1. Introduction
Climate conditions, such as temperature and precipitation, are among many important factors that determine the spatial and temporal distribution of vectors and vector-borne diseases. Changes in climate influence the occurrence of vector-borne diseases in the following three major ways: (a) reproduction, development, and survival of vectors, which in turn drive the distribution and abundance of vectors; (b) blood seeking activity of vectors; and c) rates of pathogen amplification, through development, multiplication, and survival within vectors [
1,
2]. In addition, climate conditions may affect the distribution, abundance, behavior, phenology of reproduction, and migration of vertebrate hosts [
3,
4]. Therefore, climate change will drive dramatic alterations in the spatial and temporal distribution and overall incidence of vector-borne diseases. Besides these direct effects of climate change on vector-borne diseases, climate change can also lead to substantial alterations in landscape, which in turn influence the distribution and abundance of hosts, vectors, and vector-borne pathogens [
1,
5]. Without taking these ecological effects of climate change and their interactions into consideration, projections of the potential effects of climate change on vector-borne diseases will remain inaccurate [
1,
5].
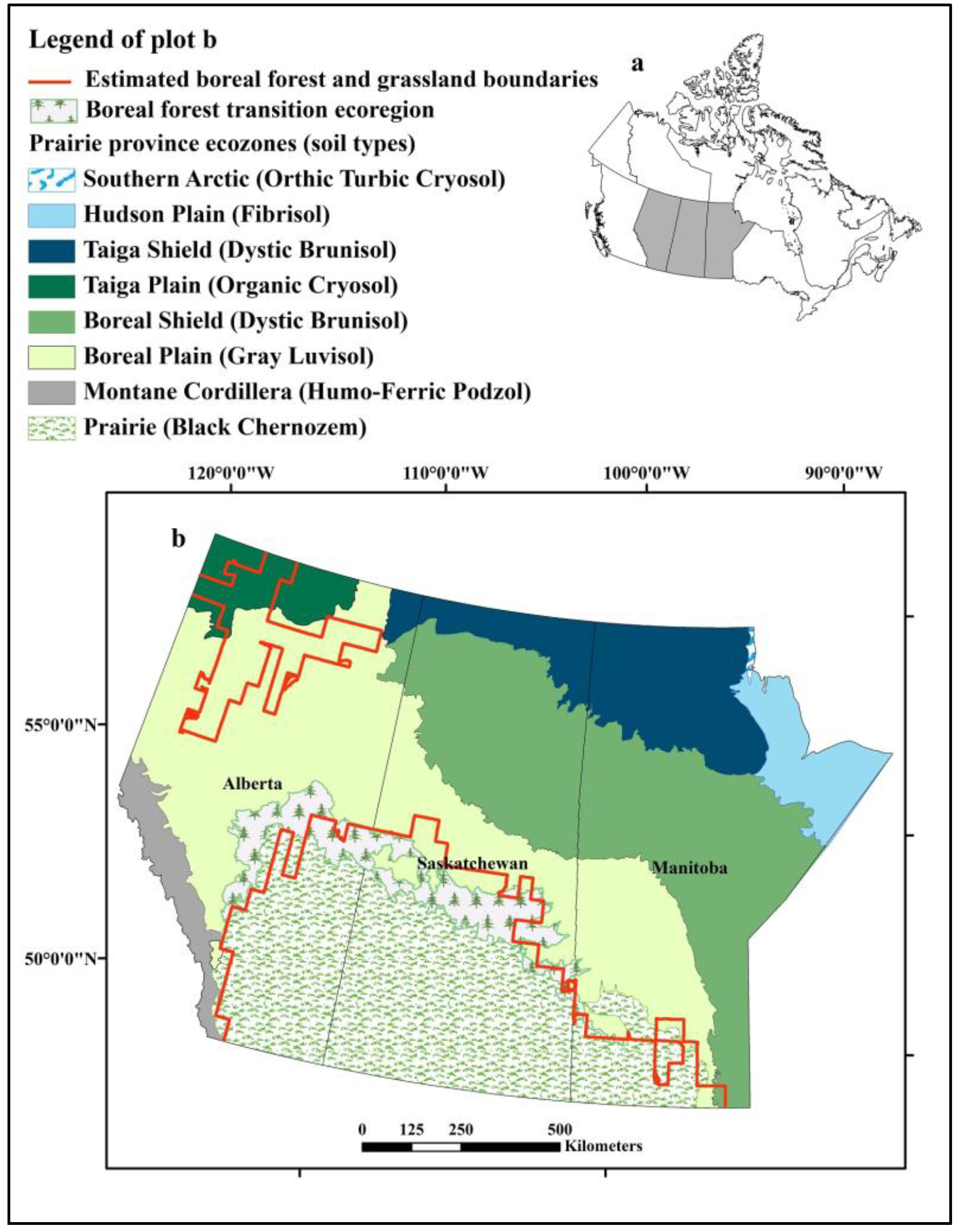
West Nile virus (WNV) from the family Flaviviridae, genus Flavivirus was introduced into the Western Hemisphere in 1999 [
6]. Since that time the Canadian prairies, grassland ecozone in the southern parts of the provinces of Manitoba, Saskatchewan, and Alberta (
Figure 1), have generally had the highest human incidence of clinical cases of WNV infection in Canada. During the 2007 epidemic season, a total of 2,215 confirmed clinical cases of WNV infection were reported in Canadians, of which 98% occurred in the prairie provinces, including 1,285 in Saskatchewan, 578 in Manitoba, and 318 in Alberta [
7]. As a newly introduced vector-borne disease affecting a wide range of vertebrate hosts, WNV remains a significant concern for public health and wildlife conservation in the Canadian prairies.
In the Canadian prairies, the mosquito species
Culex tarsalis Coquillett is the principal vector for WNV [
8,
9] This mosquito species is one of the most competent WNV vectors evaluated to date in laboratory studies [
10] and is the predominant potential vector species in the Canadian prairies during the summer WNV season [
8]. The southern boundary of the boreal forest transition zone is identified as the northernmost limit of WNV distribution in western North America [
8], although
Cx. tarsalis has been recorded further north [
11]. Climate, particularly temperature and precipitation, and habitat preference determine the distribution of
Cx. tarsalis in western North America [
12]. Grassland and agriculture area are the preferred land cover type for
Cx. tarsalis in the Canadian prairies [
8,
9,
13] and other regions of the Great Plains [
12,
14]. Stagnant water bodies with high organic content are favored sites for oviposition by
Cx. tarsalis [
8,
15]. In the Canadian prairies, larvae of
Cx. tarsalis are commonly found in many temporary water bodies, such as artificial containers, water-filled hoof prints, and weedy roadside ditches [
8,
12]. Furthermore, large water bodies and running water are not suitable for larval development due to the disturbance and lower nutrition concentration [
16]. Studies have found that the percentage of wetland is not associated with the abundance of
Cx. tarsalis and WNV risk in the Canadian prairies and northern Great Plains [
13,
14,
17,
18].
Figure 1.
Distribution of ecozones and soil types in the prairie provinces of Alberta, Saskatchewan, and Manitoba, Canada and the boreal forest and prairie boundaries estimated using baseline climate conditions. (a) Location of prairie provinces (grey shading) in Canada. (b) Enlargement of Prairie provinces and distribution of ecozones.
Figure 1.
Distribution of ecozones and soil types in the prairie provinces of Alberta, Saskatchewan, and Manitoba, Canada and the boreal forest and prairie boundaries estimated using baseline climate conditions. (a) Location of prairie provinces (grey shading) in Canada. (b) Enlargement of Prairie provinces and distribution of ecozones.
Several biological features of
Cx. tarsalis facilitate the transmission of WNV including its capacity to: vertically transmit WNV to its offspring [
19]; produce multiple generations per season; and take multiple blood meals during each generation [
8]. Because it feeds on both avian and mammalian hosts,
Cx. tarsalis plays the role of a “bridging vector” transmitting WNV from its enzootic cycle to humans and other mammalian species [
20,
21,
22]. Besides WNV,
Cx. tarsalis is the primary competent vector for the St. Louis and Western Equine Encephalitis viruses in North America [
8,
23,
24].
Changes in future climate will not only influence the distribution of vectors and pathogens, but also the habitat suitability for vectors [
25]. Therefore, the assessment of possible effects of climate change on grassland distribution is critical for predicting the occurrence of
Cx. tarsalis and WNV under future climate change. In a warmer and possibly drier future climate, current grassland habitat in the Canadian prairie ecozone might be replaced by the grassland flora found in the United States, and the boreal forest in the northern prairie provinces might be replaced by aspen parkland and grassland [
26,
27,
28]. The southern boundary of the boreal forest fits very closely with the zero isoline of the annual climate moisture index, estimated by mean annual precipitation minus potential evapotranspiration (PET) [
29].
In the present study, we integrated empirically derived, biologically-relevant temperature thresholds for
Cx. tarsalis survival and WNV development, and statistical models in order to predict the effects of climate change on the distribution and abundance of
Cx. tarsalis and WNV in the Canadian prairies, one of the most highly endemic regions in North America. Furthermore, we took into account potential changes in landscape as a result of climate change, including predicting the distribution of grassland habitat under future climate changes [
26,
27]. Our objectives were to assess and predict the potential effects of climate change on the abundance of
Cx. tarsalis and infection rate of WNV in
Cx. tarsalis in the Canadian prairie ecozone, under the assumption that competent avian amplifying hosts will continue to persist in this region. In addition, we explored the possibility of northward expansion of
Cx. tarsalis and WNV out of their current distribution area in the Canadian prairie ecozone.
4. Discussion
Climate change is expected to influence the distribution of both vectors and vector borne pathogens, and contribute to the expansion or shifting of endemic regions [
25,
40,
41]. This study demonstrates the potential for substantial expansion of the transmission season and geographic distribution of a recently introduced vector-borne disease in a highly endemic region of North America as a result of rapid climate and landscape change.
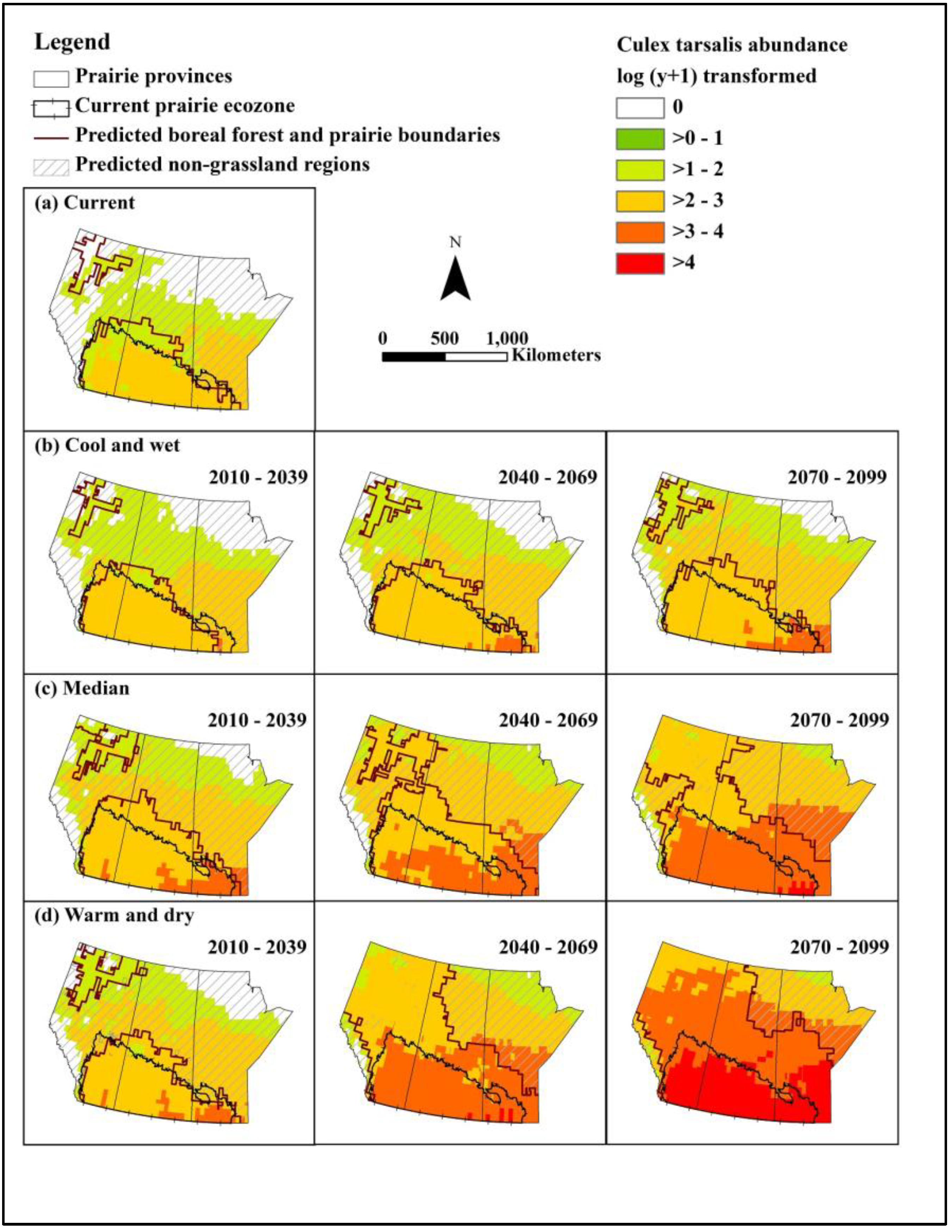
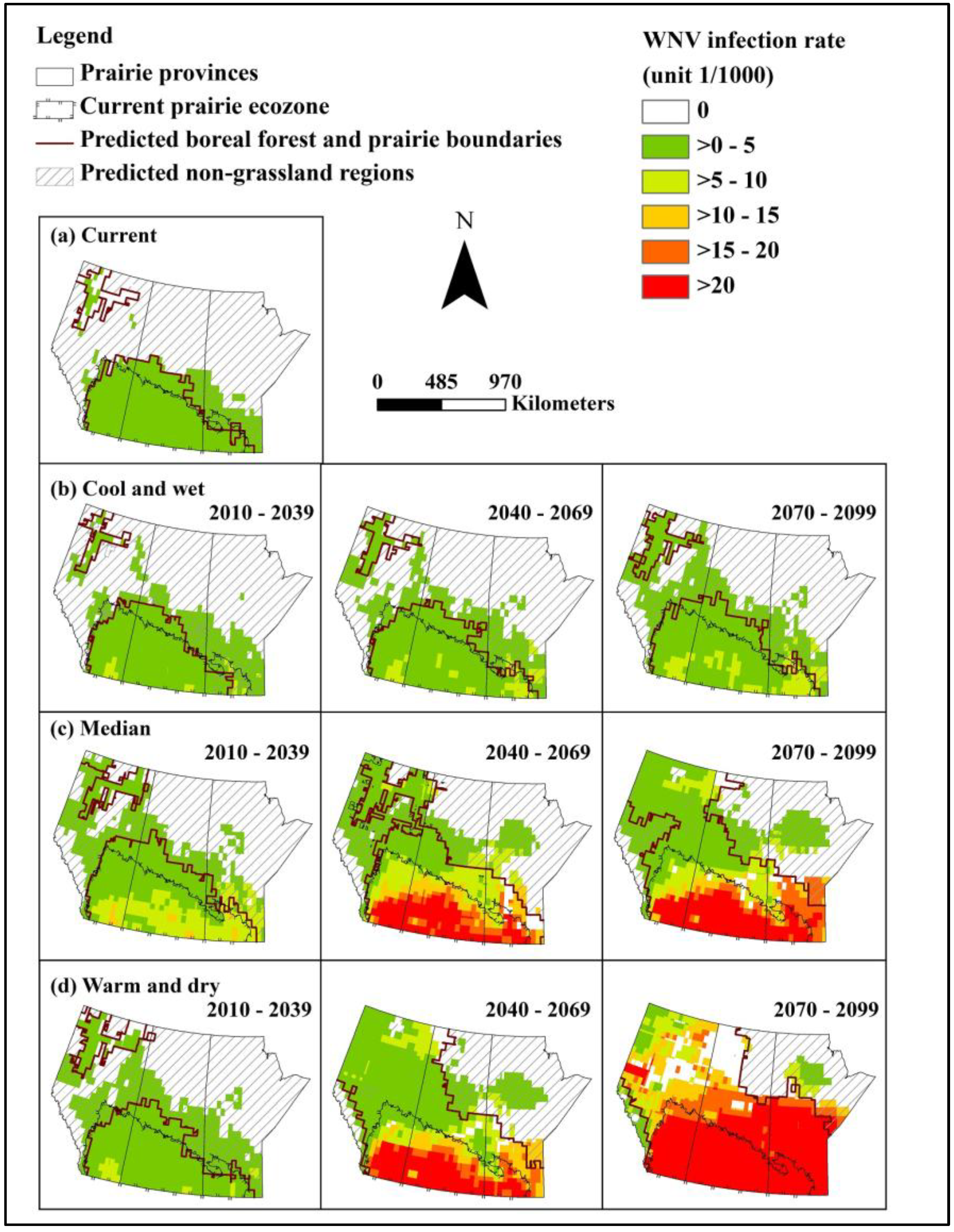
We constructed models for predicting abundance of the primary mosquito vector Cx. tarsalis and WNV infection rate, and populated these with data from baseline and selected future climate scenarios to assess the effects of climate and landscape change on WNV in the Canadian prairie ecozone. Under even the most optimistic of scenarios, WNV will undergo northern range expansion and extension of the transmission season by the 2050s. Based on a middle-of-the-road scenario, approximately half to two/thirds of the northern portion of the prairie provinces will have a climate newly suitable for WNV transmission by the 2050s. Under the most extreme warming conditions, peak mosquito infection rates could be 30 times that of baseline, representing a substantial increase in infection pressure for people and animals alike.
Although higher temperatures may lead to increased mosquito mortality and thus represent a natural check on viral amplification, our results suggest that mean monthly temperatures will not exceed the upper threshold for survival of adult female
Cx. tarsalis. In addition, mosquitoes may select cooler microhabitats if temperatures exceed tolerances. Therefore, the observed temporal and spatial distribution of WNV in the Canadian prairies will remain primarily determined by the lower temperature limitation for WNV amplification in
Cx. tarsalis (estimated to be 14.3 °C) [
37]. Laboratory experiments demonstrate that the temperature threshold for survival of
Cx. tarsalis is generally between 14 °C and 35 °C, and within this range, temperature is positively correlated with development rate of vector [
36,
42]. Therefore, climate change will lead to higher development rates for vector without a compensatory increase in mosquito mortality. Moreover, increased temperatures will also increase the infection rate of WNV in
Cx. tarsalis, especially in the southern part of the Canadian prairies.
Many factors besides climate are important determinants of the distribution and incidence of vector borne diseases, such as habitat suitability for competent vectors [
43]. Changes in future climate could also induce shifts in habitat distribution and affect habitat suitability for vectors [
25,
40]. In the current study, we used the constructed model to predict the distribution of grassland habitat under current and selected future outcome scenarios. Northward expansion of grassland has been predicted in Western Canada in a future of climate change, with boreal forest replaced by aspen parkland and grassland, and current Canadian grassland types replaced by those found in the U.S. Great Plains [
26,
27,
28]. These latitudinal shifts in vegetation zones will create more suitable habitat for
Cx. tarsalis in the northern part of the prairie provinces, while maintaining suitable habitat in the current Canadian prairie ecozone. However, the spatial expansion of
Cx. tarsalis and WNV distribution in the prairie provinces will lag behind the shifts of vegetation zones.
The predicted distribution of grassland revealed another smaller area located in northern Alberta where the climate is appropriate for grassland habitat (
Figure 1,
Figure 2). Isolated grasslands resembling mixed prairie communities of the northern Great Plains are observed in this area [
44,
45,
46]. In addition, recent studies have also revealed
Cx. tarsalis in the region of these grassland remnants, which extend into the southern Northwest Territories, although no WNV was detected [
11]. These empirical observations validated our prediction that if grassland habitat is available,
Cx. tarsalis can already establish in the northern regions of the prairie provinces under current and projected future climate conditions; however, the activity of WNV remains low or nonexistent in these regions under current climate conditions. Moreover, the Canadian prairie represents the northernmost edge of WNV distribution in the western hemisphere. As
Cx. tarsalis, WNV, and other arboviruses expand northward out of their current endemic area into regions where humans, domestic livestock, and wildlife lack immunity, these vector-borne diseases may emerge in these newly vulnerable populations [
47,
48].
Temperature increases and the ecological impact of climate change are predicted to be greater in temperate and polar regions than in tropical regions [
47,
49,
50]. Increasing environmental temperature shortens the maturation time required for
Cx. tarsalis and the extrinsic incubation period of West Nile virus. Furthermore, it also accelerates the mosquito gonadotrophic cycle and affects mosquito survival. Although beyond the scope of the current study, these relationships will influence virus transmission by increasing the contact rate between
Cx. tarsalis and competent vertebrate hosts [
37,
51].
Although we have demonstrated that changing climate and habitat will drastically alter the current distribution and abundance of a newly-introduced vector-borne disease, a number of other factors will also affect the ecology of WNV, and will in turn be affected by climate conditions. These factors include ability of hosts to migrate, disperse and adapt to changing local environments, host resistance to disease, biotic interactions, evolutionary change, other anthropogenic alternations of environment, and efforts of disease control [
25,
52]. Future models addressing how these factors will affect the ecology of WNV are critically needed. In addition, climate change predictions are themselves subject to uncertainty in terms of the magnitude and scale of physical and socioeconomic drivers, which will need to be addressed to more accurately predict changes in the ecology of vectors and vector-borne diseases. Finally, the precise lag time of habitat change (
i.e., from boreal forest to grassland) and subsequent dispersal of vectors and hosts to newly suitable habitat remain unclear. Therefore, in order to validate our predictions and improve the predictive ability of these models, further monitoring of distribution and abundance of
Cx. tarsalis and WNV is recommended, especially in regions that we have identified as vulnerable to range expansion and enhanced endemic amplification within the next 20–100 years.