Environmental Factors Correlated with Culturable Enterococci Concentrations in Tropical Recreational Waters: A Case Study in Escambron Beach, San Juan, Puerto Rico
Abstract
:1. Introduction
2. Materials and Methods
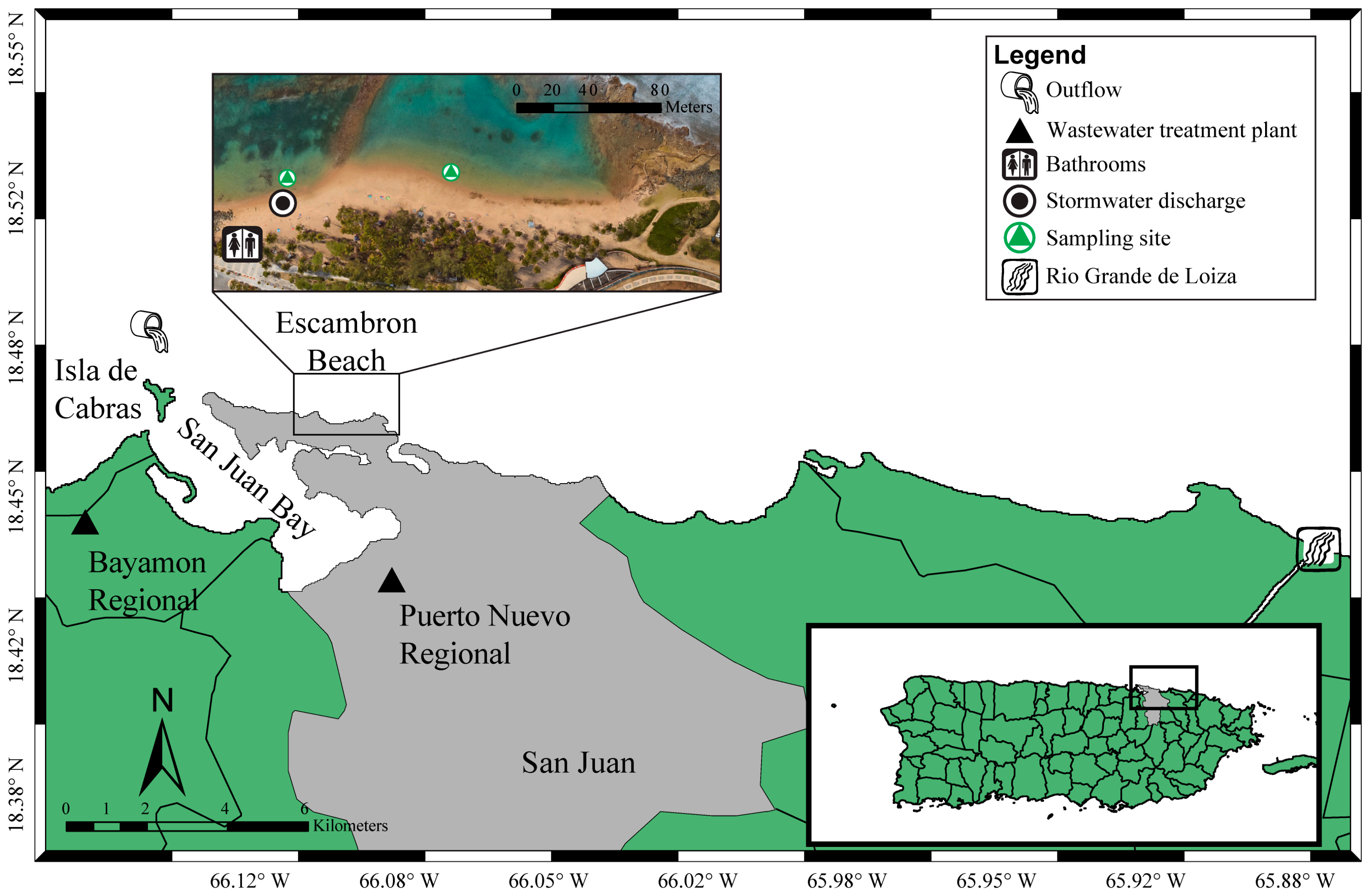
2.1. Escambron Beach, San Juan, Puerto Rico
2.2. Culturable Enterococci Data
2.3. Satellite-Derived and In Situ Environmental Data
2.4. Data Identified as Below the Limit of Detection
2.5. Non-Parametric Statistical Analyses
3. Results
3.1. Modeling Culturable Enterococci Using Akaike Information Criterion Model and Correlation Analyses
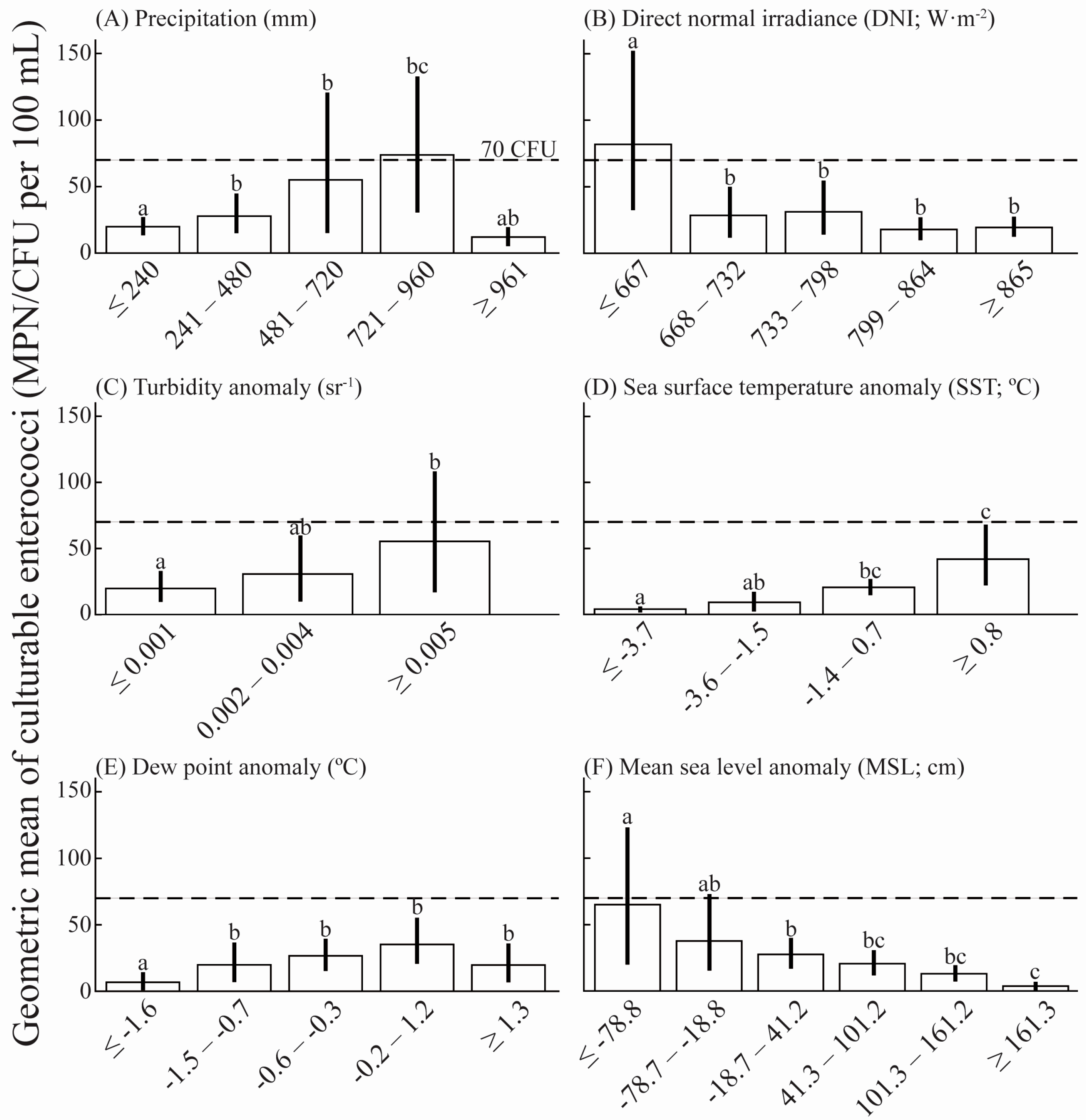
3.2. Environmental Variables Influence on Culturable Enterococci
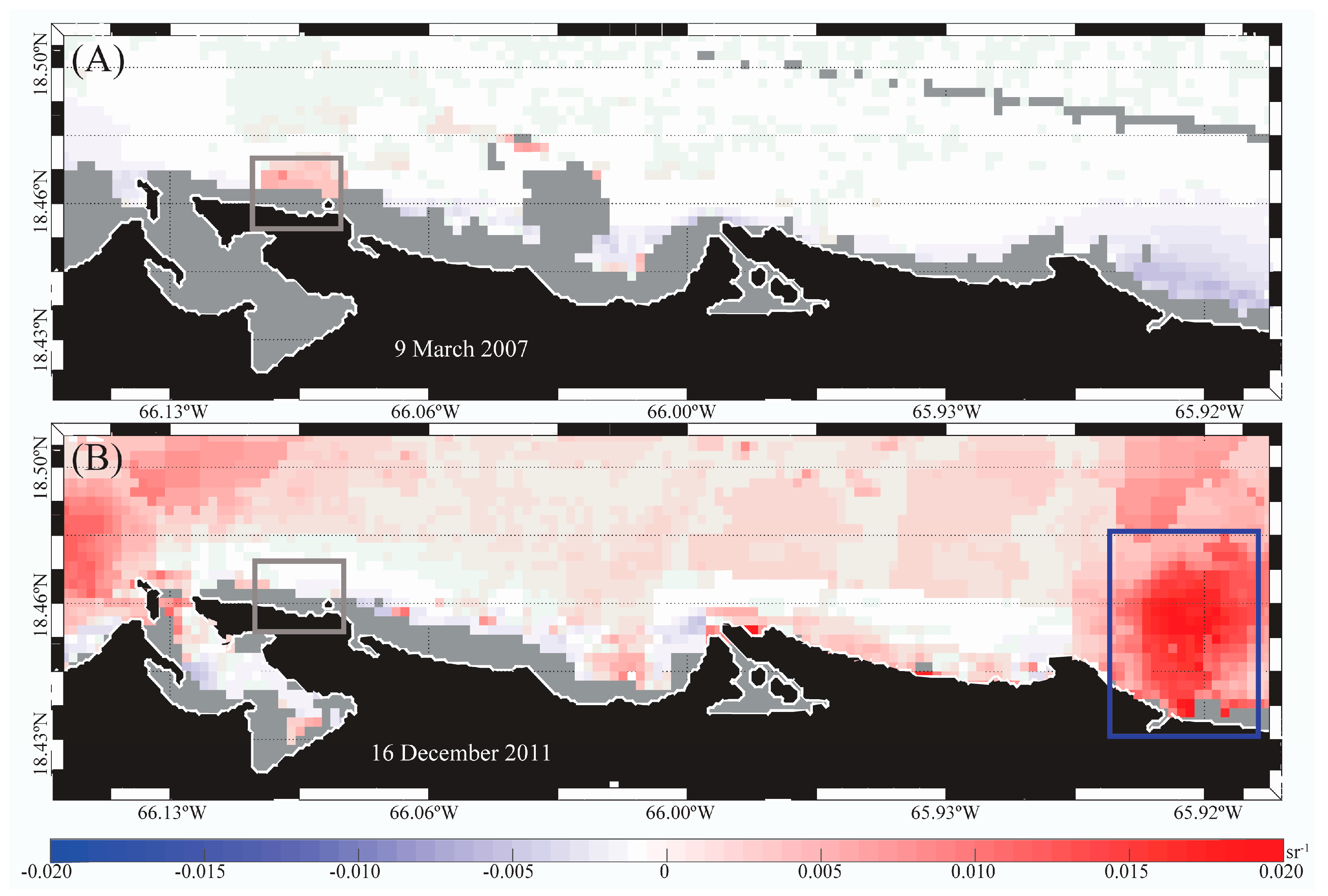
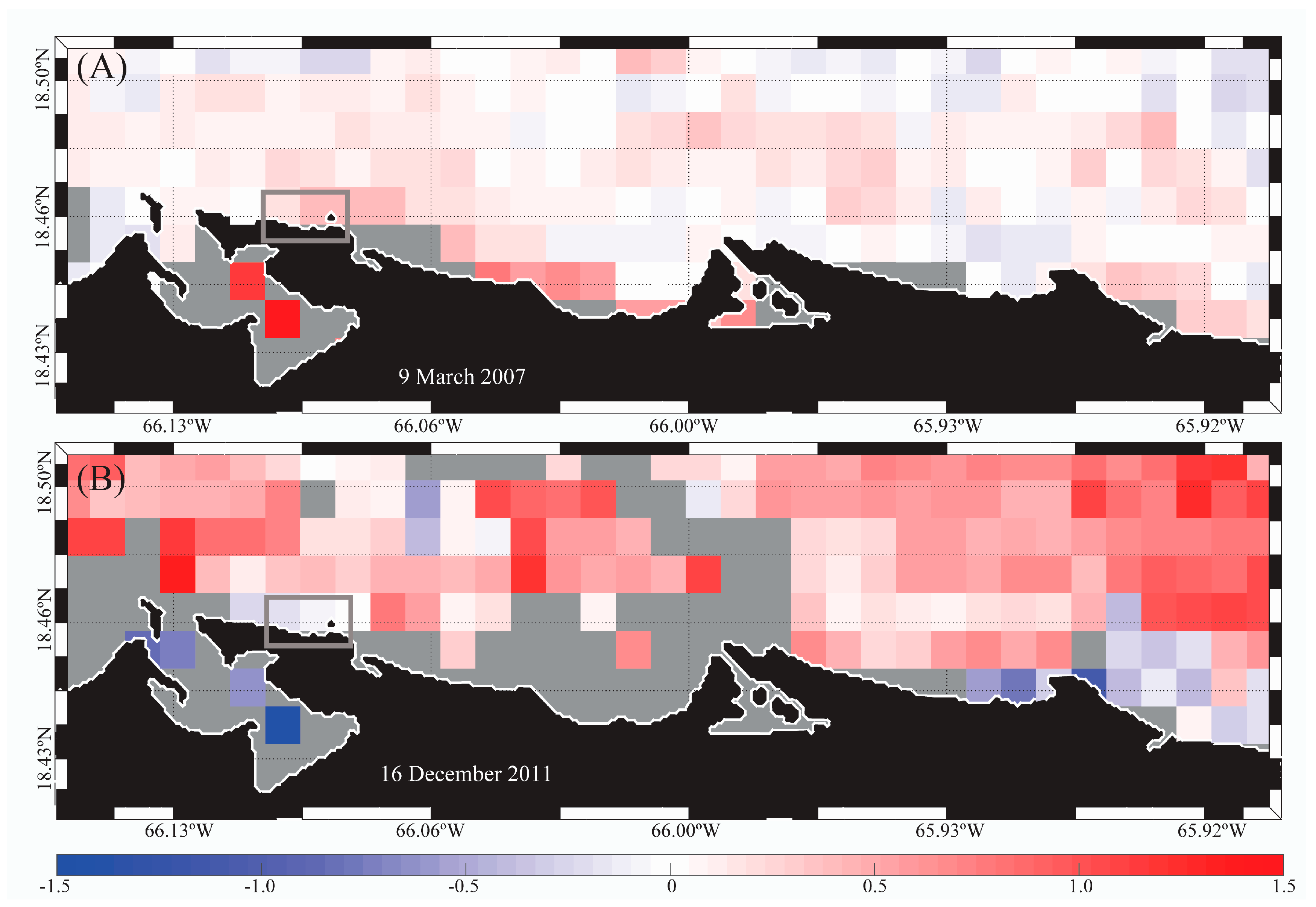
3.3. Satellite-Derived SST and Turbidity Anomaly Images to Anticipate Potential Beach Advisories
4. Discussion
4.1. Environmental Factors Associated with Culturable Enterococci Variability
4.2. Sanitation Infrastructure, Human Fecal Pollution, and Culturable Enterococci Variability
4.3. Satellite-Derived SST and Turbidity Anomaly Images, and PREQB Beach Advisories
4.4. Future Work
5. Conclusions
- Environmental factors (i.e., direct normal irradiance (DNI), mean sea level (MSL), precipitation, turbidity, and sea surface temperature (SST)) explained 20% of the enterococci variability observed in Escambron Beach surface waters during 2005–2015.
- Identified time-lags for the different environmental factors helped better understand variability in culturable enterococci in Escambron Beach surface waters due to environmental factors.
- Increased enterococci concentrations were observed at strong positive SST and turbidity anomalies; conversely, these concentrations decreased with increased DNI and MSL anomalies in Escambron Beach surface waters.
- Specific ranges of precipitation (i.e., 481–960 mm) promoted increased enterococci concentrations, potentially due to urban run-off (e.g., resuspension of solids, soil runoff, and non-human sources), combined sewer overflow events, and/or increased leaching of septic tanks.
- The combined effects of environmental factors can help model culturable enterococci concentrations and understand ranges where these would exceed recommended 2014 US EPA BAV at Escambron Beach.
- Satellite-derived data can improve beach water quality assessments, potentially reducing in-situ sampling efforts as this data is readily available, and help identify events for early warning systems and improve beach advisory.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lugo, A.E.; González, O.M.R.; Pedraza, C.R. The Rio Piedras Watershed and Its Surrounding Environment; USDA Forest Service: Washington, DC, USA, 2011.
- Diaz, M.N. Evaluacion de la Intensidad de Lluvia y Su Efecto en la Presencia de Bacterias Fecales en Las Playas del Norte de Puerto Rico; Ciencias en Gerencia Ambiental; Universidad Metropolitana: San Juan, Puerto Rico, 2007. [Google Scholar]
- Soderberg, C.-A.P. Agua potable y saneamiento para comunidades aisladas: Un imperativo social y ambiental para Puerto Rico. Rev. Int. Desastres Nat. Accid. Infraestruct. Civ. 2008, 8, 1–3. [Google Scholar]
- Quiñones, F. Impacto ambiental de pozos sépticos en puerto rico y su diseño y control. Dimensión Revista Colegio Ingenieros Agrimensores Puerto Rico 2012, 1, 16–22. [Google Scholar]
- Garcia-Montiel, D.C.; Verdejo-Ortiz, J.C.; Santiago-Bartolomei, R.; Vila-Ruiz, C.P.; Santiago, L.; Melendez-Ackerman, E. Food sources and accessibility and waste disposal patterns across an urban tropical watershed: Implications for the flow of materials and energy. Ecol. Soc. 2014, 19, 37. [Google Scholar] [CrossRef]
- Colford, J.M.; Wade, T.J.; Schiff, K.C.; Wright, C.C.; Griffith, J.F.; Sandhu, S.K.; Burns, S.; Sobsey, M.; Lovelace, G.; Weisberg, S.B. Water quality indicators and the risk of illness at beaches with nonpoint sources of fecal contamination. Epidemiology 2007, 18, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Pruss, A. Review of epidemiological studies on health effects from exposure to recreational water. Int. J. Epidemiol. 1998, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Boehm, A.B.; Sassoubre, L.M. Enterococci as Indicators of Environmental Fecal Contamination. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Gilmore, M.S., Clewell, D.B., Ike, Y., Shankar, N., Eds.; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014; pp. 1–17. [Google Scholar]
- Colford, J.M.; Schiff, K.C.; Griffith, J.F.; Yau, V.; Arnold, B.F.; Wright, C.C.; Gruber, J.S.; Wade, T.J.; Burns, S.; Hayes, J.; et al. Using rapid indicators for Enterococcus to assess the risk of illness after exposure to urban runoff contaminated marine water. Water Res. 2012, 46, 2176–2186. [Google Scholar] [CrossRef] [PubMed]
- Haile, R.W.; Witte, J.S.; Gold, M.; Cressey, R.; McGee, C.; Millikan, R.C.; Glasser, A.; Harawa, N.; Ervin, C.; Harmon, P.; et al. The health effects of swimming in ocean water contaminated by storm drain runoff. Epidemiology 1999, 10, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.K.; McIntyre, D.; Noble, R.T. Characterizing fecal contamination in stormwater runoff in coastal North Carolina, USA. Water Res. 2010, 44, 4186–4194. [Google Scholar] [CrossRef] [PubMed]
- Soller, J.; Bartrand, T.; Ravenscroft, J.; Molina, M.; Whelan, G.; Schoen, M.; Ashbolt, N. Estimated human health risks from recreational exposures to stormwater runoff containing animal faecal material. Environ. Model. Softw. 2015, 72, 21–32. [Google Scholar] [CrossRef]
- Lamparelli, C.C.; Pogreba-Brown, K.; Verhougstraete, M.; Sato, M.I.Z.; de Castro Bruni, A.; Wade, T.J.; Eisenberg, J.N. Are fecal indicator bacteria appropriate measures of recreational water risks in the tropics: A cohort study of beach goers in Brazil? Water Res. 2015, 87, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Cordero, L.; Norat, J.; Mattei, H.; Nazario, C. Seasonal variations in the risk of gastrointestinal illness on a tropical recreational beach. J. Water Health 2012, 10, 579–593. [Google Scholar] [CrossRef] [PubMed]
- Rochelle-Newall, E.; Nguyen, T.M.H.; Le, T.P.Q.; Sengteheuanghoung, O.; Ribolzi, O. A short review of fecal indicator bacteria in tropical aquatic ecosystems: Knowledge gaps and future directions. Front. Microbiol. 2015, 6, 308. [Google Scholar] [CrossRef] [PubMed]
- Boehm, A.B.; Ashbolt, N.J.; Colford, J.M.; Dunbar, L.E.; Fleming, L.E.; Gold, M.A.; Hansel, J.A.; Hunter, P.R.; Ichida, A.M.; McGee, C.D.; et al. A sea change ahead for recreational water quality criteria. J. Water Health 2009, 7, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, R.S.; Solo-Gabriele, H.M.; Byappanahalli, M.N.; Kirs, M. US Recreational water quality criteria: A vision for the future. Int. J. Environ. Res. Public Health 2015, 12, 7752–7776. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Recreational Water Quality Criteria; US EAP: Washington, DC, USA, 2012. Available online: https://www.epa.gov/wqc/microbial-pathogenrecreational-water-quality-criteria (accessed on 17 July 2017).
- PREQB. Beach Monitoring and Public Notification Program-Performance criteria 2016–2017. 2016. Available online: https://www2.pr.gov/agencias/jca/Documents/Monitoreo%20de%20Playas/Performance%20Criteria/BEACH_PERFORMANCE_CRITERIA_PREQB_2016-2017_final-draft.pdf (accessed on 19 July 2017).
- United States Environmental Protection Agency. National Beach Guidance and Required Performance Criteria for Grants. 2014. Available online: https://www.epa.gov/sites/production/files/2014-07/documents/beach-guidance-final-2014.pdf (accessed on 19 July 2017).
- United States Environmental Protection Agency. Beaches Environmental Assessment and Coastal Health Act of 2000; Public Law; US EAP: Washington, DC, USA, 2000; pp. 106–284.
- PREQB. Water Quality Standards Regulation of Puerto Rico. 2010. Available online: http://www.gobierno.pr/NR/rdonlyres/5A9F2F2E-94AE-4C69-8453-CA08D616ED7D/0/Reg_Estdares_Calidad_Agua_2010.pdf (accessed on 15 September 2017).
- Bachoon, D.S.; Markand, S.; Otero, E.; Perry, G.; Ramsubaugh, A. Assessment of non-point sources of fecal pollution in coastal waters of Puerto Rico and Trinidad. Mar. Pollut. Bull. 2000, 60, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Nazario, E.E.; Santiago-Rodriguez, T.M.; Toranzos, G.A. Prospective epidemiological pilot study on the morbidity of bathers exposed to tropical recreational waters and sand. J. Water Health 2014, 12, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Rodriguez, T.M.; Tremblay, R.L.; Toledo-Hernandez, C.; Gonzalez-Nieves, J.E.; Ryu, H.; Domingo, J.W.S.; Toranzos, G.A. Microbial quality of tropical inland waters and effects of rainfall events. Appl. Environ. Microbiol. 2012, 78, 5160–5169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Deng, Z.Q.; Rusch, K.A. Development of predictive models for determining enterococci levels at Gulf Coast beaches. Water Res. 2012, 46, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Maraccini, P.A.; Mattioli, M.C.M.; Sassoubre, L.M.; Cao, Y.P.; Griffith, J.F.; Ervin, J.S.; Van De Werfhorst, L.C.; Boehm, A.B. Solar inactivation of enterococci and Escherichia coli in natural waters: Effects of water absorbance and depth. Environ. Sci. Technol. 2016, 50, 5068–5076. [Google Scholar] [CrossRef] [PubMed]
- He, L.M.; He, Z.L. Water quality prediction of marine recreational beaches receiving watershed baseflow and stormwater runoff in southern California, USA. Water Res. 2008, 42, 2563–2573. [Google Scholar] [CrossRef] [PubMed]
- Shibata, T.; Solo-Gabriele, H.M.; Sinigalliano, C.D.; Gidley, M.L.; Plano, L.R.; Fleisher, J.M.; Wang, J.D.; Elmir, S.M.; He, G.; Wright, M.E. Evaluation of conventional and alternative monitoring methods for a recreational marine beach with nonpoint source of fecal contamination. Environ. Sci. Technol. 2010, 44, 8175–8181. [Google Scholar] [CrossRef] [PubMed]
- Maraccini, P.A.; Ferguson, D.M.; Boehm, A.B. Diurnal variation in Enterococcus species composition in polluted ocean water and a potential role for the Enterococcal Carotenoid in protection against photoinactivation. Appl. Environ. Microbiol. 2012, 78, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Zayas, J.R.; Cuevas, E.; Mayol-Bracero, O.L.; Donoso, L.; Trebs, I.; Figueroa-Nieves, D.; McDowell, W.H. Urban influences on the nitrogen cycle in Puerto Rico. Biogeochemistry 2006, 79, 109–133. [Google Scholar] [CrossRef]
- Puerto Rico Environmental Quality Board. Total Maximum Daily Loads (TMDL) Rio Grande de Loiza Watershed; Puerto Rico Environmental Quality Board: San Juan, Puerto Rico, 2007.
- Puerto Rico Environmental Quality Board. Carga Máxima Total Diaria de Bacterias Coliformes Fecales (TMDL) Para Unidades de Evaluación, Puerto Rico; Departamento de Planificación y Protección Ambiental: San Juan, Puerto Rico, 2011.
- United States Environmental Protection Agency. Method 1600: Enterococci in Water by Membrane Filtration Using Membrane-Enterococcus Indoxyl-B-D-Glucoside Agar (mEI); US EPA: Washington, DC, USA, 2006.
- Byappanahalli, M.N.; Nevers, M.B.; Korajkic, A.; Staley, Z.R.; Harwood, V.J. Enterococci in the environment. Microbiol. Mol. Biol. Rev. 2012, 76, 685–706. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Q.; Hu, C.M.; Muller-Karger, F. Monitoring turbidity in Tampa Bay using MODIS/Aqua 250-m imagery. Remote Sens. Environ. 2007, 109, 207–220. [Google Scholar] [CrossRef]
- Chen, Z.Q.; Hu, C.M.; Muller-Karger, F.E.; Luther, M.E. Short-term variability of suspended sediment and phytoplankton in Tampa Bay, Florida: Observations from a coastal oceanographic tower and ocean color satellites. Estuar. Coast. Shelf Sci. 2010, 89, 62–72. [Google Scholar] [CrossRef]
- Aurin, D.; Mannino, A.; Franz, B. Spatially resolving ocean color and sediment dispersion in river plumes, coastal systems, and continental shelf waters. Remote Sens. Environ. 2013, 137, 212–225. [Google Scholar] [CrossRef]
- Moreno-Madrinan, M.J.; Al-Hamdan, M.Z.; Rickman, D.L.; Muller-Karger, F.E. Using the surface reflectance MODIS Terra product to estimate turbidity in Tampa Bay, Florida. Remote Sens. 2010, 2, 2713–2728. [Google Scholar] [CrossRef]
- Schang, C.; Henry, R.; Kolotelo, P.A.; Prosser, T.; Crosbie, N.; Grant, T.; Cottam, D.; O’Brien, P.; Coutts, S.; Deletic, A.; et al. Evaluation of techniques for measuring microbial hazards in bathing waters: A comparative study. PLoS ONE 2016, 11, e0155848. [Google Scholar] [CrossRef] [PubMed]
- Leskinen, S.D.; Harwood, V.J.; Lim, D.V. Rapid dead-end ultrafiltration concentration and biosensor detection of enterococci from beach waters of Southern California. J. Water Health 2016, 7, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Boehm, A.B. Enterococci concentrations in diverse coastal environments exhibit extreme variability. Environ. Sci. Technol. 2017, 41, 8227–8232. [Google Scholar] [CrossRef]
- Blanchet, F.G.; Legendre, P.; Borcard, D. Forward selection of explanatory variables. Ecology 2008, 89, 2623–2632. [Google Scholar] [CrossRef] [PubMed]
- Burnham, K.P.; Anderson, D.R. Kullback-Leibler information as a basis for strong inference in ecological studies. Wildl. Res. 2001, 28, 111–119. [Google Scholar] [CrossRef]
- Godinez-Dominguez, E.; Freire, J. Information-theoretic approach for selection of spatial and temporal models of community organization. Mar. Ecol. Prog. Ser. 2003, 253, 17–24. [Google Scholar] [CrossRef]
- Jones, D. Fathom Toolbox for Matlab: Software for Multivariate Ecological and Oceanographic Data Analysis; College of Marine Science, University of South Florida: St. Petersburg, FL, USA, 2015; Available online: http://www.marine.usf.edu/user/djones/ (accessed on 10 October 2017).
- Herrera, A.; Suarez, P. Bacterial markers as tools for coastal water environmental quality measurement. Interciencia 2005, 30, 171–176. [Google Scholar]
- Mote, B.L.; Turner, J.W.; Lipp, E.K. Persistence and growth of the fecal indicator bacteria enterococci in detritus and natural estuarine plankton communities. Appl. Environ. Microbiol. 2012, 78, 2569–2577. [Google Scholar] [CrossRef] [PubMed]
- Viau, E.J.; Goodwin, K.D.; Yamahara, K.M.; Layton, B.A.; Sassoubre, L.M.; Burns, S.L.; Tong, H.I.; Wong, S.H.C.; Lu, Y.A.; Boehm, A.B. Bacterial pathogens in Hawaiian coastal streams-associations with fecal indicators, land cover, and water quality. Water Res. 2011, 45, 3279–3290. [Google Scholar] [CrossRef] [PubMed]
- Aranda, D.; Lopez, J.V.; Solo-Gabriele, H.M.; Fleisher, J.M. Using probabilities of enterococci exceedance and logistic regression to evaluate long term weekly beach monitoring data. J. Water Health 2016, 14, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Enns, A.A.; Vogel, L.J.; Abdelzaher, A.M.; Solo-Gabriele, H.M.; Plano, L.R.W.; Gidley, M.L.; Phillips, M.C.; Klaus, J.S.; Piggot, A.M.; Feng, Z.X.; et al. Spatial and temporal variation in indicator microbe sampling is influential in beach management decisions. Water Res. 2012, 46, 2237–2246. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Reniers, A.; Haus, B.; Solo-Gabriele, H.; Fiorentino, L.; Olascoaga, M.; MacMahan, J. Modeling microbial water quality at a beach impacted by multiple non-point sources. Coast. Eng. Proc. 2012, 1, 74. [Google Scholar] [CrossRef]
- Wright, M.E.; Abdelzaher, A.M.; Solo-Gabriele, H.M.; Elmir, S.; Fleming, L.E. The inter-tidal zone is the pathway of input of enterococci to a subtropical recreational marine beach. Water Sci. Technol. 2011, 63, 542–549. [Google Scholar] [CrossRef] [PubMed]
- Perez-Villalona, H.; Cornwell, J.C.; Ortiz-Zayas, J.R.; Cuevas, E. Sediment denitrification and nutrient fluxes in the San José Lagoon, a tropical lagoon in the highly urbanized San Juan Bay Estuary, Puerto Rico. Estuar. Coasts 2015, 38, 2259–2278. [Google Scholar] [CrossRef]
- Ramirez, A.; De Jesús-Crespo, R.; Martinó-Cardona, D.M.; Martínez-Rivera, N.; Burgos-Caraballo, S. Urban streams in Puerto Rico: What can we learn from the tropics? J. N. Am. Benthol. Soc. 2009, 28, 1070–1079. [Google Scholar] [CrossRef]
- Zhang, W.W.; Wang, J.Y.; Fan, J.F.; Gao, D.L.; Ju, H.Y. Effects of rainfall on microbial water quality on Qingdao No. 1 Bathing Beach, China. Mar. Pollut. Bull. 2013, 66, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Shehane, S.D.; Harwood, V.J.; Whitlock, J.E.; Rose, J.B. The influence of rainfall on the incidence of microbial faecal indicators and the dominant sources of faecal pollution in a Florida river. J. Appl. Microbiol. 2005, 98, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.X.; Reniers, A.; Haus, B.K.; Solo-Gabriele, H.M. Modeling sediment-related enterococci loading, transport, and inactivation at an embayed nonpoint source beach. Water Resour. Res. 2013, 49, 693–712. [Google Scholar] [CrossRef]
- Ioan, C.; Robescu, D.N. Effect of increased inflow and dilution on the activated sludge properties and viability of microbial community. UPB Sci. Bull. 2015, 77, 197–204. [Google Scholar]
- Kleinheinz, G.T.; McDermott, C.M.; Hughes, S.; Brown, A. Effects of rainfall on E. coli concentrations at Door County, Wisconsin beaches. Int. J. Microbiol. 2009, 2009, 876050. [Google Scholar] [CrossRef] [PubMed]
- Boehm, A.B.; Yamahara, K.M.; Love, D.C.; Peterson, B.M.; McNeill, K.; Nelson, K.L. Covariation and photoinactivation of traditional and novel indicator organisms and human viruses at a sewage-impacted marine beach. Environ. Sci. Technol. 2009, 43, 8046–8052. [Google Scholar] [CrossRef] [PubMed]
- Grant, S.B.; Sanders, B.F.; Boehm, A.B.; Redman, J.A.; Kim, J.H.; Mrse, R.D.; Chu, A.K.; Gouldin, M.; McGee, C.D.; Gardiner, N.A.; et al. Generation of enterococci bacteria in a coastal saltwater marsh and its impact on surf zone water quality. Environ. Sci. Technol. 2001, 35, 2407–2416. [Google Scholar] [CrossRef] [PubMed]
- Yamahara, K.M.; Walters, S.P.; Boehm, A.B. Growth of enterococci in unaltered, unseeded beach sands subjected to tidal wetting. Appl. Environ. Microbiol. 2009, 75, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Gast, R.J.; Gorrell, L.; Raubenheimer, B.; Elgar, S. Impact of erosion and accretion on the distribution of enterococci in beach sands. Cont. Shelf Res. 2011, 31, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Dodds, W.K.; Gudder, D.A. The ecology of Cladophora. J. Phycol. 1992, 28, 415–427. [Google Scholar] [CrossRef]
- Ishii, S.; Yan, T.; Shively, D.A.; Byappanahalli, M.N.; Whitman, R.L.; Sadowsky, M.J. Cladophora (Chlorophyta) spp. harbor human bacterial pathogens in nearshore water of Lake Michigan. Appl. Environ. Microbiol. 2006, 72, 4545–4553. [Google Scholar] [CrossRef] [PubMed]
- Lamb, J.B.; van de Water, J.; Bourne, D.G.; Altier, C.; Hein, M.Y.; Fiorenza, E.A.; Abu, N.; Jompa, J.; Harvell, C.D. Seagrass ecosystems reduce exposure to bacterial pathogens of humans, fishes, and invertebrates. Science 2017, 355, 731–733. [Google Scholar] [CrossRef] [PubMed]
- Whitman, R.L.; Shively, D.A.; Pawlik, H.; Nevers, M.B.; Byappanahalli, M.N. Occurrence of Escherichia coli and enterococci in Cladophora (Chlorophyta) in nearshore water and beach sand of Lake Michigan. Appl. Environ. Microbiol. 2003, 69, 4714–4719. [Google Scholar] [CrossRef] [PubMed]
- Serrano, E.; Moreno, B.; Solaun, M.; Aurrekoetxea, J.J.; Ibarluzea, J. The influence of environmental factors on microbiological indicators of coastal water pollution. Water Sci. Technol. 1998, 38, 195–199. [Google Scholar]
- Soupir, M.L.; Mostaghimi, S.; Dillaha, T. Attachment of Escherichia coli and Enterococci to Particles in Runoff. J. Environ. Qual. 2010, 39, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Landmeyer, J.E.; Garigen, T.J. Relation between Enterococcus Concentrations and Turbidity in Fresh and Saline Recreational Waters, Coastal Horry County, South Carolina, 2003–04; US Geological Survey: Reston, VA, USA, 2016.
- Phillips, L.; Liss, P. The Physical Chemistry of Air-Sea Gas Exchange. In The Sea Surface and Global Change; Cambrigde University Press: Cambridge, UK, 1997; pp. 207–250. [Google Scholar]
- Byappanahalli, M.N.; Whitman, R.L.; Shively, D.A.; Nevers, M.B. Linking non-culturable (qPCR) and culturable enterococci densities with hydrometeorological conditions. Sci. Total Environ. 2010, 408, 3096–3101. [Google Scholar] [CrossRef] [PubMed]
- Nevers, M.B.; Whitman, R.L. Nowcast modeling of Escherichia coli concentrations at multiple urban beaches of southern Lake Michigan. Water Res. 2005, 39, 5250–5260. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Phanikumar, M.S.; Molloy, S.L.; Whitman, R.L.; Shively, D.A.; Nevers, M.B.; Schwab, D.J.; Rose, J.B. Modeling the transport and inactivation of E. coli and enterococci in the near-shore region of Lake Michigan. Environ. Sci. Technol. 2006, 40, 5022–5028. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Draft National Pollutant Discharge Elimination System (NPDES) Permit to Discharge into the Waters of the United States; US EPA: Washington, DC, USA, 1994; p. 15.
- Huang, H.; Fergen, R.E.; Proni, J.R.; Tsai, J.J. Probabilistic analysis of ocean outfall mixing zones. J. Environ. Eng. 1996, 122, 359–367. [Google Scholar] [CrossRef]
- Isleib, R.; Thuman, A. Verification of large scale watershed modeling analysis using small subwatershed models. In Proceedings of the 2010 Watershed Management: Innovations in Watershed Management under Land Use and Climate Change, Madison, WI, USA, 23–27 August 2010; pp. 1405–1416. [Google Scholar]
- Murphy, S.F.; Stallard, R.F. Hydrology and climate of four watersheds in eastern Puerto Rico. In Water Quality and Landscape Processes of Four Watersheds in Eastern Puerto Rico; U.S. Geological Survey: Reston, VA, USA, 2012; Volume 100, pp. 43–84. [Google Scholar]
- Crowther, J.; Kay, D.; Wyer, M.D. Relationships between microbial water quality and environmental conditions in coastal recreational waters: The Fylde coast, UK. Water Res. 2001, 35, 4029–4038. [Google Scholar] [CrossRef]
- Boehm, A.B.; Fuhrman, J.A.; Mrse, R.D.; Grant, S.B. Tiered approach for identification of a human fecal pollution source at a recreational beach: Case study at Avalon Cay, Catalina Island, California. Environ. Sci. Technol. 2003, 37, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Fleisher, J.M.; Fleming, L.E.; Solo-Gabriele, H.M.; Kish, J.K.; Sinigalliano, C.D.; Plano, L.; Elmir, S.M.; Wang, J.D.; Withum, K.; Shibata, T.; et al. The BEACHES Study: Health effects and exposures from non-point source microbial contaminants in subtropical recreational marine waters. Int. J. Epidemiol. 2010, 39, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- Mavani, J.; Chen, L.; Joksimovic, D.; Li, S. Development and Testing of Data Driven Nowcasting Models of Beach Water Quality; City University of New York: New York, NY, USA, 2014; p. 318. [Google Scholar]
| Variable | Pearson’s Correlation Coefficient (r) | Lag |
|---|---|---|
| Mean sea level | −0.19 | 9th to 11th day (mean) |
| Direct normal irradiance | −0.24 | 1 day |
| Sea surface temperature | 0.12 | 5th to 9th day (mean) |
| Dew point | 0.19 | 7 days (mean) |
| Turbidity | 0.25 | 1 day |
| Precipitation | 0.22 | 4 days (accumulated) |
| Variable | r2 | r2-Adjusted | AIC |
|---|---|---|---|
| Precipitation | 0.08 | 0.08 | 59.81 |
| Mean sea level | 0.13 | 0.12 | 48.76 |
| Direct normal irradiance | 0.16 | 0.15 | 39.19 |
| Sea surface temperature | 0.19 | 0.17 | 32.79 |
| Turbidity | 0.21 | 0.19 | 26.76 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laureano-Rosario, A.E.; Symonds, E.M.; Rueda-Roa, D.; Otis, D.; Muller-Karger, F.E. Environmental Factors Correlated with Culturable Enterococci Concentrations in Tropical Recreational Waters: A Case Study in Escambron Beach, San Juan, Puerto Rico. Int. J. Environ. Res. Public Health 2017, 14, 1602. https://doi.org/10.3390/ijerph14121602
Laureano-Rosario AE, Symonds EM, Rueda-Roa D, Otis D, Muller-Karger FE. Environmental Factors Correlated with Culturable Enterococci Concentrations in Tropical Recreational Waters: A Case Study in Escambron Beach, San Juan, Puerto Rico. International Journal of Environmental Research and Public Health. 2017; 14(12):1602. https://doi.org/10.3390/ijerph14121602
Chicago/Turabian StyleLaureano-Rosario, Abdiel E., Erin M. Symonds, Digna Rueda-Roa, Daniel Otis, and Frank E. Muller-Karger. 2017. "Environmental Factors Correlated with Culturable Enterococci Concentrations in Tropical Recreational Waters: A Case Study in Escambron Beach, San Juan, Puerto Rico" International Journal of Environmental Research and Public Health 14, no. 12: 1602. https://doi.org/10.3390/ijerph14121602





