Application of Solid Phase Extraction on Multiwalled Carbon Nanotubes of Some Heavy Metal Ions to Analysis of Skin Whitening Cosmetics Using ICP-AES
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instrument
2.2. Reagent and Standard Solution
| RF generator power (w) | 1,150 W |
| Coolant gas flow rate | 12 L·min−1 |
| Auxiliary gas | 0.5 L·min−1 |
| Pump rate | 25 rpm |
| Plasma view | Axial |
| Number of measurements | 3 |
| Analytical Wavelengths (nm) | As (189.00); Bi (223.00) |
| Cd (228.80 ); Pb (220.30) | |
| Hg (184.95); Ti (334.90) |
2.3. Collection of Samples
2.4. Preparation of Microcolumn
2.5. Sample Preparations
2.5.1. Preparations of Standard Solution
2.5.2. Application to Microwave Digested Samples
| Step | (min) Time | (W) Power | Temp. (°C) |
|---|---|---|---|
| 1 | 15 | 450 | 195 |
| 2 | 2.0 | 0 | 190 |
| 3 | 10 | 300 | 195 |
| 4 | 15 | 350 | 195 |
2.5.3. General Preconcentration Procedure
3. Results and Discussion
3.1. Effect of pH on Adsorption
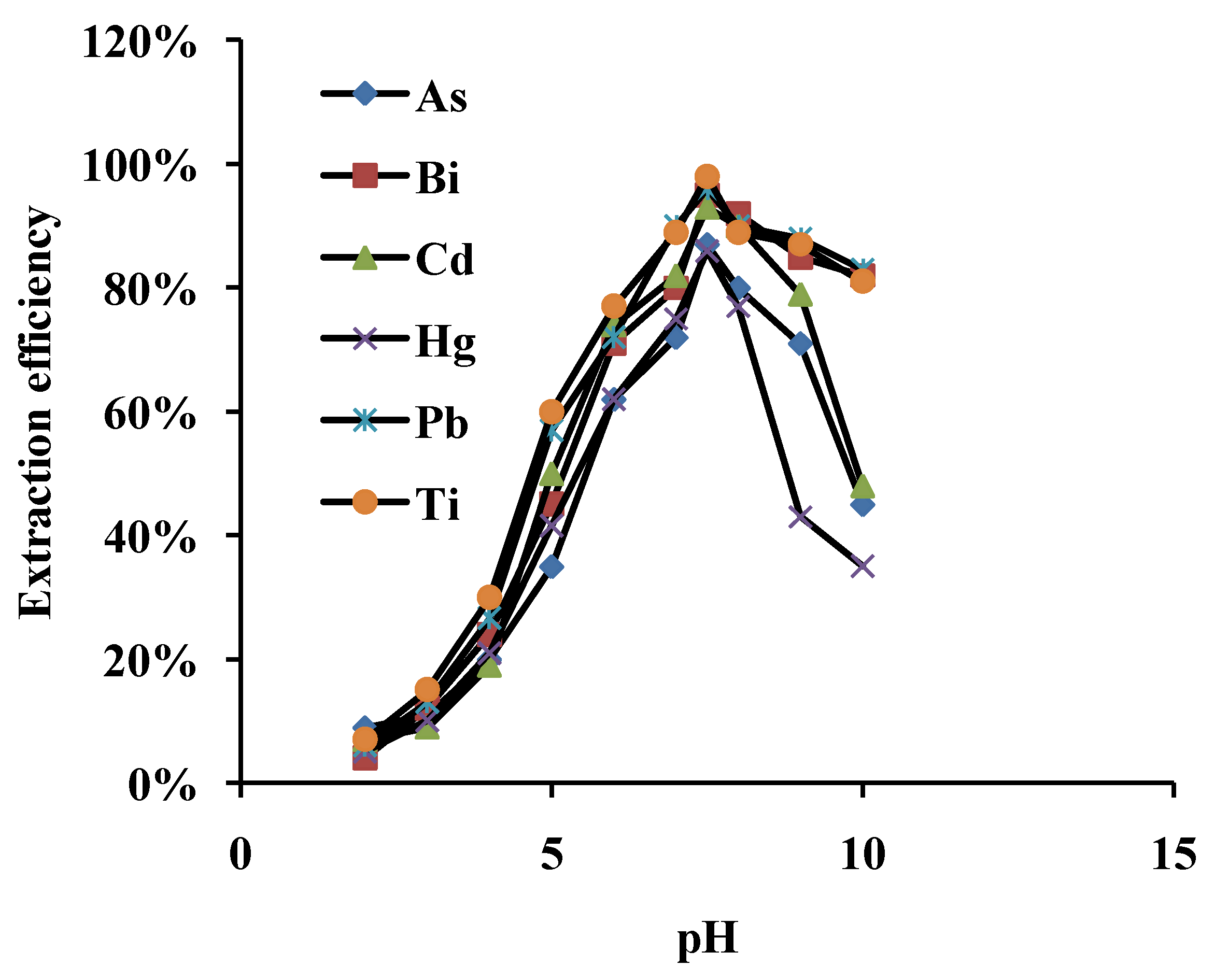
3.2. Effect of Sample Flow Rate
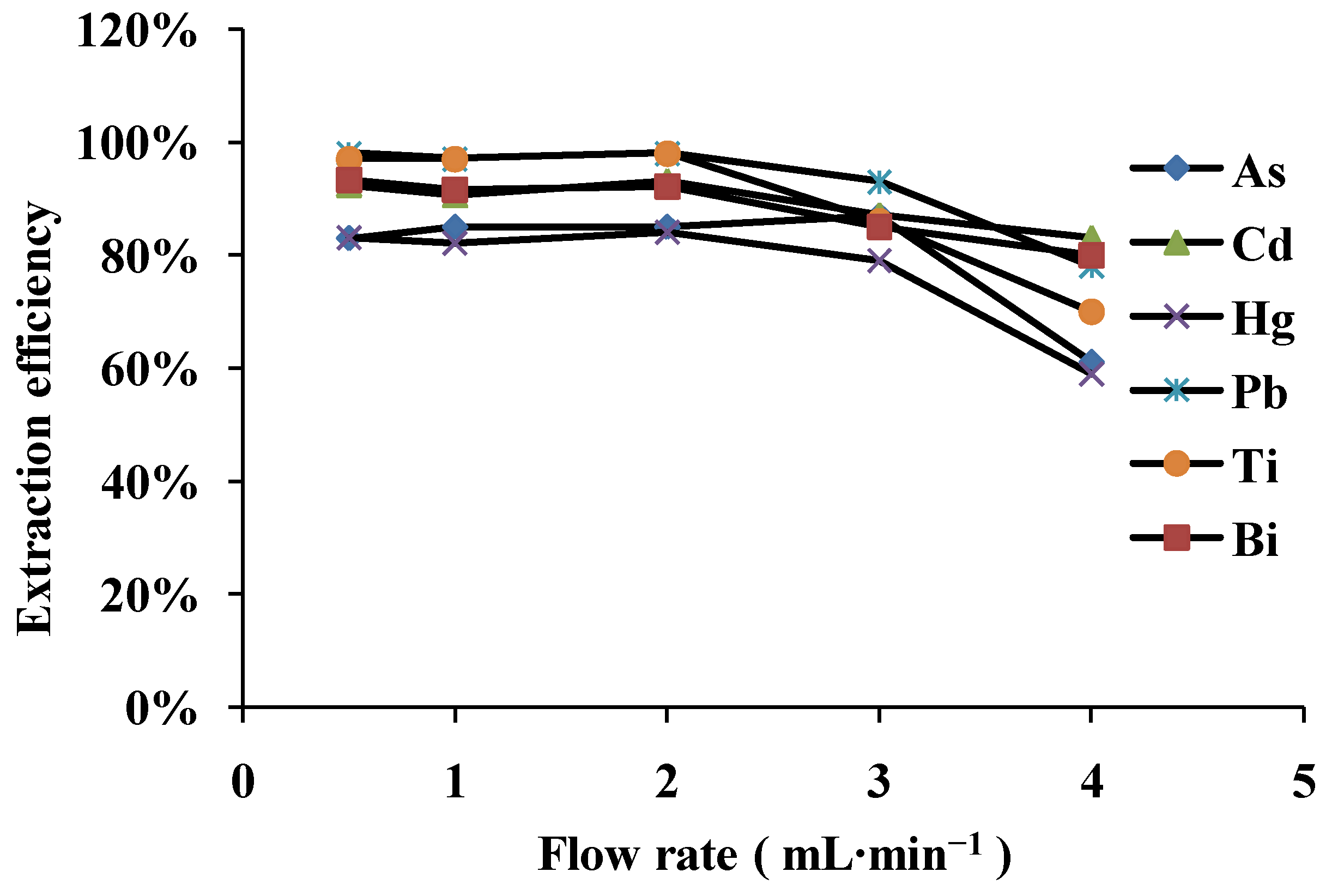
3.3. The Optimization of Elution Conditions
3.3.1. Type of Eluent
| Type of eluent | Extraction efficiency% | |||||
|---|---|---|---|---|---|---|
| As | Bi | Cd | Hg | Pb | Ti | |
| HCl 0.5 mol·L−1 | 20.2 | 25.4 | 18.1 | 13.2 | 21.5 | 31 |
| HNO3 0.5 mol·L−1 | 88.0 | 90.0 | 78.0 | 65.3 | 73.3 | 83 |
| HCl 1.0 mol·L−1 | 29.2 | 33.0 | 35.0 | 29.0 | 40.8 | 85 |
| HNO3 1.0 mol·L−1 | 92.0 | 103 | 93.3 | 90.1 | 96.3 | 95 |
3.3.2. Concentration of Eluent and Volume

| Volume of HNO3 eluent | Extraction efficiency | |||||
|---|---|---|---|---|---|---|
| As | Bi | Cd | Hg | Pb | Ti | |
| 1 mL | 87.0 | 83.0 | 90.0 | 88.0 | 90.8 | 91.0 |
| 2 mL | 92.0 | 103 | 93.3 | 90.1 | 96.3 | 95.0 |
| 3 mL | 92.6 | 97.0 | 92.0 | 93.0 | 93.6 | 95.0 |
| 4 mL | 92.3 | 98.5 | 91.8 | 89.7 | 93.0 | 94.2 |
3.3.3. Flow Rate
3.4. Adsorption Capacity

3.5. Column Reuse
3.6. Applications of the Presented Procedure
| Element | NIST SRM 1570a Spinach leaves (mg·kg−1) a | |
|---|---|---|
| Certified value | Our value | |
| As | 0.068 ± 0.012 | 0.070 ± 0.009 |
| Cd | 2.89 ± 0.070 | 2.790 ± 0. 1 |
| Hg | 0.030 ± 0.003 | 0.028 ± 0.007 |
| Pb | 0.2 | 0.192 ± 0.02 |
3.7. Performance of the Presented Procedure
| Metal | Added | Yang Chin sample | Magical mix sample | Nivea Lotion sample | |||
|---|---|---|---|---|---|---|---|
| (μg) | Found (μg) | Recovery (%) | Found (μg) | Recovery (%) | Found (μg) | Recovery (%) | |
| As | 0 | 0.987 | - | 0.810 | - | 0.825 | - |
| 2.5 | 3.250 | 90.5 | 3.110 | 92.0 | 3.090 | 90.6 | |
| Bi | 0 | BDL | - | 26.630 | - | 0.412 | - |
| 2.5 | 2.610 | 104.4 | 29.200 | 103.0 | 2.920 | 100.3 | |
| Cd | 0 | 0.218 | - | 0.041 | - | 0.055 | - |
| 2.5 | 2.510 | 91.7 | 2.600 | 102.0 | 2.310 | 90.2 | |
| Hg | 0 | BDL | - | 41.720 | - | 1.200 | - |
| 2.5 | 2.250 | 90.0 | 43.980 | 90.4 | 3.440 | 89.6 | |
| Pb | 0 | 23.880 | - | 21.370 | - | 77.360 | - |
| 2.5 | 26.200 | 93.0 | 23.650 | 91.2 | 79.950 | 103.6 | |
| Ti | 0 | 16.080 | - | 72.890 | - | 0.431 | - |
| 2.5 | 18.450 | 95.0 | 75.300 | 96.4 | 2.900 | 98.8 | |
3.7.1. Detection Limits and Precision
3.8. Analytical Ions in Real Sample
| Adsorbent | Method | Element | D. L. (ng·mL−1) | RSD (%) | Reference |
|---|---|---|---|---|---|
| Multiwalled carbon nanotubes | ICP-AES | Cd, Pb | 0.30, 1.80 | 1.63, 1.90 | This work |
| Amberlite XAD-4 resin coated with dithiocarbamates | ICP-AES | Cd, Pb | 0.70, 1.70 | 2.56, 3.03 | [14] |
| Multiwalled carbon nanotubes | AAS | Pb | 8.00 | 2.50 | [11] |
| Amberlite XAD copolymer resins | FAAS | Cd, Pb | 1.80, 6.31 | 0.14–20.36 | [15] |
| Multiwalled carbon nanotubes | FAAS | Pb | 1.00 | 4.62 | [10] |
| modified silica gel | ICP-AES | Pb | 1.23 | 3.50 | [16] |
| modified Activated Carbon | ICP-AES | Pb | 0.36 | 1.90 | [17] |
| n | As concentration (* mean ± #SD) | Bi concentration (* mean ± #SD) | Cd concentration (* mean ± #SD) | Hg concentration (* mean ± #SD) | Pb concentration (* mean ± #SD) | Ti concentration (* mean ± #SD) |
|---|---|---|---|---|---|---|
| 1 | 12.12 ± 0.04 | 1.45 ± 0.02 | 0.675 ± 0.008 | 1.70 ± 0.018 | 21.36 ± 0.150 | 11.27 ± 0.008 |
| 2 | 2.40 ± 0.05 | 273.45 ± 8.00 | 0.180 ± 0.007 | BDL | 252.00 ± 7.500 | 1,086.00 ± 15.00 |
| 3 | 4.49 ± 0.04 | 2.68 ± 0.03 | BDL | BDL | 219.16 ± 1.250 | 2.52 ± 0.080 |
| 4 | 1.28 ± 0.02 | 4.39± 0.01 | 1.100± 0.010 | 2.31 ± 0.007 | 102.30 ± 0.200 | 86.04 ± 0.470 |
| 5 | 3.15 ± 0.07 | BDL | 0.600 ± 0.007 | 9.66 ± 0.080 | 445.50 ± 2.250 | 1,025.20 ± 150 |
| 6 | 6.80 ± 0.09 | BDL | BDL | 22.78 ± 0.085 | 280.80 ± 1.610 | 204.10 ± 1.700 |
| 7 | 2.89 ± 0.08 | 11,842.50 ± 30.00 | 0.189 ± 0.009 | 2,745.00 ± 8.100 | 78.60 ± 0.150 | 56.30 ± 0.240 |
| 8 | 8.25 ± 0.14 | 2.12 ± 0.024 | 0.555 ± 0.008 | 24.03 ± 0.150 | 386.80 ± 3.750 | 11.25 ± 0.150 |
| 9 | 3.10 ± 0.03 | BDL | 0.320 ± 0.002 | 8.500 ± 0.052 | 279.90 ± 1.800 | 856.80 ± 4.000 |
| 10 | 3.09 ± 0.06 | BDL | 0.832 ± 0.008 | 10.21 ± 0.160 | 693.00 ± 0.520 | 299.60 ± 1.050 |
| 11 | 0.84 ± 0.01 | 2.25 ± 0.016 | 5.220 ± 0.026 | 1.00 ± 0.036 | 5.52 ± 0.046 | 2.78 ± 0.044 |
| 12 | 2.78 ± 0.09 | 59.43 ± 0.216 | BDL | 1.289 ± 0.018 | 109.50 ± 0.180 | 110.43 ± 0.540 |
| 13 | 11.71 ± 0.06 | 2.02 ± 0.052 | 0.500 ± 0.002 | BDL | 632.60 ± 2.250 | 3.33 ± 0.037 |
| 14 | 2.37 ± 0.01 | 0.68 ± 0.018 | 0.720± 0.003 | 1.77 ± 0.022 | 40.50 ± 1.280 | 3.53 ± 0.025 |
| 15 | 1.98 ± 0.03 | BDL | 0.171 ± 0.009 | 29.63 ± 0.160 | 288.45 ± 1.350 | 1,865.60 ± 19.200 |
| 16 | BDL | BDL | 0.720 ± 0.008 | BDL | 8.00 ± 0.080 | 3.73 ± 0.016 |
| 17 | 6.14 ± 0.07 | 1,959.20 ± 5.600 | 0.243 ± 0.001 | 2.10 ± 0.008 | BDL | 2.62 ± 0.016 |
| 18 | 0.34 ± 0.01 | 1.83 ± 0.025 | 0.76 ± 0.001 | BDL | 5.52 ± 0.170 | 7.65 ± 0.086 |
| 19 | 15.36 ± 0.12 | 13.81 ± 0.400 | 0.825 ± 0.007 | 1.95 ± 0.015 | 6.75 ± 0.050 | 2.90 ± 0.025 |
| 20 | 9.87 ± 0.07 | BDL | 2.060 ± 0.007 | BDL | 238.80 ± 0.900 | 160.80 ± 1.760 |
| 21 | 8.08 ± 0.05 | 266.40 ± 5.060 | 0.409 ± 0.010 | 417.16 ± 9.450 | 299.20 ± 1.680 | 728.96 ± 4.460 |
| 22 | 5.78 ± 0.12 | 383.00 ± 3.250 | 0.330 ± 0.010 | 4.34 ± 0.090 | 9.87 ± 0.197 | 310.00 ± 2.550 |
| 23 | 1.68 ± 0.04 | BDL | 0.500 ± 0.010 | 3.33 ± 0.033 | 21.56 ± 0.1950 | 228.10 ± 2.500 |
| 24 | 1.44 ± 0.01 | BDL | 0.150 ± 0.015 | BDL | 794.25 ± 9.750 | 841.20 ±6.000 |
| 25 | 14.76 ± 0.10 | 47.06 ± 2.740 | 0.082 ± 0001 | 2.60 ± 0.037 | 313.00 ± 1.700 | 43.74 ± 1.560 |
| 26 | 7.81 ± 0.05 | 847.50 ± 8.900 | 0.831 ± 0.023 | BDL | 106.30 ±1.300 | 678.30 ± 7.120 |
| 27 | 8.52 ± 0.15 | BDL | 0.520 ± 0.009 | BDL | 9.60 ± 0.312 | 2,749.00 ± 53.130 |
| 28 | 1.99 ± 0.03 | BDL | 0.328 ± 0.008 | 16.49 ± 0.024 | 114.70 ± 0.560 | 2,262.40 ± 9.600 |
| 29 | 5.54 ± 0.16 | BDL | BDL | 151.95 ± 0.450 | BDL | 37.80 ± 0.075 |
| 30 | 2.41 ± 0.03 | 59.95 ± 0.140 | 0.120 ± 0.001 | 9.68 ± 0.080 | 196.92 ± 0.490 | 241.10 ± 1.680 |
| 31 | 8.70 ± 0.05 | BDL | 0.232 ± 0020 | BDL | 116.70 ± 0.150 | 652.50 ± 3.370 |
| 32 | 2.18 ± 0.09 | BDL | 0.285 ± 0.008 | 0.63 ± 0.020 | 23.50 ± 0.220 | 352.05 ± 6.520 |
| 33 | 3.55 ± 0.08 | BDL | BDL | 5.21± 0.100 | 141.64 ± 0.440 | 437.48 ± 2.960 |
| 34 | 2.06 ± 0.020 | 0.61 ± 0.020 | 0.090 ± 0.001 | 9.39 ± 0.060 | 3.37 ± 0.010 | 9.00 ± 0.075 |
| Heavy metal | Concentration range (µg·g−1) | Ref. |
|---|---|---|
| As, Bi, Cd, | (0.34–15.36), (0.615–1,1842.5), (0.09–5.2), | This work |
| Hg, Pb, Ti | (0.637–2,745), (3.37–794.25), (2.52–2749) | |
| Hg | (878–36,000) | [18] |
| Hg | (2.46–23,222) | [19] |
| Hg | (660–57,000) | [20] |
| Pb | (87–123) | [21] |
| Hg, As, Cd, Pb | (0.04–2.183), (0.690–3.683), (0.625–1.875), (1.470–33.1) | [7] |
| Hg, As | (5,700–126,000), (522–161,600) | [22] |
4. Conclusions
Acknowledgements
References
- Chisvert, A.; Sisternes, J.; Balaguer, A.; Salvador, A. A gas chromatography-mass spectrometric method to determinate skin-whitening agents in cosmetic products. Talanta 2010, 81, 530–536. [Google Scholar] [CrossRef]
- Balaguer, A.; Salvador, A.; Chisvert, A. A rapid and reliable size-exclusion chromatographic method for determination of kojicdipalmitate in skin-whitening cosmetic products. Talanta 2008, 75, 407–411. [Google Scholar] [CrossRef]
- Al-Saleh, I.; Shinwari, N.; El-Doush, G.; Billedo, F. Comparison of mercury levels in various tissues of albino and pigmented mice treated with two different brands of mercury skin lightening creams. BioMetals 2004, 17, 167–175. [Google Scholar] [CrossRef]
- Al-Saleh, I.; Shinwari, N.; Al-Amodi, M. Accumulation of mercury in ovaries of mice after the application of skin-lightening creams. Biol. Trace Elem. Res. 2009, 131, 43–54. [Google Scholar] [CrossRef]
- Uram, E.; Bischofer, B.P.; Hagermann, S. Market Analysis of Some Mercury-Containing Products and Their Mercury-Free Alternatives in Selected Regions; Gesellschaft für Anlagenund Reaktoricherheit (GRS) mbH: Berlin, Germany, 2010; pp. 1–127. [Google Scholar]
- Al-Saleh, I.; Al-Doush, I. Mercury content in skin-lightening cream and potential hazards to the health of Saudi women. J. Toxicol. Environ. Health 1997, 51, 123–130. [Google Scholar]
- Sukender, K.; Jaspreet, S.; Sneha, D.; Munish, G. AAS estimation of heavy metals and trace elements in Indian herbal cosmetic preparations. Res. J. Chem. Sci. 2012, 2, 46–51. [Google Scholar]
- Li, Y.-H.; Wang, S.; Luan, Z.; Ding, J.; Xu, C.; Wu, D. A adsorption of cadmium(II) from aqueous solution by surface oxidized carbon nanotubes. Carbon 2003, 41, 1057–1062. [Google Scholar]
- Chen, S.; Liu, C.; Yang, M.; Lu, D.; Zhu, L.; Wang, Z. Solid-phase extraction of Cu, Co and Pb on oxidized single-walled carbon nanotubes and their determination by inductively coupled plasma mass spectrometry. J. Hazard. Mater. 2009, 170, 247–251. [Google Scholar] [CrossRef]
- Zhao, X.; Song, N.; Jia, Q.; Zhou, W. Determination of Cu, Zn, Mn, and Pb by microcolumn packed with multiwalled carbon nanotubes on-line coupled with flame atomic absorption spectrometry. Microchim. Acta 2009, 166, 329–335. [Google Scholar] [CrossRef]
- Ozcan, S.; Satiroglu, N.; Soylak, M. Column solid phase extraction of iron(III), copper(II), manganese(II) and lead(II) ions food and water samples on multiwalled carbon nanotubes. Food Chem. Toxicol. 2010, 48, 2401–2406. [Google Scholar] [CrossRef]
- Cai, Y.; Jiang, G.; Liu, J.; Zhou, Q. Multiwalled carbon nanotubes as a solid phase extraction adsorbent for the determination of bisphenol A, 4-n-nonylphenol, and 4-tert-octylphenol. Anal. Chem. 2003, 75, 2517–2521. [Google Scholar] [CrossRef]
- Chen, S.; Xiao, M.; Lu, D.; Wang, Z. The use of carbon nanofibers microcolumn preconcentration for inductively coupled plasma mass spectrometry determination of Mn, Co and Ni. Spectrochimica Acta B 2007, 62, 1216–1221. [Google Scholar]
- Ramesh, A.; Mohan, K.R.; Seshaiah, K. Preconcentration of trace metals on Amberlite XAD-4 resin coated with dithiocarbamates and determination by inductively coupled plasma-atomic emission spectrometry in saline matrices. Talanta 2002, 57, 243–252. [Google Scholar] [CrossRef]
- Derya, K.; Andrew, F.; Steve, J. Determination of trace heavy metals in soil and sediments by atomic spectrometry following preconcentration with Schiff bases on Amberlite XAD-4. J. Hazard. Mater. 2009, 165, 1165–1169. [Google Scholar] [CrossRef]
- Zhang, L.; Zhai, Y.; Chang, X.; He, Q. Determination of trace metals in natural samples by ICP-OES after preconcentration on modified silica gel and on modified silica nanoparticles. Microchim. Acta 2009, 165, 319–327. [Google Scholar] [CrossRef]
- Tu, Z.; He, Q.; Chang, X.; Hu, Z.; Gao, R. 1-(2-Formamidoethyl)-3-phenylurea functionalized activated carbon for selective solid-phase extraction and preconcentration of metal ions. Analytica Chimica Acta 2009, 649, 252–257. [Google Scholar] [CrossRef]
- Peregrino, C.P.; Moreno, M.V.; Miranda, S.V.; Alma, D.; Rubio, O. Levels mercury levels in locally manufactured Mexican skin-lightening Creams. Int. J. Environ. Res. Public Health 2011, 8, 2516–2523. [Google Scholar] [CrossRef]
- I-Ashban, R.M.; Barratt, D.A.; Shah, A.H. Mercury contents of skin lightening creams market in Saudi Arabia. J. Saudi Chem. Soc. 2006, 10, 383–388. [Google Scholar]
- Sin, K.W.; Tsang, H.F. Large-scale mercury exposure due to a cream cosmetic, community wide case series. Hong Kong Med. J. 2003, 9, 29–34. [Google Scholar]
- Nnorom, I.C.; Gwf, J.C.; Nnorom, C.G. Trace metal contents of facial (make-up) cosmetics commonly used in Nigeria. Afr. J. Biotechnol. 2005, 4, 1133–1138. [Google Scholar]
- Bogusz, M.J.; Tufail, M.; Hassan, H. How natural are “natural herbal remedies”? A Saudi perspective. Adverse Drug React. Toxicol. Rev. 2002, 21, 219–229. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
ALqadami, A.A.; Abdalla, M.A.; ALOthman, Z.A.; Omer, K. Application of Solid Phase Extraction on Multiwalled Carbon Nanotubes of Some Heavy Metal Ions to Analysis of Skin Whitening Cosmetics Using ICP-AES. Int. J. Environ. Res. Public Health 2013, 10, 361-374. https://doi.org/10.3390/ijerph10010361
ALqadami AA, Abdalla MA, ALOthman ZA, Omer K. Application of Solid Phase Extraction on Multiwalled Carbon Nanotubes of Some Heavy Metal Ions to Analysis of Skin Whitening Cosmetics Using ICP-AES. International Journal of Environmental Research and Public Health. 2013; 10(1):361-374. https://doi.org/10.3390/ijerph10010361
Chicago/Turabian StyleALqadami, Ayoub A., Mohammad Abulhassan Abdalla, Zeid A. ALOthman, and Kamal Omer. 2013. "Application of Solid Phase Extraction on Multiwalled Carbon Nanotubes of Some Heavy Metal Ions to Analysis of Skin Whitening Cosmetics Using ICP-AES" International Journal of Environmental Research and Public Health 10, no. 1: 361-374. https://doi.org/10.3390/ijerph10010361




