First Year Growth in Relation to Prenatal Exposure to Endocrine Disruptors — A Dutch Prospective Cohort Study
Abstract
:1. Introduction
2. Methods
2.1. Study Procedures and Subjects
2.2. First Year Growth
2.3. Chemical Exposure
2.4. Data-Analysis
2.5. Covariates
3. Results
| Boys (n = 56) | Girls (n = 33) | |
|---|---|---|
| Weight, age = 1 month (kg) | 4.68 (3.00–5.87) | 4.25 (3.21–5.38) |
| Weight, age = 11 months (kg) | 10.13 (8.49–12.85) | 8.92 (6.88–10.55) |
| Weight gain, first year (kg) | 5.31 (4.16–7.59) | 4.46 (3.41–5.60) |
| Height, age = 1 month (cm) | 54.8 (51.0–59.0) | 53.9 (51.0–59.9) |
| Height, age = 11 months (cm) | 76.6 (71.0–81.4) | 74.6 (71.0–77.0) |
| Growth, first year (cm) | 21.5 (18.0–25.0) | 19.7 (16.0–23.3) |
| BMI, age = 1 month (kg/m2) | 15.5 (12.8–18.7) | 14.8 (12.0–17.0) |
| BMI, age = 11 months (kg/m2) | 17.1 (15.6–21.0) | 16.2 (13.3–18.5) |
| BMI change, first year (kg/m2) | 1.8 (−1.6–5.2) | 1.6 (−0.8–3.2) |
| Head circumference, age = 1 month (cm) | 38.0 (34.2–40.6) | 37.3 (35.1–40.1) |
| Head circumference, age = 11 months (cm) | 46.8 (43.4–49.5) | 45.2 (42.6–48.0) |
| Head circumference change, first year (cm) | 9.0 (7.9–11.2) | 8.1 (6.7–10.4) |
| Birth weight (g) | 3624.5 (491.7) | 3564.2 (397.5) |
| Gestational age (weeks) | 39.7 (1.5) | 40.1 (1.0) |
| Parity (nulliparous, %) | 23 (42.6%) | 10 (30.3%) |
| Height mother (cm) | 171.4 (5.7) | 171.9 (4.6) |
| Height father (cm) | 185.2 (6.8) | 183.6 (6.2) |
| BMI mother (start pregnancy, kg/m2) | 23.6 (3.3) | 23.0 (4.2) |
| BMI father (kg/m2) | 24.0 (3.0) | 25.0 (2.8) |
| Age mother (years) | 30.3 (3.8) | 31.9 (3.5) |
| Age father (years) | 32.4 (5.6) | 34.2 (4.2) |
| Breastfeeding | ||
| No breastfeeding | 4 (7.7%) | 3 (10.7%) |
| 0–3 months breastfeeding | 12 (23.1%) | 7 (25.0%) |
| >3 months breastfeeding | 36 (69.2%) | 18 (64.3%) |
| Education mother (bachelor/master, %) | 41 (75.9%) | 19 (59.4%) |
| Smoking (first trimester, yes, %) | 2 (3.7%) | 2 (6.1%) |
| Alcohol (first trimester, yes, %) | 3 (2.7%) | 2 (6.6%) |
| Compound | n | Mean | Median | Range | LOQ | <LOQ (%) | |
|---|---|---|---|---|---|---|---|
| PCB-153 | |||||||
| Cord plasma | ng/L | 52 | 35.94 | 29.14 | 22.63–96.00 | 21–43 | 55.8 |
| DDE | |||||||
| Cord plasma | ng/L | 52 | 115.61 | 83.00 | 28.28–470.00 | 33–73 | 23.1 |
| Breast milk | ng/L | 24 | 2527.92 | 1950.00 | 400.00–1,1390.00 | 9.20–13.00 | 0 |
| Total a | ng/L | 76 | 102.82 | 74.50 | 14.53–470.00 | 15.8 | |
| MECPP | |||||||
| Cord plasma | ng/mL | 61 | 0.31 | 0.27 | 0.11–1.00 | 0.13–0.28 | 6.6 |
| MEHHP | |||||||
| Cord plasma | ng/mL | 61 | 0.32 | 0.26 | 0.10–1.00 | 0.14–0.27 | 11.5 |
| MEOHP | |||||||
| Cord plasma | ng/mL | 61 | 0.29 | 0.22 | 0.12–0.87 | 0.17–0.33 | 26.2 |
| PFOA | |||||||
| Cord plasma | ng/L | 61 | 940.2 | 870.0 | 300–2700 | 50–140 | 0 |
| PFOS | |||||||
| Cord plasma | ng/L | 61 | 1611.0 | 1600.0 | 570–3200 | 44–140 | 0 |
| Compound | Q1 | n | Q2 | n | Q3 | n | Q4 | n |
|---|---|---|---|---|---|---|---|---|
| DDE (Total) (ng/L) | <44.74 | 19 | 44.74–74.50 | 19 | 74.51–113.11 | 19 | >113.11 | 19 |
| MECPP (ng/mL) | <0.16 | 15 | 016–0.24 | 15 | 0.25–0.34 | 15 | >0.34 | 15 |
| MEHHP (ng/mL) | <0.20 | 16 | 0.20–0.23 | 17 | 0.24–0.37 | 11 | >0.37 | 17 |
| MEOHP (ng/mL) | <0.13 | 17 | 0.13–0.20 | 17 | 0.21–0.34 | 13 | >0.34 | 14 |
| PFOA (ng/L) | <591 | 17 | 591–870 | 14 | 871–1100 | 15 | >1100 | 15 |
| PFOS (ng/L) | <1001 | 18 | 1001–1600 | 15 | 1601–2000 | 14 | >2000 | 14 |
3.1. BMI
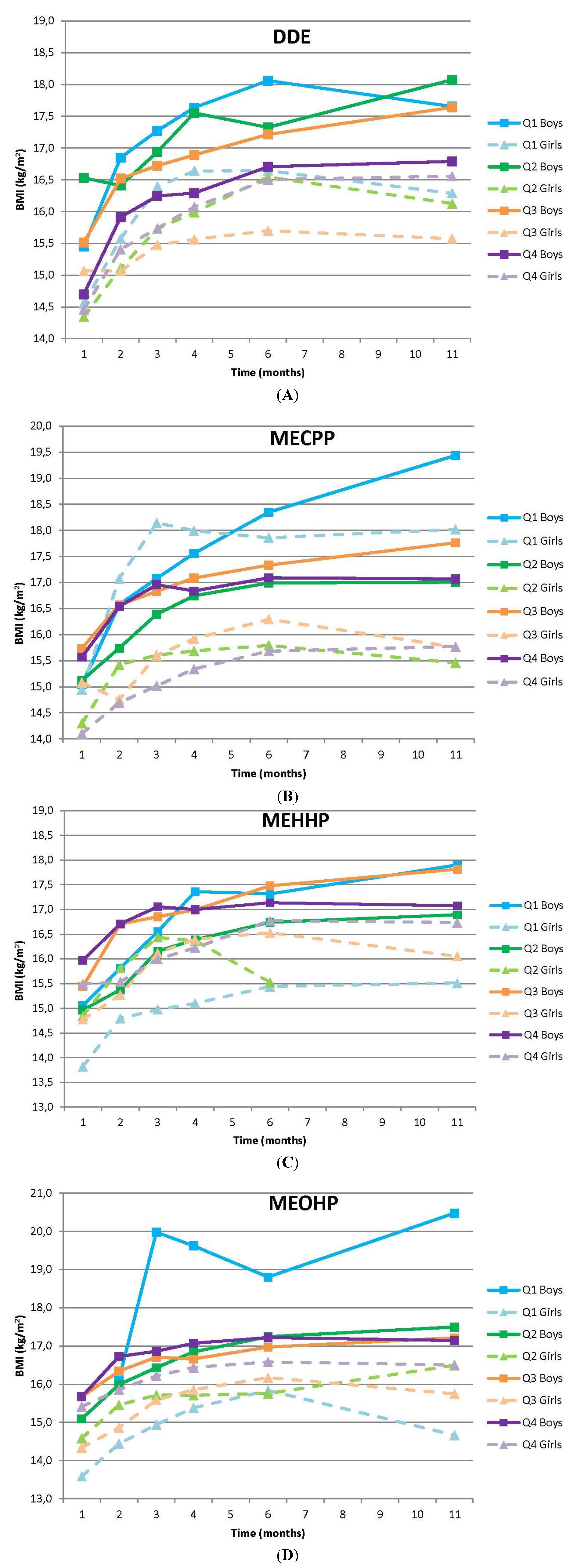
| EDC | Effect | BMI | Weight | Height | Head Circumference |
|---|---|---|---|---|---|
| p | p | p | p | ||
| DDE | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| DDE | 0.293 | 0.647 | 0.910 | 0.778 | |
| Sex | <0.001 * | <0.001 * | 0.024 * | <0.001 * | |
| Time × DDE | 0.078 * | 0.84 | 0.490 | 0.721 | |
| DDE × sex | 0.420 | 0.205 | 0.484 | 0.905 | |
| Time × DDE × sex | 0.298 | 0.695 | 0.134 | <0.001 * | |
| PFOS | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| PFOS | 0.586 | 0.802 | 0.975 | 0.649 | |
| Sex | <0.001 * | <0.001 * | 0.040 * | <0.001 * | |
| Time × PFOS | 0.878 | 0.910 | 0.485 | 0.800 | |
| PFOS × sex | 0.655 | 0.425 | 0.749 | 0.485 | |
| Time × PFOS × sex | 0.679 | 0.617 | 0.321 | 0.742 | |
| PFOA | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| PFOA | 0.813 | 0.350 | 0.045 * | 0.774 | |
| Sex | <0.001 * | <0.001 * | 0.019 * | <0.001 * | |
| Time × PFOA | 0.389 | 0.126 | <0.001 * | 0.001 * | |
| PFOA × sex | 0.242 | 0.169 | 0.165 | 0.467 | |
| Time × PFOA × sex | 0.348 | 0.812 | 0.152 | 0.387 | |
| MECPP | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| MECPP | 0.051 | 0.265 | 0.726 | 0.314 | |
| Sex | 0.004 * | <0.001 * | 0.002 * | 0.003 * | |
| Time × MECPP | 0.117 | 0.090 * | 0.944 | 0.604 | |
| MECPP × sex | 0.496 | 0.277 | 0.185 | 0.047 * | |
| Time × MECPP × sex | 0.204 | 0.929 | 0.978 | 0.886 | |
| MEHHP | Time | <0.001 * | <0.001 | <0.001 * | <0.001 * |
| MEHHP | 0.279 | 0.204 | 0.133 | 0.144 | |
| Sex | 0.005 * | 0.002 | 0.163 | 0.012 * | |
| Time × MEHHP | 0.593 | 0.962 | 0.294 | 0.100 * | |
| MEHHP × sex | 0.383 | 0.326 | 0.073 * | 0.197 | |
| Time × MEHHP × sex | 0.127 | 0.485 | 0.011 * | 0.018 * | |
| MEOHP | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| MEOHP | 0.315 | 0.151 | 0.112 | 0.119 | |
| Sex | <0.001 * | 0.001 * | 0.390 | <0.001 * | |
| Time × MEOHP | 0.152 | 0.948 | 0.084 * | 0.335 | |
| MEOHP × sex | 0.104 | 0.831 | 0.207 | 0.253 | |
| Time × MEOHP × sex | 0.029 * | 0.037 * | <0.001 * | 0.240 | |
| PCB-153 (>LOQ vs. <LOQ) | Time | <0.001 * | <0.001 * | <0.001 * | <0.001 * |
| PCB | 0.203 | 0.544 | 0.598 | 0.729 | |
| Sex | <0.001 * | <0.001 * | 0.001 * | <0.001 * | |
| Time × PCB | 0.491 | 0.518 | 0.946 | 0.861 | |
| PCB × sex | 0.338 | 0.738 | 0.115 | 0.586 | |
| Time × PCB × sex | 0.808 | 0.775 | 0.086 * | 0.102 |
3.2. Weight and Height
3.3. Head Circumference
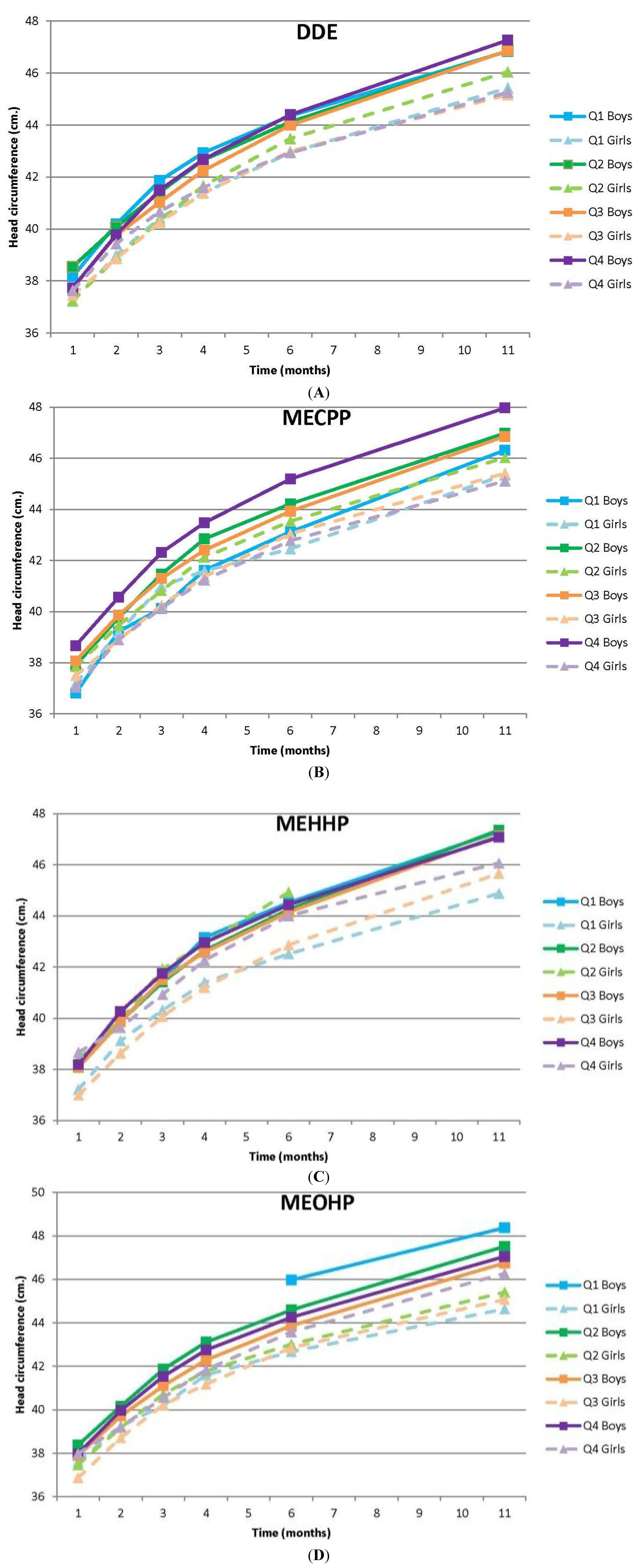
4. Discussion
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflict of Interest
References
- Hanson, M.; Gluckman, P. Developmental origins of noncommunicable disease: Population and public health implications. Am. J. Clin. Nutr. 2011, 94, 1754S–1758S. [Google Scholar] [CrossRef]
- Roseboom, T.; de Rooij, S.; Painter, R. The dutch famine and its long-term consequences for adult health. Early Hum. Dev. 2006, 82, 485–491. [Google Scholar] [CrossRef]
- Bergvall, N.; Iliadou, A.; Johansson, S.; de Faire, U.; Kramer, M.S.; Pawitan, Y.; Pedersen, N.L.; Lichtenstein, P.; Cnattingius, S. Genetic and shared environmental factors do not confound the association between birth weight and hypertension: A study among swedish twins. Circulation 2007, 115, 2931–2938. [Google Scholar]
- Hales, C.N.; Barker, D.J.; Clark, P.M.; Cox, L.J.; Fall, C.; Osmond, C.; Winter, P.D. Fetal and infant growth and impaired glucose tolerance at age 64. BMJ 1991, 303, 1019–1022. [Google Scholar] [CrossRef]
- Yu, Z.B.; Han, S.P.; Zhu, G.Z.; Zhu, C.; Wang, X.J.; Cao, X.G.; Guo, X.R. Birth weight and subsequent risk of obesity: A systematic review and meta-analysis. Obes. Rev. 2011, 12, 525–542. [Google Scholar] [CrossRef]
- Labayen, I.; Moreno, L.A.; Ruiz, J.R.; Gonzalez-Gross, M.; Warnberg, J.; Breidenassel, C.; Ortega, F.B.; Marcos, A.; Bueno, M. Small birth weight and later body composition and fat distribution in adolescents: The avena study. Obesity (Silver Spring) 2008, 16, 1680–1686. [Google Scholar] [CrossRef]
- Monteiro, P.O.; Victora, C.G. Rapid growth in infancy and childhood and obesity in later life—A systematic review. Obes. Rev. 2005, 6, 143–154. [Google Scholar] [CrossRef]
- Odegaard, A.O.; Choh, A.C.; Nahhas, R.W.; Towne, B.; Czerwinski, S.A.; Demerath, E.W. Systematic examination of infant size and growth metrics as risk factors for overweight in young adulthood. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Gittner, L.S.; Ludington-Hoe, S.M.; Haller, H.S. Utilising infant growth to predict obesity status at 5 years. J. Paediatr. Child. Health 2013, 49, 564–574. [Google Scholar] [CrossRef]
- Mendez, M.A.; Garcia-Esteban, R.; Guxens, M.; Vrijheid, M.; Kogevinas, M.; Goni, F.; Fochs, S.; Sunyer, J. Prenatal organochlorine compound exposure, rapid weight gain and overweight in infancy. Environ. Health Perspect. 2011, 119, 272–278. [Google Scholar]
- Smink, A.; Ribas-Fito, N.; Garcia, R.; Torrent, M.; Mendez, M.A.; Grimalt, J.O.; Sunyer, J. Exposure to hexachlorobenzene during pregnancy increases the risk of overweight in children aged 6 years. Acta Paediatr. 2008, 97, 1465–1469. [Google Scholar] [CrossRef]
- Valvi, D.; Mendez, M.A.; Martinez, D.; Grimalt, J.O.; Torrent, M.; Sunyer, J.; Vrijheid, M. Prenatal concentrations of polychlorinated biphenyls, DDE, and DDT and overweight in children: A prospective birth cohort study. Environ. Health Perspect. 2011, 120, 451–457. [Google Scholar] [CrossRef]
- Valvi, D.; Mendez, M.A.; Garcia-Esteban, R.; Ballester, F.; Ibarluzea, J.; Goni, F.; Grimalt, J.O.; Llop, S.; Marina, L.S.; Vizcaino, E.; et al. Prenatal exposure to persistent organic pollutants and rapid weight gain and overweight in infancy. Obesity (Silver Spring, MD) 2014, 22, 488–496. [Google Scholar] [CrossRef]
- Halldorsson, T.I.; Rytter, D.; Haug, L.S.; Bech, B.H.; Danielsen, I.; Becher, G.; Henriksen, T.B.; Olsen, S.F. Prenatal exposure to perfluorooctanoate and risk of overweight at 20 years of age: A prospective cohort study. Environ. Health Perspect. 2012, 120, 668–673. [Google Scholar]
- Garced, S.; Torres-Sanchez, L.; Cebrian, M.E.; Claudio, L.; Lopez-Carrillo, L. Prenatal dichlorodiphenyldichloroethylene (DDE) exposure and child growth during the first year of life. Environ. Res. 2012, 113, 58–62. [Google Scholar]
- Karmaus, W.; Osuch, J.R.; Eneli, I.; Mudd, L.M.; Zhang, J.; Mikucki, D.; Haan, P.; Davis, S. Maternal levels of dichlorodiphenyl-dichloroethylene (DDE) may increase weight and body mass index in adult female offspring. Occup. Environ. Med. 2009, 66, 143–149. [Google Scholar]
- Gladen, B.C.; Klebanoff, M.A.; Hediger, M.L.; Katz, S.H.; Barr, D.B.; Davis, M.D.; Longnecker, M.P. Prenatal DDT exposure in relation to anthropometric and pubertal measures in adolescent males. Environ. Health Perspect. 2004, 112, 1761–1767. [Google Scholar]
- Andersen, C.S.; Fei, C.; Gamborg, M.; Nohr, E.A.; Sorensen, T.I.; Olsen, J. Prenatal exposures to perfluorinated chemicals and anthropometry at 7 years of age. Am. J. Epidemiol. 2013, 178, 921–927. [Google Scholar] [CrossRef]
- De Cock, M.; van de Bor, M. Obesogenic effects of endocrine disruptors, what do we know from animal and human studies? Environ. Int. 2014, 70c, 15–24. [Google Scholar] [CrossRef]
- RIVM. DDT en Ddt-Derivaten; RIVM: Bilthoven, The Netherlands, 2010. [Google Scholar]
- RIVM. Fact Sheet—Chloorbenzenen; RIVM: Bilthoven, The Netherlands, 2011. [Google Scholar]
- WHO. The Use of DDT in Malaria Vector Control—Who Position Statement; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- EC. Polychlorinated Biphenyls and Polychlorinated Terphenyls (PCBs/PCTs). Available online: http://ec.europa.eu/environment/waste/pcbs/index.htm (accessed on 30 August 2013).
- EFSA. European food safety authority; Results of the monitoring of non dioxin-like PCBs in food and feed. EFSA J. 2010, 8, 1701:1–1701:35. [Google Scholar]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R., Jr.; Lee, D.H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and endocrine-disrupting chemicals: Low-dose effects and nonmonotonic dose responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef]
- Woodruff, T.J.; Zota, A.R.; Schwartz, J.M. Environmental chemicals in pregnant women in the united states: Nhanes 2003–2004. Environ. Health Perspect. 2011, 119, 878–885. [Google Scholar] [CrossRef]
- Verhulst, S.L.; Nelen, V.; Hond, E.D.; Koppen, G.; Beunckens, C.; Vael, C.; Schoeters, G.; Desager, K. Intrauterine exposure to environmental pollutants and body mass index during the first 3 years of life. Environ. Health Perspect. 2009, 117, 122–126. [Google Scholar] [CrossRef]
- Luzardo, O.P.; Mahtani, V.; Troyano, J.M.; Alvarez de la Rosa, M.; Padilla-Perez, A.I.; Zumbado, M.; Almeida, M.; Burillo-Putze, G.; Boada, C.; Boada, L.D. Determinants of organochlorine levels detectable in the amniotic fluid of women from tenerife island (Canary islands, Spain). Environ. Res. 2009, 109, 607–613. [Google Scholar] [CrossRef]
- Foster, W.; Chan, S.; Platt, L.; Hughes, C. Detection of endocrine disrupting chemicals in samples of second trimester human amniotic fluid. J. Clin. Endocrinol. Metab. 2000, 85, 2954–2957. [Google Scholar] [CrossRef]
- Newbold, R.R. Impact of environmental endocrine disrupting chemicals on the development of obesity. Hormones (Athens) 2010, 9, 206–217. [Google Scholar] [CrossRef]
- Wan, Y.; Choi, K.; Kim, S.; Ji, K.; Chang, H.; Wiseman, S.; Jones, P.D.; Khim, J.S.; Park, S.; Park, J.; et al. Hydroxylated polybrominated diphenyl ethers and bisphenol a in pregnant women and their matching fetuses: Placental transfer and potential risks. Environ. Sci. Technol. 2010, 44, 5233–5239. [Google Scholar] [CrossRef]
- Hanson, M.; Godfrey, K.M.; Lillycrop, K.A.; Burdge, G.C.; Gluckman, P.D. Developmental plasticity and developmental origins of non-communicable disease: Theoretical considerations and epigenetic mechanisms. Prog. Biophys. Mol. Biol 2011, 106, 272–280. [Google Scholar] [CrossRef]
- Hou, L.; Zhang, X.; Wang, D.; Baccarelli, A. Environmental chemical exposures and human epigenetics. Int. J. Epidemiol. 2012, 41, 79–105. [Google Scholar] [CrossRef]
- Netherlands, C.S. Gebruik Medische Voorzieningen; t/m 2009. Available online: http://statline.cbs.nl/StatWeb/publication/?DM=SLNL&PA=7042MC&D1=390–425&D2=(l-9)-l&VW=T (accessed on 5 June 2014).
- IBM. IBM Spss Statistics for Windows, Version 20.0; IBM Corp.: Armonk, NY, USA, 2011.
- Govarts, E.; Nieuwenhuijsen, M.; Schoeters, G.; Ballester, F.; Bloemen, K.; de Boer, M.; Chevrier, C.; Eggesbo, M.; Guxens, M.; Kramer, U.; et al. Birth weight and prenatal exposure to polychlorinated biphenyls (PCBs) and dichlorodiphenyldichloroethylene (DDE): A meta-analysis within 12 European birth cohorts. Environ. Health Perspect. 2012, 120, 162–170. [Google Scholar]
- WHO. World Health Organization Child Growth Standards BMI-for-Age. Available online: http://www.who.int/childgrowth/standards/bmi_for_age/en/ (accessed on 27 February 2014).
- Schmidt, J.S.; Schaedlich, K.; Fiandanese, N.; Pocar, P.; Fischer, B. Effects of di(2-ethylhexyl) phthalate (DEHP) on female fertility and adipogenesis in C3H/N mice. Environ. Health Perspect. 2012, 120, 1123–1129. [Google Scholar] [CrossRef]
- Desvergne, B.; Feige, J.N.; Casals-Casas, C. Ppar-mediated activity of phthalates: A link to the obesity epidemic? Mol. Cell. Endocrinol. 2009, 304, 43–48. [Google Scholar] [CrossRef]
- Wolff, M.S.; Engel, S.M.; Berkowitz, G.S.; Ye, X.; Silva, M.J.; Zhu, C.; Wetmur, J.; Calafat, A.M. Prenatal phenol and phthalate exposures and birth outcomes. Environ. Health Perspect. 2008, 116, 1092–1097. [Google Scholar] [CrossRef]
- Suzuki, Y.; Niwa, M.; Yoshinaga, J.; Mizumoto, Y.; Serizawa, S.; Shiraishi, H. Prenatal exposure to phthalate esters and PAHs and birth outcomes. Environ. Int. 2010, 36, 699–704. [Google Scholar] [CrossRef]
- Philippat, C.; Mortamais, M.; Chevrier, C.; Petit, C.; Calafat, A.M.; Ye, X.; Silva, M.J.; Brambilla, C.; Pin, I.; Charles, M.A.; et al. Exposure to phthalates and phenols during pregnancy and offspring size at birth. Environ. Health Perspect. 2012, 120, 464–470. [Google Scholar]
- Snijder, C.A.; Roeleveld, N.; Te Velde, E.; Steegers, E.A.; Raat, H.; Hofman, A.; Jaddoe, V.W.; Burdorf, A. Occupational exposure to chemicals and fetal growth: The generation R study. Hum. Reprod. 2012, 27, 910–920. [Google Scholar] [CrossRef]
- WHO. World Health Organization Child Growth Standards Head Circumference-for-Age. Available online: http://www.who.int/childgrowth/standards/hc_for_age/en/ (accessed on 18 March 2014).
- Cupul-Uicab, L.A.; Hernandez-Avila, M.; Terrazas-Medina, E.A.; Pennell, M.L.; Longnecker, M.P. Prenatal exposure to the major ddt metabolite 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene (DDE) and growth in boys from Mexico. Environ. Health Perspect. 2010, 110, 595–603. [Google Scholar]
- Tomatis, L.; Turusov, V.; Day, N.; Charles, R.T. The effect of long-term exposure to DDT on CF-1 mice. Int. J. Cancer 1972, 10, 489–506. [Google Scholar] [CrossRef]
- Andersen, C.S.; Fei, C.; Gamborg, M.; Nohr, E.A.; Sorensen, T.I.; Olsen, J. Prenatal exposures to perfluorinated chemicals and anthropometric measures in infancy. Am. J. Epidemiol. 2010, 172, 1230–1237. [Google Scholar] [CrossRef]
- Tang-Peronard, J.L.; Heitmann, B.L.; Andersen, H.R.; Steuerwald, U.; Grandjean, P.; Weihe, P.; Jensen, T.K. Association between prenatal polychlorinated biphenyl exposure and obesity development at ages 5 and 7 y: A prospective cohort study of 656 children from the faroe islands. Am. J. Clin. Nutr. 2014, 99, 5–13. [Google Scholar] [CrossRef]
- Hatch, E.; Nelson, J.; Qureshi, M.M.; Weinberg, J.; Moore, L.; Singer, M.; Webster, T. Association of urinary phthalate metabolite concentrations with body mass index and waist circumference: A cross-sectional study of NHANES data, 1999–2002. Environ. Health 2008, 7, 27. [Google Scholar] [CrossRef]
- Padmanabhan, V.; Sarma, H.N.; Savabieasfahani, M.; Steckler, T.L.; Veiga-Lopez, A. Developmental reprogramming of reproductive and metabolic dysfunction in sheep: Native steroids vs. environmental steroid receptor modulators. Int. J. Androl. 2010, 33, 394–404. [Google Scholar] [CrossRef]
- Lin, Y.; Wei, J.; Li, Y.; Chen, J.; Zhou, Z.; Song, L.; Wei, Z.; Lv, Z.; Chen, X.; Xia, W.; et al. Developmental exposure to di(2-ethylhexyl) phthalate impairs endocrine pancreas and leads to long-term adverse effects on glucose homeostasis in the rat. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E527–E538. [Google Scholar] [CrossRef]
- Reynolds, R.M. Glucocorticoid excess and the developmental origins of disease: Two decades of testing the hypothesis—2012 curt richter award winner. Psychoneuroendocrinology 2012, 38, 1–11. [Google Scholar] [CrossRef]
- Antunes-Fernandes, E.C.; Bovee, T.F.; Daamen, F.E.; Helsdingen, R.J.; van den Berg, M.; van Duursen, M.B. Some OH-PCBs are more potent inhibitors of aromatase activity and (anti-) glucocorticoids than non-dioxin like (NDL)-PCBs and MeSO(2)-PCBs. Toxicol. Lett. 2011, 206, 158–165. [Google Scholar] [CrossRef]
- Sargis, R.M.; Johnson, D.N.; Choudhury, R.A.; Brady, M.J. Environmental endocrine disruptors promote adipogenesis in the 3T3-L1 cell line through glucocorticoid receptor activation. Obesity 2010, 18, 1283–1288. [Google Scholar] [CrossRef]
- Maisonet, M.; Terrell, M.L.; McGeehin, M.A.; Christensen, K.Y.; Holmes, A.; Calafat, A.M.; Marcus, M. Maternal concentrations of polyfluoroalkyl compounds during pregnancy and fetal and postnatal growth in british girls. Environ. Health Perspect. 2012, 120, 1432–1437. [Google Scholar] [CrossRef]
- Schonbeck, Y.; Talma, H.; van Dommelen, P.; Bakker, B.; Buitendijk, S.E.; Hirasing, R.A.; van Buuren, S. Increase in prevalence of overweight in dutch children and adolescents: A comparison of nationwide growth studies in 1980, 1997 and 2009. PLoS One 2011, 6, e27608. [Google Scholar] [CrossRef]
- Eurostat. Eurostat Education and Training; Eurostat: Luxembourg, Luxembourg, 2012. [Google Scholar]
- Nieminen, P.; Lehtiniemi, H.; Huusko, A.; Vahakangas, K.; Rautio, A. Polychlorinated biphenyls (PCBs) in relation to secondary sex ratio—A systematic review of published studies. Chemosphere 2013, 91, 131–138. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Cock, M.; De Boer, M.R.; Lamoree, M.; Legler, J.; Van de Bor, M. First Year Growth in Relation to Prenatal Exposure to Endocrine Disruptors — A Dutch Prospective Cohort Study. Int. J. Environ. Res. Public Health 2014, 11, 7001-7021. https://doi.org/10.3390/ijerph110707001
De Cock M, De Boer MR, Lamoree M, Legler J, Van de Bor M. First Year Growth in Relation to Prenatal Exposure to Endocrine Disruptors — A Dutch Prospective Cohort Study. International Journal of Environmental Research and Public Health. 2014; 11(7):7001-7021. https://doi.org/10.3390/ijerph110707001
Chicago/Turabian StyleDe Cock, Marijke, Michiel R. De Boer, Marja Lamoree, Juliette Legler, and Margot Van de Bor. 2014. "First Year Growth in Relation to Prenatal Exposure to Endocrine Disruptors — A Dutch Prospective Cohort Study" International Journal of Environmental Research and Public Health 11, no. 7: 7001-7021. https://doi.org/10.3390/ijerph110707001




