Effect of Chlorine Exposure on the Survival and Antibiotic Gene Expression of Multidrug Resistant Acinetobacter baumannii in Water
Abstract
:1. Introduction
2. Experimental Section
2.1. A. baumannii Strains and Growth Conditions
2.2. A. baumannii Survival Assay
2.3. Antibiotic Resistance Gene Expression
2.3.1. RNA Isolation and cDNA Synthesis
2.3.2. Real-Time Quantitative PCR (RT-qPCR)
| Gene | Sequence (5’→3’) | Function |
|---|---|---|
| adeA (F) | TGACCGACCAATGCACCTT | Efflux pump |
| (R) | GCAACAGTTCGAGCGCCTAT | |
| adeB (F) | CCGATGACGTATCGAAGTTAGGA | Efflux pump |
| (R) | CCGATGACGTATCGAAGTTAGGA | |
| adeC (F) | ACGGCCCCAGAAGTCTAGTTC | Efflux pump |
| (R) | CGATTAACCCCAATAACCCAGTT | |
| adeM (F) | GGTACATGGAAGCCCAGTTCTT | Efflux pump |
| (R) | CCACTTTCTCTTGCCATTGCT | |
| blaP (F) | ACACTAGGAGAAGCCATGAAGCTT | Beta-lactam resistanceAntibiotics |
| (R) | GCATGAGATCAAGACCGATACG | |
| cmr (F) | CTATTTGAATTTGCGGTTTATATTGG | Chloramphenicol resistance |
| (R) | TGCACTTACACCGAAATCTTCAG | |
| ami (F) | TGATCCCGTAAATGAGTTGAATTG | Aminoglycoside resistance |
| (R) | GCGGGCAAATGTGATGGTA | |
| sul1 (F) | GGCATGACAATAGGGCAGTTG | Sulphonamide resistance |
| (R) | CCAAAAAGTAGATGATAATACCGGTAAA | |
| tetA (F) | CTGCGCGATCTGGTTCACT | Tetracycline resistance |
| (R) | GCATACAGCGCCAGCAGAA | |
| mdrp (F) | GTACGGCTTCTAGACCCACCATTTT | Multiple drug resistanceprotein |
| (R) | ACAAAGAGCCGTGCACAGTTT | |
| rRNA-16S(F) | TCGCTAGTAATCGCGGATCACGCTGGCGGC | Endogenous control |
| rRNA-16S(R) | GACGGGCGGTGTGTACAAG |
2.4. Statistical Analysis
3. Results and Discussion
3.1. Effect of Chlorine on A. baumannii Survival
| Free Chlorine (ppm) | A. baumannii Counts (mean ± SD **) | |||
|---|---|---|---|---|
| 30 seconds | 60 seconds | 90 seconds | 120 seconds | |
| 0 | 7.34 ± 0.24 | 7.38 ± 0.23 | 7.37 ± 0.26 | 7.35 ± 0.23 |
| 0.2 | 7.37 ± 0.15 | 7.39 ± 0.18 | 7.37 ± 0.18 | 7.39 ± 0.20 |
| 1 | 7.35 ± 0.18 | 7.34 ± 0.18 | 7.33 ± 0.20 | 7.34 ± 0.22 |
| 2 | 7.27 ± 0.51 | 7.27 ± 0.53 | 7.25 ± 0.50 | 7.20 ± 0.55 |
| 3 | 7.28 ± 0.48 | 7.29 ± 0.47 | 7.26 ± 0.46 | 7.24 ± 0.50 |
| 4 | 7.28 ± 0.46 | 7.29 ± 0.50 | 7.25 ± 0.47 | 7.24 ± 0.49 |
3.2. Effect of Chlorine on A. baumannii Antibiotic Resistance Genes
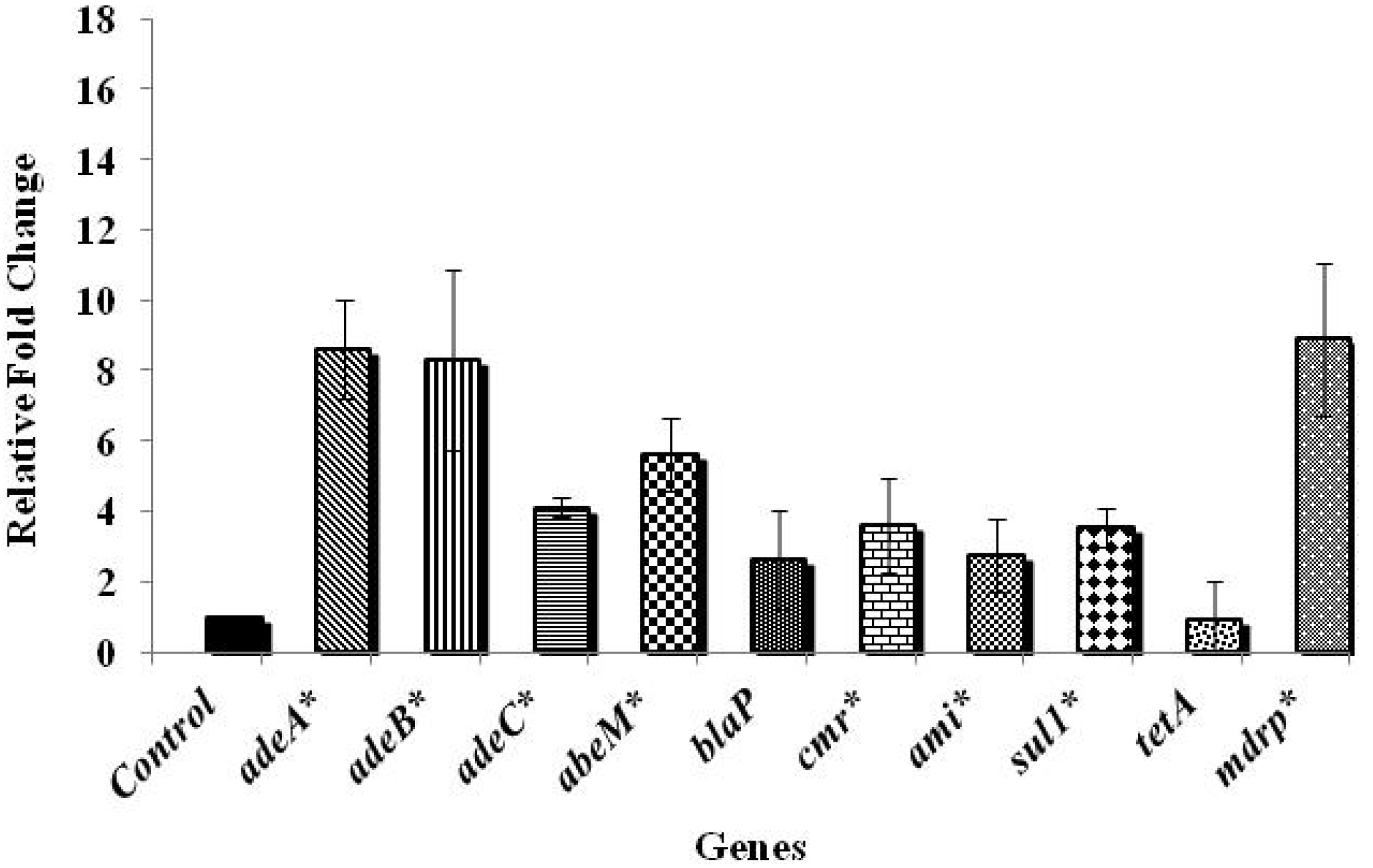
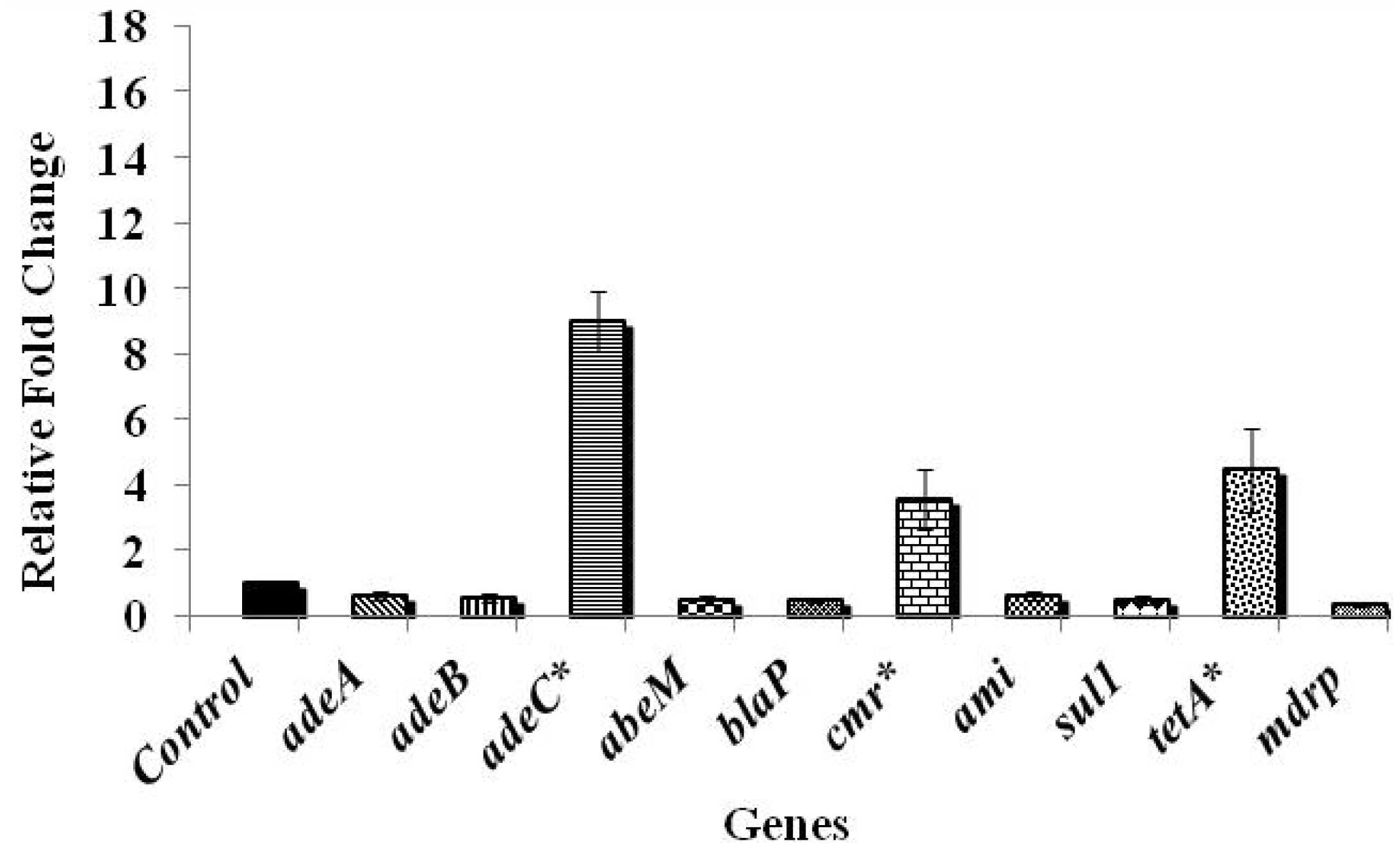
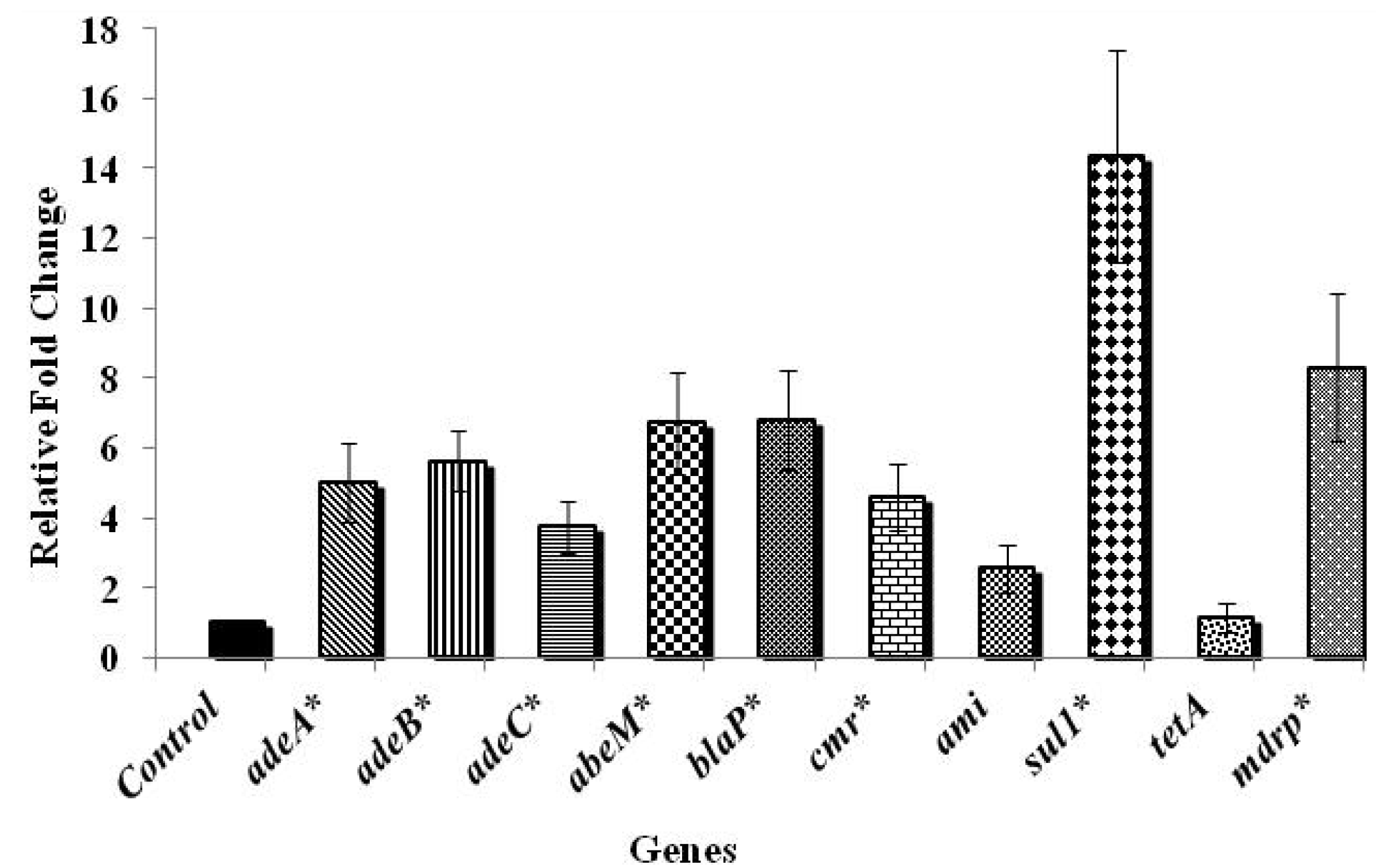
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dijkshoorn, L.; Nemec, A.; Seifert, H. An increasing threat in hospitals: Multidrug-resistant Acinetobacter baumannii. Nat. Rev. Microbiol. 2007, 5, 939–951. [Google Scholar] [CrossRef]
- Giamarellou, H. Multidrug-resistant gram-negative bacteria: How to treat and for how long. Int. J. Antimicrob. Agents 2010, 36, S50–S54. [Google Scholar] [CrossRef]
- Karageorgopoulos, D.E.; Falagas, M.E. Current control and treatment of multidrug-resistant Acinetobacter baumannii infections. Lancet Infect. Dis. 2008, 8, 751–762. [Google Scholar] [CrossRef]
- Marti, S.; Rodriguez-Bano, J.; Catel-Ferreira, M.; Jouenne, T.; Vila, J.; Seifert, H.; De, E. Biofilm formation at the solid-liquid and air-Liquid interfaces by Acinetobacter Species. BMC Res. Notes 2011, 4. [Google Scholar] [CrossRef]
- Peleg, A.Y.; Seifert, H.; Paterson, D.L. Acinetobacter baumannii: Emergence of a successful pathogen. Clin. Microbiol. Rev. 2008, 21, 538–582. [Google Scholar] [CrossRef]
- Perez, F.; Hujer, A.M.; Hujer, K.M.; Decker, B.K.; Rather, P.N.; Bonomo, R.A. Global challenge of multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2007, 51, 3471–3484. [Google Scholar] [CrossRef]
- Gaddy, J.A.; Tomaras, A.P.; Actis, L.A. The Acinetobacter baumannii 19606 OmpA protein plays a role in biofilm formation on abiotic surfaces and in the interaction of this pathogen with eukaryotic cells. Infect. Immun. 2009, 77, 3150–3160. [Google Scholar] [CrossRef]
- Moultrie, D.; Hawker, J.; Cole, S. Factors associated with multidrug-resistant Acinetobacter transmission: An integrative review of the literature. AORN J. 2011, 94, 27–36. [Google Scholar] [CrossRef]
- Pour, N.K.; Dusane, D.H.; Dhakephalkar, P.K.; Zamin, F.R.; Zinjarde, S.S.; Chopade, B.A. Biofilm formation by Acinetobacter baumannii strains isolated from urinary tract infection and urinary catheters. FEMS Immunol. Med. Microbiol. 2011, 62, 328–338. [Google Scholar] [CrossRef]
- Shin, J.H.; Lee, H.W.; Kim, S.M.; Kim, J. Proteomic analysis of Acinetobacter baumannii in biofilm and planktonic growth mode. J. Microbiol. 2009, 47, 728–735. [Google Scholar] [CrossRef]
- Urban, C.; Segal-Maurer, S.; Rahal, J.J. Considerations in control and treatment of nosocomial infections due to multidrug-resistant Acinetobacter baumannii. Clin. Infect. Dis. 2003, 36, 1268–1274. [Google Scholar] [CrossRef]
- Wilks, M.; Wilson, A.; Warwick, S.; Price, E.; Kennedy, D.; Ely, A.; Millar, M.R. Control of an outbreak of multidrug-resistant Acinetobacter baumannii-calcoaceticus colonization and infection in an intensive care unit (ICU) without closing the ICU or placing patients in isolation. Infect. Control. Hosp. Epidemiol. 2006, 27, 654–658. [Google Scholar] [CrossRef]
- Baumann, P. Isolation of Acinetobacter from soil and water. J. Bacteriol. 1968, 96, 39–42. [Google Scholar]
- Berlau, J.; Aucken, H.M.; Houang, E.; Pitt, T.L. Isolation of Acinetobacter spp. including A. baumannii from vegetables: Implications for hospital-acquired infections. J. Hosp. Infect. 1999, 42, 201–204. [Google Scholar] [CrossRef]
- Fournier, P.E.; Richet, H. The epidemiology and control of Acinetobacter baumannii in health care facilities. Clin. Infect. Dis. 2006, 42, 692–699. [Google Scholar] [CrossRef]
- Munoz-Price, L.S.; Weinstein, R.A. Acinetobacter infection. N. Engl. J. Med. 2008, 358, 1271–1281. [Google Scholar] [CrossRef]
- Mussi, M.A.; Limansky, A.S.; Viale, A.M. Acquisition of resistance to carbapenems in multidrug-resistant clinical strains of Acinetobacter baumannii: Natural insertional inactivation of a gene encoding a member of a novel family of beta-barrel outer membrane proteins. Antimicrob. Agents Chemother. 2005, 49, 1432–1440. [Google Scholar] [CrossRef]
- Cateau, E.; Verdon, J.; Fernandez, B.; Hechard, Y.; Rodier, M.H. Acanthamoeba sp. promotes the survival and growth of Acinetobacter baumannii. FEMS Microbiol. Lett. 2011, 319, 19–25. [Google Scholar] [CrossRef]
- Ferreira, A.E.; Marchetti, D.P.; de Oliveira, L.M.; Gusatti, C.S.; Fuentefria, D.B.; Corcao, G. Presence of OXA-23-producing isolates of Acinetobacter baumannii in wastewater from hospitals in southern Brazil. Microb. Drug Resist. 2011, 17, 221–227. [Google Scholar] [CrossRef]
- Zhang, C.; Qiu, S.; Wang, Y.; Qi, L.; Hao, R.; Liu, X.; Shi, Y.; Hu, X.; An, D.; Li, Z.; et al. Higher isolation of NDM-1 producing Acinetobacter baumannii from the sewage of the hospitals in Beijing. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Kempf, M.; Rolain, J.M. Emergence of resistance to carbapenems in Acinetobacter baumannii in Europe: Clinical impact and therapeutic options. Int. J. Antimicrob. Agents 2012, 39, 105–114. [Google Scholar] [CrossRef] [Green Version]
- Ngwenya, N.; Ncube, E.J.; Parsons, J. Recent advances in drinking water disinfection: Successes and challenges. Rev. Environ. Contam. Toxicol. 2013, 222, 111–170. [Google Scholar]
- Shrivastava, R.; Upreti, R.K.; Jain, S.R.; Prasad, K.N.; Seth, P.K.; Chaturvedi, U.C. Suboptimal chlorine treatment of drinking water leads to selection of multidrug-resistant Pseudomonas aeruginosa. Ecotoxicol. Environ. Saf. 2004, 58, 277–283. [Google Scholar] [CrossRef]
- Guidelines for Drinking Water Quality. Available online: http://www.who.int/water_sanitation_health/publications/2011/dwq_guidelines/en/index.html (accessed on 1 November 2013).
- Basic Information about Disinfectants in Drinking Water: Chloramine, Chlorine and Chlorine Dioxide. Available online: http://water.epa.gov/drink/contaminants/basicinformation/disinfectants.cfm (accessed on 1 November 2013).
- Healthy Swimming or Recreational Water. Available online: http://www.cdc.gov/healthywater/swimming/pools/disinfection-team-chlorine-ph.html (accessed on 1 November 2013).
- LeChevallier, M.W.; Babcock, T.M.; Lee, R.G. Examination and characterization of distribution system biofilms. Appl. Environ. Microbiol. 1987, 53, 2714–2724. [Google Scholar]
- Ridgway, H.F.; Olson, B.H. Chlorine resistance patterns of bacteria from two drinking water distribution systems. Appl. Environ. Microbiol. 1982, 44, 972–987. [Google Scholar]
- Means, E.G.; Hanami, L.; Ridgway, H.F.; Olson, B.H. Enumeration of bacteria in potable water distribution systems: Evaluation of media and plating techniques. J. Am. Water Works Assoc. 1981, 53, 585–590. [Google Scholar]
- Armstrong, J.L.; Shigeno, D.S.; Calomiris, J.J.; Seidler, R.J. Antibiotic-resistant bacteria in drinking water. Appl. Environ. Microbiol. 1981, 42, 277–283. [Google Scholar]
- Armstrong, J.L.; Calomiris, J.J.; Seidler, R.J. Selection of antibiotic-resistant standard plate count bacteria during water treatment. Appl. Environ. Microbiol. 1982, 44, 308–316. [Google Scholar]
- Murray, G.E.; Tobin, R.S.; Junkins, B.; Kushner, D.J. Effect of chlorination on antibiotic resistance profiles of sewage-related bacteria. Appl. Environ. Microbiol. 1984, 48, 73–77. [Google Scholar]
- Zhao, T.; Doyle, M.P.; Zhao, P.; Blake, P.; Wu, F.M. Chlorine inactivation of Escherichia coli O157:H7 in water. J. Food Prot. 2001, 64, 1607–1609. [Google Scholar]
- Adams, M.D.; Goglin, K.; Molyneaux, N.; Hujer, K.M.; Lavender, H.; Jamison, J.J.; MacDonald, I.J.; Martin, K.M.; Russo, T.; Campagnari, A.A.; et al. Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii. J. Bacteriol. 2008, 24, 8053–8064. [Google Scholar]
- Kollanoor-Johny, A.; Mattson, T.; Baskaran, S.A.; Amalaradjou, M.A.; Babapoor, S.; March, B.; Valipe, S.; Darre, M.; Hoagland, T.; Schreiber, D.; et al. Reduction of Salmonella enterica serovar enteritidis colonization in 20 day old broiler chickens by the plant-derived compounds trans-cinnamaldehyde and eugenol. Appl. Environ. Microbiol. 2012, 78, 2981–2987. [Google Scholar] [CrossRef]
- Farkas-Himsley, H. Killing of chlorine-resistant bacteria by chlorine-bromine solutions. Appl. Microbiol. 1964, 12, 1–6. [Google Scholar]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar]
- Corvec, S.; Caroff, N.; Espaze, E.; Giraudeau, C.; Drugeon, H.; Reynaud, A. AmpC cephalosporinase hyperproduction in Acinetobacter baumannii clinical strains. J. Antimicrob. Chemother. 2003, 52, 629–635. [Google Scholar]
- Shi, P.; Jia, S.; Zhang, X.X.; Zhang, T.; Cheng, S.; Li, A. Metagenomic Insights into chlorination effects on microbial antibiotic resistance in drinking water. Water Res. 2013, 47, 111–120. [Google Scholar]
- Molecular Investigation of the Chlorine and Antibiotic Resistance Mechanisms of Escherichia coli Isolated from Natural Water Sources in Western Cape. Available online: http://scholar.sun.ac.za/handle/10019.1/2779 (accessed on 1 November 2013).
- Xi, C.; Zhang, Y.; Marrs, C.F.; Ye, W.; Simon, C.; Foxman, B.; Nriagu, J. Prevalence of antibiotic resistance in drinking water treatment and distribution systems. Appl. Environ. Microbiol. 2009, 75, 5714–5718. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karumathil, D.P.; Yin, H.-B.; Kollanoor-Johny, A.; Venkitanarayanan, K. Effect of Chlorine Exposure on the Survival and Antibiotic Gene Expression of Multidrug Resistant Acinetobacter baumannii in Water. Int. J. Environ. Res. Public Health 2014, 11, 1844-1854. https://doi.org/10.3390/ijerph110201844
Karumathil DP, Yin H-B, Kollanoor-Johny A, Venkitanarayanan K. Effect of Chlorine Exposure on the Survival and Antibiotic Gene Expression of Multidrug Resistant Acinetobacter baumannii in Water. International Journal of Environmental Research and Public Health. 2014; 11(2):1844-1854. https://doi.org/10.3390/ijerph110201844
Chicago/Turabian StyleKarumathil, Deepti Prasad, Hsin-Bai Yin, Anup Kollanoor-Johny, and Kumar Venkitanarayanan. 2014. "Effect of Chlorine Exposure on the Survival and Antibiotic Gene Expression of Multidrug Resistant Acinetobacter baumannii in Water" International Journal of Environmental Research and Public Health 11, no. 2: 1844-1854. https://doi.org/10.3390/ijerph110201844
APA StyleKarumathil, D. P., Yin, H.-B., Kollanoor-Johny, A., & Venkitanarayanan, K. (2014). Effect of Chlorine Exposure on the Survival and Antibiotic Gene Expression of Multidrug Resistant Acinetobacter baumannii in Water. International Journal of Environmental Research and Public Health, 11(2), 1844-1854. https://doi.org/10.3390/ijerph110201844





