Resistance Genes, Phage Types and Pulsed Field Gel Electrophoresis Pulsotypes in Salmonella enterica Strains from Laying Hen Farms in Southern Italy
Abstract
:1. Introduction
2. Methods
2.1. Samples and Bacterial Strains
2.2. Clonality—PFGE and Phage Typing
2.3. Antimicrobial Susceptibility Testing
2.4. Antimicrobial Resistance Genes/Genetic Elements and Conjugation Assays
3. Results and Discussion
3.1. Salmonella Prevalence in Samples and Farms
3.2. Clonality Study

| PulseNet Europe | Serovar | Phage type | Place/Year | Farm | Sample | Resistance | Class 1 integrons | SGI1 ‡ | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| nomenclature | (n. of strains) | (n. of strains) † | pattern (strain) * | ||||||||
| intI1 | gene | Resistance | left | right | |||||||
| cassette(s) | gene (s) | junction | junction | ||||||||
| SENTXB.0001 | Enteritidis (2) | PT1 (1) | Apulia/1998 | A | Workers’ shoes | Su | - | - | sul2 | - | - |
| PT37 (1) | “ | B | Feces | " | - | - | " | - | - | ||
| SENTXB.0001 | Enteritidis (1) | PT8 (1) | Apulia/2001 | I | Eggs | " | - | - | " | - | - |
| SENTXB.0001 | Enteritidis (2) | PT4 (1) | Apulia/2003 | J | Feces | " | - | - | " | - | - |
| PT14b (1) | “ | K | Eggs | " | - | - | " | - | - | ||
| SENTXB.0001 | Enteritidis (4) | PT14b (2) | Apulia/2004 | L | Feces; eggs | " | - | - | " | - | - |
| PT14b (1); NT (1) | “ | C | Feces Eggs | " | - | - | " | - | - | ||
| SENTXB.0001 | Enteritidis (1) | NT (1) | Apulia/2005 | M | Feces | " | - | - | " | - | - |
| SENTXB.0001 | Enteritidis (2) | PT14b (1) | Apulia/2006 | L | Feces | " | - | - | " | - | - |
| NT (1) | N | Egg grader | " | - | - | " | - | - | |||
| SENTXB.0001 | Enteritidis (1) | NT (1) | " | O | Eggs | ANxSSuT (SE402) | - | - | blaTEM-1; strAB; sul2; tet(A) | - | - |
| STYMXB.0147 | Typhimurium (3) | DT193 (2) | Apulia/1999 | C | Eggs;eggs grader | Su | - | - | sul2 | - | - |
| NT (1) | “ | C | Eggs | " | - | - | " | - | - | ||
| STYMXB.0147 | Typhimurium (4) | DT193 (1) | Apulia/2000 | C | Feces | " | - | - | " | - | - |
| DT193 (1) | " | F | Feces | " | - | - | " | - | - | ||
| DT104 (1) | " | G | Egg grader | " | - | - | " | - | - | ||
| U302 (1) | " | H | Selection room floor | " | - | - | " | - | - | ||
| STYMXB.0147 | Typhimurium (1) | DT104 (1) | Apulia/2006 | E | Feces | " | - | - | " | - | - |
| STYMXB.0147 | Typhimurium (1) | DT193 (1) | Basilicata/1999 | D | Eggs | " | - | - | " | - | - |
| STYMXB.0110 | Typhimurium (1) | DT208 (1) | Apulia/2000 | E | Feces | SSuT (ST256) | + | aadA1 | sul1; sul2; tet(C) | - | - |
| STYMXB.0061 | Typhimurium (1) | DT104 (1) | Basilicata/2007 | P | Feces | ACSSuT (ST425) | + | aadA2; blaPSE-1 | sul1; floR; tet(G) | + | + |
3.3. Antimicrobial Susceptibility Study
3.4. Genetic Elements and Resistance Genes
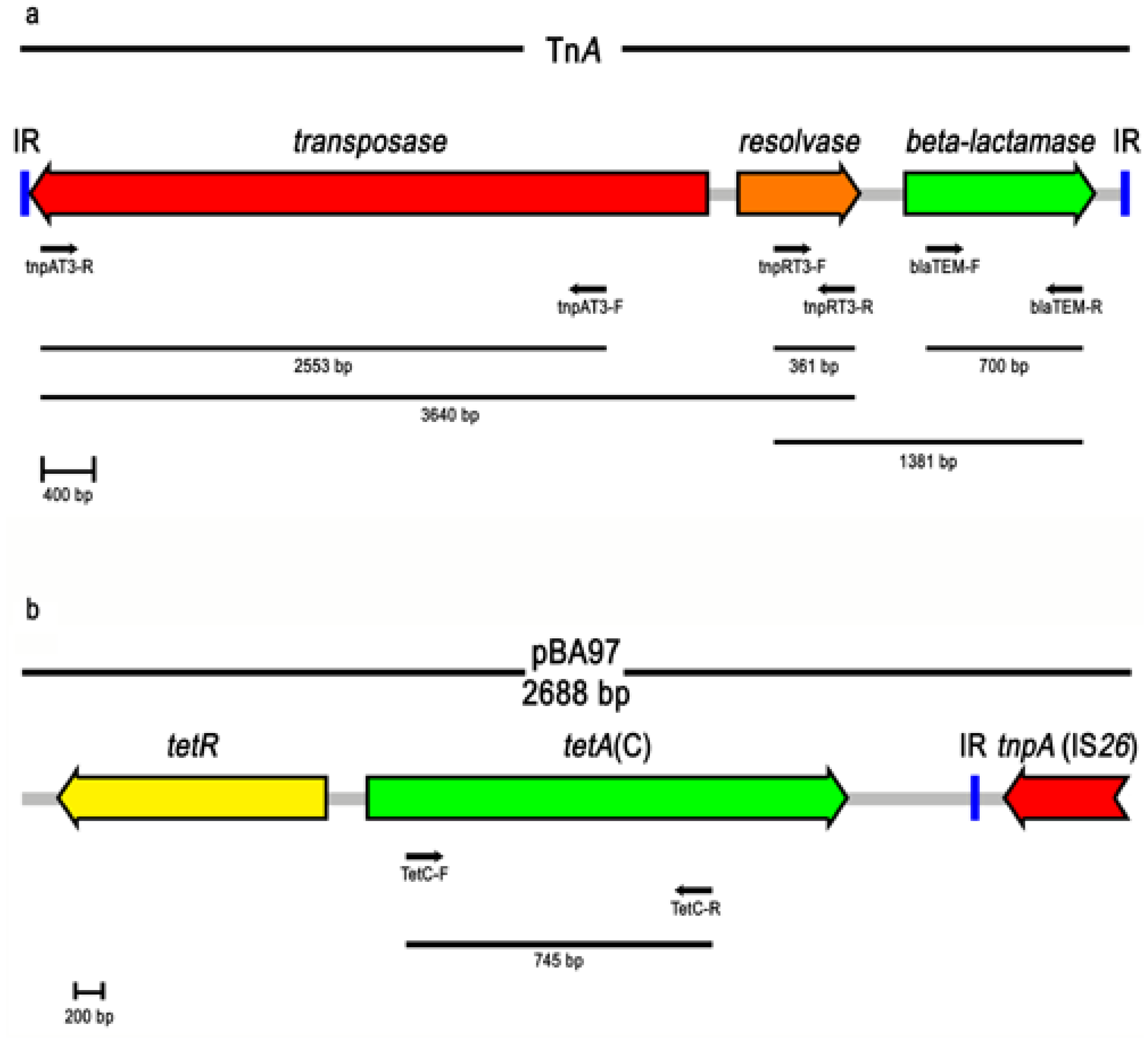
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Herikstad, H.; Motarjemi, Y.; Tauxe, R.V. Salmonella surveillance: A global survey of public health serotyping. Epidemiol. Infect. 2002, 129, 1–8. [Google Scholar]
- Woo, Y.K. Finding the sources of Korean Salmonella enterica serovar Enteritidis PT 4 isolates by pulsed-field gel electrophoresis. J. Microbiol. 2005, 43, 424–429. [Google Scholar]
- Velge, P.; Cloeckaert, A.; Barrow, P. Emergence of Salmonella epidemics: The problems related to Salmonella enterica serotype Enteritidis and multiple antibiotic resistance in other major serotypes. Vet. Res. 2005, 36, 267–288. [Google Scholar] [CrossRef]
- Collard, J.M.; Bertrand, S.; Dierick, K.; Godard, C.; Wildemauwe, C.; Vermeersch, K.; Duculot, J.; van Immerseel, F.; Pasmans, F.; Imberechts, H.; et al. Drastic decrease of Salmonella Enteritidis isolated from humans in belgium in 2005, shift in phage types and influence on foodborne outbreaks. Epidemiol. Infect. 2008, 136, 771–781. [Google Scholar]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Gast, R.; Humphrey, T.J.; van Immerseel, F. Mechanisms of egg contamination by Salmonella Enteritidis. FEMS Microbiol. Rev. 2009, 33, 718–738. [Google Scholar] [CrossRef]
- Okamura, M.; Miyamoto, T.; Kamijima, Y.; Tani, H.; Sasai, K.; Baba, E. Differences in abilities to colonize reproductive organs and to contaminate eggs in intravaginally inoculated hens and in vitro adherences to vaginal explants between Salmonella enteritidis and other Salmonella serovars. Avian Dis. 2001, 45, 962–971. [Google Scholar] [CrossRef]
- Gebreyes, W.A.; Davies, P.R.; Morrow, W.E.; Funk, J.A.; Altier, C. Antimicrobial resistance of Salmonella isolates from swine. J. Clin. Microbiol. 2000, 38, 4633–4636. [Google Scholar]
- Glenn, L.M.; Lindsey, R.L.; Frank, J.F.; Meinersmann, R.J.; Englen, M.D.; Fedorka-Cray, P.J.; Frye, J.G. Analysis of antimicrobial resistance genes detected in multidrug-resistant Salmonella enterica serovar Typhimurium isolated from food animals. Microb. Drug Resist. 2011, 17, 407–418. [Google Scholar] [CrossRef]
- Boyd, D.; Peters, G.A.; Cloeckaert, A.; Boumedine, K.S.; Chaslus-Dancla, E.; Imberechts, H.; Mulvey, M.R. Complete nucleotide sequence of a 43-kilobase genomic island associated with the multidrug resistance region of Salmonella enterica serovar Typhimurium DT104 and its identification in phage type DT120 and serovar Agona. J. Bacteriol. 2001, 183, 5725–5732. [Google Scholar] [CrossRef]
- Doublet, B.; Boyd, D.; Mulvey, M.R.; Cloeckaert, A. The Salmonella genomic island 1 is an integrative mobilizable element. Mol. Microbiol. 2005, 55, 1911–1924. [Google Scholar] [CrossRef]
- Dionisi, A.M.; Graziani, C.; Lucarelli, C.; Filetici, E.; Villa, L.; Owczarek, S.; Caprioli, A.; Luzzi, I. Molecular characterization of multidrug-resistant strains of Salmonella enterica serotype Typhimurium and monophasic variant (S. 4,[5],12:i:-) isolated from human infections in Italy. Foodborne Pathog. Dis. 2009, 6, 711–717. [Google Scholar] [CrossRef]
- European Food Safety Authority. Technical specifications on the harmonised monitoring and reporting of antimicrobial resistance in Salmonella, Campylobacter and indicator Escherichia coli and Enterococcus spp. Bacteria transmitted through food. EFSA J. 2012, 10, 2742.
- Swaminathan, B.; Barrett, T.J.; Hunter, S.B.; Tauxe, R.V. Pulsenet: The molecular subtyping network for foodborne bacterial disease surveillance, United States. Emerg. Infect. Dis. 2001, 7, 382–389. [Google Scholar]
- International Organization for Standardization (ISO), Microbiology of Food Animal Feeding Stuffs Horizontal Method for the Detection of Salmonella Spp; International Organization for Standardization: Geneva, Switzerland, 2002.
- Popoff, M.Y.; Bockemuhl, J.; Gheesling, L.L. Supplement 2002 (no. 46) to the Kauffmann-White scheme. Res. Microbiol. 2004, 155, 568–570. [Google Scholar] [CrossRef]
- Tenover, F.C.; Arbeit, R.D.; Goering, R.V.; Mickelsen, P.A.; Murray, B.E.; Persing, D.H.; Swaminathan, B. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: Criteria for bacterial strain typing. J. Clin. Microbiol. 1995, 33, 2233–2239. [Google Scholar]
- Ribot, E.M.; Fair, M.A.; Gautom, R.; Cameron, D.N.; Hunter, S.B.; Swaminathan, B.; Barrett, T.J. Standardization of pulsed-field gel electrophoresis protocols for the subtyping of Escherichia coli O157:H7, Salmonella, and Shigella for PulseNet. Foodborne Pathog. Dis. 2006, 3, 59–67. [Google Scholar] [CrossRef]
- Centers fo Disease Control and Prevention (CDC). Pulsenet home. Available online: http://www.cdc.gov/pulsenet.
- Ward, L.R.; de Sa, J.D.; Rowe, B. A phage-typing scheme for Salmonella enteritidis. Epidemiol. Infect. 1987, 99, 291–294. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI), Performance Standards for Antimicrobial Disk Susceptibility Tests, 9th ed.; CLSI: Villanova, PA, USA, 2006.
- Clinical and Laboratory Standards Institute (CLSI), Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria tha Grow Aerobically; CLSI: Villanova, PA, USA, 2006.
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef]
- Camarda, A.; Pupillo, A.; Pugliese, N.; Circella, E.; Dionisi, A.M.; Ricci, A.; Pazzani, C. Phenotypic and genetic traits of Salmonella enterica subsp. Serovar Typhimurium strains causing salmonellosis foci in rabbit farms from Southern Italy in 1999–2003. Res. Vet. Sci. 2013, 94, 394–398. [Google Scholar] [CrossRef]
- Pugliese, N.; Maimone, F.; Scrascia, M.; Materu, S.F.; Pazzani, C. SXT-related integrating conjugative element and incC plasmids in Vibrio cholerae O1 strains in Eastern Africa. J. Antimicrob. Chemother. 2009, 63, 438–442. [Google Scholar] [CrossRef]
- Ye, J.; McGinnis, S.; Madden, T.L. BLAST: improvements for better sequence analysis. Nucleic Acids Res. 2006, 34, W6–W9. Available online: http://www.ncbi.nlm.nih.gov/BLAST. [Google Scholar]
- Sherburne, C.K.; Lawley, T.D.; Gilmour, M.W.; Blattner, F.R.; Burland, V.; Grotbeck, E.; Rose, D.J.; Taylor, D.E. The complete DNA sequence and analysis of R27, a large incHI plasmid from Salmonella typhi that is temperature sensitive for transfer. Nucleic Acids Res. 2000, 28, 2177–2186. [Google Scholar] [CrossRef]
- Carattoli, A.; Bertini, A.; Villa, L.; Falbo, V.; Hopkins, K.L.; Threlfall, E.J. Identification of plasmids by PCR-based replicon typing. J. Microbiol. Methods 2005, 63, 219–228. [Google Scholar] [CrossRef]
- Peters, T.M.; Berghold, C.; Brown, D.; Coia, J.; Dionisi, A.M.; Echeita, A.; Fisher, I.S.; Gatto, A.J.; Gill, N.; Green, J.; et al. Relationship of pulsed-field profiles with key phage types of Salmonella enterica serotype Enteritidis in Europe: results of an international multi-centre study. Epidemiol. Infect. 2007, 135, 1274–1281. [Google Scholar]
- Threlfall, E.J. Epidemic Salmonella typhimurium DT 104-a truly international multiresistant clone. J. Antimicrob. Chemother. 2000, 46, 7–10. [Google Scholar] [CrossRef]
- Guerin, P.J.; Nygard, K.; Siitonen, A.; Vold, L.; Kuusi, M.; de Jong, B.; Rottingen, J.A.; Alvseike, O.; Olsson, A.; Lassen, J.; et al. Emerging Salmonella Enteritidis anaerogenic phage type 14b: Outbreak in Norwegian, Swedish and Finnish travellers returning from Greece. Euro Surveill. 2006, 11, 61–66. [Google Scholar]
- Janmohamed, K.; Zenner, D.; Little, C.; Lane, C.; Wain, J.; Charlett, A.; Adak, B.; Morgan, D. National outbreak of Salmonella Enteritidis phage type 14b in England, September to December 2009: Case-control study. Euro Surveill. 2011, 16, 1–6. [Google Scholar]
- Hrivniakova, L.; Schmid, D.; Luckner-Hornischer, A.; Lassnig, H.; Kornschober, C.; Angermayer, J.; Allerberger, F. Salmonellosis outbreak due to Salmonella Enteritidis phage type 14b resistant to nalidixic acid, Austria, September 2010. Euro Surveill. 2011, 16, 1–7. [Google Scholar]
- Gatto, A.J.; Peters, T.M.; Green, J.; Fisher, I.S.; Gill, O.N.; O'Brien, S.J.; Maguire, C.; Berghold, C.; Lederer, I.; Gerner-Smidt, P.; et al. Distribution of molecular subtypes within Salmonella enterica serotype Enteritidis phage type 4 and S. Typhimurium definitive phage type 104 in nine European countries, 2000–2004: Results of an international multi-centre study. Epidemiol. Infect. 2006, 134, 729–736. [Google Scholar]
- Majtanova, L.; Majtan, J.; Majtan, V. Trends in phage types of Salmonella enterica serovars Enteritidis and Typhimurium isolated in Slovakia from 1995 to 2009. Diagn. Microbiol. Infect. Dis. 2011, 69, 454–456. [Google Scholar] [CrossRef]
- Rabsch, W.; Tschape, H.; Baumler, A.J. Non-typhoidal salmonellosis: Emerging problems. Microbes Infect. 2001, 3, 237–247. [Google Scholar] [CrossRef]
- Threlfall, E.J. Antimicrobial drug resistance in Salmonella: Problems and perspectives in food- and water-borne infections. FEMS Microbiol. Rev. 2002, 26, 141–148. [Google Scholar] [CrossRef]
- Dos Reis, E.M.; Rodrigues Ddos, P.; de Freitas-Almeida, A.C.; Hofer, E. Prevalence of R-type ACSSuT in strains of Salmonella serovar Typhimurium DT193 isolated from human infections in Brazil. Rev. Panam. Salud Publica 2011, 29, 387–392. [Google Scholar]
- Michael, G.B.; Butaye, P.; Cloeckaert, A.; Schwarz, S. Genes and mutations conferring antimicrobial resistance in Salmonella: An update. Microbes Infect. 2006, 8, 1898–1914. [Google Scholar] [CrossRef]
- Mulvey, M.R.; Boyd, D.A.; Olson, A.B.; Doublet, B.; Cloeckaert, A. The genetics of Salmonella genomic island 1. Microbes Infect. 2006, 8, 1915–1922. [Google Scholar] [CrossRef]
- Nogrady, N.; Toth, A.; Kostyak, A.; Paszti, J.; Nagy, B. Emergence of multidrug-resistant clones of Salmonella infantis in broiler chickens and humans in Hungary. J. Antimicrob. Chemother. 2007, 60, 645–648. [Google Scholar] [CrossRef]
- Bailey, J.K.; Pinyon, J.L.; Anantham, S.; Hall, R.M. Distribution of the blaTEM gene and blaTEM-containing transposons in commensal Escherichia coli. J. Antimicrob. Chemother. 2011, 66, 745–751. [Google Scholar] [CrossRef]
- Heffron, F.; McCarthy, B.J.; Ohtsubo, H.; Ohtsubo, E. DNA sequence analysis of the transposon Tn3: Three genes and three sites involved in transposition of Tn3. Cell 1979, 18, 1153–1163. [Google Scholar] [CrossRef]
- Soufi, L.; Saenz, Y.; Vinue, L.; Abbassi, M.S.; Ruiz, E.; Zarazaga, M.; Ben Hassen, A.; Hammami, S.; Torres, C. Escherichia coli of poultry food origin as reservoir of sulphonamide resistance genes and integrons. Int. J. Food Microbiol. 2011, 144, 497–502. [Google Scholar] [CrossRef]
- Van Duijkeren, E.; Houwers, D.J. A critical assessment of antimicrobial treatment in uncomplicated Salmonella enteritis. Vet. Microbiol. 2000, 73, 61–73. [Google Scholar] [CrossRef]
- Thaker, M.; Spanogiannopoulos, P.; Wright, G.D. The tetracycline resistome. Cell Mol. Life Sci. 2010, 67, 419–431. [Google Scholar] [CrossRef]
- Douadi, B.; Thong, K.-L.; Watanabe, H.; Puthucheary, S.D. Characterization of drug resistant Salmonella enterica Serotype Typhimurium by Antibiograms, Plasmids, Integrons, Resistance Genes and PFGE. J. Microbiol. Biotechn. 2010, 20, 1042–1052. [Google Scholar] [CrossRef]
- Ma, M.; Wang, H.; Yu, Y.; Zhang, D.; Liu, S. Detection of antimicrobial resistance genes of pathogenic Salmonella from swine with DNA microarray. J. Vet. Diagn. Invest. 2007, 19, 161–167. [Google Scholar] [CrossRef]
- Chalmers, G.; Kozak, G.K.; Hillyer, E.; Reid-Smith, R.J.; Boerlin, P. Low minimum inhibitory concentrations associated with the tetracycline-resistance gene tet(C) in Escherichia coli. Can. J. Vet. Res. 2010, 74, 145–148. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI), Performance Standards for Antimicrobial Susceptibility Testing; Eighteen Informational Supplement; CLSI: Villanova, PA, USA, 2008.
- Ford, P.J.; Avison, M.B. Evolutionary mapping of the SHV beta-lactamase and evidence for two separate IS26-dependent blaSHV mobilization events from the Klebsiella pneumoniae chromosome. J. Antimicrob. Chemother. 2004, 54, 69–75. [Google Scholar] [CrossRef]
- Guerra, B.; Junker, E.; Schroeter, A.; Malorny, B.; Lehmann, S.; Helmuth, R. Phenotypic and genotypic characterization of antimicrobial resistance in German Escherichia coli isolates from cattle, swine and poultry. J. Antimicrob. Chemother. 2003, 52, 489–492. [Google Scholar] [CrossRef]
- Suzuki, H.; Yano, H.; Brown, C.J.; Top, E.M. Predicting plasmid promiscuity based on genomic signature. J. Bacteriol. 2010, 192, 6045–6055. [Google Scholar] [CrossRef]
- Antunes, P.; Mourao, J.; Machado, J.; Peixe, L. First description of qnrS1-incN plasmid in a ST11 Salmonella Enteritidis clinical isolate from Portugal. Diagn. Microbiol. Infect. Dis. 2011, 69, 463–465. [Google Scholar] [CrossRef]
- Frost, J.A.; Ward, L.R.; Rowe, B. Acquisition of a drug resistance plasmid converts Salmonella enteritidis phage type 4 to phage type 24. Epidemiol. Infect. 1989, 103, 243–248. [Google Scholar] [CrossRef]
- Soufi, L.; Saenz, Y.; de Toro, M.; Abbassi, M.S.; Rojo-Bezares, B.; Vinue, L.; Bouchami, O.; Touati, A.; Ben Hassen, A.; Hammami, S.; et al. Phenotypic and genotypic characterization of Salmonella enterica recovered from poultry meat in Tunisia and identification of new genetic traits. Vector Borne Zoonotic Dis. 2012, 12, 10–16. [Google Scholar] [CrossRef]
- Parsons, D.J.; Orton, T.G.; D'Souza, J.; Moore, A.; Jones, R.; Dodd, C.E. A comparison of three modelling approaches for quantitative risk assessment using the case study of Salmonella spp. in poultry meat. Int. J. Food Microbiol. 2005, 98, 35–51. [Google Scholar] [CrossRef] [Green Version]
- Gaffga, N.H.; Barton Behravesh, C.; Ettestad, P.J.; Smelser, C.B.; Rhorer, A.R.; Cronquist, A.B.; Comstock, N.A.; Bidol, S.A.; Patel, N.J.; Gerner-Smidt, P.; et al. Outbreak of salmonellosis linked to live poultry from a mail-order hatchery. N. Engl. J. Med. 2012, 366, 2065–2073. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Camarda, A.; Pugliese, N.; Pupillo, A.; Oliva, M.; Circella, E.; Dionisi, A.M.; Ricci, A.; Legretto, M.; Caroli, A.; Pazzani, C. Resistance Genes, Phage Types and Pulsed Field Gel Electrophoresis Pulsotypes in Salmonella enterica Strains from Laying Hen Farms in Southern Italy. Int. J. Environ. Res. Public Health 2013, 10, 3347-3362. https://doi.org/10.3390/ijerph10083347
Camarda A, Pugliese N, Pupillo A, Oliva M, Circella E, Dionisi AM, Ricci A, Legretto M, Caroli A, Pazzani C. Resistance Genes, Phage Types and Pulsed Field Gel Electrophoresis Pulsotypes in Salmonella enterica Strains from Laying Hen Farms in Southern Italy. International Journal of Environmental Research and Public Health. 2013; 10(8):3347-3362. https://doi.org/10.3390/ijerph10083347
Chicago/Turabian StyleCamarda, Antonio, Nicola Pugliese, Antonia Pupillo, Marta Oliva, Elena Circella, Anna Maria Dionisi, Antonia Ricci, Marilisa Legretto, Anna Caroli, and Carlo Pazzani. 2013. "Resistance Genes, Phage Types and Pulsed Field Gel Electrophoresis Pulsotypes in Salmonella enterica Strains from Laying Hen Farms in Southern Italy" International Journal of Environmental Research and Public Health 10, no. 8: 3347-3362. https://doi.org/10.3390/ijerph10083347








