Though not Reservoirs, Dogs might Transmit Leptospira in New Caledonia
Abstract
:1. Introduction
2. Experimental Section
2.1. DNA Extraction
2.2. Molecular Tools
2.3. Leptospira Genotyping by Sequencing
2.4. Serology
| Species | Serogroup | serovar | Strain |
|---|---|---|---|
| L. interrogans | Australis | Australis | Ballico |
| L. interrogans | Autumnalis | Autumnalis | Akiyami A |
| L. borgpetersenii | Ballum | castellonis | Castellon 3 |
| L. interrogans | Bataviae | Bataviae | Van Tienen |
| L. interrogans | Canicola | Canicola | Hond Utrecht |
| L. interrogans | Icterohaemorrhagiae | Icterohaemorrhagiae | Verdun |
| L. interrogans | Icterohaemorrhagiae | Copenhagenii | Winjberg |
| L. noguchi | Panama | Panama | CZ 214 K |
| L. interrogans | Pomona | Pomona | Pomona |
| L. interrogans | Pyrogenes | Pyrogenes | Salinem |
| L. borgpetersenii | Tarassovi | Tarassovi | Mitis Johnson |
| L. biflexa | Semarranga | Patoc | Patoc I |
3. Results
3.1. Leptospira Carriage
| Mammal | Source | Sample size | PCR inhibition | Positive | Negative | Prevalence | 95% CI |
|---|---|---|---|---|---|---|---|
| Deer * | Hunting | 85 | 9 | 25 | 167 | 13.02 | [8.26–17.78] |
| Slaughtered | 107 | ||||||
| Pig * | Feral | 94 | 0 | 6 | 88 | 6.38 | [1.44–11.32] |
| Farmed | 138 | 1 | 14 | 123 | 10.22 | [5.15–15.29] | |
| Dog | Urban pound (kidney) | 82 | 4 | 3 | 75 | 4.4 | [0.19–8.61] |
| Tribes (urine) | 13 | 0 | 1 | 12 |
3.2. Strain Identification by Genotyping
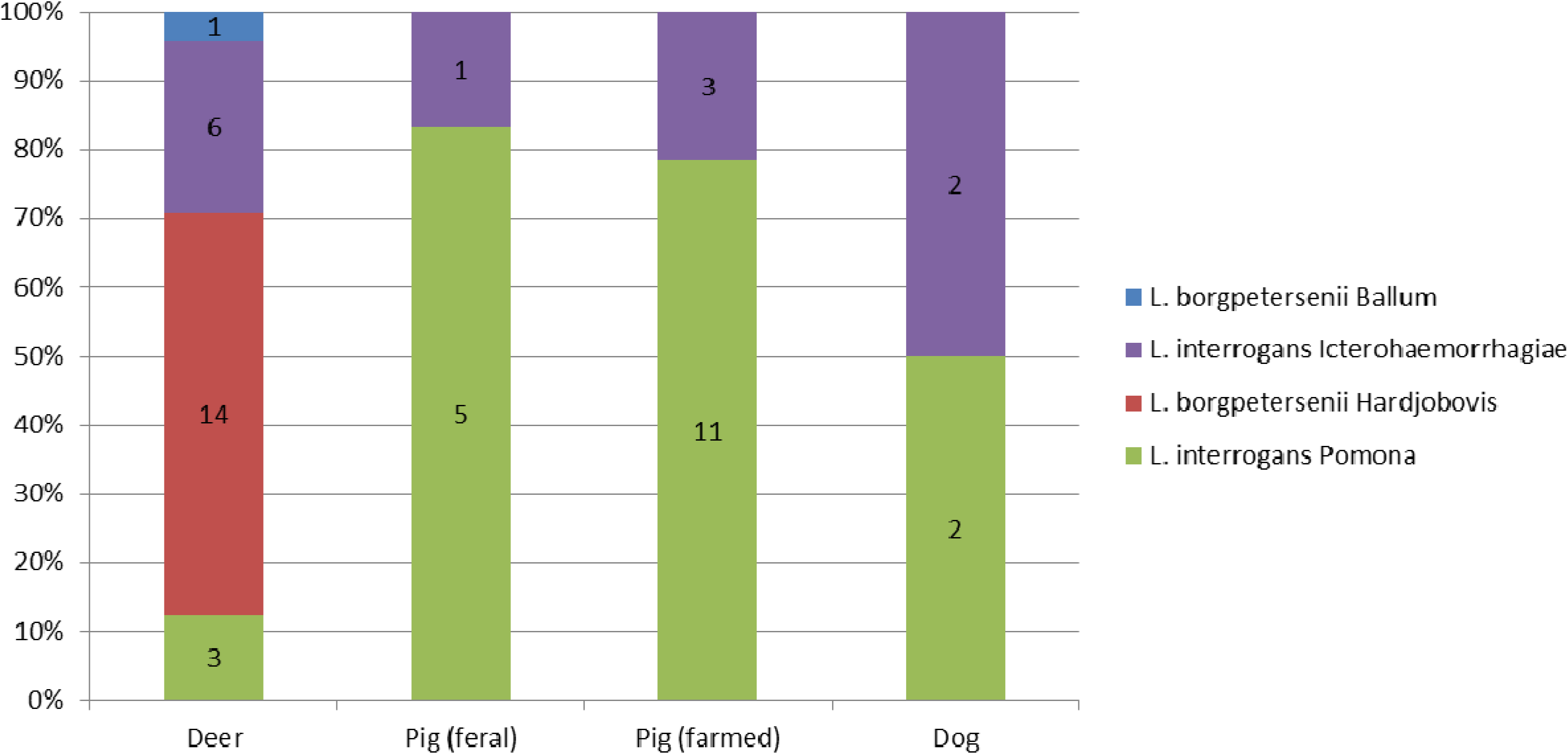
3.3. MAT on Dog Sera
| Putative Serogroup | Australis | Icterohaemorrhagiae | Canicola | Pyrogenes | Unknown * |
|---|---|---|---|---|---|
| Urban dogs (n = 31) | 0 | 4 | 0 | 0 | 1 |
| Rural dogs (n = 47) | 3 | 11 | 0 | 1 | 4 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Berlioz-Arthaud, A.; Kiedrzynski, T.; Singh, N.; Yvon, J.F.; Roualen, G.; Coudert, C.; Uluiviti, V. Multicentre survey of incidence and public health impact of leptospirosis in the Western Pacific. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 714–721. [Google Scholar]
- Victoriano, A.F.; Smythe, L.D.; Gloriani-Barzaga, N.; Cavinta, L.L.; Kasai, T.; Limpakarnjanarat, K.; Ong, B.L.; Gongal, G.; Hall, J.; Coulombe, C.A.; Yanagihara, Y.; Yoshida, S.I.; Adler, B. Leptospirosis in the Asia Pacific region. BMC Infect. Dis. 2009, 9. [Google Scholar] [CrossRef]
- Adler, B.; de la Pena Moctezuma, A. Leptospira and leptospirosis. Vet. Microbiol. 2009, 140, 287–296. [Google Scholar]
- Baranton, G.; Old, I.G. The spirochaetes: A different way of life. Bull. Inst. Pasteur. 1995, 93, 63–95. [Google Scholar]
- Levett, P.N. Leptospirosis. Clin. Microbiol. Rev. 2001, 14, 296–326. [Google Scholar] [CrossRef]
- Babudieri, B. Animal reservoirs of leptospires. Ann. N.Y. Acad. Sci. 1958, 70, 393–413. [Google Scholar] [CrossRef]
- Desvars, A. Epidémiologie d'une zoonose, la leptospirose, dans deux îles de l'Océan indien, la Réunion et Mayotte–Etude comparée du rôle de différentes espèces sauvages et domestiques. Ph.D. Thesis, Université de la Rénuion, Saint Denis, La Réunion, 26 April 2012. [Google Scholar]
- Berlioz-Arthaud, A.; Merien, F.; Baranton, G. Laboratory based human leptospirosis surveillance in New Caledonia (2001–2005). Bull. Soc. Pathol. Exot. 2007, 100, 133–138. (In French) [Google Scholar]
- Goarant, C.; Marchal, C.; Gourinat, A.-C. La surveillance de la leptospirose en Nouvelle-Calédonie: approche pluridisciplinaire d’une zoonose endémique. Bulletin épidémiologique, santé animale et alimentation 2011, 43, 22–25. [Google Scholar]
- Gargominy, O.; Bouchet, P.; Pascal, M.; Jaffré, T.; Tourneur, J.-C. Conséquences des introductions d’espèces animales et végétales sur la biodiversité en Nouvelle-Calédonie. Rev. Ecol. (Terre Vie) 1996, 51, 375–402. [Google Scholar]
- Salaün, L.; Merien, F.; Gurianova, S.; Baranton, G.; Picardeau, M. Application of multilocus variable-number tandem-repeat analysis for molecular typing of the agent of leptospirosis. J. Clin. Microbiol. 2006, 44, 3954–3962. [Google Scholar]
- Perez, J.; Goarant, C. Rapid Leptospira identification by direct sequencing of the diagnostic PCR products in New Caledonia. BMC Microbiol. 2010, 10. [Google Scholar] [CrossRef]
- Andre-Fontaine, G. Canine leptospirosis—Do we have a problem? Vet. Microbiol. 2006, 117, 19–24. [Google Scholar] [CrossRef]
- Vinetz, J.M. Leptospirosis. Curr. Opin. Infect. Dis. 2001, 14, 527–538. [Google Scholar] [CrossRef]
- Perez, J.; Brescia, F.; Becam, J.; Mauron, C.; Goarant, C. Rodent abundance dynamics and leptospirosis carriage in an area of hyper-endemicity in New Caledonia. PLoS Negl. Trop. Dis. 2011, 5. [Google Scholar] [CrossRef]
- Goarant, C. Leptospirosis: Time to move to molecular epidemiology: Comments on “Reassessment of MLST schemes for Leptospira spp. typing worldwide” by Varni and colleagues. Infect. Genet. Evol. 2014, 21, 484–485. [Google Scholar] [CrossRef]
- Levett, P.N.; Morey, R.E.; Galloway, R.L.; Turner, D.E.; Steigerwalt, A.G.; Mayer, L.W. Detection of pathogenic leptospires by real-time quantitative PCR. J. Med. Microbiol. 2005, 54, 45–49. [Google Scholar]
- Stoddard, R.A.; Gee, J.E.; Wilkins, P.P.; McCaustland, K.; Hoffmaster, A.R. Detection of pathogenic leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn. Microbiol. Infect. Dis. 2009, 64, 247–255. [Google Scholar]
- Merien, F.; Portnoi, D.; Bourhy, P.; Charavay, F.; Berlioz-Arthaud, A.; Baranton, G. A rapid and quantitative method for the detection of Leptospira species in human leptospirosis. FEMS Microbiol. Lett. 2005, 249, 139–147. [Google Scholar]
- Thornley, C.N.; Baker, M.G.; Weinstein, P.; Maas, E.W. Changing epidemiology of human leptospirosis in New Zealand. Epidemiol. Infect. 2002, 128, 29–36. [Google Scholar]
- Goldstein, R.E. Canine leptospirosis. Vet. Clin. North Am. 2010, 40, 1091–1101. [Google Scholar] [CrossRef]
- Weekes, C.C.; Everard, C.O.; Levett, P.N. Seroepidemiology of canine leptospirosis on the island of Barbados. Vet. Microbiol. 1997, 57, 215–222. [Google Scholar] [CrossRef]
- Jansen, A.; Schoneberg, I.; Frank, C.; Alpers, K.; Schneider, T.; Stark, K. Leptospirosis in Germany, 1962–2003. Emerg. Infect. Dis. 2005, 11, 1048–1054. [Google Scholar]
- Trevejo, R.T.; Rigau-Perez, J.G.; Ashford, D.A.; McClure, E.M.; Jarquin-Gonzalez, C.; Amador, J.J.; de los Reyes, J.O.; Gonzalez, A.; Zaki, S.R.; Shieh, W.J.; et al. Epidemic leptospirosis associated with pulmonary hemorrhage—Nicaragua, 1995. J. Infect. Dis. 1998, 178, 1457–1463. [Google Scholar]
- Douglin, C.P.; Jordan, C.; Rock, R.; Hurley, A.; Levett, P.N. Risk factors for severe leptospirosis in the parish of St. Andrew, Barbados. Emerg. Infect. Dis. 1997, 3, 78–80. [Google Scholar]
- Ghneim, G.S.; Viers, J.H.; Chomel, B.B.; Kass, P.H.; Descollonges, D.A.; Johnson, M.L. Use of a case-control study and geographic information systems to determine environmental and demographic risk factors for canine leptospirosis. Vet. Res. 2007, 38, 37–50. [Google Scholar]
- Langston, C.E.; Heuter, K.J. Leptospirosis: A re-emerging zoonotic disease. Vet. Clin. North Am. 2003, 33, 791–807. [Google Scholar]
- Yoak, A.J.; Reece, J.F.; Gehrt, S.D.; Hamilton, I.M. Disease control through fertility control: Secondary benefits of animal birth control in Indian street dogs. Prev. Vet. Med. 2014, 113, 152–156. [Google Scholar]
- Minke, J.M.; Bey, R.; Tronel, J.P.; Latour, S.; Colombet, G.; Yvorel, J.; Cariou, C.; Guiot, A.L.; Cozette, V.; Guigal, P.M. Onset and duration of protective immunity against clinical disease and renal carriage in dogs provided by a bi-valent inactivated leptospirosis vaccine. Vet. Microbiol. 2009, 137, 137–145. [Google Scholar]
- Prescott, J.F.; McEwen, B.; Taylor, J.; Woods, J.P.; Abrams-Ogg, A.; Wilcock, B. Resurgence of leptospirosis in dogs in Ontario: Recent findings. Can. Vet. J. 2002, 43, 955–961. [Google Scholar]
- Mayer-Scholl, A.; Luge, E.; Draeger, A.; Nockler, K.; Kohn, B. Distribution of Leptospira Serogroups in dogs from Berlin, Germany. Vector Borne Zoonotic Dis. 2013, 13, 200–202. [Google Scholar] [CrossRef]
- Ellis, W.A. Control of canine leptospirosis in Europe: Time for a change? Vet. Rec. 2010, 167, 602–605. [Google Scholar] [CrossRef]
- Davis, M.A.; Evermann, J.F.; Petersen, C.R.; Vanderschalie, J.; Besser, T.E.; Huckabee, J.; Daniels, J.B.; Hancock, D.D.; Leslie, M.; Baer, R. Serological survey for antibodies to Leptospira in dogs and raccoons in Washington State. Zoonoses Public Health 2008, 55, 436–442. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gay, N.; Soupé-Gilbert, M.-E.; Goarant, C. Though not Reservoirs, Dogs might Transmit Leptospira in New Caledonia. Int. J. Environ. Res. Public Health 2014, 11, 4316-4325. https://doi.org/10.3390/ijerph110404316
Gay N, Soupé-Gilbert M-E, Goarant C. Though not Reservoirs, Dogs might Transmit Leptospira in New Caledonia. International Journal of Environmental Research and Public Health. 2014; 11(4):4316-4325. https://doi.org/10.3390/ijerph110404316
Chicago/Turabian StyleGay, Noellie, Marie-Estelle Soupé-Gilbert, and Cyrille Goarant. 2014. "Though not Reservoirs, Dogs might Transmit Leptospira in New Caledonia" International Journal of Environmental Research and Public Health 11, no. 4: 4316-4325. https://doi.org/10.3390/ijerph110404316
APA StyleGay, N., Soupé-Gilbert, M.-E., & Goarant, C. (2014). Though not Reservoirs, Dogs might Transmit Leptospira in New Caledonia. International Journal of Environmental Research and Public Health, 11(4), 4316-4325. https://doi.org/10.3390/ijerph110404316




