Effects of Diesel Engine Exhaust Origin Secondary Organic Aerosols on Novel Object Recognition Ability and Maternal Behavior in BALB/C Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Generation of Secondary Organic Aerosol (SOA)
2.3. Experimental Schedule
| Chamber | Particles | Temperature | Relative Humidity | |||
|---|---|---|---|---|---|---|
| Size (nm) | Particles Number (cm−3) | Concentration (µg/m3) | (°C) | (%) | ||
| Clean air | -- | 2.90 ± 0.43 | 15.70 ± 0.63 | 21.99 ± 0.08 | 52.99 ± 0.38 | |
| DE-SOA | 32.78 ± 0.87 | 3.16 × 106 ± 6.18 × 104 | 113.99 ± 3.06 | 22.08 ± 0.10 | 50.77 ± 0.68 | |
| DE | 31.48 ± 0.71 | 3.28× 106 ± 4.52 × 104 | 97.69 ± 3.60 | 22.02 ± 0.07 | 51.35 ± 0.65 | |
| Chamber | Gaseous Compounds | |||||
| CO (ppm) | SO2 (ppm) | NOx (ppm) | NO2 (ppm) | NO (ppm) | CO2 (%) | |
| Clean air | 0.33 ± 0.05 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.05 ± 0.00 |
| DE-SOA | 2.61 ± 0.08 | 0.00 ± 0.00 | 1.39 ± 0.02 | 1.09 ± 0.03 | 0.29 ± 0.03 | 0.07 ± 0.00 |
| DE | 2.52 ± 0.08 | 0.01 ± 0.00 | 1.33 ± 0.03 | 0.43 ± 0.01 | 0.90 ± 0.02 | 0.07 ± 0.00 |
2.4. Novel Object Recognition Test
2.5. Maternal Behavior Test
2.6. Quantification of the mRNA Expression Levels
2.7. Measurement of Plasma 8-Hydroxy-2’Deoxyguanosine (8OHdG) Concentration
2.8. Immunohistochemistry
2.9. Statistical Analysis
3. Results
3.1. Body and Organ Weights of Male Mice after Novel Object Recognition Test
3.2. Effect of Diesel Exhaust (DE) or Diesel Exhaust Origin Secondary Organic Aerosol (DE-SOA) Exposure on Novel Object Recognition Test
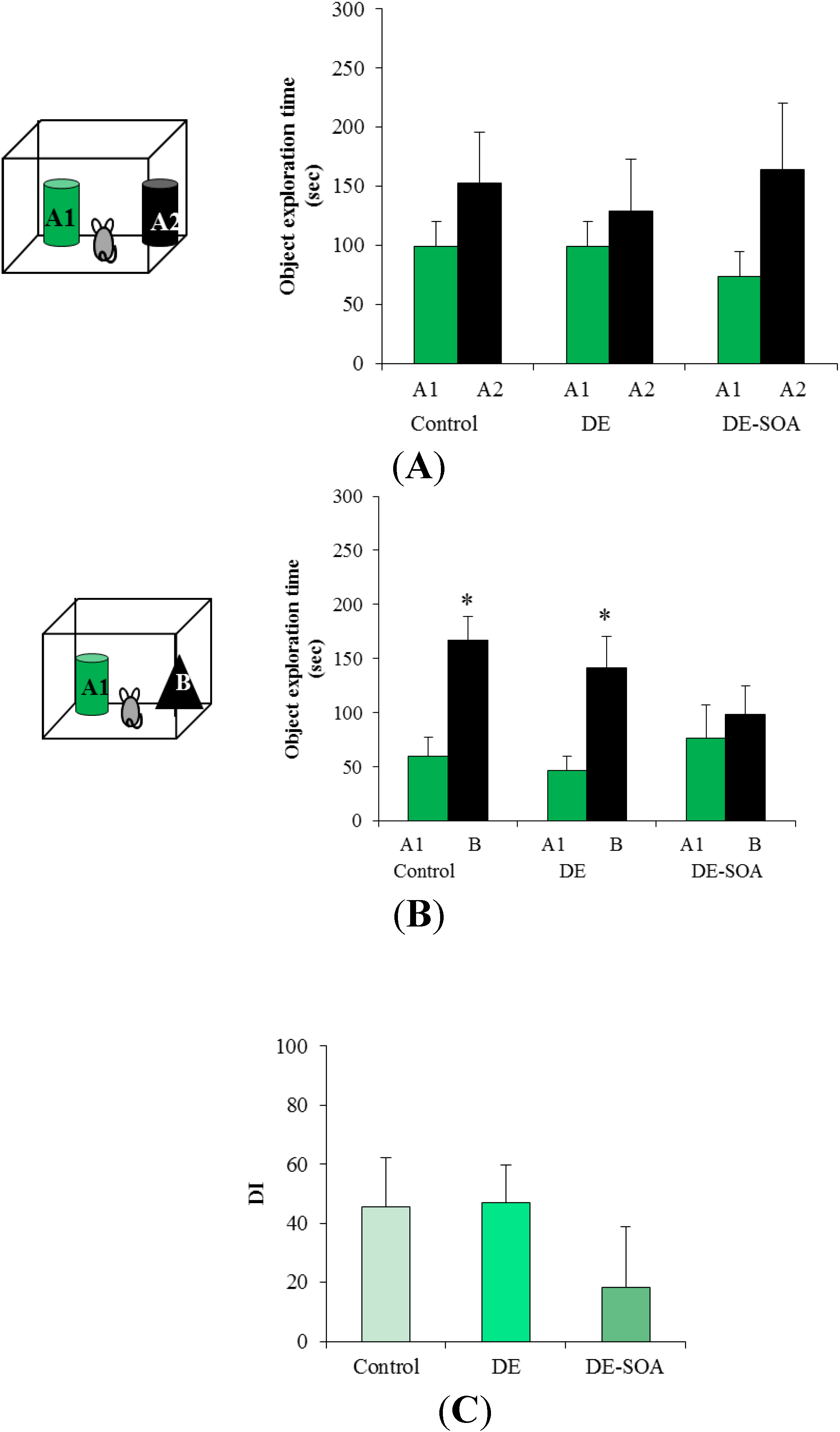
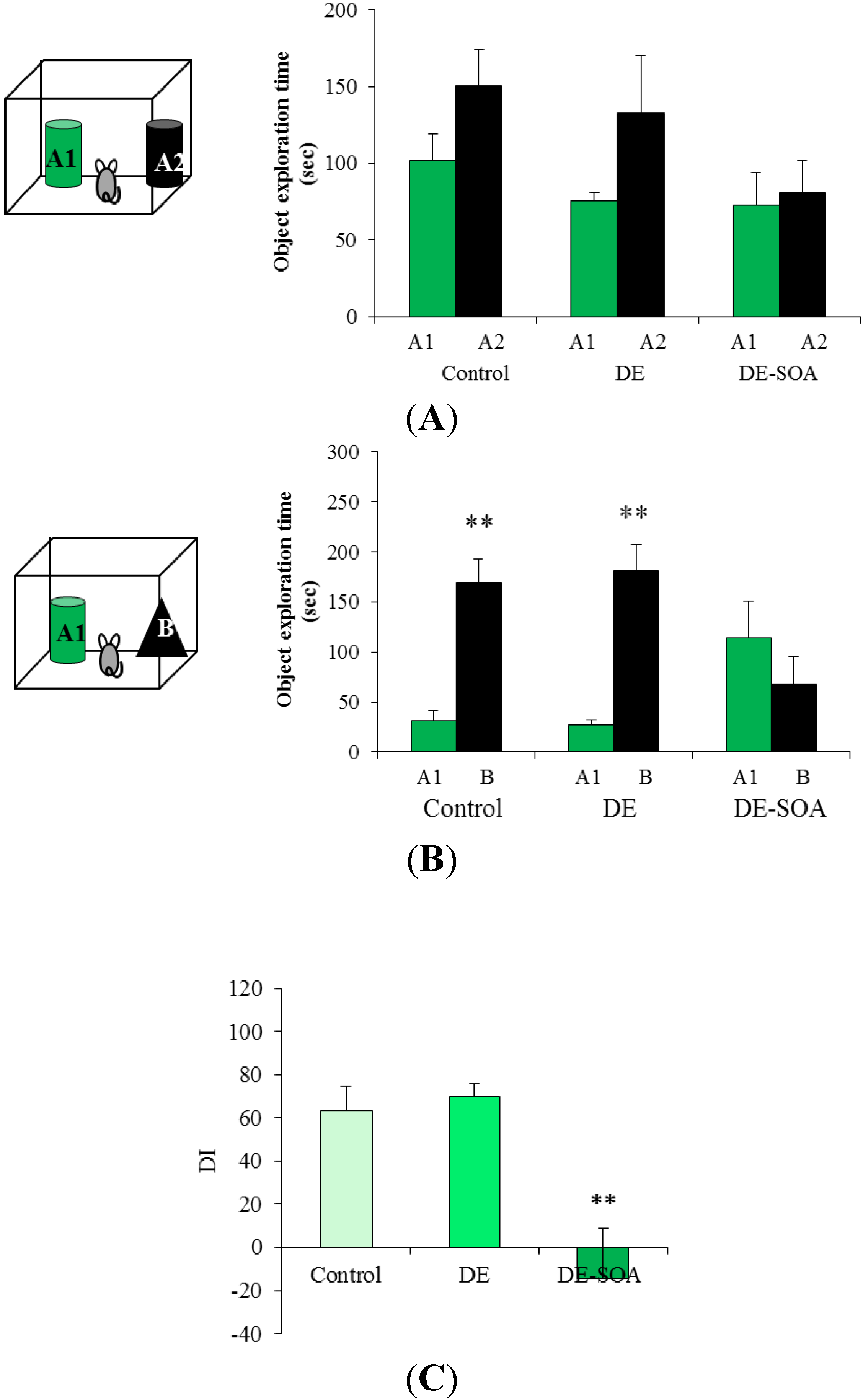
3.3. Effect of Diesel Exhaust (DE) or Diesel Exhaust Origin Secondary Organic Aerosol (DE-SOA) Exposure on the mRNA Expressions of NMDA Receptor Subunits in the Hippocampus
3.4. Effect of Diesel Exhaust (DE) or Diesel Exhaust Origin Secondary Organic Aerosol (DE-SOA) Exposure on Plasma 8OHdG Concentration

3.5. Effect of Diesel Exhaust (DE) or Diesel Exhaust Origin Secondary Organic Aerosol (DE-SOA) Exposure on Maternal Behavior
| Maternal Behavior | Control (%) | DE (%) | DE-SOA (%) |
|---|---|---|---|
| Nesting | 100 | 75 | 50 * |
| Licking | 100 | 87.5 | 87.5 |
| Crouching | 87.5 | 75 | 12.5 ** |
| Retrieving | 12.5 | 37.5 | 12.5 |
| Maternal Behavior | Control (%) | DE (%) | DE-SOA (%) |
|---|---|---|---|
| Nesting | 100 | 62.5 | 87.5 |
| Licking | 100 | 75 | 100 |
| Crouching | 100 | 87.5 | 87.5 |
| Retrieving | 0 | 0 | 0 |
3.6. Body and Organ Weights for Maternal Behavior
| Maternal Data | Control (n = 8) | DE (n = 8) | DE-SOA (n = 8) |
|---|---|---|---|
| % of successful pregnancy | 100% | 100% | 100% |
| % of delayed pregnancy (more than 1 week after mating) | 0% | 10% | 20% |
| Mother eats pup | 0% | 12.5% | 25% |
| Range of number of pup | 5–6 | 5–6 | 4–5 |
| Sex Ratio | male > female | male = female | male = female |
3.7. Effect of Diesel Exhaust (DE) or Diesel Exhaust Origin Secondary Organic Aerosol (DE-SOA) Exposure on Maternal Behavior-Related Gene Expressions in the Hypothalamus
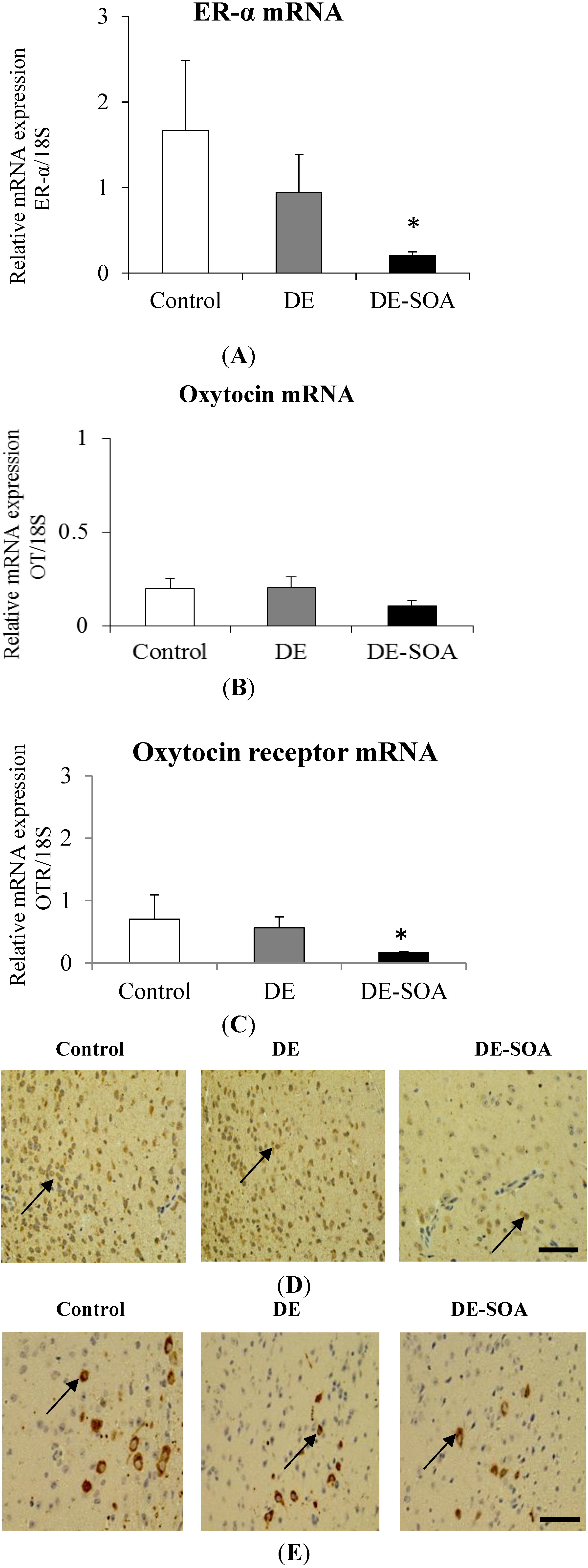
4. Discussion
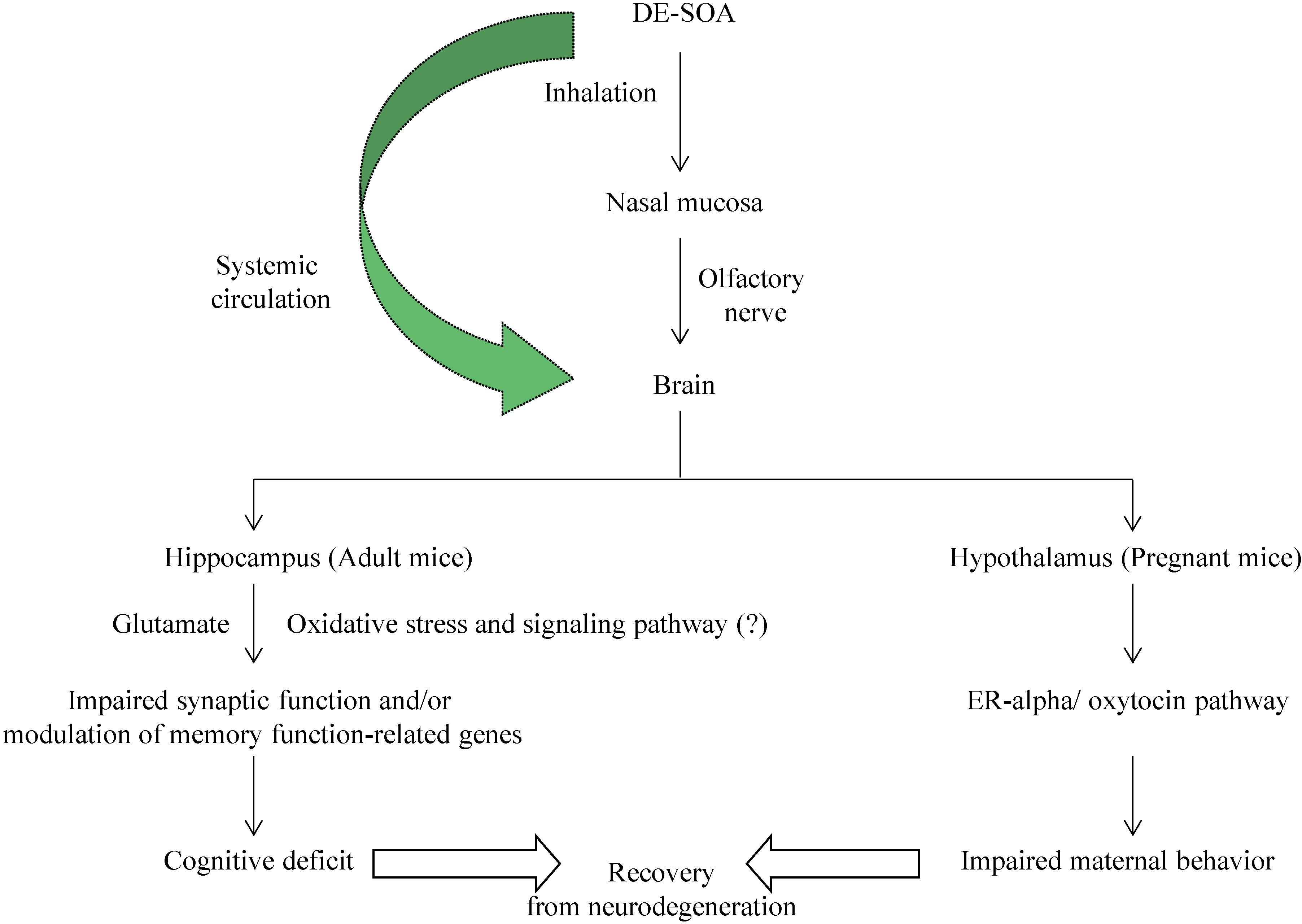
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dockery, D.W. Epidemiologic study design for investigating respiratory health effects of complex air pollution mixtures. Environ. Health Perspect. 1993, 101, 187–191. [Google Scholar]
- Penttinen, P.; Timonen, K.L.; Tittanen, P.; Mirme, A.; Ruuskanen, J.; Pekkanen, J. Ultrafine particles in urban air and respiratory health among adult asthmatics. Euro. Respir. J. 2001, 17, 428–435. [Google Scholar]
- Peters, A.; Döring, A.; Wichmann, H.E; Koenig, W. Increased plasma viscosity during an air pollution episode: A link to mortality? Lancet 1997, 349, 1582–1587. [Google Scholar]
- Pope, C.A., III; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA 2002, 287, 1132–1141. [Google Scholar]
- Kilburn, K.H. Effects of diesel exhaust on neurobehavioral and pulmonary functions. Arch. Environ. Health 2000, 55, 11–17. [Google Scholar]
- Cruts, B.; van Etten, L.; Tornqvist, H.; Blomberg, A.; Sandstrom, T.; Mills, N.L.; Borm, P.J. Exposure to diesel exhaust induces changes in EEG in human volunteers. Part. Fibre Toxicol. 2008, 5. [Google Scholar] [CrossRef]
- Virtanen, A.; Joutsensaari, J.; Koop, T.; Kannosto, J.; Yli-Pirilä, P.; Leskinen, J.; Mäkelä, J.M.; Holopainen, J.K.; Pöschl, U.; Kulmala, M.; Worsnop, D.R.; Laaksonen, A. An amorphous solid state of biogenic secondary organic aerosol particles. Nature 2012, 467, 824–827. [Google Scholar]
- Youssefi, S.; Waring, M.S. Predicting secondary organic aerosol formation from terpenoid ozonolysis with varying yields in indoor environments. Indoor Air 2012, 22, 415–426. [Google Scholar]
- Wang, H.; He, C.; Morawska, L.; McGarry, P.; Johnson, G. Ozone-initiated particle formation, particle aging, and precursors in a laser printer. Environ. Sci. Technol. 2012, 46, 704–712. [Google Scholar]
- Win-Shwe, T.T.; Fujitani, Y.; Sone, H.; Furuyama, A.; Nitta, H.; Hirano, S. Effects of acute single intranasal instillation of secondary organic aerosol on neurological and immunological biomarkers in the brain and lung of BALB/c mice. J. Toxicol. Sci. 2013, 38, 71–82. [Google Scholar]
- Win-Shwe, T.T.; Mitsushima, D.; Yamamoto, S.; Fujitani, Y.; Funabashi, T.; Hirano, S.; Fujimaki, H. Extracellular glutamate level and NMDA receptor subunit expression in mouse olfactory bulb following nanoparticle-rich diesel exhaust exposure. Inhal. Toxicol. 2009, 21, 828–836. [Google Scholar]
- Win-Shwe, T.T.; Yamamoto, S.; Fujitani, Y.; Hirano, S.; Fujimaki, H. Spatial learning and memory function-related gene expression in the hippocampus of mouse exposed to nanoparticle-rich diesel exhaust. Neurotoxicology 2008, 29, 940–947. [Google Scholar]
- Win-Shwe, T.T.; Fujimaki, H. Nanoparticles and neurotoxicity. Int. J. Mol. Sci. 2011, 12, 6267–6280. [Google Scholar]
- Win-Shwe, T.T.; Yamamoto, S.; Fujitani, Y.; Hirano, S.; Fujimaki, H. Nanoparticle-rich diesel exhaust affects hippocampal-dependent spatial learning and NMDA receptor subunit expression in female mice. Nanotoxicology 2012, 6, 543–553. [Google Scholar]
- Win-Shwe, T.T.; Fujimaki, H.; Fujitani, Y.; Hirano, S. Novel object recognition ability in female mice following exposure to nanoparticle-rich diesel exhaust. Toxicol. Appl. Pharmacol. 2012, 262, 355–362. [Google Scholar]
- Bos, I.; De Boever, P.; Emmerechts, J.; Buekers, J.; Vanoirbeek, J.; Meeusen, R.; VanPoppel, M.; Nemery, B.; Nawrot, T.; Panis, L.I. Changed gene expression in brains of mice exposed to traffic in a highway tunnel. Inhal. Toxicol. 2012, 24, 676–686. [Google Scholar]
- Calderon-Garciduenas, L.; Reed, W.; Maronpot, R.R.; Henriquez-Roldan, C.; Delgado-Chavez, R.; Calderon-Garciduenas, A.; Dragustinovis, I.; Franco-Lira, M.; Aragon-Flores, M.; Solt, A.C.; Altenburg, M.; Torres-Jardon, R.; Swenberg, J.A. Brain inflammation and Alzheimer’s like pathology in individuals exposed to severe air pollution. Toxicol. Pathol. 2004, 32, 650–658. [Google Scholar]
- Calderon-Garciduenas, L.; Solt, A.C.; Henriquez-Roldan, C.; Torres-Jardon, R.; Nuse, B.; Herritt, L.; Villarreal-Calderon, R.; Osnaya, N.; Stone, I.; Garcia, R.; Brooks, D.M.; Gonzalez-Maciel, A.; Reynoso-Robles, R.; Delgado-Chavez, R.; Reed, W. Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood-brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-synuclein in children and young adults. Toxicol. Pathol. 2008, 36, 289–310. [Google Scholar]
- Calderon-Garciduenas, L.; Mora-Tiscareno, A.; Ontiveros, E.; Gomez-Garza, G.; Barragan-Mejia, G.; Broadway, J.; Chapman, S.; Valencia-Salazar, G.; Jewells, V.; Maronpot, R.R.; Henriquez-Roldan, C.; Perez-Guille, B.; Torres-Jardon, R.; Herrit, L.; Brooks, D.; Osnaya-Brizuela, N.; Monroy, M.E.; Gonzalez-Maciel, A.; Reynoso-Robles, R.; Villarreal-Calderon, R.; Solt, A.C.; Engle, R.W. Air pollution, cognitive deficits and brain abnormalities: a pilot study with children and dogs. Brain Cogn. 2008, 68, 117–127. [Google Scholar]
- Calderon-Garciduenas, L.; Engle, R.; Mora-Tiscareno, A.; Styner, M.; Gomez-Garza, G.; Zhu, H.; Jewells, V.; Torres-Jardon, R.; Romero, L.; Monroy-Acosta, M.E.; Bryant, C.; Gonzalez-Gonzalez, L.O.; Medina-Cortina, H.; D’Angiulli, A. Exposure to severe urban air pollution influences cognitive outcomes, brain volume and systemic inflammation in clinically healthy children. Brain Cogn. 2011, 77, 345–355. [Google Scholar]
- Chen, J.C.; Schwartz, J. Neurobehavioral effects of ambient air pollution on cognitive performance in US adults. Neurotoxicology 2009, 30, 231–239. [Google Scholar]
- Ranft, U.; Schikowski, T.; Sugiri, D.; Krutmann, J.; Kramer, U. Long-term exposure to traffic-related particulate matter impairs cognitive function in the elderly. Environ. Res. 2009, 109, 1004–1011. [Google Scholar]
- Weuve, J.; Puett, R.C.; Schwartz, J.; Yanosky, J.D.; Laden, F.; Grodstein, F. Exposure to particulate air pollution and cognitive decline in older women. Arch. Intern. Med. 2012, 172, 219–227. [Google Scholar]
- Harrison, R.M.; Yin, J. Particulate matter in the atmosphere: which particle properties are important for its effects on health? Sci. Total Environ. 2000, 249, 85–101. [Google Scholar]
- Pope, C.A., 3rd; Dockery, D.W. Health effects of fine particulate air pollution: lines that connect. J. Air Waste Manag. Assoc. 2006, 56, 709–742. [Google Scholar]
- Fujitani, Y.; Hirano, S.; Kobayashi, S.; Tanabe, K.; Suzuki, A.; Furuyama, A.; Kobayashi, T. Characterization of dilution conditions for diesel nanoparticle inhalation studies. Inhal. Toxicol. 2009, 21, 200–209. [Google Scholar]
- Ennaceur, A.; Delacour, J. A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav. Brain Res. 1988, 31, 47–59. [Google Scholar]
- Loft, S.; Fischer-Nielsen, A.; Jeding, I.B.; Vistisen, K.; Poulsen, H.E. 8-Hydroxydeoxyguanosine as a urinary biomarker of oxidative DNA damage. J. Toxicol. Environ. Health 1993, 140, 391–404. [Google Scholar]
- Stern, J.M. Offspring-induced nurturance: animal-human parallels. Dev. Psychobiol. 1997, 31, 19–37. [Google Scholar]
- Fahrbach, S.E.; Morrell, J.I.; Pfaff, D.W. Possible role for endogenous oxytocin in estrogen-facilitated maternal behavior in rats. Neuroendocrinology 1985, 40, 526–532. [Google Scholar]
- Ogawa, S.; Eng, V.; Taylor, J.; Lubahn, D.B.; Korach, K.S.; Pfaff, D.W. Roles of estrogen receptor-alpha gene expression in reproduction-related behaviors in female mice. Endocrinology 1998, 139, 5070–5081. [Google Scholar]
- Ribeiro, A.C.; Musatov, S.; Shteyler, A.; Simanduyev, S.; Arrieta-Cruz, I.; Ogawa, S.; Pfaff, D.W. siRNA silencing of estrogen receptor-α expression specifically in medial preoptic area neurons abolishes maternal care in female mice. Proc. Natl. Acad. Sci. USA 2012, 109, 16324–16329. [Google Scholar]
- Yu, S.; Bhave, P.V.; Dennis, R.L.; Mathur, R. Seasonal and regional variations of primary and secondary organic aerosols over the continental United States: semi-empirical estimates and model evaluation. Environ. Sci. Technol. 2007, 41, 4690–4697. [Google Scholar]
- Castro, L.M.; Pio, C.A.; Harrison, R.M.; Smith, D.J.T. Carbonaceous aerosol in urban and rural European atmospheres: estimation of secondary organic carbon concentrations. Atmos. Environ. 1999, 33, 2771–2781. [Google Scholar]
- Lim, H.J.; Turpin, B.J. Origins of primary and secondary organic aerosol in Atlanta: results of time-resolved measurements during the Atlanta Supersite Experiment. Environ. Sci. Technol. 2002, 36, 4489–4496. [Google Scholar]
- von Stackelberg, K.; Buonocore, J.; Bhave, P.V.; Schwartz, J.A. Public health impacts of secondary particulate formation from aromatic hydrocarbons in gasoline. Environ. Health 2013, 12. [Google Scholar] [CrossRef]
- McDonald, J.D.; Doyle-Eisele, M.; Kracko, D.; Lund, A.; Surratt, J.D.; Hersey, S.P.; Seinfeld, J.H.; Rohr, A.C.; Knipping, E.M. Cardiopulmonary response to inhalation of secondary organic aerosol derived from gas-phase oxidation of toluene. Inhal. Toxicol. 2012, 24, 689–697. [Google Scholar]
- Diaz, E.A.; Lemos, M.; Coull, B.; Long, M.S.; Rohr, A.C.; Ruiz, P.; Gupta, T.; Kang, C.M.; Godleski, J.J. Toxicological evaluation of realistic emission source aerosols (TERESA)—Power plant studies: assessment of breathing pattern. Inhal. Toxicol. 2011, 23 (Suppl 2), 42–59. [Google Scholar]
- Delfino, R.J.; Staimer, N.; Tjoa, T.; Polidori, A.; Arhami, M.; Gillen, D.L.; Kleinman, M.T.; Vaziri, N.D.; Longhurst, J.; Zaldivar, F.; Sioutas, C. Circulating biomarkers of inflammation, antioxidant activity, and platelet activation are associated with primary combustion aerosols in subjects with coronary artery disease. Environ. Health Perspect. 2008, 116, 898–906. [Google Scholar]
- Monyer, H.; Burnashev, N.; Laurie, D.J.; Sakmann, B.; Seeburg, P.H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 1994, 12, 529–540. [Google Scholar]
- Carroll, R.C.; Zukin, R.S. NMDA-receptor trafficking and targeting: implications for synaptic transmission and plasticity. Trends Neurosci. 2002, 25, 571–577. [Google Scholar]
- Johnson, J.W.; Ascher, P. Glycine potentiates the NMDA response in cultured mouse brain neurons. Nature 1987, 325, 529–531. [Google Scholar]
- Clements, J.D.; Westbrook, G.L. Activation kinetics reveal the number of glutamate and glycine binding sites on the N-methyl-D-aspartate receptor. Neuron 1991, 7, 605–613. [Google Scholar]
- Lemos, M.; Diaz, E.A.; Gupta, T.; Kang, C.M.; Ruiz, P.; Coull, B.A.; Godleski, J.J.; Gonzalez-Flecha, B. Cardiac and pulmonary oxidative stress in rats exposed to realistic emissions of source aerosols. Inhal. Toxicol. 2011, 23 (Suppl 2), 75–83. [Google Scholar]
- Kalinichev, M.; Rosenblatt, J.S.; Morrell, J.I. The medial preoptic area, necessary for adult maternal behavior in rats, is only partially established as a component of the neural circuit that supports maternal behavior in juvenile rats. Behav. Neurosci. 2000, 114, 196–210. [Google Scholar]
- Numan, M. The role of the medial preoptic area in the regulation of maternal behavior in the rat. Ann. NY Acad. Sci. 1986, 474, 226–233. [Google Scholar]
- Numan, M. Hypothalamic neural circuits regulating maternal responsiveness toward infants. Behav. Cogn. Neurosci. Rev. 2006, 5, 163–190. [Google Scholar]
- Peña, C.J.; Neugut, Y.D.; Champagne, F.A. Developmental timing of the effects of maternal care on gene expression and epigenetic regulation of hormone receptor levels in female rats. Endocrinology 2013, 154, 4340–4351. [Google Scholar]
- Pilger, A.; Rüdiger, H.W. 8-Hydroxy-2'-deoxyguanosine as a marker of oxidative DNA damage related to occupational and environmental exposures. Int. Arch. Occup. Environ. Health 2006, 80, 1–15. [Google Scholar]
- Young, L.J.; Wang, Z.; Donaldson, R.; Rissman, E.F. Estrogen receptor alpha is essential for induction of oxytocin receptor by estrogen. Neuroreport 1998, 9, 933–936. [Google Scholar]
- Noirot, E. Changes in responsiveness to young in the adult mouse. V. Priming. Anim. Behav. 1969, 17, 542–546. [Google Scholar]
- Noirot, E. Serial order of maternal responses in mice. Anim. Behav. 1969, 17, 547–550. [Google Scholar]
- Wang, Z.; Storm, D.R. Maternal behavior is impaired in female mice lacking type 3 adenylyl cyclase. Neuropsychopharmacology 2011, 36, 772–781. [Google Scholar]
- Tsukue, N.; Watanabe, M.; Kumamoto, T.; Takano, H.; Takeda, K. Perinatal exposure to diesel exhaust affects gene expression in mouse cerebrum. Arch. Toxicol. 2009, 83, 985–1000. [Google Scholar]
- Suzuki, T.; Oshio, S.; Iwata, M.; Saburi, H.; Odagiri, T.; Udagawa, T.; Sugawara, I.; Umezawa, M.; Takeda, K. In utero exposure to a low concentration of diesel exhaust affects spontaneous locomotor activity and monoaminergic system in male mice. Part. Fibre Toxicol. 2010, 7, 1–8. [Google Scholar]
- Clifford, A.; Lang, L.; Chen, R. Effects of maternal cigarette smoking during pregnancy on cognitive parameters of children and young adults: a literature review. Neurotoxicol. Teratol. 2012, 34, 560–570. [Google Scholar]
- Costa, L.G.; Cole, T.B.; Coburn, J.; Chang, Y.C.; Dao, K.; Roque, P. Neurotoxicants are in the air: Convergence of human, animal and in vitro studies on the effects of air pollution on the brain. BioMed. Res. Int. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Win-Shwe, T.-T.; Fujitani, Y.; Kyi-Tha-Thu, C.; Furuyama, A.; Michikawa, T.; Tsukahara, S.; Nitta, H.; Hirano, S. Effects of Diesel Engine Exhaust Origin Secondary Organic Aerosols on Novel Object Recognition Ability and Maternal Behavior in BALB/C Mice. Int. J. Environ. Res. Public Health 2014, 11, 11286-11307. https://doi.org/10.3390/ijerph111111286
Win-Shwe T-T, Fujitani Y, Kyi-Tha-Thu C, Furuyama A, Michikawa T, Tsukahara S, Nitta H, Hirano S. Effects of Diesel Engine Exhaust Origin Secondary Organic Aerosols on Novel Object Recognition Ability and Maternal Behavior in BALB/C Mice. International Journal of Environmental Research and Public Health. 2014; 11(11):11286-11307. https://doi.org/10.3390/ijerph111111286
Chicago/Turabian StyleWin-Shwe, Tin-Tin, Yuji Fujitani, Chaw Kyi-Tha-Thu, Akiko Furuyama, Takehiro Michikawa, Shinji Tsukahara, Hiroshi Nitta, and Seishiro Hirano. 2014. "Effects of Diesel Engine Exhaust Origin Secondary Organic Aerosols on Novel Object Recognition Ability and Maternal Behavior in BALB/C Mice" International Journal of Environmental Research and Public Health 11, no. 11: 11286-11307. https://doi.org/10.3390/ijerph111111286






