Appraisal of Hygiene Indicators and Farming Practices in the Production of Leafy Vegetables by Organic Small-Scale Farmers in uMbumbulu (Rural KwaZulu-Natal, South Africa)
Abstract
:1. Introduction
2. Experimental Section
2.1. Site Location, Farmer Survey and Data Collection
2.2. Sample Collection
2.3. Microbiological Analysis
2.4. Survey Statistical Analysis
3. Results and Discussion
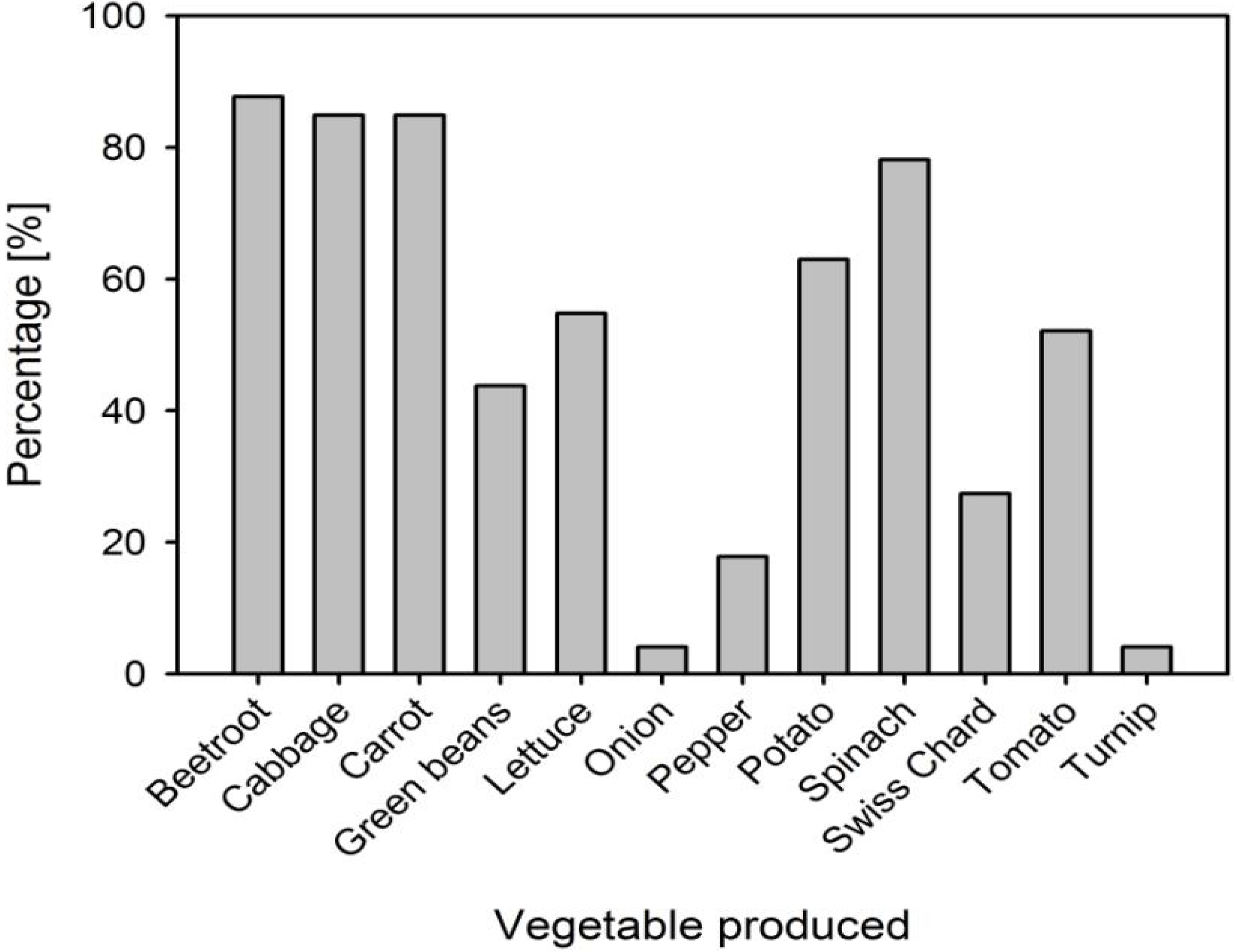
3.1. Farmer Responses on Vegetables Produced
3.2. Irrigation Water Sources Identified in Farmer Survey
| Irrigation source | Frequency ∑ = 73 | Percentage % |
|---|---|---|
| Tap water | 31 | 42.5 |
| IsiJodi River | 14 | 19.2 |
| Nongwane Dam water | 12 | 16.4 |
| Spring water | 7 | 9.6 |
| Borehole | 4 | 5.5 |
| Wetland | 3 | 4.1 |
| Tank water | 2 | 2.7 |
3.3. The Importance of Farmer Training
| Agri-Hub members (% n = 33) | Non-Agri-Hub members (% n = 40) | p-value | ||
|---|---|---|---|---|
| Hygienic practices prior to entering the garden | ||||
| Individuals washing hands and boots | 91 | 53 | <0.001 *** | |
| Individuals washing of farming equipment | 91 | 55 | <0.001 *** | |
| Individuals who acknowledge the following sources of contamination | ||||
| Contaminated water | 82 | 65 | 0.026 ** | |
| Incorrect composting techniques | 94 | 53 | <0.001 *** | |
| Poor personal hygiene | 91 | 23 | <0.001 *** | |
| Contaminated soils | 82 | 50 | 0.001 *** | |
| Contaminated equipment | 85 | 50 | 0.002 *** | |
| Type of treatment manure subjected to | ||||
| Drying of manure (umquba) | 55 | 48 | 0.305 | |
| Composting | 76 | 33 | <0.001 *** | |
| Direct use of wet/fresh manure (no treatment) | 9 | 38 | 0.009 *** | |
3.4. Physico-Chemical Characteristics of Irrigation Water Sources Tested
| Month | Irrigation Water Source | ||||
|---|---|---|---|---|---|
| Nungwane Dam water | Spring water source | IsiJodi River water | Tap water | ||
| October 2011 | Water temp. | 23.1 °C | 23.9 °C | 18.4 °C | 19.0 °C |
| Water pH | 7.72 | 6.50 | 7.64 | 7.34 | |
| Water COD | 14 mg/L | 39 mg/L | 25 mg/L | 17 mg/L | |
| November 2011 | Water temp. | 23.8 °C | 22.0 °C | 19.3 °C | 18.0 °C |
| Water pH | 7.15 | 6.51 | 7.13 | 7.79 | |
| Water COD | 17 mg/L | 36 mg/L | 36 mg/L | 14 mg/L | |
| December 2011 | Water temp. | 23.5 °C | 23.0 °C | 19.8 °C | 18.8 °C |
| Water pH | 7.52 | 6.54 | 7.45 | 7.66 | |
| Water COD | 18 mg/L | 36 mg/L | 17 mg/L | 17 mg/L | |
3.5. Hygienic Quality of Irrigation Water
| Source of irrigation water | October 2011 | November 2011 | December 2011 | |||
|---|---|---|---|---|---|---|
| MPN/100 mL | 95% confidence interval lower/upper limit * | MPN/100 mL | 95% confidence interval lower/upper limit | MPN/100 mL | 95% confidence interval lower/upper limit | |
| Total coliforms | ||||||
| Nungwane Dam | 7.90 | 2.4/25 | 7.90 | 2.4/25 | 7.90 | 2.4/25 |
| Spring water | 110.00 | 39/300 | 79.00 | 25/247 | 110.00 | 39/300 |
| IsiJodi River | 110.00 | 39/300 | 25.00 | 11/62 | 33.00 | 11/99 |
| Tap water source | n.d | - | n.d | - | n.d | - |
| Fecal coliforms | ||||||
| Nungwane Dam | 2.00 | 0.28/14 | 4.50 | 1.1/18 | 4.50 | 1.1/18 |
| Spring water | 14.00 | 5.5/34 | 4.50 | 1.1/18 | 4.50 | 1.1/18 |
| IsiJodi River | 27.00 | 11/64 | 4.50 | 1.1/18 | 7.90 | 2.4/25 |
| Tap water source | n.d | - | n.d | - | n.d | - |
| E. coli | ||||||
| Nungwane Dam | n.d | - | n.d | - | n.d | - |
| Spring water | n.d | - | n.d | - | n.d | - |
| IsiJodi River | n.d | - | n.d | - | n.d | |
| Tap water source | n.d | - | n.d | - | n.d | - |
3.6. Hygienic Quality of Compost
| Source of Compost | October 2011 | November 2011 | December 2011 | |||
|---|---|---|---|---|---|---|
| MPN/g | 95% confidence interval lower/upper limit * | MPN/g | 95% confidence interval lower/upper limit | MPN/g | 95% confidence interval lower/upper limit | |
| Total coliforms | ||||||
| Nungwane | 1,405.60 | 561.44/3,527.98 | 313.90 | 107.39/919.63 | 221.20 | 89.59/547.02 |
| Senzakahle | 1,405.60 | 561.44/3,527.98 | 278.10 | 117.46/659.73 | 27.80 | 11.74/65.95 |
| Siyazenzela | 22.10 | 8.96/54.68 | 22.10 | 8.96/54.68 | 22.10 | 8.96/54.68 |
| Jabulani | 943.50 | 349.79/2,551.18 | 140.60 | 56.12/352.65 | 22.10 | 8.96/54.68 |
| Fecal coliforms | ||||||
| Nungwane | 313.90 | 107.39/919.63 | 2.60 | 1.13/6.20 | 1.40 | 0.55/3.47 |
| Senzakahle | 221.20 | 89.59/547.02 | 140 | 0.55/3.41 | 1.40 | 0.55/3.41 |
| Siyazenzela | 2.20 | 0.88/5.32 | 1.70 | 0.65/4.41 | 2.20 | 0.88/5.32 |
| Jabulani | 27.80 | 11.74/65.05 | 1.10 | 0.39/2.95 | 1.00 | 0.39/2.95 |
| E. coli | ||||||
| Nungwane | 27.80 | 11.74/65.95 | 2.60 | 1.13/6.20 | 2.60 | 1.13/6.20 |
| Senzakahle | 22.10 | 8.96/54.68 | 2.60 | 1.13/6.20 | 1.40 | 0.55/3.47 |
| Siyazenzela | 1.40 | 0.55/3.47 | n.d | - | n.d | - |
| Jabulani | 2.60 | 1.13/6.20 | 1.10 | 0.39/2.95 | n.d | - |
3.7. The Administration of Compost
3.8. The Use of Wet and Dried Manure (Umquba)
3.9. Hygienic Quality of Leafy Vegetables
| Source of Spinach | October 2011 | November 2011 | December 2011 | |||
|---|---|---|---|---|---|---|
| MPN/g | 95% confidence interval lower/upper limit * | MPN/g | 95% confidence interval lower/upper limit | MPN/g | 95% confidence interval lower/upper limit | |
| Total coliforms | ||||||
| Nungwane | 0.50 | 0.11/1.81 | 1.10 | 0.39/2.95 | 1.40 | 0.55/3.41 |
| Senzakahle | 1.40 | 0.55/3.47 | 1.40 | 0.55/3.47 | 1.10 | 0.39/2.48 |
| Siyazenzela | 0.70 | 0.22/2.15 | 0.70 | 0.22/2.15 | 0.80 | 0.24/2.95 |
| Jabulani | 0.20 | 0.028/1.41 | 1.40 | 0.55/3.47 | 1.40 | 0.55/3.47 |
| Fecal coliforms | ||||||
| Nungwane | 0.20 | 0.028/1.41 | 0.70 | 0.22/2.15 | 0.50 | 0.11/1.81 |
| Senzakahle | 0.70 | 0.22/2.15 | 0.70 | 0.22/2.15 | 0.50 | 0.11/1.81 |
| Siyazenzela | 0.20 | 0.028/1.41 | 0.20 | 0.028/1.41 | n.d | - |
| Jabulani | n.d | - | 0.40 | 0.11/1.81 | n.d | - |
| E. coli | ||||||
| Nungwane | n.d | - | n.d | - | n.d | - |
| Senzakahle | n.d | - | n.d | - | n.d | - |
| Siyazenzela | n.d | - | n.d | - | n.d | - |
| Jabulani | n.d | - | n.d | - | n.d | - |
| Source of Lettuce | October 2011 | November 2011 | December 2011 | |||
|---|---|---|---|---|---|---|
| MPN/g | 95% confidence interval lower/upper limit * | MPN/g | 95% confidence interval lower/upper limit | MPN/g | 95% confidence interval lower/upper limit | |
| Total coliforms | ||||||
| Nungwane | 0.50 | 0.11/1.81 | 0.20 | 0.028/1.41 | 0.50 | 0.11/1.81 |
| Senzakahle | 1.40 | 0.55/3.47 | 0.70 | 0.22/2.15 | n.d | - |
| Siyazenzela | 1.40 | 0.55/3.47 | 0.90 | 0.34/2.50 | 0.70 | 0.22/2.15 |
| Jabulani | 0.70 | 0.22/2.15 | 0.70 | 0.22/2.15 | 0.70 | 0.22/2.15 |
| Fecal coliforms | ||||||
| Nungwane | n.d | - | n.d | - | 0.20 | 0.281.41 |
| Senzakahle | 0.40 | 0.11/1.62 | 0.2 | 0.028/1.41 | n.d | - |
| Siyazenzela | 0.70 | 0.22/2.15 | 0.68 | 0.22/2.15 | 0.20 | 0.028/1.41 |
| Jabulani | n.d | - | 0.20 | 0.028/1.41 | 0.40 | 0.10/1.62 |
| E. coli | ||||||
| Nungwane | n.d | - | n.d | - | n.d | - |
| Senzakahle | n.d | - | n.d | - | n.d | - |
| Siyazenzela | n.d | - | n.d | - | n.d | - |
| Jabulani | n.d | - | n.d | - | n.d | - |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Louw, A.; Vermeulen, H.; Kirsten, J.; Madevu, H. Securing small farmer participation in supermarket supply chains in South Africa. Dev. South. Afr. 2007, 24, 539–551. [Google Scholar] [CrossRef]
- Barrow, S. South African Organic Market Study; EPOPA: Bennekom, The Netherlands, 2006; pp. 5–28. [Google Scholar]
- Berger, C.N.; Sodha, S.V.; Shaw, R.K.; Griffin, P.M.; Pink, D.; Hand, P.; Frankel, G. Fresh fruit and vegetables as vehicles for the transmission of human pathogens. Environ. Microbiol. 2010, 2, 2385–2397. [Google Scholar]
- UNEP-UNCTAD, Organic Agriculture and Food Security in Africa; United Nations: New York, NY, USA, 2008.
- Wegner, L.; Zwart, G. Who Will Feed the World? The Production Challenge; Oxfam Research: Oxford, UK, 2011. [Google Scholar]
- Wettstein, N.; Hanf, J.H.; Burggraf, C. Unshakable loyalty in the food sector: Sustainable customer retention. Empirical study of organic food consumers in Germany. J Verbrauch. Lebensm. 2011, 6, 359–365. [Google Scholar] [CrossRef]
- Beuchat, L.R. Vectors and conditions for pre-harvest contamination of fruits and vegetables with pathogens causing enteric diseases. Br. Food J. 2006, 108, 38–53. [Google Scholar] [CrossRef]
- Buck, J.W.; Walcott, R.; Beuchat, L.R. Recent trends in microbiological safety of fruits and vegetables. Plant Health Prog. 2003, 121, 1–10. [Google Scholar]
- Schwaiger, K.; Helmke, K.; Hölzel, C.S.; Bauer, J. Comparative analysis of the bacterial flora of vegetables collected directly from farms and from supermarkets in Germany. Int. J. Environ. Health Res. 2011, 21, 161–172. [Google Scholar] [CrossRef]
- Oliveira, M.; Usall, J.; Vinas, L.; Anguera, M.; Gatius, F.; Abadias, S. Microbiological quality of fresh lettuce from organic and conventional production. Food Microbiol. 2010, 27, 679–684. [Google Scholar] [CrossRef]
- Brinton, W.F. Compost Quality Standards and Guidelines; New York State Association of Recyclers: New York, NY, USA, 2000. [Google Scholar]
- Shepherd, M.W.; Kim, J.; Jiang, X.; Doyle, M.P.; Erickson, M.C. Evaluation of physical coverings used to control Escherichia coli O157:H7 at the compost heap surface. Appl. Environ. Microbiol. 2011, 77, 5044–5049. [Google Scholar] [CrossRef]
- Stopforth, J.D.; Kottapalli, B.; Sofos, J.N. Interventions for Hazard Control in Foods Preharvest. In Pathogens and Toxins in Foods: Challenges and Interventions; Juneja, V.K., Sofos, J.N., Eds.; ASM Press: Washington, DC, USA, 2010; pp. 357–395. [Google Scholar]
- Chigor, V.N.; Sibanda, T.; Okoh, A.I. Studies on the bacteriological qualities of the Buffalo River and three source water dams along its course in the Eastern Cape Province of South Africa. Environ. Sci. Pollut. Res. 2013, 20, 4125–4136. [Google Scholar] [CrossRef]
- Gemmell, M.E.; Schmidt, S. Microbiological assessment of river water used for the irrigation of fresh produce in a sub-urban community in Sobantu, South Africa. Food Res. Int. 2012, 47, 300–305. [Google Scholar] [CrossRef]
- Gemmell, M.E.; Schmidt, S. Is the microbiological quality of the Msunduzi River (KwaZulu-Natal, South Africa) suitable for domestic, recreational, and agricultural purposes? Envir. Sci. Pollut. Res. 2013, 20, 6551–6562. [Google Scholar] [CrossRef]
- Gemmell, M.E.; Schmidt, S. Potential Links between Irrigation Water Quality and Microbiological Quality of Food in Subsistence Farming in KwaZulu-Natal, South Africa. In Current Research, Technology and Education Topics in Applied Microbiology and Microbial Technology; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2010; Volume 2, pp. 1190–1195. [Google Scholar]
- Deering, A.J.; Mauer, L.J.; Pruitt, R.E. Internalization of E. coli O157:H7 and Salmonella spp. in plants: A review. Food Res. Int. 2012, 45, 567–575. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on the risk posed by pathogens in food of non-animal origin. Part 1 (outbreak data analysis and risk ranking of food/pathogen combinations). EFSA J. 2013, 11. [CrossRef]
- Erickson, M.C. Microbial risks associated with cabbage, carrots, celery, onions, and Deli salads made with these produce items. Compr. Rev. Food Sci. Food Saf. 2010, 9, 602–619. [Google Scholar]
- Frank, C.; Faber, M.S.; Askar, M.; Bernard, H.; Fruth, A.; Gilsdorf, A.; Karch, M.H.; Krause, G.; Prager, R.; Pode, A.; et al. Large and ongoing outbreak of haemolytic uraemic syndrome, Germany, May 2011. Eurosurveillance 2011, 16, 1–3. [Google Scholar]
- Niehaus, A.J.; Apalata, T.; Coovadia, Y.M.; Smith, A.M.; Moodley, P. An outbreak of foodborne Salmonellosis in rural KwaZulu-Natal, South Africa. Foodborne Pathog. Dis. 2011, 8, 693–697. [Google Scholar] [CrossRef]
- Beutin, L.; Martin, A. Outbreak of Shiga toxin-producing Escherichia coli (STEC) O104:H4 infection in Germany causes a paradigm shift with regard to human pathogenicity of STEC strains. J. Food Prot. 2012, 75, 408–418. [Google Scholar] [CrossRef]
- EFSA, Tracing Seeds, in Particular Fenugreek (Trigonella foenum-graecum) Seeds, in Relation to the Shiga Toxin-Producing E. coli (STEC) O 104:H4 2011 Outbreaks in Germany and France; European Food Safety Authority: Parma, Italy, 2011.
- WHO, Excreta and Greywater Use in Agriculture. In WHO Guidelines for the Safe Use of Wastewater Excreta and Greywater; World Health Organization: Geneva, Switzerland, 2006; Volume 4.
- WHO, Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011.
- Little, C.L.; Gillespie, I.A. Prepared salads and public health. J. Appl. Microbiol. 2008, 105, 1729–1743. [Google Scholar] [CrossRef]
- Digbo, S.O.A.; Momoh, S. Sustainable agriculture in Nigeria: Contributions of soil fertility maintenance practices. J. Sustain. Agric. Environ. 2007, 9, 95–103. [Google Scholar]
- Health Canada, MFHPB-19: Enumeration of Coliforms, Faecal Coliforms and of E. coli in Foods Using the MPN Method; Health Canada: Ottawa, Canada, 2002.
- Garthright, W.E.; Blodgett, R.J. FDA’s preferred MPN methods for standard, large or unusual tests, with a spreadsheet. Food Microbiol. 2003, 20, 439–445. [Google Scholar] [CrossRef]
- Modi, A.T. What do subsistence farmers know about indigenous crops and organic farming? Preliminary experience in KwaZulu-Natal. Dev. South. Afr. 2003, 20, 675–684. [Google Scholar] [CrossRef]
- Department of Water Affairs and Forestry, Agricultural Water Use: Irrigation. In South African Water Quality Guidelines, 2nd ed.; CSIR, Environmental Services: Pretoria, South Africa, 1996; Volume 4.
- US EPA, Standards for the Use and Disposal of Sewage Sludge (40 CFR 503.32); United States Government Printing office: Washington, DC, USA, 1993.
- European Commission, Establishing Ecological Criteria for the Award of the Community Eco-Label to Soil Improvers and Growing Media (Commission Decision of 28 August 2001 (2001/688/EC)). Official Journal of the European Union, 12 September 2001; L 242/17.
- Baldwin, K.R.; Greenfield, J.T. Composting on Organic Farms; North Carolina Cooperative Extension Services: Raleigh, NC, USA, 2009. [Google Scholar]
- Ryckeboer, J.; Mergaert, J.; Vaes, K.; Klammer, S.; De Clercq, D.; Coosemans, J.; Insam, H.; Swings, J. A survey of bacteria and fungi occurring during composting and self-heating processes. Ann. Microbiol. 2003, 53, 349–410. [Google Scholar]
- Omisakin, F.; MacRae, M.; Ogden, I.D.; Strachan, N.J.C. Concentration and prevalence of Escherichia coli O157 in cattle feces at slaughter. Appl. Environ. Microbiol. 2003, 69, 2444–2447. [Google Scholar] [CrossRef]
- Hodges, J.; Kimball, A. The global diet: Trade and novel infections. Global. Health 2005, 1. [Google Scholar] [CrossRef] [Green Version]
- Department of Health, Guidelines for Environmental Health Officers on the Interpretation of Microbiological Analysis Data of Food; Department of Health, Directorate Food Control: Pretoria, South Africa, 2001.
- Department of Water Affairs and Forestry, Domestic Water Use. In South African Water Quality Guidelines, 2nd ed.; CSIR, Environmental Services: Pretoria, South Africa, 1996; Volume 1.
- Abadius, M.; Usall, J.; Oliveria, M.; Alegre, I.; Vinas, I. Efficacy of neutral electrolyzed water (NEW) for reducing microbial contamination on minimally-processed vegetables. Int. J. Food Microbiol. 2008, 123, 151–158. [Google Scholar] [CrossRef]
- Koseki, S.; Yoshida, K.; Kamitani, Y.; Isobe, S.; Itoh, K. Effect of mild heat pre-treatment with alkaline electrolyzed water on the efficacy of acidic electrolyzed water against Escherichia coli O157:H7 and Salmonella on lettuce. Food Microbiol. 2004, 21, 559–566. [Google Scholar] [CrossRef]
- Clayton, D.A.; Griffith, C.J.; Price, P.; Peters, A.C. Food handlers’ beliefs and self-reported practices. Int. J. Environ. Health Res. 2002, 12, 25–39. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mdluli, F.; Thamaga-Chitja, J.; Schmidt, S. Appraisal of Hygiene Indicators and Farming Practices in the Production of Leafy Vegetables by Organic Small-Scale Farmers in uMbumbulu (Rural KwaZulu-Natal, South Africa). Int. J. Environ. Res. Public Health 2013, 10, 4323-4338. https://doi.org/10.3390/ijerph10094323
Mdluli F, Thamaga-Chitja J, Schmidt S. Appraisal of Hygiene Indicators and Farming Practices in the Production of Leafy Vegetables by Organic Small-Scale Farmers in uMbumbulu (Rural KwaZulu-Natal, South Africa). International Journal of Environmental Research and Public Health. 2013; 10(9):4323-4338. https://doi.org/10.3390/ijerph10094323
Chicago/Turabian StyleMdluli, Fezile, Joyce Thamaga-Chitja, and Stefan Schmidt. 2013. "Appraisal of Hygiene Indicators and Farming Practices in the Production of Leafy Vegetables by Organic Small-Scale Farmers in uMbumbulu (Rural KwaZulu-Natal, South Africa)" International Journal of Environmental Research and Public Health 10, no. 9: 4323-4338. https://doi.org/10.3390/ijerph10094323




