1. Introduction
From a public health perspective [
1] the need for effective waste treatments and sludge stabilisation is ever increasing. With increased travel [
2] and tourism the potential for diffusion of infectious diseases has increased and throughout Europe and elsewhere many diseases have (re)-emerged resulting in major health, ecological, socio-economical and political consequences. The establishment of EDEN (Emerging Diseases in a changing European Environment;
http://www.eden-fp6project.net) is attempting to spread the message of the importance of developing and implementing advanced wastewater and sludge treatment systems [
3]. As an alternative to standard sludge treatment systems, a new, energy-efficient, non-chemical process, autothermal thermophilic aerobic digestion (ATAD) was introduced over the past decade or so [
4]. ATAD is a biological sludge treatment process that utilises the aerobic degradative abilities of microorganisms to convert soluble organic materials to lower energy forms [
5]. Microbial catabolic processes resulting from aerobic oxidation of biosolids, gives rise to significant heat generation, which in turn results in thermal processing of the sludge [
6–
10]. Typically ATAD processes that operate to treat domestic sludge, following primary and secondary wastewater processing, involve sludge thickening, followed by a two stage thermal treatment where the sludge is highly aerated and the sludge temperature allowed to rise naturally due to microbial metabolism in insulated reactors [
4,
6,
8]. This metabolic activity reduces the sludge solids content while the elevated temperatures should effectively stabilise the sludge and remove pathogens. In typical ATAD systems treating domestic sludge the temperature ranges between 45 °C and 65 °C, which ensures the best degradation rates while the temperature rise aids pathogen reduction. However above 60 °C, the degree of microbial diversity is markedly decreased, at least in ATAD systems treating pharmaceutical wastes, with negative consequences on the degradation process [
11].
Biosolids reuse and disposal practices are currently regulated in Ireland by EU directive [
12] and by a code of practice for biosolids reuse [
13].The EU directive suggests a treatment regime of 55 °C for 20 hours, an absence of
Salmonella in 50 g of sludge and process conditions to reduce
E. coli CFU to <500 g
−1. The pasteurisation ability of an ATAD process to produce such a class A biosolids is determined by the interplay between operational parameters, feed composition and its biodegradability, the diversity of the ATAD microbial consortia and their metabolic capacities and reactor design features such as insulation and aeration control [
9,
14] and thermotolerent coliforms and
Enterococci are most often used as indicator organisms to assess the hygienic quality of such treated organic waste [
15–
23]. In addition
Salmonella is a relatively common excreted pathogen, often present in sludge and sewage and hence is also monitored as an indicator organism [
17,
22,
24].
The effect of heat on microorganisms has mainly been studied with respect to pathogen removal in food and wastewater and it has been assumed that the death kinetics of enteric viruses and protozoa are similar [
25]. The exponential law of disinfection, referred to as “Chick’s Law” relates the survival of pathogens and other microorganisms as a function of temperature:
where
Xt is the surviving fraction following treatment,
Xo is the starting population number,
t-to is the treatment time interval,
k is the specific decay rate [
17,
25,
26]. In general, studies on the thermal effects and heat inactivation of pathogens have been carried out under laboratory conditions with application of test bacteria [
17,
27], with
Salmonella and
Escherichia spp. being used as model indicator bacteria [
28–
30]. For the pathogen
Salmonella enterica, inactivation is dependent on the strain type, and it is most effectively inactivated at 71 °C during 1.2 seconds [
27] in a nutrient-rich environment. However, inactivation of enterococci in pure culture [
17] required 40 days at 45 °C, 3 days at 50 °C, 15 hours at 55 °C, 2 hours at 60 °C, and 7 minutes at 70 °C. For pathogen inactivation in sludge [
20], it has been shown that holding for 4 hours at 55 °C or for 30 minutes at 70 °C is effective in killing most pathogens, with killing depending on the matrix or media. The heat resistance of a pathogen is highly influenced by the strain tested, the type of experimental method used, culture conditions prior to the experiment and the heating method and the recovery conditions utilised. Thus there are many features that could add to variability in pathogen recovery under real sludge treatment conditions.
For sludge, adequate pathogen reduction can be obtained at time-temperature combinations that are described in detail by U.S. EPA’s time–temperature equation [
5]. The time-temperature equation in the regulation requires that all particles be treated for a specified time at the temperature of operation. In brief, this requirement suggests a temperature and time of sludge treatment to achieve desirable quality of 65 °C for 1 hour, 75 °C for 136 seconds, 85 °C for 5.5 seconds or 55 °C for 24 hours. These requirements differ from the conditions necessary for pathogen inactivation in pure culture under laboratory conditions [
25,
30,
32], as in wastewater or sludge microorganisms are often found within organic substances or embedded in flocs, which can protect them from pasteurization [
25]. It has been demonstrated that treating domestic sludge at 60 °C for 35 minutes would reduce pathogens to acceptable levels [
33], in comparison to pasteurization at 70 °C with a retention time of 30 min while at 60 °C full inactivation of pathogens within sludge requires a holding time of 4.78 h [
5,
34].
Cell-free nucleic acid is a potentially important source of energy and nutrients in sludge ecosystems [
35,
36]. However, little is known about the identity, metabolism, and interactions of the microorganisms capable of consuming cell-free DNA originating from cell lysis or from the original fecal materials. Some may undergo genetic transformation [
37], some may be used for DNA or RNA synthesis [
35,
38] or it may give a valuable source of phosphorus depending on the ability of microorganisms to take up this macromolecule with passive transport limited to >0.6 kDa [
39,
40]. Some microorganisms are capable of degrading cell-free DNA with extracellular nucleases [
42] and then consume the hydrolyzed products [
35], while DNA that interacts with sludge material may be protected [
42]. DNA hydrolysing bacteria from aquatic environments have been described [
43,
44] and assigned to mesophilic or psychroplilic organisms in the main. In wastewater ecosystems, it can be expected that the size and availability of the cell-free DNA and how microorganisms interact with it will have a strong effect on the diversity and metabolism of micro-organisms in wastewater and sludge [
45]. There have been few reports on such activities with only one by Ruiz [
46] describing the detection of DN
ase activity in mesophilic anaerobic wastewater sludge. No data on DN
ase production by thermophilic organisms and its possible role in thermal treatment of sludge has been reported. Such DN
ases may also play a key role in preventing transmission of viruses, or mobile elements from sludge to the wider environment via gene transfer [
47] which has contributed to genome plasticity and dissemination of fitness-enhancing traits, including antibiotic resistance and virulence factors [
48–
50]. Recent studies suggest that sludge is a specific location where genetic exchange can occur [
47] and limiting such exchange may be particularly important for domestic sludge where antibiotic-resistant bacteria occur [
51–
54]. Although lysis can occur, natural transformation and DNA uptake is known to be responsible for genetic spread under mesophilic treatment conditions [
47,
55–
58] and may even occur at thermophilic processing temperatures [
59].
The first full scale ATAD plant to operate in Ireland is located in Killarney to treat locally produced primary and secondary sludge [
60]. Insufficient treatment could lead to contamination by pathogenic microorganisms when the stabilised sludge is utilised for land spread. Thus we wished to determine the suitability of this ATAD process and sludge type for pathogen reduction [
21] by monitoring the sludge seasonally using traditional culture based and molecular profiling techniques. We also wished to examine the effect of high levels of nuclease activity detected in the sludge on eliminating mobile DNA elements responsible for transmission of antibiotic resistance determinants.
4. Discussion
Although ATAD plants treating domestic sludge have been operating worldwide for many years, there has been limited systematic analysis of their actual potential to remove pathogens [
14,
15,
79]. There has always been the ‘presumption’ that because of the time-temperature relationship between sludge holding in the thermophilic reactors that this would lead to the removal of coliforms, making the sludge pathogen free and allowing its classification as a Class A Biosolids.
There are, however, many factors that could effect this presumption. These range from inadequate holding times, thermal protection of organisms by the sludge biosolids [
61] or re-growth of viable non-culturable coliforms after the sludge treatment process.
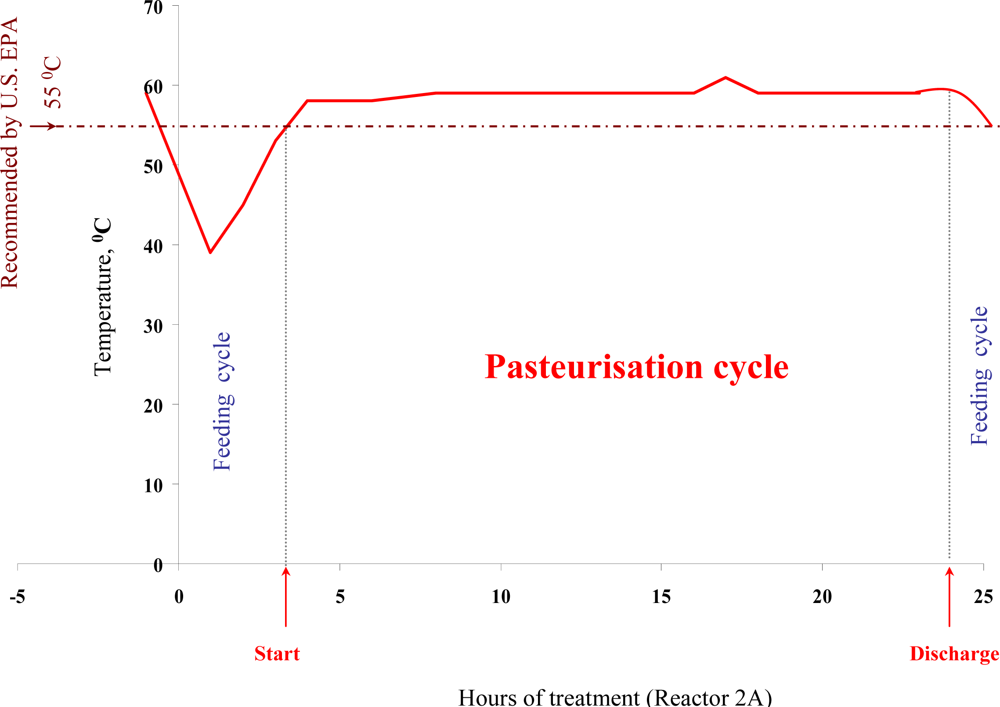
Bio-flocculation, production and embedding of microorganisms into a sludge Extra Cellular Polysaccharide (EPS) matrix, sludge structure, nutrient availability and heat distribution within reactors can all be important factors influencing the actual performance of a full-scale ATAD sludge treatment system. In addition, other factor such as climate, operation, and pathogen densities may fluctuate from site to site. Thus we have examined the thermal inactivation of pathogens in a full scale ATAD system at Killarney
de novo over an extended 15 month period. Using a two-factor Weibull model based on temperature and retention time, we were able to obtain good agreement between calculated and observed rates of kill of
Salmonella spp. and fecal coliforms (
Figure 1). Times of exposure of the sludge to inactivation temperatures were found to be sufficient to meet the requirements of both the EU and the US EPA standards [
5,
12,
13]. Our data provides evidence for pathogen reduction and also verifies their absence at the post treatment stage, during storage and prior to land spread. Autothermal processing capitalises on microbial exothermic reactions and process monitoring at the Killarney ATAD indicates that temperatures regularly reach 65 °C but can fluctuate depending on addition of new sludge and other process parameters [
60].
In general ATAD temperatures operate between 55–65 °C on a consistent basis even allowing for addition of new sludge and removal of treated matter. In addition to heat, changes in pH (
Table 2) due to the accumulation of NH
3, the presence of metabolic antagonistic compounds produced by indigenous microflora, microbiological competition for nutrients with developing thermophilic populations [
80,
81] and water content may all play a synergistic role. During ATAD high levels of ammonia are released from the degradation of proteins and as its solubility increases with temperature, this result in pH changes in the bulk water. Ammonia is highly soluble in water which is partly explained by its polarity and ability to form hydrogen bonds [
82]. In aqueous solution, ammonia acts as a weak base producing hydroxide ions by the de-protonation of water. Various authors have demonstrated that molecular ammonia has a bactericidal effect on enteric pathogen [
83–
86].
Culture based analysis of pathogens applied to ATAD sludge have limitations because of the possibility of bacteria entering a viable but non-culturable state (VBNC) as a strategy for temperature or adverse condition survival or the inability to provide growth conditions for unknown ATAD consortia. DNA based or microscopic analysis are thus needed for routine application which would provide data on the presence of pathogens, their survival during treatment and their potential out growth later during land application.