Radioactivity of Tobacco Leaves and Radiation Dose Induced from Smoking
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Radionuclide Concentrations
3.2. Effective Dose Estimate
4. Conclusions
References
- Martell, EA. Radioactivity of tobacco trichomes and insoluble cigarette smoke particles. Nature 1974, 249, 215–217. [Google Scholar]
- Tso, TC; Harley, N; Alexander, LT. Source of lead-210 and polonium-210 in tobacco. Science 1966, 153, 880–882. [Google Scholar]
- Francis, CWM; Chesters, G; Erhardt, WE. 210Polonium entry into plants. Environ. Sci. Technol 1968, 2, 690–695. [Google Scholar]
- Chakraborty, MK; Weybrew, JA. Tobacco Sci 1963, 7, 122.
- Barrera, R; Werusman, EW. Tobacco Sci 1966, 10, 157.
- Papastefanou, C. Radiological impact from atmospheric releases of 238U and 226Ra from phosphate rock processing plants. J. Environ. Radioactivity 2001, 54, 75–83. [Google Scholar]
- Papastefanou, C; Bondietti, EA. Mean residence times of atmospheric aerosols in the boundary layer as determined from 210Bi/210Pb activity ratios. J. Aerosol Sci 1991, 22, 927–931. [Google Scholar]
- Papastefanou, C; Manolopoulou, M; Charalambous, S. Radiation measurements and radioecological aspects of fallout from the Chernobyl reactor accident. J. Environ. Radioactivity 1988, 7, 49–64. [Google Scholar]
- Martell, EA. Radioactivity in cigarette smoke. N. Engl. J. Med 1982, 307, 309–313. [Google Scholar]
- Martell, EA; Sweder, KS. The roles of polonium isotopes in the etiology of the lung cancer in cigarette smokers and uranium miners. In In Proceedings of the International Conference on Radiation Hazards in Mining, USA, 4– 9 October 1981; Gomez, M, Ed.; Colorado School of Mines: Golden, CO; AIME: NY, 1982; pp. 383–389. [Google Scholar]
- Martell, EA. Tobacco radioactivity and cancer in smokers. Am. Sci 1975, 63, 404–412. [Google Scholar]
- Martell, EA; Sweder, KS. Properties of radon progeny aerosols in mainstream cigarette smoke and the alpha dose at segmental bifurcations of smokers. In In Proceedings of Radiation Research Society Special Workshop on Current Topics in Lung Dosimetry, Salt Lake City, Utah, 21–22 April 1982; US. Government Printing Office: Washington D.C, 1983; pp. 144–151. [Google Scholar]
- Radford, EP, Jr; Hunt, VR. Polonium-210: A volatile radioelement in cigarette. Science 1964, 143, 247–249. [Google Scholar]
- Winters, TH; Di Franza, JR. Radioactivity in cigarette smoking. N. Engl. J. Med 1982, 306, 364–365. [Google Scholar]
- Takizawa, Y; Zhang, L; Zhao, L. 210Pb and 210Po in tobacco- with a special focus on estimating the doses to man. J. Radioanal. Nucl. Chem. Articles 1994, 182, 119–125. [Google Scholar]
- Peres, AC; Hiromoto, G. Evaluation of 210Pb and 210Po in cigarette tobacco produced in Brazil. J. Environ. Radioactivity 2002, 62, 115–119. [Google Scholar]
- Colangelo, CH; Huguet, MR; Palacios, MA; Oliveira, AA. Levels of 210Po in some beverages and in tobacco. J. Radioanal. Nucl. Chem. Lett 1992, 166, 195–202. [Google Scholar]
- Khater, AE. Polonium-210 budget in cigarettes. J. Environ. Radioactivity 2004, 71, 33–41. [Google Scholar]
- Eisenbud, M; Gesell, T. Environmental Radioactivity, 4th ed; Academic Press: San Diego, USA, 1997. [Google Scholar]
- Kelley, TF. Polonium-210 content of mainstream cigarette smoke. Science 1965, 149, 537–538. [Google Scholar]
- Holtzman, RB; Ilcewicz, FH. Lead-210 and polonium-210 in tissues of cigarette smokers. Science 1966, 153, 1259–1260. [Google Scholar]
- Manolopoulou, M. A radioecological study of the coal power plant environment, Ph.D. Thesis. Aristotle University of Thessaloniki: Thessaloniki, Greece, 1990; 178.
- UNSCEAR United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and Effects of Ionizing Radiation; UNSCEAR: New York, NY, USA, 2000. [Google Scholar]
- Papastefanou, C; Manolopoulou, M; Stoulos, S; Ioannidou, A. Behavior of 137Cs in the environment one decade after Chernobyl. J. Radioecology 1996, 4, 9–14. [Google Scholar]
- NRPB. National Radiological Protection Board. Committed equivalent organ doses and committed effective doses from intakes of radionuclides; NRPB; Report M. Chilton, Didcot, UK, 1991; p. 288. [Google Scholar]
- UNSCEAR United Nations Scientific Committee on the Effects of Atomic Radiation. Sources and Effects of Ionizing Radiation; UNSCEAR: New York, NY, USA, 1993. [Google Scholar]
- ICRP International Commission on Radiological Protection. Age-dependent doses to members of the public from intake of radionuclides; Part 5: Compilation of ingestion and inhalation dose coefficients ICRP Publication 72. Pergamon Press: Oxford, UK, 1996. [Google Scholar]
- EC. European Commission. Investigation of a possible basis for a common approach with regard to the restoration of areas affected by lasting radiation exposure as a result of past or old practice or work activity. In CARE Final Report; Vandenhove, H, Bousher, A, Jensen, PH, Jackson, D, Lambers, B, Zeevaert, T, Eds.; European Commission DG XI Environment, Nuclear Safety and Civil Protection, under contract 96-ET-006, Brussels, Belgium, 1999. [Google Scholar]
- Fletcher, JJ. Doses from radiocesium and 40K activities found in some tobacco leaves and cigarettes. Appl. Radiat. Isot 1994, 45, 133–134. [Google Scholar]
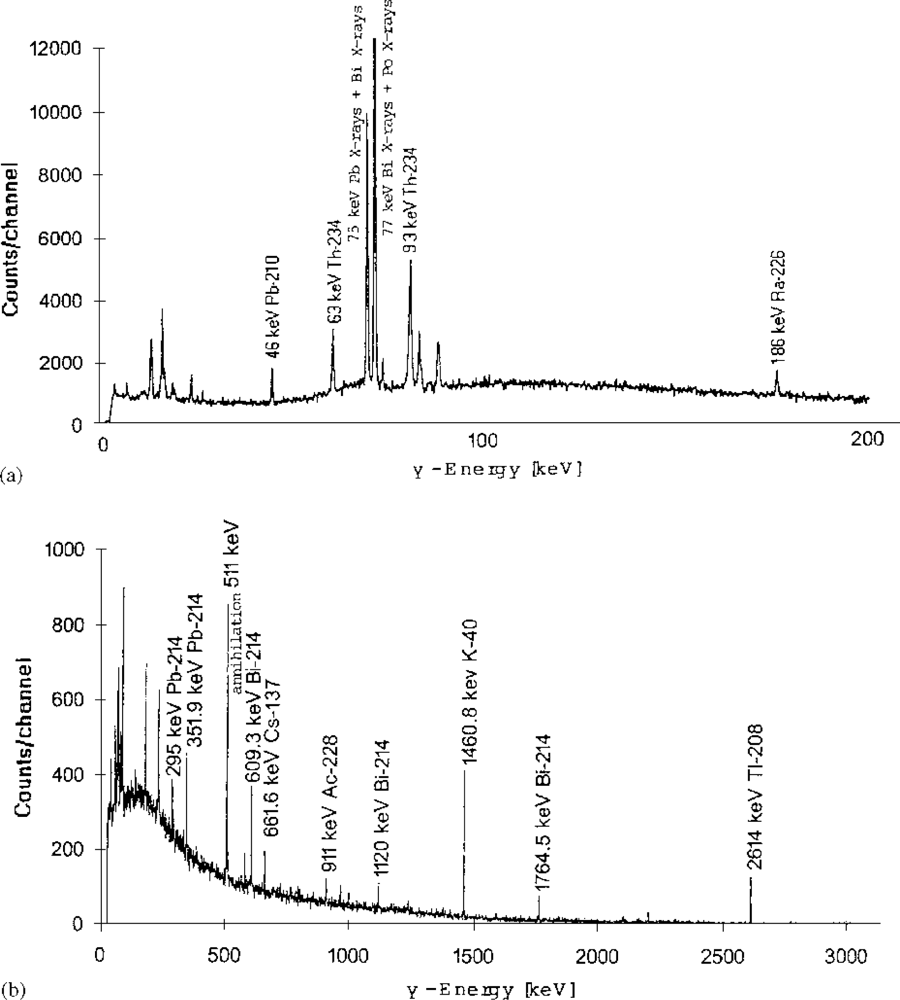
| No | Lab.No. | 226Ra | 210Pb | 228Ra | 40K | 137Cs | 134Cs |
|---|---|---|---|---|---|---|---|
| 1 | TAB-2 | 6±1 | 16±1 | 5±1 | 819±20 | 7±1 | M.D.A |
| 2 | TAB-3 | 2±1 | 16±5 | 6±1 | 897±21 | 4±1 | M.D.A |
| 3 | TAB-4 | 3±2 | 17±5 | 5±1 | 619±270 | 6±1 | M.D.A. |
| 4 | TAB-5 | 4±1 | 18±4 | 5±1 | 825±20 | 7±1 | M.D.A. |
| 5 | TAB-6 | 3±1 | 9±4 | 5±1 | 771±19 | 5±1 | M.D.A. |
| 6 | TAB-7 | 2±1 | 6±4 | 3±1 | 872±16 | 6±1 | M.D.A. |
| 7 | TAB-8 | 8±1 | 14±5 | 7±1 | 967±22 | 2±1 | M.D.A. |
| 8 | TAB-9 | 3±1 | 16±5 | 3±1 | 618±16 | 3±1 | - |
| 9 | TAB-10 | 3±1 | 18±5 | 5±1 | 745±19 | 2±1 | M.D.A. |
| 10 | TAB-11 | 3±1 | 14±4 | 4±1 | 751±14 | 3±1 | M.D.A. |
| 11 | TAB-12 | 2±1 | 17±5 | 1±1 | 756±20 | 2±1 | M.D.A |
| 12 | TAB-13 | 4±1 | 8±5 | 3±1 | 879±18 | 2±1 | M.D.A. |
| 13 | TAB-14 | 3±1 | 16±5 | 3±1 | 2080±26 | 2±1 | M.D.A. |
| 14 | TAB-15 | 2±2 | 17±4 | 3±1 | 1110±20 | M.D.A. | M.D.A. |
| 15 | TAB-16 | 3±1 | 15±5 | 4±1 | 493±16 | 1±1 | M.D.A |
| 16 | TAB-17 | 3±1 | 7±4 | 2±1 | 273±13 | 2±1 | M.D.A |
| 17 | TAB-18 | 3±1 | 17±4 | 4±1 | 503±16 | 4±1 | 0.6±0.2 |
| Radionuclide | Sv Bq−1 | Reference |
|---|---|---|
| 226Ra | 3.50x10−6 | [27–28] |
| 228Ra | 2.60x10−6 | [27–28] |
| 210Pb | 1.10x10−6 | [27–28] |
| 210Po | 3.30x10−6 | [27–28] |
| 137Cs | 8.70x10−9 | [25–26] |
| No. | Lab.No. | 226Ra (μSv y−1) | 210Pb (μSv y−1) | 228Ra (μSv y−1) | 137Cs (nSv y−1) |
|---|---|---|---|---|---|
| 1 | TAB-2 | 146.47 | 121.57 | 89.36 | 410.39 |
| 2 | TAB-3 | 50.24 | 114.89 | 98.47 | 235.68 |
| 3 | TAB-4 | 74.06 | 122.31 | 83.93 | 367.01 |
| 4 | TAB-5 | 86.33 | 134.91 | 88.83 | 410.39 |
| 5 | TAB-6 | 70.28 | 63.90 | 84.28 | 275.55 |
| 6 | TAB-7 | 55.67 | 47.00 | 53.61 | 371.69 |
| 7 | TAB-8 | 178.55 | 106.74 | 115.99 | 99.67 |
| 8 | TAB-9 | 76.89 | 119.34 | 55.01 | 175.88 |
| 9 | TAB-10 | 74.77 | 129.72 | 81.12 | 93.80 |
| 10 | TAB-11 | 65.89 | 104.52 | 72.19 | 191.12 |
| 11 | TAB-12 | 42.46 | 127.50 | 19.28 | 99.67 |
| 12 | TAB-13 | 86.80 | 56.48 | 52.74 | 123.12 |
| 13 | TAB-14 | 73.59 | 119.34 | 46.96 | 124.29 |
| 14 | TAB-15 | 51.42 | 128.98 | 51.34 | — |
| 15 | TAB-16 | 77.36 | 111.93 | 61.67 | 70.35 |
| 16 | TAB-17 | 73.12 | 47.96 | 30.84 | 89.70 |
| 17 | TAB-18 | 74.06 | 122.31 | 61.15 | 249.17 |
Share and Cite
Papastefanou, C. Radioactivity of Tobacco Leaves and Radiation Dose Induced from Smoking. Int. J. Environ. Res. Public Health 2009, 6, 558-567. https://doi.org/10.3390/ijerph6020558
Papastefanou C. Radioactivity of Tobacco Leaves and Radiation Dose Induced from Smoking. International Journal of Environmental Research and Public Health. 2009; 6(2):558-567. https://doi.org/10.3390/ijerph6020558
Chicago/Turabian StylePapastefanou, Constantin. 2009. "Radioactivity of Tobacco Leaves and Radiation Dose Induced from Smoking" International Journal of Environmental Research and Public Health 6, no. 2: 558-567. https://doi.org/10.3390/ijerph6020558
APA StylePapastefanou, C. (2009). Radioactivity of Tobacco Leaves and Radiation Dose Induced from Smoking. International Journal of Environmental Research and Public Health, 6(2), 558-567. https://doi.org/10.3390/ijerph6020558




