Ecological and Physiological Studies of Gymnodinium catenatum in the Mexican Pacific: A Review
Abstract
:1. Introduction
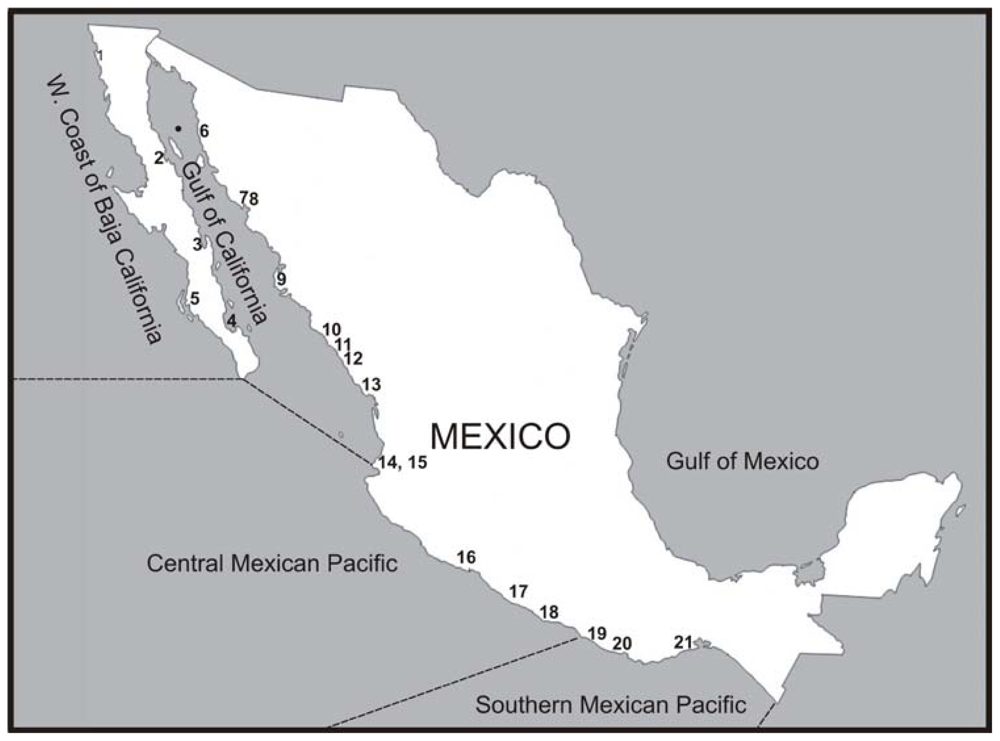
2. Distribution of Gymnodinium catenatum along the Western Coasts of Mexico and Associated Toxic Events
3. Ecological Studies
3.1. Gulf of California
3.2. West Coast of Baja California Peninsula
3.3. Central Mexican Pacific
3.4. Southern Mexican Pacific
4. Grazing Studies
5. Toxicity Studies
5.1. PST in Phytoplankton Samples Related to the Presence of G. catenatum
5.2. Toxin Content and Toxin Profile of G. catenatum S trains
5.3. Presence of PST in Mollusks Linked to HAB of G. catenatum
6. Toxic Effects of G. catenatum on Terrestrial and Marine Organisms or Toxic Effects of G. catenatum on Other Organisms
6.1. Laboratory Studies
6.2. Aquaculture Activities
7. Growth Variations of G. catenatumStrains of the Gulf of California
8. LSUrDNA Sequences
9. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Graham, HW. Gymnodinium catenatum, a new dinoflagellate from the Gulf of California. Trans. Am. Microsc. Soc 1943, 62, 259–261. [Google Scholar]
- Osorio-Tafall, BF. El Mar de Cortés y la productividad fitoplanctónica de sus aguas. Anal. Esc. Nal. Cien. Biol. IPN, Mex 1943, 3, 73–118. [Google Scholar]
- Hallegraeff, GM; Fraga, S. Anderson, DM, Cembella, AD, Hallegraeff, GM, Eds.; Bloom dynamics of the toxic dinoflagellate Gymnodinium catenatum, with emphasis on Tasmanian and Spanish coastal waters. In Physiological Ecology of Harmful Algal Blooms NATO ASI Series; Springer-Verlag: Berlin, Germany, 1998; Volume G41, pp. 59–80. [Google Scholar]
- Oshima, Y; Hasegawa, M; Yasumoto, T; Hallegraeff, G; Blackburn, S. Dinoflagellate Gymnodinium catenatum as the source of paralytic shellfish toxins in Tasmanian shellfish. Toxicon 1987, 25, 1105–1111. [Google Scholar]
- Bagnis, R; Berglund, F; Elias, PS; Van Esch, GJ; Halstead, BW; Kojima, K. Problems of toxicants in marine food products. 1. Marine Biotoxins. Bull. World Health Org 1970, 42, 69–88. [Google Scholar]
- Mee, LD; Espinosa, M; Díaz, G. Paralytic shellfish poisoning with a Gymnodinium catenatum red tide on the Pacific coast of Mexico. Mar. Environ. Res 1986, 19, 77–92. [Google Scholar]
- De la Garza-Aguilar, J. Intoxicación alimentaria por ingestión de mariscos contaminados. Sal. Pub. Mex 1983, 25, 145–150. [Google Scholar]
- Cortés-Altamirano, R; Núñez-Pasten, A. Doce años (1979–1990) de registros de mareas rojas en la Bahía de Mazatlán, Sinaloa, México. An. Inst. Cienc. Mar y Limnol. UNAM 1992, 19, 113–121. [Google Scholar]
- Cortés-Altamirano, R; Núñez-Pastén, A; Pastén-Miranda, N. Abundancia anual de Gymnodinium catenatum Graham dinoflagelado tóxico de la costa Este del Golfo de California. Ciencia y Mar 1999, 50–56. [Google Scholar]
- García-Hernández, J; García-Rico, L; Jara-Marini, ME; Barraza-Guardado, R; Hudson Weaver, A. Concentrations of heavy metals in sediment and organisms during a harmful algal bloom (HAB) at Kun Kaak Bay, Sonora, Mexico. Mar. Poll. Bull 2005, 50, 733–739. [Google Scholar]
- Gárate-Lizárraga, I; Arellano-Martínez, M; Ceballos-Vázquez, BP; Bustillos-Guzmán, JJ; López-Cortés, DJ; Hernández-Sandoval, F. Fitoplancton tóxico y presencia de toxinas paralizantes en la almeja mano de león (Nodipecten subnodosus, Sowerby, 1835) en la Bahía de Los Ángeles, B.C. Resúmenes del II Taller sobre Florecimientos Algales Nocivo, Ensenada, Mexico, 21–23 November; CICESE-CETMAR: Ensenada, Mexico, 2007; p. 20. [Google Scholar]
- Gárate-Lizárraga, I; Hernández-Orozco, ML; Band-Schmidt, CJ; Casillas-Serrano, G. Red tides along the coasts of Baja California Sur, Mexico (1984 to 2001). CICIMAR Oceánides 2001, 16, 127–134. [Google Scholar]
- Gárate-Lizárraga, I; Bustillos-Guzmán, JJ; Alonso-Rodríguez, R; Luckas, B. Comparative paralytic shellfish toxin profiles in two marine bivalves during outbreaks of Gymnodinium catenatum (Dinophyceae) in the Gulf of California. Mar. Poll. Bull 2004, 48, 378–402. [Google Scholar]
- Morquecho, ML; Lechuga-Devéze, CH. Dinoflagellates in recent sediments from Bahía Concepción, Gulf of California. Bot. Mar 2003, 46, 132–141. [Google Scholar]
- Gárate-Lizárraga, I; Bustillos-Guzmán, JJ; Erler, K; Muñetón-Gómez, MS; Luckas, B; Tripp-Quezada, A. Paralytic shellfish toxins in the chocolata clam, Megapitaria squalida (Bivalvia: Veneridae), in Bahía de La Paz, Gulf of California. Rev. Biol. Trop 2004, 52, 133–140. [Google Scholar]
- Gárate-Lizárraga, I; Bustillos-Guzmán, JJ; López-Cortés, DJ; Hernández-Sandoval, F; Erler, K; Luckas, B. Paralytic shellfish toxin profiles in net phytoplankton samples from Bahía Concepción, Gulf of California, Mexico. Mar. Poll. Bull 2006, 52, 800–815. [Google Scholar]
- Band-Schmidt, CJ; Bustillos-Guzmán, J; Gárate-Lizárraga, I; Lechuga-Devéze, CH; Reinhard, K; Luckas, B. Paralytic shellfish toxin profile in strains of the dinoflagellate Gymnodinium catenatum Graham and the scallop Argopecten ventricosus G. B. Sowerby II from Bahía Concepción, Gulf of California, Mexico. Harmful Algae 2005, 4, 21–31. [Google Scholar]
- López-Cortés, DJ; Bustillos-Guzmán, JJ; Gárate-Lizárraga, I. Unusual mortality of Krill (Crustacea: Euphausiacea) in Bahía de La Paz, Gulf of California. Pac. Sci 2006, 60, 235–242. [Google Scholar]
- López-Cortés, DJ; Bustillos-Guzmán, JJ; Gárate-Lizárraga, I; Band-Schmidt, CJ; Hernández-Sandoval, F. Dos eventos de mareas rojas durante el monitoreo de nutrientes en dos granjas atuneras. Resúmenes del II Taller sobre Florecimientos Algales Nocivos, Ensenada, Mexico, 21–23 November; CICESE-CETMAR: Ensenada, Mexico, 2007; p. 15. [Google Scholar]
- Gárate-Lizárraga, I; Band-Schmidt, CJ; López-Cortés, D; Bustillos-Guzmán, JJ; Erler, K. Bloom of Pseudo-nitzschia fraudulenta in Bahía de La Paz, Gulf of California (june-july 2006). Harmful Algae News 2007, 33, 6–7. [Google Scholar]
- Hernández-Sandoval, F; Núñez-Vázquez, EJ; Band-Schmidt, C; López-Cortés, D; Gárate-Lizárraga, I; Bustillos-Guzmán, JJ. Paralytic toxins in bivalve mollusks during a proliferation of Gymodinium catenatumin Bahía de La Paz, Mexico. Hidrobiológica 2009, 19, 245–256. [Google Scholar]
- Gárate-Lizárraga, I; Band-Schmidt, CJ; Grayeb-del Alamo, T. A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008). CICIMAR Oceánides 2009, 24, 15–29. [Google Scholar]
- Cortés-Altamirano, R; Alonso-Rodríguez, R. Mareas rojas durante 1997 en la Bahía de Mazatlán, Sinaloa, México. Ciencias del Mar, Universidad Autónoma de Sinaloa 1997, 15, 31–37. [Google Scholar]
- Alonso-Rodríguez, R; Páez-Osuna, F. Nutrients, phytoplankton and harmful algal blooms in shrimp ponds: a review with special reference to the situation in the Gulf of California. Aquaculture 2003, 219, 317–336. [Google Scholar]
- Mee, LD; Ramírez-Flores, A; Flores-Verdugo, F; González-Frías, F. Coastal upwelling and fertility of the Southern Gulf of California: Impact of the 1982–83 ENSO event. Trop. Ocean-Atmos. Newsletter 1985, 31, 9–10. [Google Scholar]
- Morey-Gaines, G. Gymnodinium catenatum Graham (Dinophyceae): morphology and affinities with armoured forms. Phycologia 1982, 21, 154–163. [Google Scholar]
- Cortés-Altamirano, R; Núñez-Pastén, A. Registros de mareas rojas en la Bahía de Mazatlán, Sin., México. Rev. Invest. Cient. UABCS 1991, 13, 1–19. [Google Scholar]
- Alonso-Rodríguez, R. Ocurrencia de mareas rojas y calidad del agua en la zona sur de la Bahía de Mazatlán, Sinaloa, México. MSc Thesis, Universidad Nacional Autónoma de México, Mexico City, Mexico, 1998. [Google Scholar]
- Ramírez-Camarena, C; Cortés-Altamirano, R; Muñoz-Cabrera, L. Mareas rojas provocadas por el dinoflagelado Gymnodinium catenatum (Gymnodinales: Gymnodiniaceae) en la Bahía de Mazatlán, Sin., México, en 1997. Rev. Biol. Trop 1999, 47, 77–80. [Google Scholar]
- Acevedo-Medina, G. Concentración y variación de la composición bioquímica de las biotoxinas del tipo PSP en el ostión de roca Striostrea prismatica Gray, 1825 (=Ostrea iridescens Hanley, 1854) en la Bahía de Mazatlán. BSc Thesis, Universidad Autónoma de Sinaloa, Mazatlán, Sinaloa, Mexico, 2005. [Google Scholar]
- Cortés-Altamirano, R; Alonso-Rodríguez, R; Sierra-Beltrán, A. Fish mortality associated with Chattonella marina and C. cf. ovata (Raphidophyceae) blooms in Sinaloa (Mexico). Harmful Algae News 2006, 31, 7–8. [Google Scholar]
- Poot-Delgado, CA. Estructura de la comunidad fitoplanctónica con énfasis en las especies tóxicas y/o nocivas de la laguna de Macapule, Sinaloa. MSc thesis, Centro Interdisciplinario de Ciencias Marinas-IPN, La Paz, B.C.S., Mexico, 2006. [Google Scholar]
- Gárate-Lizárraga, I; Siqueiros-Beltrones, DA. Infection of Ceratium furca by the parasitic dinoflagellate Amoebophrya ceratii (Amoebophridae) in the Mexican Pacific. Acta Bot. Mex 2003, 65, 1–9. [Google Scholar]
- Gárate-Lizárraga, I; Band-Schmidt, CJ; Verdugo-Díaz, G; Muñetón-Gómez, MS; Félix-Pico, EF. Funes-Rodríguez, R, Gómez-Gutiérrez, J, Palomares-García, R, Eds.; Dinoflagelados (Dinophyceae) del sistema lagunar Magdalena-Almejas. In Estudios Ecológicos de Bahía Magdalena; Instituto Politécnico Nacional: Mexico City, Mexico, 2007; pp. 145–174. [Google Scholar]
- Gómez-Villarreal, MC; Durán-Salgueiro, L. Presencia de Gymnodinium catenatum en Bahía de Banderas (Nayarit-Jalisco). Resúmenes XII Reunión Nacional de la Sociedad Mexicana de Planctología, Xalapa, Mexico, 6–9 May 2002; Universidad Veracruzana: Xalapa, Mexico, 2002. (compactdisk ). [Google Scholar]
- Castillo-Barrera, E; García-Murillo, A. Relación de organismos planctónicos presentes en florecimientos algales nocivos en Bahía de Matachen, San Blas, Nayarit. Recursos y Medio Ambiente: Memorias del XIV Congreso Nacional de Ciencia y Tecnología, Nuevo Vallarta, Mexico, 29–31 October 2007. (compact disk).
- Gómez-Villarreal, MC; Gárate-Lizárraga, I; Bustillos-Guzmán, JJ. Toxinas paralizantes en ostión de piedra, Crassostrea iridescens en Bahía de Banderas. Recursos y Medio Ambiente: Memorias del XIV Congreso Nacional de Ciencia y Tecnología, Nuevo Vallarta, 29–31 October 2007; Mexico, 2007. (compact disk). [Google Scholar]
- Figueroa-Torres, MG; Zepeda-Esquivel, MA. Mareas rojas del puerto interior, Colima, México. Scientia Naturae 2001, 3, 39–52. [Google Scholar]
- Ortíz-Lira, JH; Jiménez-Quiroz, MdC. Jiménez-Quiróz, MdC, Espino-Barr, E, Eds.; Registros de eventos de Marea Roja en Manzanillo, Colima (1986–2004). In Los Recursos Pesqueros y Acuícolas de Jalisco, Colima y Michoacán; SAGARPA, INP, and CRIP Manzanillo: Mexico, 2006; pp. 324–334. [Google Scholar]
- Blanco-Blanco, M; Aguilar-Olguín, S; Morales-Blake, A. Caracterización de una marea roja en la Bahía de Manzanillo, Colima, México. Libro de resúmenes VIII Congreso Latinoamericano sobre Ciencias del Mar, Trujillo, Peru, 17–21 October 1999; Tresierra-Aguilar, AE, Culquichicón-Malpica, ZG, Eds.; Trujillo, Peru, 1999; pp. 338–339. [Google Scholar]
- Morales-Blake, A; Hernández-Becerril, DU; Cavazos-Guerra, C. Ríos-Jara, E, Juárez-Carrillo, E, Pérez-Peña, M, López-Linares, E, Robles-Jarero, EG, Hernández-Becerril, DU, Silva-Briano, M, Eds.; Registros de mareas rojas en las bahías de Manzanillo, Colima, México. In Estudios sobre Plancton en México y el Caribe; Sociedad Mexicana de Planctología y Universidad de Guadalajara:Guadalajara: Mexico, 2000; pp. 81–82. [Google Scholar]
- González-Chan, RB; Hernández-Silva, L; Navarro-Ornelas, JG; Blanco-Padilla, MA. Proliferación algal nociva en las Bahías del Puerto de Manzanillo, Colima (marzo-mayo 2007). Recursos y Medio Ambiente: Memorias del XIV Congreso Nacional de Ciencia y Tecnología, Nuevo Vallarta, Mexico, 29–31 october 2007; 2007. (compact disk). [Google Scholar]
- Zepeda-Esquivel, MA; Meave del Castillo, ME. FAN de dinoflagelados en Manzanillo, Col., durante abril-mayo del 2007. II Taller sobre Florecimientos Algales Nocivos, Ensenada, Mexico, 21–23 November; CICESE-CETMAR: Ensenada, B.C., Mexico, 2007; p. 9. [Google Scholar]
- Gómez-Aguirre, S. Eventos de hemotalasia en México: el caso observado en la Bahía de Banderas (Abril, 2001). Rev. Esp. Cienc. Quím.-Biol 2003, 6, 5–10. [Google Scholar]
- Licea, S; Gómez-Aguirre, S; Cortés-Altamirano, R; Gómez, S. Notas sobre algunos florecimientos algales y la presencia de especies tóxicas en cinco localidades del Pacífico Mexicano (1996–1999). VIII Congreso Latinoamericano sobre Ciencias del Mar, Trujillo, Peru, 17–21 October 1999; pp. 335–337.
- Mancilla-Cabrera, E; Ramírez-Camarena, C; Muñoz-Cabrera, L. Ríos-Jara, R, Juárez-Carrillo, E, Pérez-Peña, M, López-Iriarte, E, Robles-Jarero, EG, Hernández-Becerril, DU, Silva-Briano, M, Eds.; Primer registro de Gymnodinium catenatum Graham (Gymnodiniaceae) como causante de marea roja en la Bahía de Acapulco, Gro., México. In Estudios sobre el plancton marino en México y el Caribe; Sociedad Mexicana de Planctología y Universidad de Guadalajara: Guadalajara, Mexico, 2000; pp. 85–86. [Google Scholar]
- Gárate-Lizárraga, I; Pérez-Cruz, B; Díaz-Ortíz, J; Band-Schmidt, CJ. Microalgas y biotoxinas marinas en costas mexicanas. Conversus 2008, 9, 20–24. [Google Scholar]
- Gárate-Lizárraga, I; Díaz-Ortíz, J; Pérez-Cruz, B; Alarcón-Tacuba, M; Alarcón-Romero, MA; Salgado-Sales, P; López-Silva, S. Florecimientos de Cochlodinium polykrikoides y Gymnodinium catenatum en la Bahía de Acapulco, Guerrero, México (2005–2008). Resúmenes del III Taller sobre Florecimiento Algales Nocivos, Acapulco, Mexico, November, 2009; 2009; p. 14. [Google Scholar]
- Saldate-Castañeda, O; Vázquez-Castellanos, JL; Galván, J; Sánchez-Anguiano, A; Nazar, A. Poisoning from paralytic shellfish toxins in Oaxaca, Mexico. Sal. Pub. Mex 1991, 33, 240–247. [Google Scholar]
- Ronsón-Paulín, JA. Análisis retrospectivo y posibles causas de mareas rojas tóxicas en el litoral del sureste mexicano (Guerrero, Oaxaca, Chiapas). Ciencia y Mar 1999, 9, 49–55. [Google Scholar]
- Herrera-Galindo, JE. Presencia de dinoflagelados potencialmente tóxicos en la zona aledaña a la desembocadura del río Copalita, Huatulco, Oaxaca. Resúmenes XII Congreso Nacional de Oceanografía, Huatulco, Mexico, 22–26 May 2000; p. 151.
- Herrera-Galindo, JE. Presencia de Gymnodinium catenatum Graham (1943) en la zona costera de Bahía de Huatulco-Puerto Escondido, Oaxaca, México (octubre de 1998). Resúmenes VII Congreso Nacional de Ciencia y Tecnología del Mar, Campeche, Mexico, 13–15 November 2000; p. 90.
- Gárate-Lizárraga, I; Díaz-Ortíz, J; Alarcón, M; Pérez-Cruz, B; Torres-Jaramillo, A; Alarcón-Romero, MA; López-Silva, S. Monitoreo de florecimientos algales nocivos y de toxinas paralizantes en las costas de Guerrero y Oaxaca (1992–2007). Resúmenes del II Taller sobre Florecimientos Algales Nocivos, Ensenada, Mexico, 21–23 November 2007; p. 7.
- Cortés-Altamirano, R; Sierra-Beltrán, AP; Cortés-Lara, MC. Steidinger, KA, Landsberg, JH, Tomas, RC, Vargo, GA, Eds.; Dominance and permanence of species of harmful algae forming blooms in Mazatlán Bay, Mexico (1979–2002). In Harmful Algae 2002; Florida Fish and Wildlife Conservation Commission, Florida Institute of Oceanography, and Intergovernmental Oceanographic Commission of UNESCO: St. Petersburg, FL, USA, 2004. [Google Scholar]
- Manrique, FA; Molina, RE. Presencia de mareas rojas en la Bahía de Bacochibampo, Guaymas, Sonora, México. Hidrobiologica 1997, 7, 81–86. [Google Scholar]
- Cabrera-Mancilla, E; Ramírez-Camarena, C; Muñoz-Cabrera, L; Monreal-Prado, A. Ríos-Jara, E, Juárez-Carrillo, E, Pérez-Peña, M, López-Linares, E, Robles-Jarero, EG, Hernández-Becerril, DU, Silva-Briano, M, Eds.; Primer registro de Gymnodinium catenatum Graham (Gymnodinaceae) como causante de marea roja en la Bahía de Acapulco, Gro. México. In Estudios sobre plancton en México y el Caribe; Sociedad Mexicana de Planctología y Universidad de Guadalajara: Guadalajara, Mexico, 2000; pp. 85–86. [Google Scholar]
- Góngora-González, D. Estructura microfitoplanctónica y condiciones hidrológicas relacionadas con la presencia de dinoflagelados tóxicos en Bahía Concepción, B. C. S., México. BSc Thesis, Universidad Autónoma de Baja California Sur, La Paz, B. C. S., Mexico, 2001. [Google Scholar]
- Smayda, TJ. Harmful algal blooms: Their ecophysiology and general relevance to phytoplankton blooms in the sea. Limnol. Oceanogr 1997, 42, 1137–1153. [Google Scholar]
- Smayda, TJ. Ecological features of harmful algae blooms in coastal upwelling ecosystems. S. Afr. J. Mar. Sci 2000, 22, 219–253. [Google Scholar]
- Anderson, DM; Gilbert, PM; Burkholder, JM. Harmful algal blooms and eutrophication: Nutrients sources, composition, and consequences. Estuaries 2002, 25, 704–726. [Google Scholar]
- Granéli, E. Wassmann, P, Olli, K, Eds.; Eutrophication and Harmful Algal Blooms. In Drainage Basin Nutrient Inputs and Eutrophication: An Integral Approach; University of Troms⊘: Troms⊘, Norway, 2005; Volume Chapter 7, pp. 99–112. [Google Scholar]
- Hodgkiss, IJ; Ho, KC. Are changes in N:P ratios in coastal waters the key to increased red tide blooms? Hydrobiologia 1997, 352, 141–147. [Google Scholar]
- Pinckney, JL; Paerle, HW; Tester, P; Richardson, TL. The role of nutrient loading and eutrophication in estuarine ecology. Environ. Health Persp 2001, 109, 699–706. [Google Scholar]
- Beman, MJ; Arrigo, KR; Matson, PA. Agricultural runoff fuels large phytoplankton blooms in vulnerable areas of the ocean. Nature 2005, 434, 211–214. [Google Scholar]
- Heisler, J; Gilbert, PM; Burkholder, JM; Anderson, DM; Cochlan, W; Dennison, WC; Dortch, A; Gobler, CJ; Heil, CA; Humphies, E; et al. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar]
- Herrera-Silveira, JA; Morales-Ojeda, SM. Evaluation of the health status of coastal ecosystem in southeast Mexico: Assessment of water quality, phytoplankton and submerged aquatic vegetation. Mar. Poll. Bull 2009, 59, 72–86. [Google Scholar]
- Álvarez-Borrego, S; Lara-Lara, JR. Dauphin, JP, Simoneit, BRT, Eds.; The Physical Environment and Primary Productivity of the Gulf of California. In The Gulf and Peninsular Province of the California; American Association of Petroleum Geologists: Tulsa, OK, USA, 1991; Volume 47, pp. 555–567. [Google Scholar]
- Páez-Osuna, F; Gracia, A; Flores-Verdugo, F; Lyle-Fritch, LP; Alonso-Rodríguez, R; Roque, A; Ruíz-Fernández, AC. Shrimp aquaculture development and the environment in the Gulf of California ecoregion. Mar. Poll. Bull 2003, 46, 806–815. [Google Scholar]
- Badan-Dangon, A; Koblinski, DJ; Baumgartner, T. Spring and summer in the Gulf of California: Observation of surface thermal patterns. Oceanol. Acta 1985, 8, 13–22. [Google Scholar]
- Bray, NA. Thermohaline circulation in the Gulf of California. J. Geophys. Res 1988, 93, 4993–5020. [Google Scholar]
- Santamaría-del-Ángel, E; Álvarez-Borrego, S; Muller-Karger, FE. The 1982–1984 El Niño in the Gulf of California as seen in coastal zone color scanner imagery. J. Geophys. Res 1994, 99, 7423–7431. [Google Scholar]
- Lavín, MF; Beire, E; Badan, A. Lavín, MF, Ed.; Contribuciones a la Oceanografía Física en México. Monografía #3; Estructura hidrográfica y circulación del Golfo de California: escalas estacional e interanual; Unión de Geofísica Mexicana: Ensenada, Mexico, 1997; pp. 141–171. [Google Scholar]
- Kahru, M; Marinone, SG; Lluch-Cota, SE; Pares-Sierra, A; Greg-Mitchell, B. Ocean-color variability in the Gulf of California: scales from days to ENSO. Deep-Sea Res. Part II 2004, 51, 139–146. [Google Scholar]
- Morquecho-Escamilla, ML. Fitoplancton tóxico y actividad de ficotoxinas en la almeja catarina Argopecten circularis (Sowerby, 1835) en Bahía Concepción, Golfo de California. MSc thesis, Centro Interdisciplinario de Ciencias Marinas-Instituto Politécnico Nacional, La Paz, B.C.S., Mexico, 1996. [Google Scholar]
- Morquecho, L; Lechuga-Devéze, CH. Seasonal occurrence of planktonic dinoflagellates and cyst production in relationship to environmental variables in subtropical Bahía Concepción, Gulf of California. Bot. Mar 2004, 47, 313–322. [Google Scholar]
- Blackburn, SI; Hallegraeff, GM; Bolch, CJ. Vegetative reproduction and sexual life cycle of the toxic dinoflagellate Gymnodinium catenatum Graham from Tasmania, Australia. J. Phycol 1989, 25, 577–590. [Google Scholar]
- Bravo, I; Anderson, DM. The effects of temperature, growth medium and darkness on excystment and growth of the toxic dinoflagellate Gymnodinium catenatum from northwest Spain. J. Plank. Res 1994, 16, 513–525. [Google Scholar]
- Cortés-Altamirano, R; Hernández-Becerril, DU; Luna-Soria, R. Yasumoto, T, Oshima, Y, Fukuyo, Y, Eds.; Red Tides in Mexico: A Review. In Harmful and Toxic Algal Blooms; Intergovernmental Oceanographic Commission of UNESCO: Paris, France, 1996; pp. 101–104. [Google Scholar]
- Cortés-Altamirano, R. Observaciones de mareas rojas en la Bahía de Mazatlán, Sinaloa, México. Cienc. Mar 1987, 13, 1–19. [Google Scholar]
- Alonso-Rodríguez, R; Ochoa, JL. Hydrology of winter-spring “red tides” in Bahía de Mazatán, Sinaloa, México. Harmful Algae 2004, 3, 163–171. [Google Scholar]
- Flores-Trujillo, JG; Helenes, J; Herguera, JC; Orellana-Zepeda, E. Palynological record (1483–1994) of Gymnodinium catenatum in Pescadero Basin, southern Gulf of California, Mexico. Mar. Micropal 2009, 73, 80–89. [Google Scholar]
- Cortés-Altamirano, R; Hernández-Becerril, DU; Luna-Soria, R. Evaluación y prevención de los efectos de las mareas rojas en la Bahía de Mazatlán (1994). Ciencias del Mar, Universidad Autónoma de Sinaloa 1995, 14, 10–14. [Google Scholar]
- Aguirre-Gómez, R; Álvarez, R; Salmerón-García, O. Red tide evolution in the Mazatlán Bay area from remotely sensed sea surface temperatures. Geof. Int 1999, 38, 63–71. [Google Scholar]
- Alonso-Rodríguez, R. Hidrología y condiciones ambientales que determinan la proliferación de dinoflagelados causantes de marea roja en la Bahía de Mazatlán, Sinaloa, México. DSc Thesis, Centro de Investigaciones Biológicas del Noroeste, La Paz, B.C.S., Mexico, 2004. [Google Scholar]
- Wyrki, K. Surface Currents the eastern tropical Pacific Ocean. Inter. Trop. Tuna Comm. Bull 1965, 95(5), 269–304. [Google Scholar]
- Gómez-Gutiérrez, J; Palomares-García, R; Gendron, D. Community structure of the euphauisiid population along the west coast of Baja California, Mexico, during the weak ENSO 1986–1987. Mar. Ecol. Prog. Ser 1995, 120, 41–51. [Google Scholar]
- Hernández-Trujillo, S. Variability of community structure of Copepoda related to El Niño 1982–83 and 1987–88 along the west coast of Baja California Peninsula, Mexico. Fish. Oceanogr 1999, 8(4), 284–295. [Google Scholar]
- Félix-Uraga, R; Gómez-Muñoz, VM; Quiñónez-Vázquez, C; Melo-Barrera, FL; García-Franco, W. On the existence of Pacific sardine groups off the west coast of Baja California and southern California. Calif. Coop. Ocean Fish Invest. Rep 2004, 45, 146–151. [Google Scholar]
- Gómez-Gutiérrez, J; Martínez-Gómez, S; Robinson, CJ. Influence of termo-haline fronts forced by tide on near-surface zooplankton aggregation and community structure in Bahía Magdalena, Mexico. Mar. Ecol. Prog. Ser 2007, 346, 109–125. [Google Scholar]
- Blasco, D. Red tide in the upwelling region of Baja California. Limnol. Oceanog 1977, 22, 255–263. [Google Scholar]
- Dickman, M; Zhang, F. Mid-ocean exchange of container vessel ballast water. 2: effects of vessel type in the transport of diatoms and dinoflagellates from Manzanillo, Mexico, to Hong Kong, China. Mar. Ecol. Progr. Ser 1999, 176, 253–262. [Google Scholar]
- Olivos-Ortíz, A; Sosa-Avalos, R; Torres-Orozco, E; Silva-Iñiguez, L; Galicia-Pérez, MA; Gaviño-Rodríguez, JH. Influencia de las descargas de agua continental en la concentración de nutrientes en las Bahías de Santiago y Manzanillo, Colima. Iridia 2008, 5, 15–16. [Google Scholar]
- Olivos-Ortíz, A; Quijano-Scheggia, SI; García-Uribe, G; Galicia-Pérez, MA; Gaviño-Rodríguez, JH. Distribución de los principales grupos de fitoplancton en las costas del estado de Colima durante el 2002. Rev. Cien. Tecnol. Universidad Autónoma de Ciudad Juárez 2005, 3, 13–28. [Google Scholar]
- Rodríguez-Palacio, MC; Lozano-Ramírez, C; Álvarez-Hernández, S; de Lara-Isassi, G. Registro de dos florecimientos algales nocivos (FAN) en las costas del Pacífico Mexicano. Resúmenes del II Taller sobre Florecimientos Algales Nocivos, Ensenada, Mexico, 21–23 November 2007; CICESE-CETMAR: Ensenada, Mexico, 2007; p. 5. [Google Scholar]
- Nava-Sánchez, E. Riesgo geológico en la zona costera de la Bahía de Acapulco, Guerrero. In Informe Técnico, Consejo de Recursos Minerales; Mexico City, Mexico, 2003; pp. 1–41. [Google Scholar]
- Palacios-Salgado, DS. Asociación de peces en los arrecifes rocosos de la Bahía de Acapulco, Guerrero, México. MSc, Centro Interdisciplinario de Ciencias Marinas-Instituto Politécnico Nacional, La Paz, B. C. S., Mexico, 2005. [Google Scholar]
- Gárate-Lizárraga, I; Díaz-Ortíz, J; Pérez-Cruz, B; Alarcón-Tacuba, M; Alarcón-Romero, MA; López-Silva, S. Cochlodinium polykrikoides and Gymnodinium catenatum in Bahía de Acapulco, Mexico 2005–2008. Harmful Algae News 2009, 40, 8–9. [Google Scholar]
- Núñez-Vázquez, EJ; Bustillos-Guzmán, J; Ramírez-Camarena, C; Hernández-Sandoval, F. Perfiles cromatográficos de toxinas paralizantes en moluscos bivalvos asociados a Pyrodinium bahamense var. compressum en el Pacífico Sur Mexicano. Resúmenes del II Taller sobre Florecimientos Algales Nocivos, Ensenada, Mexico, 21–23 November 2007; p. 18.
- Alonso-Rodríguez, R; Ochoa, JL; Uribe-Alcocer, M. Grazing of heterotrophic dinoflagellate Noctiluca scintillans (McCartney) Kofoid on Gymnodinium catenatum Graham. Rev. Lat. Microbiol 2005, 47, 6–10. [Google Scholar]
- Bustillos-Guzmán, JJ; Hernández-Sandoval, FE; Band-Schmidt, CJ; López-Cortés, DJ; Núñez-Vázquez, EJ; Gárate-Lizárraga, I. Grazing of the dinoflagellate Noctiluca scintillans on Gymnodinium catenatum: a laboratory study. Cienc. Mar 2010. submitted. [Google Scholar]
- Palomares-García, R; Bustillos-Guzmán, J; Band-Schmidt, CJ; López-Cortés, D; Luckas, B. Effect of the toxic dinoflagellate Gymnodinium catenatum on the grazing, egg production, and hatching success of the copepod Acartia clausi. Cienc. Mar 2006, 32, 97–119. [Google Scholar]
- Gárate-Lizárraga, I; Bustillos-Guzmán, JJ; Morquecho, L; Band-Schmidt, CJ; Alonso-Rodríguez, R; Erler, K; Luckas, B; Reyes-Salinas, A; Góngora-González, DT. Comparative paralytic shellfish toxin profiles in the strains of Gymnodinium catenatum Graham from the Gulf of California, Mexico. Mar. Poll. Bull 2005, 50, 208–236. [Google Scholar]
- Band-Schmidt, CJ; Morquecho, L; Lechuga-Devéze, C; Anderson, DM. Effects of growth medium, temperature, salinity and seawater source on the growth of Gymnodinium catenatum (Dinophyceae) from Bahía Concepción, Gulf of California. J. Plank. Res 2004, 26, 1459–1470. [Google Scholar]
- Band-Schmidt, CJ; Bustillos-Guzmán, J; Morquecho, L; Gárate-Lizárraga, I; Alonso-Rodríguez, R; Reyes-Salinas, A; Erler, K; Luckas, B. Variations of PSP toxin profiles during different growth phases in Gymnodinium catenatum (Dinophyceae) strains isolated from three locations in the Gulf of California, Mexico. J. Phycol 2006, 42, 757–768. [Google Scholar]
- Bustillos-Guzmán, JJ; Band-Schmidt, CJ; López-Cortés, DJ; Gárate-Lizárraga, I; Núñez-Vázquez, E; Hernández-Sandoval, F. Variations in growth and toxicity in Gymnodinium catenatum Graham from the Gulf of California under different ratios of nitrogen and phosphorus. Harmful Algae 2010. submitted. [Google Scholar]
- Wright, JLC. Review: Dealing with seafood toxins: present approaches and future options. Food Res. Int 1995, 28, 347–358. [Google Scholar]
- Oshima, Y; Sugino, K; Hirota, M; Yasumoto, T. Granéli, E, Sundstr⊘m, B, Elder, L, Anderson, DM, Eds.; Comparative studies on paralytic shellfish toxin profiles of dinoflagellates and bivalves. In Toxic Marine Phytoplankton; Elsevier: New York, NY, USA, 1990; pp. 391–396. [Google Scholar]
- Pérez-Cruz, B. Tasa de acumulación y depuración de toxinas paralíticas producidas por Gymnodinium catenatum (1943) en la almeja chocolata Megapitaria squalida (Sowerby, 1835). MSc Thesis, Centro Interdisciplinario de Ciencias Marinas-IPN, La Paz, B.C.S., Mexico, 2008. [Google Scholar]
- Núñez-Vázquez, EJ; Hernández-Sandoval, F; Heredia-Tapia, A; Ochoa, JL; Band-Schmidt, CJ; López-Cortés, D; Gárate-Lizárraga, I; Bustillos-Guzmán, J. Presence of paralytic toxins in the puffer fish Sphoeroides annulatus from Bahía de La Paz and Arothron meleagris from Punta Pericos, B. C. S. Resúmenes XV Reunión de la Sociedad Mexicana de Planctología A. C. VIII International Meeting of the Mexican Society of Planktology Centro Cultural Iztacala, Estado de México, Mexico, 20–24 April 2009; Facultad de Estudios Superiores Iztacala, Universidad Nacional Autónoma de México; p. 20.
- Núñez-Vázquez, EJ; Cordero-Tapia, A; Ochoa, JL; Bustillos-Guzmán, J. Candanosa-Aranda, E, Ed.; Histopatología por exposición aguda a las Biotoxinas Marinas de importancia en Salud Pública en México. In Resúmenes XVII Congreso Nacional de Patología Veterinaria. Sociedad Mexicana de Patólogos Veterinarios A.C; Facultad de Medicina Veterinaria y Zootecnia. Universidad Nacional Autónoma de México: Mexico City, Mexico, 2008; pp. 518–524. [Google Scholar]
- Pérez-Linares, J; Ochoa, JL; Gago-Martínez, A. Effects of PSP toxins in white leg shrimp Litopenaeus vannamei Boone, 1931. Tox. Chem. Food Saf 2008, 73, 671–673. [Google Scholar]
- Pérez-Linares, J; Ochoa, JL; Gago-Martínez, A. Retention and tissue damage of PSP and NSP toxins in shrimp: Is cultured shrimp a potential vector of toxins to human population? Toxicon 2009, 53, 185–195. [Google Scholar]
- Demaret, A; Sohet, K; Houvenaghel, G. Lassus, P, Arzul, G, Erard, E, Gentien, P, Marcaillou, C, Eds.; Effects of toxic dinoflagellates on the feeding and mortality of Artemia franciscana larvae. In Harmful Marine Algal Blooms; Lavoisier, Intercept: Paris, France, 1995; pp. 427–432. [Google Scholar]
- Tan, ZJ; Yan, T; You, PC; Zhou, MJ. Transfer of Paralytic Shellfish Toxins via Marine Foods Chains: A Simulated experiment. Biomed. Environ. Sci 2007, 20, 235–241. [Google Scholar]
- Estrada, N; Lagos, N; García, C; Maeda-Martínez, AN; Ascencio, F. Effect of the toxic dinoflagellate, Gymnodinium catenatum on uptake and fate of paralytic shellfish poisons in the Pacific giant lion-paw scallop Nodipecten subnodosus. Mar. Biol 2007, 151, 1205–1204. [Google Scholar]
- MacQuarrie, SP; Bricelj, VM. Behavioral and physiological responses to PSP toxins in softshell clam, Mya arenaria, populations in relation to the history of exposure to red tides. Mar. Ecol. Progr. Ser 2008, 336, 59–74. [Google Scholar]
- Bricelj, VM; Shumway, SE. Paralytic shellfish toxins in bivalve molluscs: occurrence, transfer kinetics, and biotransformation. Rev. Fish. Sci 1998, 6, 315–383. [Google Scholar]
- Estrada, N; Romero, MJ; Campa-Córdova, A; Luna, A; Ascencio, F. Effect of the toxic dinoflagellate, Gymnodinium catenatum on the hydrolytic and antioxidant enzymes, in tissues of the giant lion-paw scallop Nodipecten subnodosus. Comp. Bioch. Phys. Part C 2007, 146, 502–510. [Google Scholar]
- Estrada, N; Rodríguez-Jaramillo, C; Contreras, G; Ascencio, F. Effects of induced paralysis on hemocytes and tissues of the giant lions-paw scallop by paralyzing shellfish poison. Mar. Biol 2010, 157, 1401–1415. [Google Scholar]
- Heredia-Tapia, A; Campa-Córdova, AI; Band-Schmidt, CJ; Núñez-Vázquez, EJ; Bustillos-Guzmán, JJ. Lipid peroxidation in the bivalve mollusks Dosinia ponderosa and Crassostrea gigas exposed to the toxic dinoflagellate Gymnodinium catenatum. resúmenes XVI Reunión de la Sociedad Mexicana de Planctología A.C. y IX International Meeting of the Mexican Society of Planktology, La Paz, Mexico, 2010; p. 57.
- Alonso-Rodríguez, R; Páez-Osuna, F; Gárate-Lizárraga, I. El fitoplancton en la camaronicultura y larvicultura: importancia de un buen manejo; Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México y Comité Estatal de Sanidad Acuícola de Sinaloa: Mexico, 2004a. [Google Scholar]
- Sierra-Beltrán, AP; Lluch-Cota, DB; Lluch-Cota, SE; Cortés-Altamirano, R; Cortés-Lara, MC; Castillo-Chávez, M; Carrillo, L; Pacas, L; Vázquez, R; García-Hansen, I. Dinámica espacio-temporal de organismos precursores de mareas rojas en la costa Pacífica de América del Norte y Centroamérica. Rev. Biol. Trop 2004, 52, 99–107. [Google Scholar]
- Barraza-Guardado, R; Cortés-Altamirano, R; Sierra-Beltrán, A. Marine die-offs from Chattonella marina and Ch. cf. ovata in Kun Kaak Bay, Sonora in the Gulf of California. Harmful Algae News 2004, 25, 7–8. [Google Scholar]
- Band-Schmidt, CJ; Rojas-Posadas, D; Morquecho, L; Hernández-Saavedra, NY. Heterogeneity of LSU rDNA sequences and morphology of Gymnodinium catenatum strains in Bahía Concepción, Gulf of California, Mexico. J. Plank. Res 2008, 30, 755–763. [Google Scholar]
| Region | Year | Locality | Abundance (cells L−1) | Toxicity (μg STXeq per 100 g−1) and bivalve species | Temperature (ºC) | Ref |
|---|---|---|---|---|---|---|
| Gulf of California | 1939 | N Gulf of California | 1 × 106 | nd | 14.0–17.0 | [1] |
| 1981 | Puerto Libertad | 190 × 103 | nd | nd | [9] | |
| 2003 | Bahía Kun Kaak | Nd | nd | 25.32 ± 0.99 | [10] | |
| 2006 | Bahía de Los Ángeles | Nd | 3–54 Nodipecten subnodosus | 16.0–23.0 | [11] | |
| 1990 | Bahía Concepción | 1.8 × 102–3 × 103 | nd | nd | [12] | |
| 1999 | 5.7 × 105 | 298 Argopecten ventricosus | 18.0–25.0 | [13] | ||
| 2000 | 500–4 × 104 | 63 A. ventricosus | 18.0–25.0 | [13,14] | ||
| 1997–1998 | Bahía de La Paz | 1.60 × 10–2.6 × 102 | 0.14–5.46 Megapitaria squalida | 18.0–26.0 | [15] | |
| 2001 | Nd | 2–67 A. ventricosus PST in net phytoplankton samples | 22.0–26.0 | [16,17] | ||
| 2003 | 1–1.20 × 103 | nd | 18.0–22.0 | [16,18] | ||
| 2006 | 1.0–3.6 × 103 | 3–4.5 M. squalida 4–9 Dosinia ponderosa | 18.5–26.5 | [19,20] | ||
| 2007 | 6–2.39 × 106 | 0.40–37.74 M. squalida, M. aurantiaca, D. ponderosa. Modiolus capax, Pinna rugosa, P. multicostata | 20.0–21.9 | [21] | ||
| 2008 | 8–79 × 103 | nd | 20.0–24.0 | [22] | ||
| 1995–1996 | Shrimp culture pond, Sinaloa | ? | ? | ? | [23] | |
| 1997 | 15 × 103 | 40 Crassostrea iridescens. Nauplii and adult mortality of L. vannamei | nd | [15,24] | ||
| 2001 | 18–528 × 103 | 29 oyster Nauplii mortality of L. stylirostris. No toxins detected in nauplii | nd | [23] | ||
| 1979* | Cruz de Elota, Punta Piaxtla, Bahía de Mazatlán, and Teacapan, Sinaloa | 240–6.6 × 106 | 〈20–7640 C. iridescens and Donax sp. extensive fish | 21.60 | [7,25,26] | |
| 1981 | Bahía de Mazatlán | 35–544 × 103 | nd | 19.74–20.52 | [9] | |
| 1985 | 65 × 103 | nd | 22.04 | [9,27] | ||
| 1986 | 170–940 × 103 | nd | 20.64–22.34 | |||
| 1988** | 1000 × 103 | 20.94 | ||||
| 1994–1995 | 1.2–2.2 × 105 | nd | 21.14–22.54 | [28] | ||
| 1996 | 3856–5000 × 103 | nd | 21.0–32.9 | [23] | ||
| 1997 | 3856–5000 × 103 | 〈35 Ostrea iridescens | nd | [29] | ||
| 2001 | 1.5–196 × 103 | 39.40 C. iridescens | 16.5–25.0 | [9,15] | ||
| 2003–2004 | 71–115 × 103 | 63–1315 | 19.0–24.0 | [30] | ||
| 2006 | 148 × 103 | nd | 21.2–22.7 | [31] | ||
| 2005 | Laguna de Macapule | 38.8 × 103 | nd | nd | [32] | |
| W. Coast of B. California Peninsula | 1996 | West Coast of Baja California | 1.2–4.2 × 102 | nd | 13.0–17.0 | [33] |
| 2005–2006? | Bahía Magdalena-Almejas | Presence in net phytoplankton samples | negative Anadara tuberculosa | nd | [34] | |
| Central Mexican Pacific | 1979 | San Blas and Puerto Vallarta | Nd | 〈20 Crassosstrea cortesiensis | nd | [25,35] |
| 2005 | Bahía de Matachén | 1010 × 103 | nd | nd | [36] | |
| 1999 | Bahía de Manzanillo (Puerto Interior) | 2.5–3.8 × 106 | 11–13 C. iridescens | nd | [37,38] | |
| 1989 | Bahía de Manzanillo and Santiago | 5000 × 103 | nd | nd | [39] | |
| 2002 | 832 × 103 | nd | nd | |||
| 2000 | Bahía de Manzanillo | >3500 × 103 | 235 Oyster | 23.0–25.0 | [40,41] | |
| 2007 | Bahía Banderas | 3.53 × 103–3.8 × 106 | 29–235.28 C. iridescens | nd | [42,43] | |
| 2001 | 450–2134 × 103 | 11–13 C. iridescens | 23.0 | [37, 44] | ||
| 1999 | Bahía de Acapulco | 0.01–78 × 106 | 120–209 O. iridescens | nd | [45,46] | |
| 2005 | 6.29 × 103 | 25–217 | nd | [47] | ||
| 2006 | 10 × 106 | 112 | nd | [47–49] | ||
| 2007 | 3 × 103–13 × 106 | 1152 | nd | |||
| South Mexican Pacific | 1989 | Salina Cruz to Chiapas | Nd | Presence of PSP | nd | [50] |
| 1998 | Puerto Escondido to Huatulco | 4–10 × 106 | 〈80 | nd | [51,52] | |
| 2001 | Laguna Corralero-Alotengo, Oaxaca | Nd | 24–1456 Mussel | nd | [53] | |
| 2006 | Gulf of Tehuantepec, Coasts of Salina Cruz | 13 × 103 | nd | nd | [38] |
| Toxin | Bahía de Mazatlán | Bahía Concepción | Bahía de La Paz | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2001 Apr | 2001 May | 2002 | 2002 | 2007 | ||||||||||
| 18 Jan | 30 Jan | 14 Feb | 6 Mar | 17 Apr | 2 May | 21 May | 8 Jul | 15 Mar | 12 Mar | 20 Aug | 7 Mar | |||
| STX | 64.3 | - | - | - | - | 1.0 | - | 3.2 | - | 34.6 | - | 1.4 | 0–31 | |
| neoSXT | - | - | - | - | - | - | - | - | 7.8 | - | - | - | - | 0–25 |
| GTX2 | - | - | - | - | - | - | - | - | 9.0 | - | - | - | - | 0–5.4 |
| GTX3 | - | - | - | - | - | - | - | - | 2.6 | - | - | - | - | 0–3.6 |
| dcSTX | 17.4 | 9.0 | 62.9 | 8.3 | - | |||||||||
| dcGTX2 | 22.6 | 2.0 | 78.2 | 47.3 | 88.6 | 73.9 | 66.8 | 70.2 | 1.6 | 72.8 | 37.0 | 37.1 | 52.5 | - |
| dcGTX3 | 27.4 | 3.6 | 21.8 | 25.8 | 11.4 | 26.1 | 18.5 | 29.8 | 0.4 | 27.2 | 12.0 | - | 18.5 | 0–1.1 |
| B 1 | 4.4 | - | - | - | - | 0.8 | - | - | - | 7.4 | - | 5.7 | 0–5.4 | |
| B 2 | - | 1.0 | - | - | - | - | ||||||||
| C 1 | - | 16.0 | - | - | 9.4 | - | 62.6 | - | - | - | 10.5 | 0–37.0 | ||
| C 2 | 50.0 | 7.4 | - | 10.9 | - | - | 2.5 | - | 12.8 | - | - | - | 3.2 | 53.8–68.9 |
| 1.5 ng STXeq cell−1 | ||||||||||||||
| Strain Origin | Media | STX | neoSTX | GTX2 | GTX3 | dcSTX | dcGTX2 | dcGTX3 | B 1 | B 2 | C 3 | C 1 | C 2 | C 4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BACO | GSe | 0–0.6 | 6.6–12.1 | nd | nd | 2.1–2.8 | 3.3–4.7 | 0.9–1.4 | 0.7–0.9 | 3.7–7.6 | 1.6–2.9 | 50.9–55.9 | 18.6–22.8 | 0.1–0.6 |
| BAPAZ | 0–0.8 | 25.7–35.2 | 6.3–24.2 | 1.6–2.9 | 0.5–0.9 | 0.4–0.8 | 4.1–12.9 | 1.6–7.1 | 19.7–33.1 | 7.0–15.2 | 0–2.7 | |||
| BAMAZ | 0.1–0.6 | 29.2–46.3 | 1.1–3.4 | 2.2–3.8 | 0.7–1.4 | 0.2–0.4 | 7.5–15.3 | 2.3–4.3 | 23.7–31.7 | 8.6–13.5 | 0.5–1.7 | |||
| BACO | f/2 | nd | 0–5.1 | 0–0.3 | 0–0.1 | 19.4–43.2 | 19.3–43.1 | 6.5–11.7 | 0–0.2 | 0–1.5 | nd | 12.8–39.9 | 4.6–38.7 | nd |
| Toxin | Bahía Concepción Argopecten ventricosus May 99, 00, 01 | Bahía de Mazatlán Crassostrea iridescens April, 01 | Bahía de La Paz Megapitaria squalida Dec, 01–Aug, 02 |
|---|---|---|---|
| STX | 0–0.92 | - | 0–38.69 |
| neoSXT | 0–9.00 | 5.71 | - |
| GTX2 | 0–31.17 | - | 0–41.19 |
| GTX3 | 0–4.79 | 0.89 | 0–16.02 |
| dcSTX | 0–41.62 | 18.54 | 0–62.90 |
| dcGTX2 | 0–40.67 | 2.52 | 0–52.45 |
| dcGTX3 | 0–59.33 | 3.21 | 0–39.77 |
| B 1 | 0–42.34 | 9.80 | 0–7.40 |
| B 2 | 0–1.94 | 1.91 | - |
| C 1 | 0–54.61 | 37.89 | 0–52.54 |
| C 2 | 0–47.72 | 19.50 | 0–35.07 |
| C3 | 0–3.32 | - | - |
| C4 | 0–3.98 | - | - |
| Organism tested | Strain | Culture conditions | Total toxicity (pgSTXeq cell−1 or μg eq. STX) | Toxin profile | Effects | Ref. | |
|---|---|---|---|---|---|---|---|
| Mammals | Mouse model, Mus musculus (BALB/c and CD1mice) | GCCV-6 | f/2+Se, 33 ‰, 25 ± 1 ºC, 12:12 L:O, 150 μEm2 s−1, Fernbach flasks | 0.2, 0.3 μg eq.STX | STX, dcSTX GTX-2,3 dcGTX-2,3 C1–2 | Clinical signs: dyspnea, paralysis, convulsions, jump, respiratory failure, and death. In necropsy a pronounced isquemic zone only in liver border was detected. Histopathological changes: cerebellar injury (Purkinje cell degeneration). | [110] |
| Crustaceans | Acartia clausi | GCCV-14 | f/2 + Se 10−8M, 33– 34‰, 20 ºC, 12:12 L:O, 150 μEm2 s−1, 2 L flasks | 60 | dcSTX, dcGTX-2, 3,C1–2 | No adverse effects. | [101] |
| Litopenaeu s vannamei | GCCV-6 | f/2, 33‰, 26 ± 1 ºC, 12:12 L:O, 150 μEm2 s−1, Fernbach flasks | nd | STX, dcSTX, neoSTX, GTX-1,2,3,4 | Paralysis of antennae and pereiopods, disequilibrium, atypical swimming. Slow and irregular movements of gills, pleopods, and maxillipeds. Heart and brain severely damaged; juvenile shrimp more susceptible than adult animals. In chronic exposure: gastric glands and muscle retained paralytic toxins for a longer period, histological damages were observed in the heart, gastric gland, and brain tissue. | [111,112] | |
| Mollusks | Artemia salina (adults and metanauplii) | GSe, 33‰, 23 ± 1 ºC, 12:12 L:O, 150 μEm2 s−1, Fernbach flasks | nd | STX, dcSTX GTX-2,3, dcGTX-2,3 C1–2 | 20% and 10% mortality in adults and metanauplii, respectively. Clinical signs: Adults: erratic swimming (circles), spasms, convulsions, and death. Metanauplii: erratic swimming and death. | this study | |
| Nodipecten subnodosus | GSe medium, 32‰, 21 ºC, 16:8 L:O, 70 W, 20 L flasks | 2–5 | GTX | At high food concentrations, juvenile showed production of pseudofeces, partial shell valve closure, and reduction in feeding. An increase of antioxidant and hydrolytic enzymes mainly in gills and the digestive gland. Melanization in gills, mantle, and labial palps. | [115,116] | ||
| Megapitaria squalida | GCCV-7 | f/2 + Se 10−8M, 33–34‰, 22 ºC, 12:12 L:O, 150 μEm2 s−1, 10 L flasks | 26–28 | STX, dcSTX, neoSTX, dcGTX-3, 4, C1–2 | No adverse effects. | [108] | |
| Strain Code | Source | Growth Rate (div day−1) | Maximum Cell Density (Cells mL−1) | Media | Temperature (ºc) | Salinity (psu) | Light Intensity (μmol m2 s−1) | Light/Dark Cycle | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| GCCQ-1 | 0.74 ± 0.07 | 1619 ± 252 | |||||||
| GCCV-2 | BACO | 0.70 ± 0.07 | 1090 ± 270 | ||||||
| GCCV-4 | 0.82 ± 0.09 | 3393 ± 836 | |||||||
| GCPV-1 | BAPAZ | 0.74 ± 0.06 | 1631 ± 152 | GSe | 20 ± 1 | 35 | 150 | 12,12 | [104] |
| GCPV-2 | 0.77 ± 0.05 | 1421 ± 290 | |||||||
| GCMV-1 | BAMAZ | 0.81 ± 0.02 | 2063 ± 226 | ||||||
| GCMV-2 | 0.82 ± 0.03 | 1865 ± 516 | |||||||
| 0.14–0.21 | nd | f/2 + Se 10−8 M | 15–29 | 30 | 230 | 10,14 | |||
| 0.24 | nd | 26–30 | 150 | 10,14 | |||||
| GCCV-10 | BACO | 0.28–0.31 | nd | 20 | 28–38 | 150 | 12,12 | [103] | |
| 0.15–0.19 | 1559–1970 | f/2 + Se 10−6, 10−7, 10−8 M | 35 | 150 | 12,12 |
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Band-Schmidt, C.J.; Bustillos-Guzmán, J.J.; López-Cortés, D.J.; Gárate-Lizárraga, I.; Núñez-Vázquez, E.J.; Hernández-Sandoval, F.E. Ecological and Physiological Studies of Gymnodinium catenatum in the Mexican Pacific: A Review. Mar. Drugs 2010, 8, 1935-1961. https://doi.org/10.3390/md8061935
Band-Schmidt CJ, Bustillos-Guzmán JJ, López-Cortés DJ, Gárate-Lizárraga I, Núñez-Vázquez EJ, Hernández-Sandoval FE. Ecological and Physiological Studies of Gymnodinium catenatum in the Mexican Pacific: A Review. Marine Drugs. 2010; 8(6):1935-1961. https://doi.org/10.3390/md8061935
Chicago/Turabian StyleBand-Schmidt, Christine J., José J. Bustillos-Guzmán, David J. López-Cortés, Ismael Gárate-Lizárraga, Erick J. Núñez-Vázquez, and Francisco E. Hernández-Sandoval. 2010. "Ecological and Physiological Studies of Gymnodinium catenatum in the Mexican Pacific: A Review" Marine Drugs 8, no. 6: 1935-1961. https://doi.org/10.3390/md8061935






