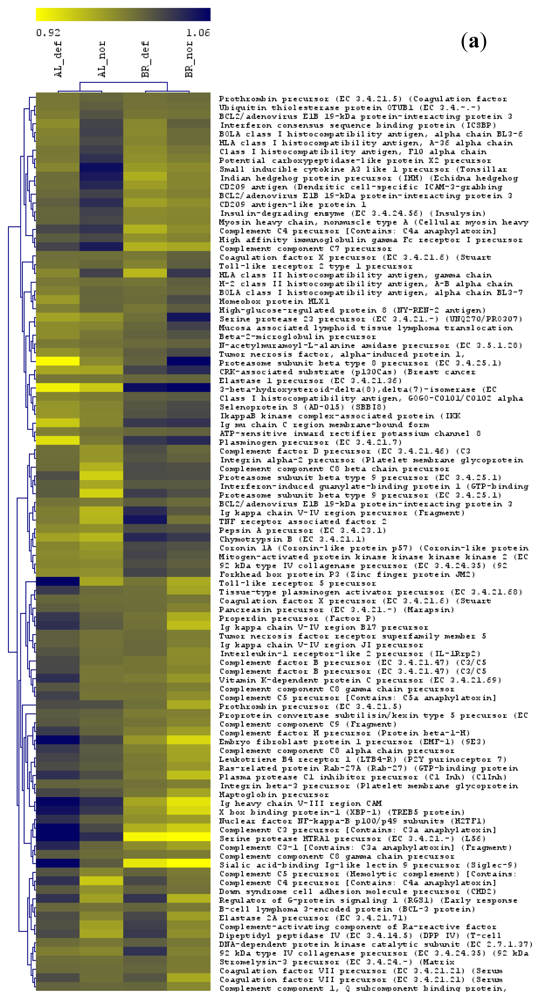
2.8. Hierarchical Clustering
2.9. Real-Time qPCR Analysis
3. Results and Discussion
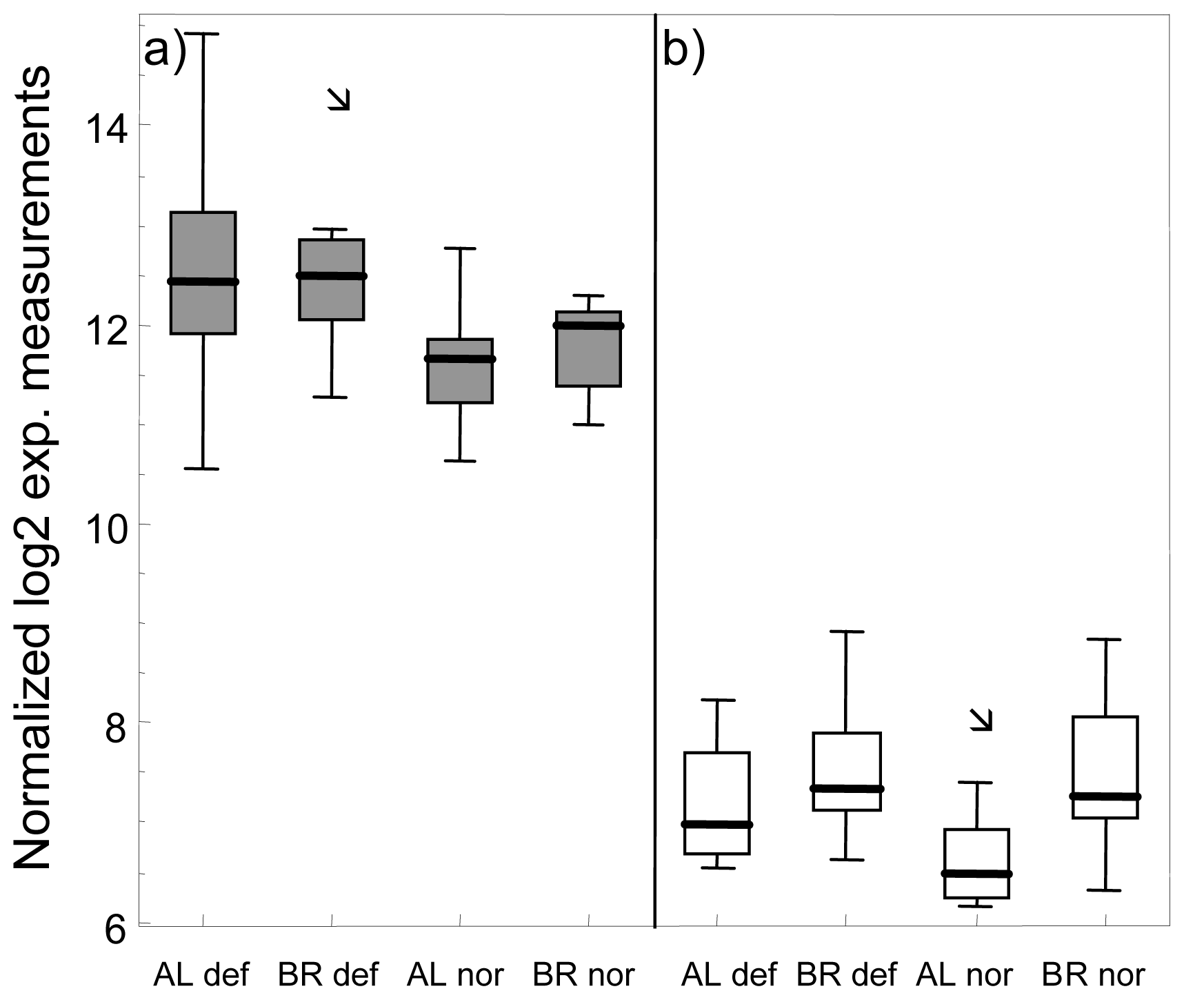
3.1. Gene-by-Gene Analysis
3.2. GO Analysis
3.3. Deviation in Regulation of Immune System Processes
3.4. Autoimmune Disease?
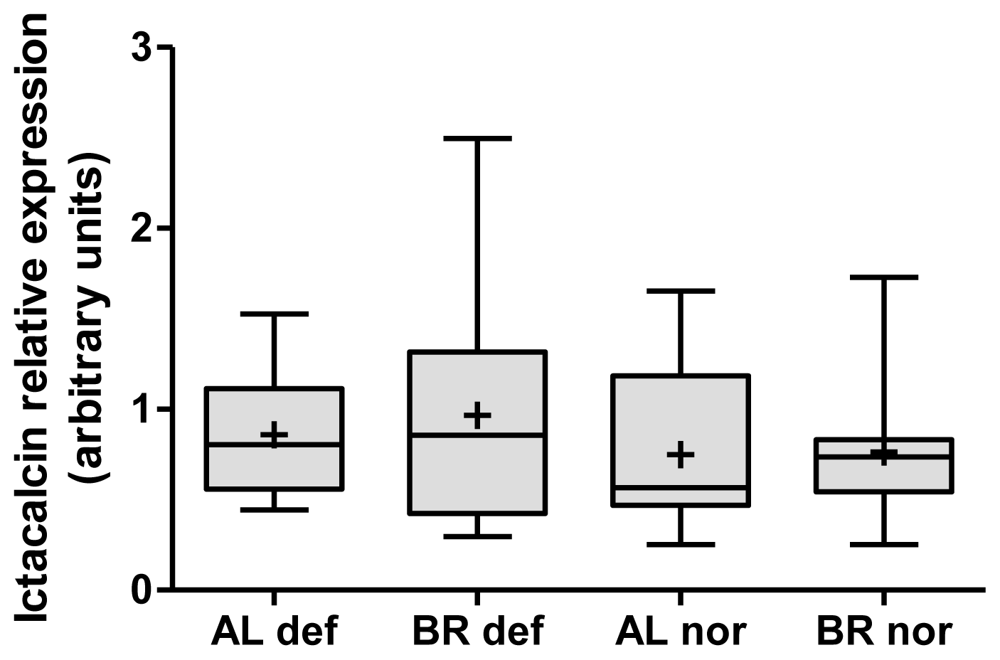
3.5. Indications of Ictacalcin
4. Conclusions
Supplementary Materials
ijerph-08-02706-s001.docAcknowledgements
References and Notes
- Bernet, D; Wahli, T; Kueng, C; Segner, H. Frequent and unexplained gonadal abnormalities in whitefish (central alpine Coregonus sp.) from an alpine oligotrophic lake in Switzerland. Dis. Aquat. Org 2004, 61, 137–148. [Google Scholar]
- Bittner, D; Bernet, D; Wahli, T; Segner, H; Kung, C; Largiader, CR. How normal is abnormal? Discrimination between deformations and natural variation in gonad morphology of European whitefish Coregonus lavaretus. J. Fish Biol 2009, 74, 1594–1614. [Google Scholar]
- Allen, Y; Scott, AP; Matthiessen, P; Haworth, S; Thain, JE; Feist, S. Survey of estrogenic activity in United Kingdom estuarine and coastal waters and its effects on gonadal development of the flounder Platichthys flesus. Environ. Toxicol. Chem 1999, 18, 1791–1800. [Google Scholar]
- Van der Ven, LTM; Wester, PW; Vos, JG. Histopathology as a tool for the evaluation of endocrine disruption in zebrafish (Danio rerio). Environ. Toxicol. Chem 2003, 22, 908–913. [Google Scholar]
- Vethaak, AD; Lahr, J; Schrap, SM; Belfroid, AC; Rijs, GBJ; Gerritsen, A; de Boer, J; Bulder, AS; Grinwis, GCM; Kuiper, RV; et al. An integrated assessment of estrogenic contamination and biological effects in the aquatic environment of The Netherlands. Chemosphere 2005, 59, 511–524. [Google Scholar]
- Schafers, C; Teigeler, M; Wenzel, A; Maack, G; Fenske, M; Segner, H. Concentration- and time-dependent effects of the synthetic estrogen, 17 alpha-ethinylestradiol, on reproductive capabilities of the zebrafish, Danio rerio. J. Toxicol. Environ. Health, Part A 2007, 70, 768–779. [Google Scholar]
- Bernet, D; Liedtke, A; Bittner, D; Eggen, RIL; Kipfer, S; Küng, C; Largiader, CR; Suter, MJ-F; Wahli, T; Segner, H. Gonadal malformations in whitefish from Lake Thun: defining the case and evaluating the role of EDCs. Chimia 2008, 62, 383–388. [Google Scholar]
- Bogdal, C; Naef, M; Schmid, P; Kohler, M; Zennegg, M; Bernet, D; Scheringer, M; Hungerbuhler, K. Unexplained gonad alterations in whitefish (Coregonus spp.) from Lake Thun, Switzerland: Levels of persistent organic pollutants in different morphs. Chemosphere 2009, 74, 434–440. [Google Scholar]
- Kipfer, S; Segner, H; Wenger, M; Wahli, T; Bernet, D. Long-term estrogen exposure of whitefish Coregonus lavaretus induces intersex but not Lake Thun-typical gonad malformations. Dis. Aquat. Org 2009, 84, 43–56. [Google Scholar]
- Bittner, D. Gonad Deformations in Whitefish (Coregonus spp.) from Lake Thun, Switzerland-A Population Genetic and Transcriptomic Approach. PhD Thesis, University of Bern, Bern, Switzerland, 2009. [Google Scholar]
- Bernet, D; Wahli, T; Küng, C; Zieri, HR; Segner, H. Zooplankton as the key factor for the development of macroscopical gonad deformation in whitefish (Coregonus lavaretus) from Lake Thun, Switzerland. Aquatic Biology 2011. under revision. [Google Scholar]
- Cossins, AR; Crawford, DL. Opinion-Fish as models for environmental genomics. Nat. Rev. Genet 2005, 6, 324–333. [Google Scholar]
- Denslow, ND; Garcia-Reyero, N; Barber, DS. Fish ‘n’ chips: the use of microarrays for aquatic toxicology. Mol. BioSyst 2007, 3, 172–177. [Google Scholar]
- Miller, KM; Maclean, N. Teleost microarrays: development in a broad phylogenetic range reflecting diverse applications. J. Fish Biol 2008, 72, 2039–2050. [Google Scholar]
- Jin, W; Riley, RM; Wolfinger, RD; White, KP; Passador-Gurgel, G; Gibson, G. The contributions of sex, genotype and age to transcriptional variance in Drosophila melanogaster. Nat. Genet 2001, 29, 389–395. [Google Scholar]
- Oleksiak, MF; Churchill, GA; Crawford, DL. Variation in gene expression within and among natural populations. Nat. Genet 2002, 32, 261–266. [Google Scholar]
- Ranz, JM; Castillo-Davis, CI; Meiklejohn, CD; Hartl, DL. Sex-dependent gene expression and evolution of the Drosophila transcriptome. Science 2003, 300, 1742–1745. [Google Scholar]
- Townsend, JP; Cavalieri, D; Hartl, DL. Population genetic variation in genome-wide gene expression. Mol. Biol. Evol 2003, 20, 955–963. [Google Scholar]
- Williams, TD; Gensberg, K; Minchin, SD; Chipman, JK. A DNA expression array to detect toxic stress response in European flounder (Platichthys flesus). Aquat. Toxicol 2003, 65, 141–157. [Google Scholar]
- Oleksiak, MF; Roach, JL; Crawford, DL. Natural variation in cardiac metabolism and gene expression in Fundulus heteroclitus. Nat. Genet 2005, 37, 67–72. [Google Scholar]
- Fisher, MA; Oleksiak, MF. Convergence and divergence in gene expression among natural populations exposed to pollution. BMC Genomics 2007, 8, 108. [Google Scholar]
- Olohan, LA; Li, W; Wulff, T; Jarmer, H; Gracey, AY; Cossins, AR. Detection of anoxia-responsive genes in cultured cells of the rainbow trout Oncorhynchus mykiss (Walbaum), using an optimized, genome-wide oligoarray. J. Fish Biol 2008, 72, 2170–2186. [Google Scholar]
- Giger, T; Excoffier, L; Amstutz, U; Day, PJR; Champigneulle, A; Hansen, MM; Kelso, J; Largiader, CR. Population transcriptomics of life-history variation in the genus Salmo. Mol. Ecol 2008, 17, 3095–3108. [Google Scholar]
- Bernatchez, L. Ecological theory of adaptive radiation An empirical assessment from Coregonine fishes (Salmoniformes). In Evolution Illuminated, Salmon and Their Relatives; Hendry, AP, Stearns, SC, Eds.; Oxford University Press: New York NY, USA, 2004; pp. 175–207. [Google Scholar]
- Patterns of morphological changes and hybridization between sympatric whitefish morphs (Coregonus spp.) in a Swiss lake: a role for eutrophication? Mol. Ecol 2010, 19, 2152–2167.
- Berg, A; Grimaldi, E. A critical interpretation of the scale structures used for determination of annuli in fish growth studies. Mem. Ist. Ital. Idrobiol 1967, 21, 225–239. [Google Scholar]
- Rannala, B; Mountain, JL. Detecting immigration by using multilocus genotypes. Proc. Natl. Acad. Sci. USA 1997, 94, 9197–9201. [Google Scholar]
- Paetkau, D; Slade, R; Burden, M; Estoup, A. Genetic assignment methods for the direct, real-time estimation of migration rate: A simulation-based exploration of accuracy and power. Mol. Ecol 2004, 13, 55–65. [Google Scholar]
- Piry, S; Alapetite, A; Cornuet, JM; Paetkau, D; Baudouin, L; Estoup, A. GENECLASS2: A software for genetic assignment and first-generation migrant detection. J. Hered 2004, 95, 536–539. [Google Scholar]
- Renn, SCP; Aubin-Horth, N; Hofmann, HA. Biologically meaningful expression profiling across species using heterologous hybridization to a cDNA microarray. BMC Genomics 2004, 5, 42. [Google Scholar]
- Workman, C; Jensen, LJ; Jarmer, H; Berka, R; Gautier, L; Nielsen, HB; Saxild, HH; Nielsen, C; Brunak, S; Knudsen, S. A new non-linear normalization method for reducing variability in DNA microarray experiments. Genome Biol 2002, 16, 0041–0048. [Google Scholar]
- Sahai, H; Ageel, MI. The Analysis of Variance: Fixed, Random and Mixed Models; Birkhäuser: Boston, MA, USA, 2000. [Google Scholar]
- Excoffier, L; Smouse, PE; Quattro, JM. Analysis of molecular variance inferred from metric distances among DNA haplotypes-application to human mitochondrial-DNA restriction data. Genetics 1992, 131, 479–491. [Google Scholar]
- Benjamini, Y; Hochberg, Y. Controlling the false discovery rate-a practical and powerful approach to multiple testing. J. R. Stat. Soc. Series B 1995, 57, 289–300. [Google Scholar]
- Harris, MA; Clark, JI; Ireland, A; Lomax, J; Ashburner, M; Collins, R; Eilbeck, K; Lewis, S; Mungall, C; Richter, J; et al. The Gene Ontology (GO) project in 2006. Nucleic Acids Res 2006, 34, D322–D326. [Google Scholar]
- Camon, E; Barrell, D; Lee, V; Dimmer, E; Apweiler, R. The gene ontology annotation (GOA) database-An integrated resource of GO annotations to the UniProt knowledgebase. In Silico Biol 2004, 4, 5–6. [Google Scholar]
- Sokal, RR; Rohlf, FJ. Biometry: The Principles and Practices of Statistics in Biological Research, 3rd ed; W.H. Freeman: San Francisco, CA, USA, 1994. [Google Scholar]
- Prufer, K; Muetzel, B; Do, HH; Weiss, G; Khaitovich, P; Rahm, E; Paabo, S; Lachmann, M; Enard, W. FUNC: A package for detecting significant associations between gene sets and ontological annotations. BMC Bioinf 2007, 8, 41. [Google Scholar]
- Hartigan, J. Clustering Algorithms; Wiley: New York, NY, USA, 1975. [Google Scholar]
- Sneath, PHA; Sokal, RR. Numerical Taxonomy; Freeman: San Francisco, CA, USA, 1973. [Google Scholar]
- Saeed, AI; Sharov, V; White, J; Li, J; Liang, W; Bhagabati, N; Braisted, J; Klapa, M; Currier, T; Thiagarajan, M; et al. TM4: A free, open-source system for microarray data management and analysis. Biotechniques 2003, 34, 374–390. [Google Scholar]
- Paffl, W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 2001, 29, e9. [Google Scholar]
- St-Cyr, J; Derome, N; Bernatchez, L. The transcriptomics of life-history trade-offs in whitefish species pairs (Coregonus sp.). Mol. Ecol 2008, 17, 1850–1870. [Google Scholar]
- Cuesta, A; Esteban, MA; Meseguer, J. Effects of different stressor agents on gilthead seabream natural cytotoxic activity. Fish Shellfish Immunol 2003, 15, 433–441. [Google Scholar]
- Yin, Z; Lam, TJ; Sin, YM. The Effects of crowding stress on the nonspecific immune-response in fancy carp (Cyprinus-Carpio L). Fish Shellfish Immunol 1995, 5, 519–529. [Google Scholar]
- Ortuno, J; Esteban, MA; Meseguer, J. Effects of short-term crowding stress on the gilthead seabream (Sparus aurata L.) innate immune response. Fish Shellfish Immunol 2001, 11, 187–197. [Google Scholar]
- Douglas, SE. Microarray studies of gene expression in fish. OMICS: J. Integr. Biol 2006, 10, 474–489. [Google Scholar]
- Hook, SE; Skillman, AD; Gopalan, B; Small, JA; Schultz, IR. Gene expression profiles in rainbow trout, Onchorynchus mykiss, exposed to a simple chemical mixture. Toxicol. Sci 2008, 102, 42–60. [Google Scholar]
- Roberge, C; Paez, DJ; Rossignol, O; Guderley, H; Dodson, J; Bernatchez, L. Genome-wide survey of the gene expression response to saprolegniasis in Atlantic salmon. Mol. Immunol 2007, 44, 1374–1383. [Google Scholar]
- Williams, TD; Diab, A; Ortega, F; Sabine, VS; Godfrey, RE; Falciani, F; Chipman, JK; George, SG. Transcriptomic responses of European flounder (Platichthys flesus) to model toxicants. Aquat. Toxicol 2008, 90, 83–91. [Google Scholar]
- Gasque, P. Complement: a unique innate immune sensor for danger signals. Mol. Immunol 2004, 41, 1089–1098. [Google Scholar]
- Boshra, H; Li, J; Sunyer, JO. Recent advances on the complement system of teleost fish. Fish Shellfish Immunol 2006, 20, 239–262. [Google Scholar]
- Jacquiersarlin, MR; Gabert, FM; Villiers, MB; Colomb, MG. Modulation of antigen-processing and presentation by covalently-linked complement C3b fragment. Immunology 1995, 84, 164–170. [Google Scholar]
- Villiers, MB; Villiers, CL; JacquierSarlin, MR; Gabert, FM; Journet, AM; Colomb, MG. Covalent binding of C3b to tetanus toxin: Influence on uptake/internalization of antigen by antigen-specific and non-specific B cells. Immunology 1996, 89, 348–355. [Google Scholar]
- Boackle, SA; Morris, MA; Holers, VM; Karp, DR. Complement opsonization is required for presentation of immune complexes by resting peripheral blood B cells. J. Immunol 1998, 161, 6537–6543. [Google Scholar]
- Oertelt, S; Selmi, C; Invernizzi, P; Podda, M; Gershwin, ME. Genes and goals: An approach to microarray analysis in autoimmunity. Autoimmun. Rev 2005, 4, 414–422. [Google Scholar]
- Pernis, AB. Rho GTPase-mediated pathways in mature CD4* T cells. Autoimmun. Rev 2009, 8, 199–203. [Google Scholar]
- Moll, R; Bahn, H; Bayerl, C; Moll, I. Cellular adhesion molecules and components of the extracellular matrix as target structures of autoimmunity. Pathologe 1996, 17, 254–61. [Google Scholar]
- Clement, TM; Anway, MD; Uzumcu, M; Skinner, MK. Regulation of the gonadal transcriptome during sex determination and testis morphogenesis: Comparative candidate genes. Reproduction 2007, 134, 455–472. [Google Scholar]
- von Schalburg, KR; Cooper, GA; Yazawa, R; Davidson, WS; Koop, BF. Microarray analysis reveals differences in expression of cell surface and extracellular matrix components during development of the trout ovary and testis. Comp. Biochem. Physiol., Part D: Genomics Proteomics 2008, 3, 78–90. [Google Scholar]
- Mastellos, D; Lambris, JD. Complement: More than a ‘guard’ against invading pathogens? Trends Immunol 2002, 23, 485–491. [Google Scholar]
- Riley-Vargas, RC; Lanzendorf, S; Atkinson, JP. Targeted and restricted complement activation on acrosome-reacted spermatozoa. J. Clin. Invest 2005, 115, 1241–1249. [Google Scholar]
- von Schalburg, KR; Rise, ML; Brown, GD; Davidson, WS; Koop, BF. A comprehensive survey of the genes involved in maturation and development of the rainbow trout ovary. Biol. Reprod 2005, 72, 687–699. [Google Scholar]
- Von Schalburg, KR; McCarthy, SP; Rise, ML; Hutson, JC; Davidson, WS; Koop, BF. Expression of morphogenic genes in mature ovarian and testicular tissues: Potential stem-cell niche markers and patterning factors. Mol. Reprod. Dev 2006, 73, 142–152. [Google Scholar]
- Herskowitz, A; Ahmed-Ansari, A; Neumann, DA; Beschorner, WE; Rose, NR; Soule, LM; Burek, CL; Sell, KW; Baughman, KL. Induction of major histocompatibility complex antigens within the myocardium of patients with active myocarditis: A nonhistologic marker of myocarditis. J. Am. Coll. Cardiol. Found 1990, 15, 624–632. [Google Scholar]
- Holers, VM. Complement receptors and the shaping of the natural antibody repertoire. Springer Semin. Immunopathol 2005, 26, 405–423. [Google Scholar]
- Toskos, GC; Fleming, SD. Complement in Autoimmunity; Karger: Basel, Switzerland, 2004. [Google Scholar]
- Song, W-C. Complement in Autoimmunity; Karger: Basel, Switzerland, 2004. [Google Scholar]
- Koppang, EO; Bjerkas, I; Haugarvoll, E; Chan, EKL; Szabo, NJ; Ono, N; Akikusa, B; Jirillo, E; Poppe, TT; Sveier, H; et al. Vaccination-induced systemic autoimmunity in farmed Atlantic salmon. J. Immunol 2008, 181, 4807–4814. [Google Scholar]
- Secombes, CJ; Lewis, AE; Laird, LM; Needham, EA; Priede, IG. Role of autoantibodies in the autoimmune-response to testis in rainbow-trout (Salmo-Gairdneri). Immunology 1985, 56, 409–415. [Google Scholar]
- Lou, YH; Takahashi, H. The blood-testis barrier and its breakdown following immunization to testis material in the Nile tilapia, Oreochromis-Niloticus. Cell Tissue Res 1989, 258, 491–498. [Google Scholar]
- Laird, LM; Wilson, AR; Holliday, FGT. Lesions observed in the testis of precociously maturing male atlantic salmon, Salmo-Salar L. J. Fish Biol 1980, 17, 343–348. [Google Scholar]
- Fiorini, C; Tilloy-Ellul, A; Chevalier, S; Charuel, C; Pointis, G. Sertoli cell junctional proteins as early targets for different classes of reproductive toxicants. Reprod. Toxicol 2004, 18, 413–421. [Google Scholar]
- Banerjee, BD; Chakraborti, A; Suke, SG; Ahmed, RS; Tripathi, AK. Xenobiotic-induced immune alterations: Implications in health and disease. Indian J. Biochem. Biophys 2008, 45, 7–15. [Google Scholar]
- Bigazzi, PE. Autoimmunity caused by xenobiotics. Toxicology 1997, 119, 1–21. [Google Scholar]
- Bettini, E; Porta, AR; Dahmen, N; Wang, H; Margolis, FL. Expressed sequence tags (Est) identify genes preferentially expressed in catfish chemosensory tissues. Mol. Brain Res 1994, 23, 285–291. [Google Scholar]
- Porta, AR; Bettini, E; Buiakova, OI; Baker, H; Danho, W; Margolis, FL. Molecular cloning of ictacalcin: A novel calcium-binding protein from the channel catfish, Ictalurus punctatus. Mol. Brain. Res 1996, 41, 81–89. [Google Scholar]
- Cao, ZJ; Tanguay, RL; McKenzie, D; Peterson, RE; Aiken, JM. Identification of a putative calcium-binding protein as a dioxin-responsive gene in zebrafish and rainbow trout. Aquat. Toxicol 2003, 63, 271–282. [Google Scholar]
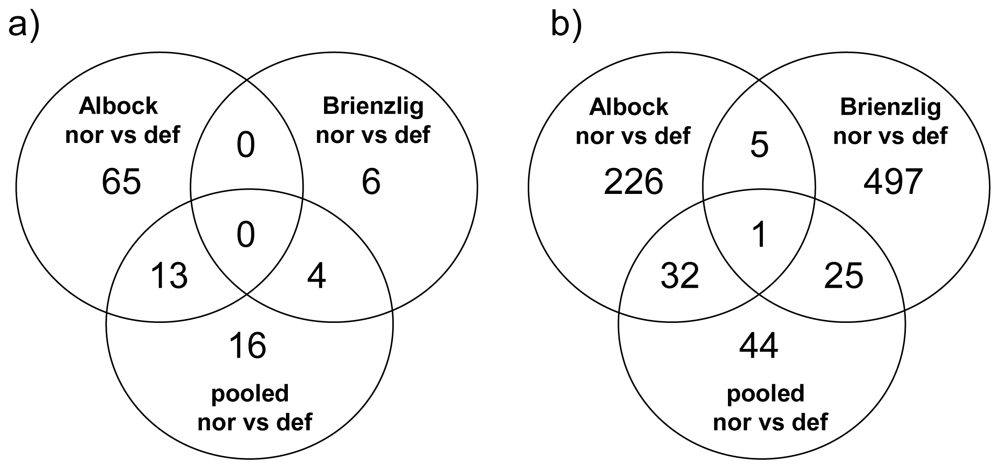
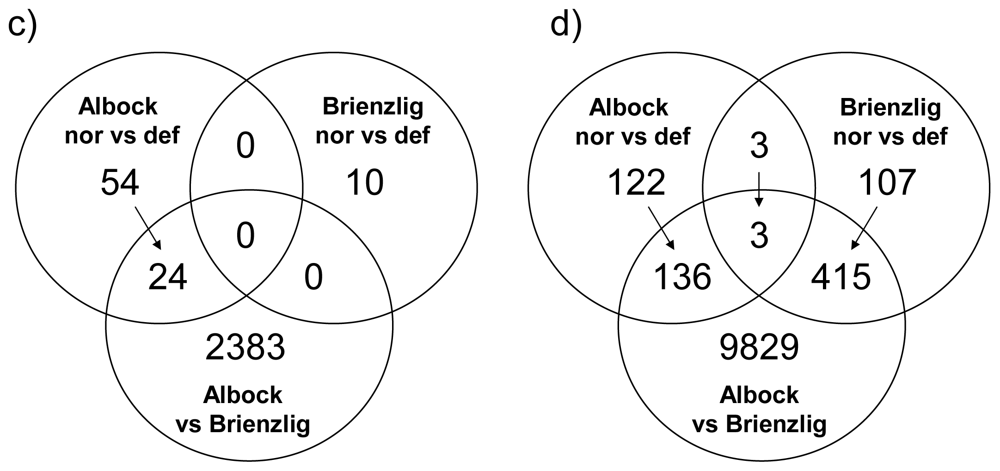

| Liver: GEXP = 7,013 | Comparison | 5% | FDR | 1% | FDR | FDR ≤ 5% |
|---|---|---|---|---|---|---|
| A | 346 (0.049) | 1 | 78 (0.011) | 0.88 | 0 | |
| B | 74 (0.011) | 1 | 10 (0.001) | 1 | 0 | |
| C | 177 (0.025) | 1 | 33 (0.005) | 1 | 0 | |
| D | 3,407 (0.486) | 0.1 | 2,407 (0.343) | 0.03 | 2,764 (0.394) | |
| Head kidney: GEXP = 17,834 | Comparison | 5% | FDR | 1% | FDR | FDR ≤ 5% |
| A | 1,162 (0.065) | 0.77 | 264 (0.015) | 0.68 | 0 | |
| B | 2,548 (0.143) | 0.35 | 528 (0.030) | 0.31 | 0 | |
| C | 651 (0.037) | 0.95 | 102 (0.006) | 0.95 | 0 | |
| D | 12,416 (0.696) | 0.01 | 10,382 (0.582) | 0.02 | 12,820 (0.719) | |
| (a) | Liver: Albock normal vs. Albock deformed | ||||
|---|---|---|---|---|---|
| GO root node | Name | GO ID | Genes | P | |
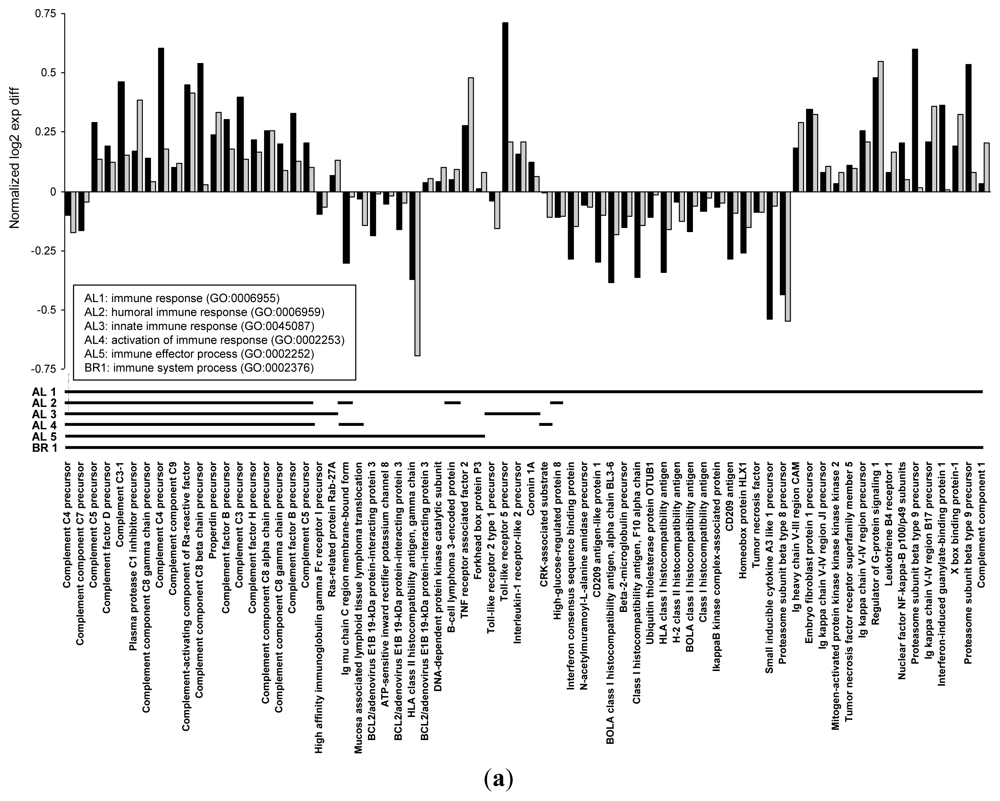
| biological_process | immune response | GO:0006955 | 70 | 0.000 | |
| biological_process | humoral immune response | GO:0006959 | 22 | 0.001 | |
| biological_process | innate immune response | GO:0045087 | 25 | 0.001 | |
| biological_process | activation of immune response | GO:0002253 | 22 | 0.002 | |
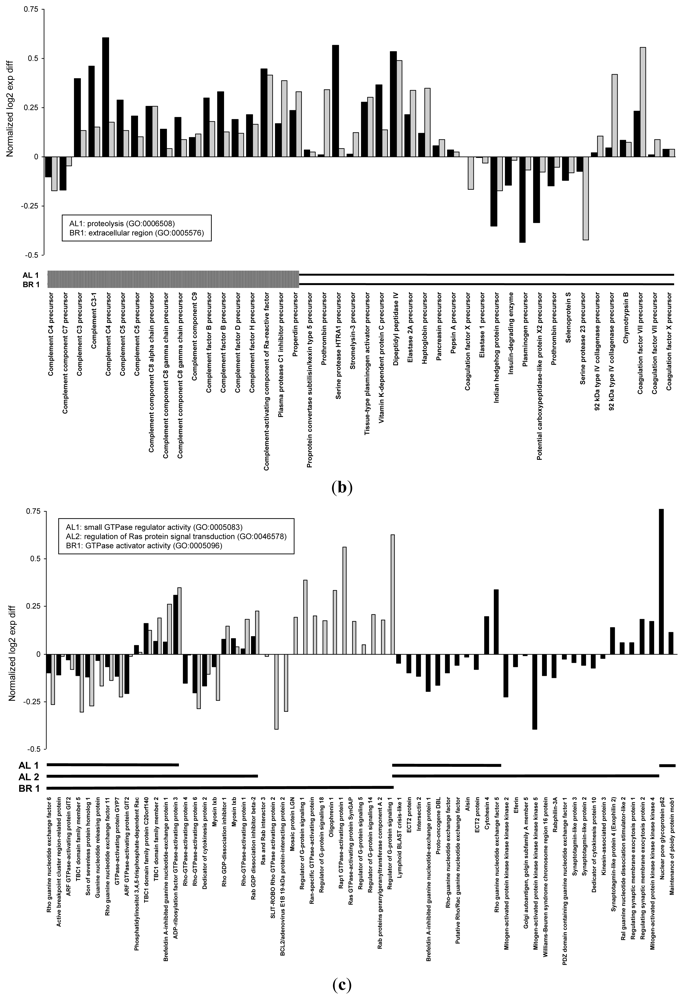
| biological_process | Proteolysis | GO:0006508 | 160 | 0.004 | |
| cellular_component | integral to plasma membrane | GO:0005887 | 60 | 0.009 | |
| biological_process | immune effector process | GO:0002252 | 32 | 0.009 | |
| (b) | Liver: Brienzlig normal vs. deformed | ||||
| GO root node | Name | GO ID | Genes | P | |
| cellular_component | extracellular region | GO:0005576 | 197 | 0.002 | |
| cellular_component | proteinaceous extracellular matrix | GO:0005578 | 31 | 0.002 | |
| biological_process | immune system process | GO:0002376 | 107 | 0.006 | |
| biological_process | cell adhesion | GO:0007155 | 85 | 0.009 | |
| (c) | Head kidney: Albock normal vs. deformed | ||||
| GO root node | Name | GO ID | Genes | P | |
| biological_process | regulation of cell proliferation | GO:0042127 | 114 | 0.000 | |
| biological_process | positive regulation of cell proliferation | GO:0008284 | 54 | 0.000 | |
| biological_process | embryonic development | GO:0009790 | 86 | 0.000 | |
| biological_process | protein import into nucleus | GO:0006606 | 23 | 0.001 | |
| molecular_function | ATP-dependent helicase activity | GO:0008026 | 20 | 0.001 | |
| biological_process | cell migration | GO:0016477 | 52 | 0.001 | |
| biological_process | embryonic development ending in birth or egg hatching | GO:0009792 | 58 | 0.002 | |
| molecular_function | small GTPase regulator activity | GO:0005083 | 49 | 0.004 | |
| biological_process | regulation of Ras protein signal transduction | GO:0046578 | 27 | 0.004 | |
| biological_process | cellular protein complex assembly | GO:0043623 | 41 | 0.009 | |
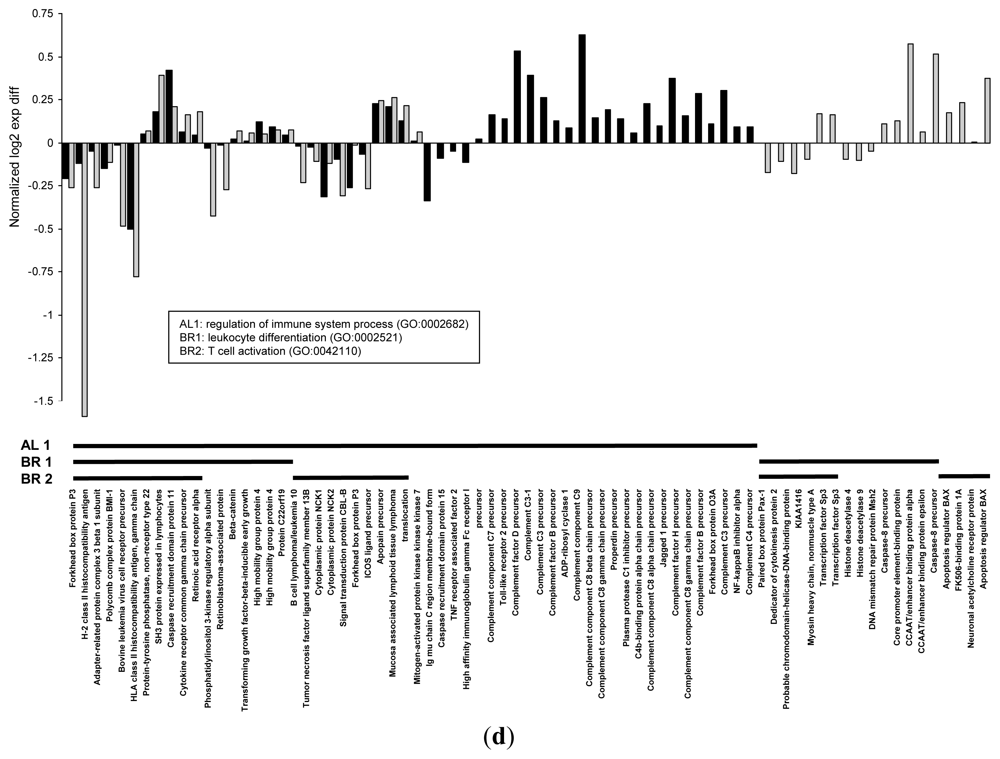
| biological_process | regulation of immune system process | GO:0002682 | 45 | 0.009 | |
| biological_process | central nervous system development | GO:0007417 | 45 | 0.010 | |
| (d) | Head kidney: Brienzlig normal vs. deformed | ||||
| GO root node | Name | GO ID | Genes | P | |
| molecular_function | GTPase activator activity | GO:0005096 | 36 | 0.003 | |
| biological_process | leukocyte differentiation | GO:0002521 | 32 | 0.006 | |
| biological_process | gamete generation | GO:0007276 | 40 | 0.006 | |
| biological_process | T cell activation | GO:0042110 | 31 | 0.008 | |
| cellular_component | soluble fraction | GO:0005625 | 32 | 0.009 | |
| biological_process | muscle contraction | GO:0006936 | 28 | 0.010 | |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bittner, D.; Cossins, A.R.; Segner, H.; Excoffier, L.; Largiadèr, C.R. Identification of Candidate Genes and Physiological Pathways Involved in Gonad Deformation in Whitefish (Coregonus spp.) from Lake Thun, Switzerland. Int. J. Environ. Res. Public Health 2011, 8, 2706-2733. https://doi.org/10.3390/ijerph8072706
Bittner D, Cossins AR, Segner H, Excoffier L, Largiadèr CR. Identification of Candidate Genes and Physiological Pathways Involved in Gonad Deformation in Whitefish (Coregonus spp.) from Lake Thun, Switzerland. International Journal of Environmental Research and Public Health. 2011; 8(7):2706-2733. https://doi.org/10.3390/ijerph8072706
Chicago/Turabian StyleBittner, David, Andrew R. Cossins, Helmut Segner, Laurent Excoffier, and Carlo R. Largiadèr. 2011. "Identification of Candidate Genes and Physiological Pathways Involved in Gonad Deformation in Whitefish (Coregonus spp.) from Lake Thun, Switzerland" International Journal of Environmental Research and Public Health 8, no. 7: 2706-2733. https://doi.org/10.3390/ijerph8072706





