Characterization of a Novel Serine Protease Inhibitor Gene from a Marine Metagenome
Abstract
:1. Introduction
2. Results and Discussion
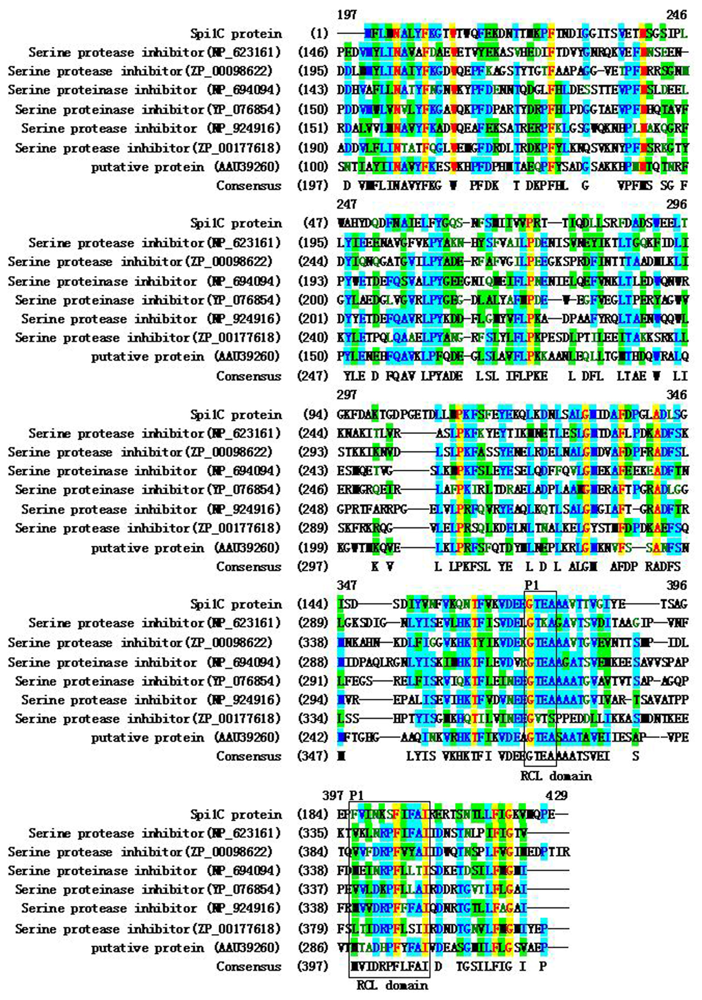
2.1. Cloning and Sequence Analysis of a Novel Serpin Gene
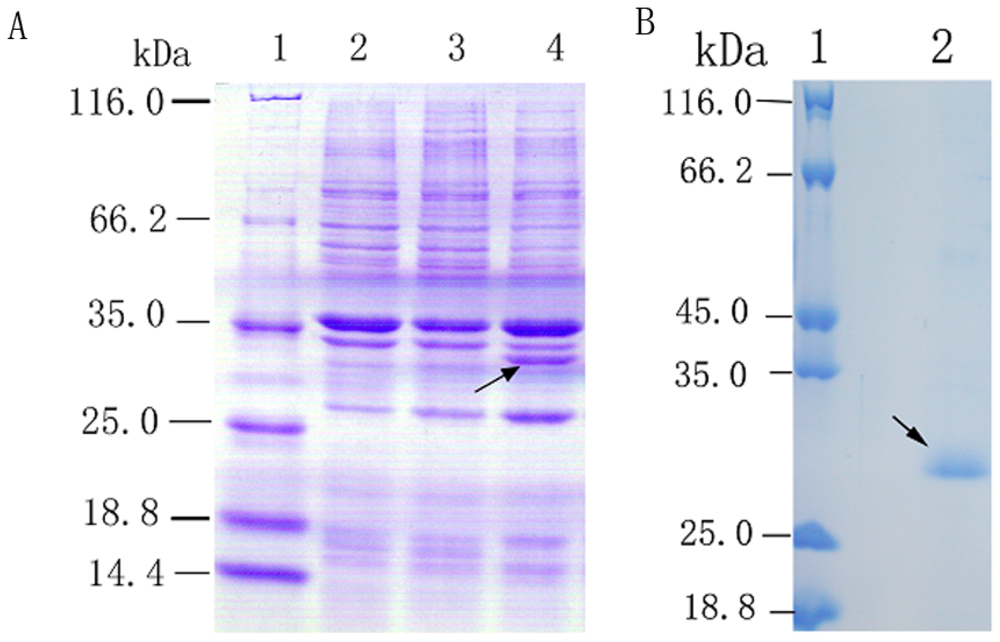
2.2. Overexpression and Purification of Recombinant Spi1C Protein
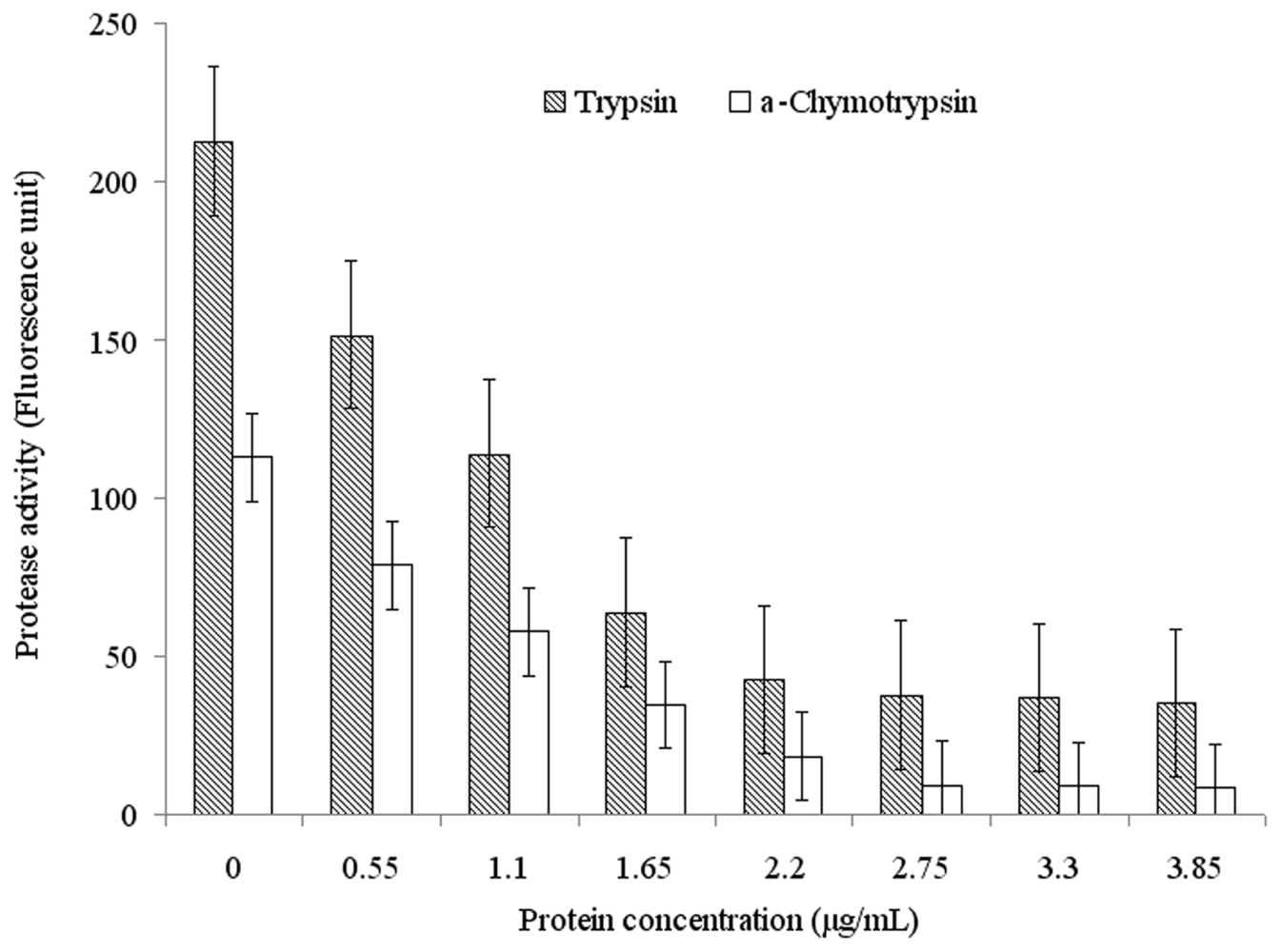
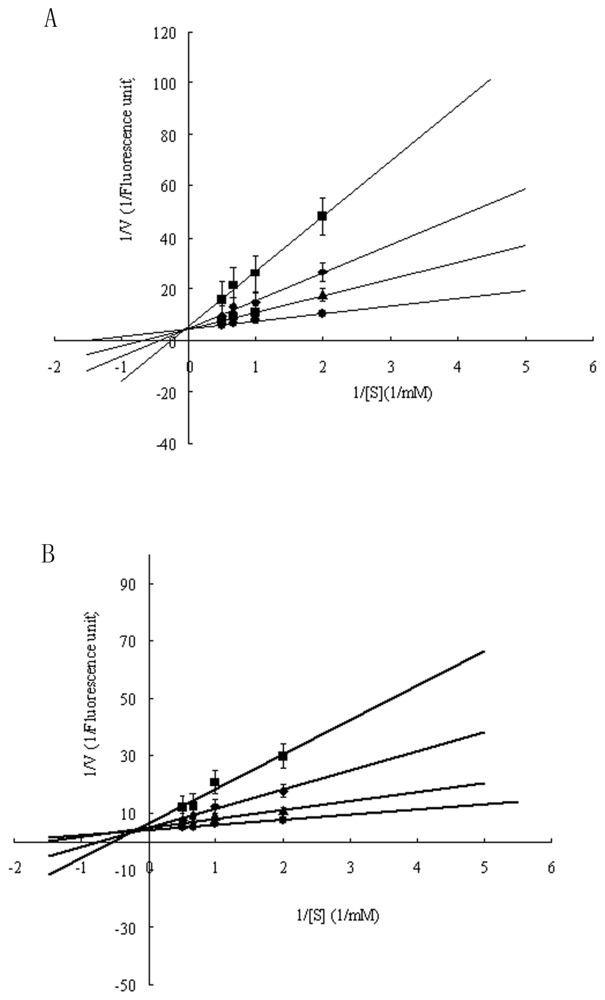
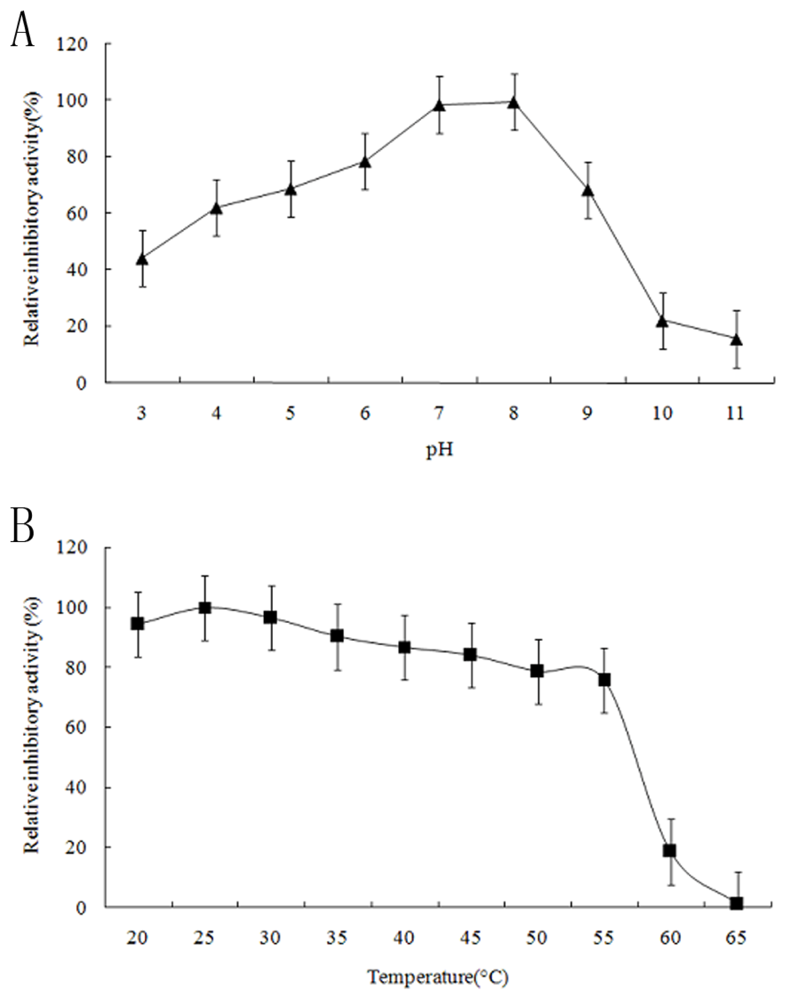
2.3. Functional Characterization of Recombinant Spi1C Protein
3. Experimental Section
3.1. Plasmid Vectors, Bacterium Strains, and Growth Conditions
3.2. DNA Manipulation and Protein Analysis
3.3. DNA Sequence Analysis, Database Search, and Gene Structure Characterization
3.4. Overexpression and Purification of the Recombinant Serpin Protein
3.5. Characterization of the Serpin Gene Spi1C
3.5.1. Elastase Inhibition Assay
3.5.2. α-Chymotrypsin Inhibition Assay
3.5.3. Trypsin Inhibition Assay
3.6. Nucleotide Sequence Accession Number
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Gettins, P. Serpin structure, mechanism, and function. Chem. Rev 2002, 102, 4751–4803. [Google Scholar]
- Silverman, GA; Whisstock, JC; Bottomley, SP; Huntington, JA; Kaiserman, D; Luke, CJ; Pak, SC; Reichhart, JM; Bird, PI. Serpins flex their muscle: Putting the clamps on proteolysis in diverse biological systems. J. Biol. Chem 2010, 285, 24299–24305. [Google Scholar]
- Gooptu, B; Lomas, DA. Conformational pathology of the serpins: Themes, variations, and therapeutic strategies. Annu. Rev. Biochem 2009, 78, 147–176. [Google Scholar]
- Belorgey, D; Hägglöf, P; Karlsson-Li, S; Lomas, DA. Protein misfolding and the serpinopathies. Prion 2007, 1, 15–20. [Google Scholar]
- Gettins, PGW; Olson, ST. Exosite determinants of serpin specificity. J. Biol. Chem 2009, 284, 20441–20445. [Google Scholar]
- Davis, AE, III; Mejia, P; Lu, F. Biological activities of C1 inhibitor. Mol. Immunol 2008, 45, 4057–4063. [Google Scholar]
- Dementiev, A; Dobo, J; Gettins, PGW. Active-site distortion is sufficient for proteinase inhibition by serpins. J. Biol. Chem 2006, 281, 3452–3457. [Google Scholar]
- Quan, LT; Caputo, A; Bleackley, RC; Pickup, DJ; Salvesen, GS. Granzyme B is inhibited by the cowpox virus serpin cytokine response modifier A. J. Biol. Chem 1995, 270, 10377–10379. [Google Scholar]
- Schick, C; Pemberton, PA; Shi, GP; Kamachi, Y; Cataltepe, S; Bartuski, AJ; Gornstein, ER; Brömme, D; Chapman, HA; Silverman, GA. Cross-class inhibition of the cysteine proteinases cathepsins K, L, and S by the serpin squamous cell carcinoma antigen 1: A kinetic analysis. Biochemistry 1998, 37, 5258–5266. [Google Scholar]
- Scheidig, AJ; Hynes, TR; Pelletier, LA; Wells, JA; Kossiakoff, AA. Crystal structures of bovine chymotrypsin and trypsin complexed to the inhibitor domain of Alzheimer’s amyloid beta-protein precursor (APPI) and basic pancreatic trypsin inhibitor (BPTI): Engineering of inhibitors with altered specificities. Protein Sci 1997, 6, 1806–1824. [Google Scholar]
- Nagahara, A; Nakayama, M; Oka, D; Tsuchiya, M; Kawashima, A; Mukai, M; Nakai, Y; Takayama, H; Nishimura, K; Jo, Y; Nagai, A; Okuyama, A; Nonomura, N. SERPINE2 is a possible candidate promotor for lymph node metastasis in testicular cancer. Biochem. Biophys. Res. Commun 2010, 391, 1641–1646. [Google Scholar]
- Kennedy, J; Flemer, B; Jackson, SA; Lejon, DP; Morrissey, JP; O’Gara, F; Dobson, AD. Marine metagenomics: New tools for the study and exploitation of marine microbial metabolism. Mar. Drugs 2010, 8, 608–628. [Google Scholar]
- Newton, RJ; Griffin, LE; Bowles, KM; Meile, C; Gifford, S; Givens, CE; Howard, EC; King, E; Oakley, CA; Reisch, CR; Rinta-Kanto, JM; Sharma, S; Sun, S; Varaljay, V; Vila-Costa, M; Westrich, JR; Moran, MA. Genome characteristics of a generalist marine bacterial lineage. ISME J 2010, 4, 784–798. [Google Scholar] [Green Version]
- Johnson, DJ; Langdown, J; Huntington, JA. Molecular basis of factor IXa recognition by heparin-activated antithrombin revealed by a 1.7-A structure of the ternary complex. Proc. Natl. Acad. Sci. USA 2010, 107, 645–650. [Google Scholar]
- Jean, F; Stella, K; Thomas, L; Liu, G; Xiang, Y; Thomas, G. alpha1-Antitrypsin Protland, a bioengineered serpin highly selective for furin: Application as an antipathogenic agent. Proc. Natl. Acad. Sci. USA 1998, 95, 7293–7298. [Google Scholar]
- Jiang, C; Wu, LL; Zhao, GC; Shen, PH; Jin, K; Hao, ZY; Li, SX; Ma, GF; Luo, FF; Hu, GQ; Kang, WL; Bi, YL; Qin, XM; Tang, XL; Wu, B. Identification and characterization of a novel fumarase gene by metagenome expression cloning from marine microorganisms. Microb. Cell Fact 2010, 9, 91. [Google Scholar]
- Yooseph, S; Nealson, KH; Rusch, DB; McCrow, JP; Dupont, CL; Kim, M; Johnson, J; Montgomery, R; Ferriera, S; Beeson, K; Williamson, SJ; Tovchigrechko, A; Allen, AE; Zeigler, LA; Sutton, G; Eisenstadt, E; Rogers, YH; Friedman, R; Frazier, M; Venter, JC. Genomic and functional adaptation in surface ocean planktonic prokaryotes. Nature 2010, 468, 60–66. [Google Scholar]
- Singh, BK. Exploring microbial diversity for biotechnology: The way forward. Trends Biotechnol 2010, 28, 111–116. [Google Scholar]
- Edwards, JL; Smith, DL; Connolly, J; McDonald, JE; Cox, MJ; Joint, I; Edwards, C; McCarthy, AJ. Identification of carbohydrate metabolism genes in the metagenome of a marine biofilm community shown to be dominated by gammaproteobacteria and bacteroidetes. Genes 2010, 1, 371–384. [Google Scholar]
- Torres-Castillo, JA; Jacobo, CM; Blanco-Labra, A. Characterization of a highly stable trypsin-like proteinase inhibitor from the seeds of Opuntia streptacantha (O. streptacantha Lemaire). Phytochemistry 2009, 70, 1374–1381. [Google Scholar]
- Yang, X; Wang, Y; Lu, Z; Zhai, L; Jiang, J; Liu, J; Yu, H. A novel serine protease inhibitor from the venom of Vespa bicolor Fabricius. Comp. Biochem. Physiol. B Biochem. Mol. Biol 2009, 153, 116–120. [Google Scholar]
- Lu, J; Yang, H; Yu, H; Gao, W; Lai, R; Liu, J; Liang, X. A novel serine protease inhibitor from Bungarus fasciatus venom. Peptides 2008, 29, 369–374. [Google Scholar]
- Laskowski, M, Jr; Kato, I. Protein inhibitors of proteinases. Annu. Rev. Biochem 1980, 49, 593–626. [Google Scholar]
- Bode, W; Huber, R. Natural protein proteinase inhibitors and their interaction with proteinases. Eur. J. Biochem 1992, 204, 433–451. [Google Scholar]
- Cheng, YC; Yan, FJ; Chang, LS. Taiwan cobra chymotrypsin inhibitor: Cloning, functional expression and gene organization. Biochim. Biophys. Acta 2005, 1747, 213–220. [Google Scholar]
- Irving, JA; Pike, RN; Lesk, AM; Whisstock, JC. Phylogeny of the serpin superfamily: Implications of patterns of amino acid conservation for structure and function. Genome Res 2000, 10, 1845–1864. [Google Scholar]
- Olson, ST; Gettins, PG. Regulation of proteases by protein inhibitors of the serpin superfamily. Prog. Mol. Biol. Transl. Sci 2011, 99, 185–240. [Google Scholar]
- Sambrook, J; Russell, DW. Molecular Cloning: A Laboratory Manual, 3rd ed; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, Suffolk, NY, USA, 2001. [Google Scholar]
- Schägger, H; von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem 1987, 166, 368–379. [Google Scholar]
- ExPASy Translate. Available online: http://www.expasy.org/tools/dna.html accessed on 20 August 2010.
- NCBI Entres. Available online: http://www.ncbi.nlm.nih.gov/Entrez/ accessed on 10 August 2010.
- Tamura, K; Dudley, J; Nei, M; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol 2007, 24, 1596–1599. [Google Scholar]
- Barrett, AJ. A new assay for cathepsin B1 and other thiol proteinases. Anal. Biochem 1972, 47, 280–293. [Google Scholar]
- Kang, SH; Fuchs, MS. An improvement in the Hummel chymotrypsin assay. Anal. Biochem 1973, 54, 262–265. [Google Scholar]
- Ahsan, MN; Watabe, S. Kinetic and structural properties of two isoforms of trypsin isolated from the viscera of Japanese anchovy, Engraulis japonicus. J. Protein Chem 2001, 20, 49–58. [Google Scholar]
- Kristiansson, E; Hugenholtz, P; Dalevi, D. ShotgunFunctionalizeR: An R-package for functional comparison of metagenomes. Bioinformatics 2009, 25, 2737–2738. [Google Scholar]

© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jiang, C.-J.; Hao, Z.-Y.; Zeng, R.; Shen, P.-H.; Li, J.-F.; Wu, B. Characterization of a Novel Serine Protease Inhibitor Gene from a Marine Metagenome. Mar. Drugs 2011, 9, 1487-1501. https://doi.org/10.3390/md9091487
Jiang C-J, Hao Z-Y, Zeng R, Shen P-H, Li J-F, Wu B. Characterization of a Novel Serine Protease Inhibitor Gene from a Marine Metagenome. Marine Drugs. 2011; 9(9):1487-1501. https://doi.org/10.3390/md9091487
Chicago/Turabian StyleJiang, Cheng-Jian, Zhen-Yu Hao, Rong Zeng, Pei-Hong Shen, Jun-Fang Li, and Bo Wu. 2011. "Characterization of a Novel Serine Protease Inhibitor Gene from a Marine Metagenome" Marine Drugs 9, no. 9: 1487-1501. https://doi.org/10.3390/md9091487



