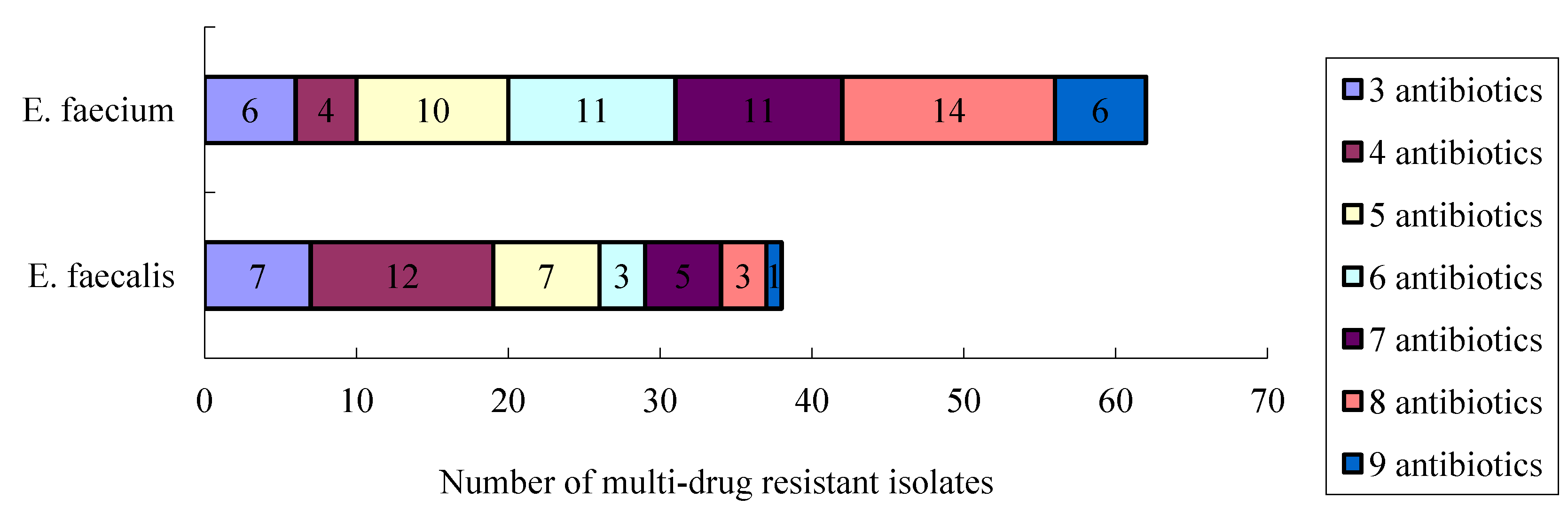
4. Discussion
Due to the spread of enterococcal antimicrobial resistance [
8,
9], the tracing of the infectious sources is of great significance for the control of enterococcal infections and its spreading. Among the 289 enterococcal strainss isolated from a tertiary-care pediatric hospital in Mexico City during an 18-month period,
E. faecalis and
E. faecium comprised 81.2% of the total isolates, and antimicrobial resistance in
Enterococcus spp. was found to be common [
10]. Of the 415 enterococcal isolates obtained from clinical samples between January 1999 and 31 December 2001 in the Mubarak Al-Kabeer, Amiri, Adan, Ibn Sina and Maternity hospitals in Kuwait,
E. faecalis (85.3%) and
E. faecium (7.7%) accounted for 93% of the samples [
11]. Salem-Bekhit and colleagues identified 69.2%
E. faecalis and 11.3%
E. faecium in 206 enterococcal species obtained from the clinical samples in Riyadh hospitals of King Saud University, Saudi Arabia [
12].
Maschieto
et al. reported that the distribution of
Enterococcus spp. isolated from the intestinal tracts of patients from a university hospital in Brazil was
E. faecium (34%), followed by
E. faecalis (33%),
E. gallinarum (23.7%),
E. casseliflavus (5.2%),
E. avium (1%), and
E. hirae (1%) [
13]. In China,
E. faecium and
E. faecalis were also found to be predominant in the enterococci isolated from clinical specimens [
14,
15,
16,
17]. Similar to these findings, the current study showed that
E. faecium (58.7%) and
E. faecalis (33%) were predominant in the 1157 clinical isolates of
Enterococcus species isolated from our hospital. However, the present study involved a large sample size, compared the antimicrobial resistance in enterococcal strains isolated from different departments of the hospital, and investigated the efflux mechanism of resistance in enterococci, which is rarely reported previously. The
Enterococcus species were mainly isolated from the urinary system clinical specimens, which was in agreement with the detection of
Enterococcus species isolated from the First Affiliated Hospital of Chongqing Medical University [
18]. In addition,
Enterococcus species were found to be predominantly isolated from the burns department, ICU and pediatrics department, which was associated with the patients’ critical illness, long-term antibiotic use and decline in immune function [
19].
Enterococcus species are found to be intrinsically resistant to cephalosporins and aminoglycosides. Even though bacteria were found to be sensitive to these drugs in
in-vitro experiments, unsatisfactory efficacy was found in clinical practice [
2,
20,
21]. Multiple-antimicrobial resistance has been widely reported in
Enterococcus species [
22,
23,
24,
25].
In the current study, a significantly higher prevalence of resistance to penicillin, ampicillin, rifampicin, ciprofloxacin, levofloxacin, fosfomycin, erythromycin and furadantin was detected in
E. faecium than in
E. faecalis (
p < 0.05), while a greater prevalence of resistance to chloramphenicol, quinupristin/dalfopristin, minocycline and tetracycline was found in
E. faecalis than in
E. faecium (
p < 0.05). In addition, a low prevalence of resistance to linezolid, vancomycin and teicoplanin was detected in both
E. faecium and
E. faecalis. Therefore, linezolid, vancomycin and teicoplanin are currently widely used drugs for the effective treatment of enterococcal infections [
22,
23,
26]. Quinupristin/dalfopristin, a novel streptogramin antibiotic agent, has been widely used for the treatment of vancomycin-resistant enterococcal infections in USA and Europe, and a high therapeutic efficacy has been achieved [
27,
28,
29]. The mechanism of action of the agent is found to involve early and late stage inhibition of bacterial protein synthesis [
30,
31], however, the drug shows poor efficacy against
E. faecalis [
32,
33]. High rates of resistance to quinupristin-dalfopristin have been detected in enterococci isolated from poultry production environments [
34], chickens [
35], and clinical specimens [
36,
37,
38]. In the present study, the prevalence of quinupristin-dalfopristin resistance was 81.2% in
E. faecalis, which was significantly higher than that the 1.8% in
E. faecium (
p < 0.05). In addition, quinupristin/dalfopristin has been recommended by CLSI for the treatment of vancomycin-resistant enterococcal infections. Since antimicrobial resistance varies in
Enterococcus species, there is a great need to identify enterococcal strains to the species level, which would facilitate the appropriate selection of antibiotics.
Like previous reports [
16,
17], our findings also found that the prevalence of antimicrobial resistance varied in the enterococci isolated from different departments of the hospital. A lower prevalence of antibiotic resistance was detected in the enterococci isolated from the department of pediatrics as compared to those from other departments of the hospital, while a high prevalence of penicillin resistance was found, which may be associated with the frequent application of penicillin, a commonly used drug in pediatrics. A high prevalence of antimicrobial resistance was found in the enterococci isolated from the burns department and ICU of the hospital, which may be attributed to the patients’ critical illness, poor immunity and long-term antibiotic use, or the habit of the antibiotic use [
19].
To understand the shift of antimicrobial resistance in enterococci in our hospital, we compared the results from this study to the distribution of antimicrobial resistance in enterococci isolated from clinical specimens during the period from January 2007 through December 2009 [
39], and found a great rise in the number of enterococcal isolates, in which
E. faecium was still predominant, but its constituent ratio increased. In addition, the enterococcal isolates were still resistant to more than 40% of the commonly used antibiotics; however, no significant rise was found in the prevalence of antimicrobial resistance. In the current study, we identified 10 linezolid-resistant enterococcal strains, which were not detected in the enterococci isolated between 2007 and 2009. It is considered that the continuous antibiotic pressure causes the secondary resistance to linezolid in enterococci [
40].
Reserpine has been proved to reduce the MIC of fluoroquinolones against antimicrobial-resistant bacteria [
41,
42,
43]. It is found that the combination of the multidrug efflux inhibitor reserpine and fluoroquinolone enhances the sensitivity of fluoroquinolone-resistant
Streptococcus pneumonia and
Staphylococcus aureus to fluoroquinolones [
44]. Our findings showed that reserpine treatment caused a significant reduction in the resistance to the three fluoroquinolones ciprofloxacin, gatifloxacin and levofloxacin in
Enterococcus species, and the MIC of fluoroquinolones was reduced by over 2-fold in 72% of the enterococcal isolates.
In the current study, the emeA gene was detected in 73.8% of the ciprofloxacin-resistant enterococci, 76.7% of the gatifloxacin-resistant enterococci, and 75.8% of the levofloxacin-resistant enterococci, respectively, suggesting the presence of other mechanisms involved in the resistance of enterococci to the three fluoroquinolones in addition to drug efflux, and such a gene was present in 41.4% of the ciprofloxacin-sensitive enterococci, 45.7% of the gatifloxacin-sensitive enterococci, and 44.8% of the levofloxacin-sensitive enterococci, respectively, indicating no expression of the emeA gene in some enterococcal isolates. In addition, the occurrence of the emeA gene was significantly greater in the fluoroquinolone-resistant enterococci than that in the fluoroquinolone-sensitive enterococci (p < 0.05), indicating that the distribution of the emeA gene was associated with the resistance to the three fluoroquinolones in the Enterococcus species.
It is indicated that the resistance of enterococci to β-lactam is caused by the production of β-lactamase, which is encoded by the
TEM gene, or modification in the penicillin-binding proteins (PBPs) [
45,
46]. In the current study, a high prevalence of penicillin resistance was detected in
E. faecium, while a low prevalence was found in
E. faecalis, and the occurrence of the
TEM gene was 95.1% and 47.4% in
E. faecium and
E. faecalis, respectively. The aminoglycosides resistance in enterococci is mainly attributable to the production of aminoglycoside-modifying enzymes [
47]. Currently, over 30 aminoglycoside modifying enzymes have been identified, in which bifunctional 6'-aminoglycoside acetyltransferase (AAC(6')) 2"-aminoglycoside phosphotransferase (APH(2")) enzyme encoded by the
aac(6')/aph(2") gene is the most common one, which eliminates the synergistic effect between penicillin or glycopeptide antibiotics and aminoglycosides [
48]. Our findings showed that the occurrence of the
aac(6')/aph(2") gene, the
aph(3')-III gene that encodes aminoglycoside 3'-type III phosphotransferase (APH(3')-III), the
ant(6)-I gene that encodes 6-nucleotidyltransferase I (ANT(6)-I) and the
ant(2")-I gene that encodes aminoglycoside- 2"-O-nucleotidyltransferase I (ANT(2")-I) was 62%, 26%, 13% and 36% in the 100 multiple-drug resistant enterococcal isolates, respectively. The resistance of enterococci to tetracyclines is mainly caused by the binding of the
tetM gene-encoded ribosomal protection proteins to the ribosome, thereby avoiding the effect of tetracyclines [
49]. In the current study, the prevalence of tetracycline resistance gene was 31.6% and 30.1% in
E. faecalis and
E. faecium, respectively. It is therefore considered that the resistance of
Enterococcus species to β-lactam, aminoglycosides and tetracyclines is attributable to the presence of the gene that encodes the corresponding enzymes. Two mechanisms are considered to be responsible for macrolides resistance in enterococci, including the change in the target site of erythromycin mediated by the
erm gene, and
mef gene-mediated antimicrobial efflux [
50,
51].
ermB gene is the predominant type of erm gene in enterococci [
50]. Our findings showed that the occurrence of the
ermB gene was 71.1% and 62.1% in
E. faecalis and
E. faecium, respectively, indicating that the macrolides resistance in the
Enterococcus species isolated from Ningxia region is mainly associated with the presence of the
ermB gene. Like previous studies [
52,
53], the
mefA gene was detected in enterococci in the current study; however, Liang
et al. [
54] detected the
mefA gene in 9 of 53 clinical isolates of
Enterococcus species, which may be due to the regional variation in the occurrence of the
mefA gene in enterococci.
It is indicated that the resistance to glycopeptides in enterococci is mainly caused by the alteration of peptidoglycan precursors on the cell wall of enterococci, which leads to the failure of the glycopeptides to inhibiting the synthesis of the cell walls of enterococci, thereby resulting in the emergence of glycopeptide resistance [
55,
56]. In the present study, the
vanA gene was detected in all of the 5 vancomycin-resistant isolates of enterococci. Vancomycin-resistant enterococci may transfer the
vanA gene to
S. aureus, which leads to the emergence of vancomycin-resistant
S. aureus, thereby resulting in more difficulty in the clinical treatment of enterococcal infections [
5]. Therefore, vancomycin should be used cautiously in the clinical therapy of enterococcal infections, and the management of vancomycin-resistant enterococci should be improved [
22,
57].