Demospongic Acids Revisited
Abstract
:1. Introduction
2. What Exactly Is a Demospongic Acid?
- the presence of the ever-mentioned 5,9-diunsaturation pattern cannot be considered as characteristic of “demospongic acids” due to the elongation process during their biosynthetic pathways, and diunsaturations such as 5,9-, 7,11-, 9,13-, 11,15-, 17,21-, 19,23- 21,25- and 23,27- can be considered as being similar [18], but other dienoic patterns with short chains such as 6,11-18:2 and 6,11-20:2 have also been considered as “demospongic” acids [19]. Furthermore, several “demospongic acids” display E and Z configurations for Δ5 and Δ9 double bonds [20].
3. Occurrence of “Demospongic” Acids among Other Organisms
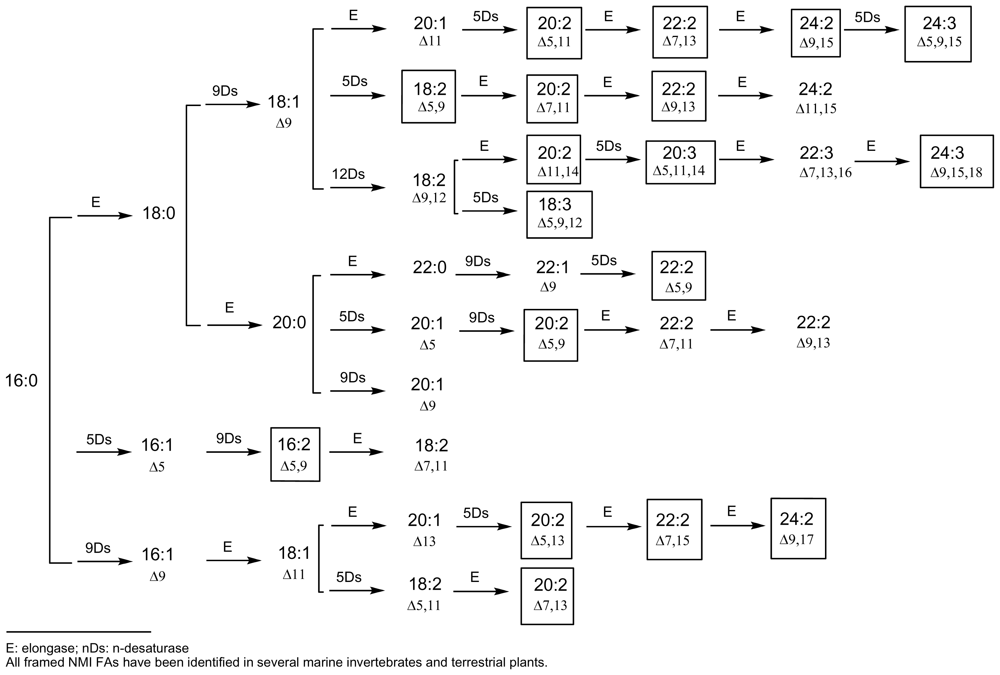
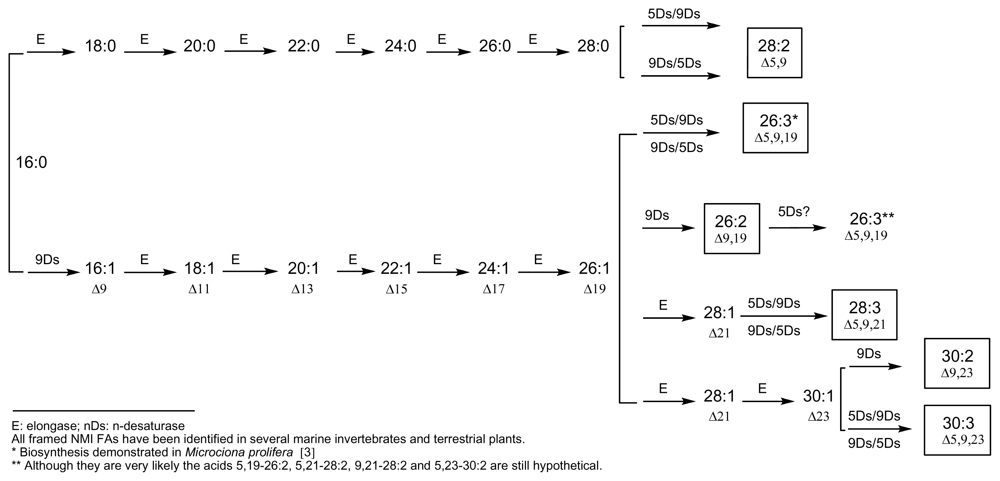
4. Towards a Classification of Non-Methylene-Interrupted Fatty Acids?
5. Conclusion
References
- Litchfield, C; Morales, RW. Harrison, FW, Cowden, RR, Eds.; Are demospongiae membranes unique among living organisms? In Aspects of Sponge Biology; Academic Press: New York, NY, USA, 1976; pp. 183–200. [Google Scholar]
- Litchfield, C; Tyszkiewicz, J; Dato, V. 5,9,23-Triacontatrienoic acid, principal fatty acid of the marine sponge Chondrilla nucula. Lipids 1980, 15, 200–202. [Google Scholar]
- Djerassi, C; Lam, W-K. Sponge phospholipids. Acc. Chem. Res 1991, 24, 69–75. [Google Scholar]
- Barnathan, G; Kornprobst, J-M. Baudimant, G, Guézennec, J, Roy, P, Samain, J-F, Eds.; Fatty acids from marine organisms: recent research developments. In Marine Lipids; Editions Ifremer: Plouzané, France, 2000; pp. 35–43. [Google Scholar]
- Kornprobst, J-M. Encyclopedia of Marine Natural Products; Wiley-VCH: Weinheim, Germany, 2010; Volume 2, pp. 538–555. [Google Scholar]
- Carballeira, NM. New advances in fatty acids as antimalarial, antimycobacterial and antifungal agents. Prog. Lipid Res 2008, 47, 50–61. [Google Scholar]
- Wolff, RL; Christie, WW; Marpeau, AM. Reinvestigation of the polymethylene-interrupted 18:2 and 20:2 acids of Ginkgo biloba seed lipids. J. Am. Oil. Chem. Soc 1999, 76, 273–276. [Google Scholar]
- Wolff, RL; Deluc, LG; Marpeau, AM. Conifer seeds: oil content and fatty acid compositions. J. Am. Oil. Chem. Soc 1996, 73, 765–771. [Google Scholar]
- Ando, Y; Kawabata, Y; Narukawa, K; Ota, T. Demospongic acids of the marine sponge Halichondria panicea from the coast of Hokkaido, Japan. Fish. Sci 1998, 64, 136–139. [Google Scholar]
- Rezanka, T; Sigler, K. Odd-numbered very-long-chain fatty acids from the microbial, animal and plant kingdoms. Prog. Lipid Res 2009, 48, 206–238. [Google Scholar]
- Bergé, JP; Barnathan, G. Fatty acids from lipids of marine organisms: molecular biodiversity, roles as biomarkers, biologically active compounds, and economical aspects. Adv. Biochem. Eng. Biotechnol 2005, 96, 49–125. [Google Scholar]
- Johnson, DW. Contemporary clinical usage of LC/MS: analysis of biologically important carboxylic acids. Clin. Biochem 2005, 38, 351–361. [Google Scholar]
- Leonard, AE; Pereira, SL; Sprecher, H; Huang, Y-S. Elongation of long-chain fatty acids. Prog. Lipid Res 2004, 43, 36–54. [Google Scholar]
- Tsydendambaev, VD; Christie, WW; Brechany, EY; Vereshchagin, AG. Identification of unusual fatty acids of four alpine plant species from the Pamir. Phytochemistry 2004, 65, 2695–2703. [Google Scholar]
- Moldovan, Z; Jover, E; Bayona, JM. Gas chromatographic and mass spectrometric methods for the characterization of long-chain fatty acids – Application to wool wax extract. Anal. Chim. Acta 2002, 465, 359–378. [Google Scholar]
- Steinberg, SJ; Wang, SJ; Kim, DG; Mihalik, SJ; Watkins, PA. Human very-long-chain acyl-CoA synthetase: cloning, topography, and relevance to branched-chain fatty acid metabolism. Biochem. Biophys. Res. Commun 1999, 257, 615–621. [Google Scholar]
- Rezanka, T. Very-long-chain fatty acids from the animal and plant kingdoms. Prog. Lipid Res 1989, 28, 147–187. [Google Scholar]
- Christie, WW. AOCS Lipid Library. http://lipidlibrary.aocs.org/Lipids/fa_conj+/index.htm (accessed on 6 July 2010).
- Kulkarni, BA; Chattopadhyay, S; Chattopadhyay, A; Mamdapur, VR. Synthesis of the demospongic compounds, (6Z,11Z)-octadecadienoic acid and (6Z,11Z)-eicosadienoic acid. Molecules 1997, 2, 3–6. [Google Scholar]
- Carballeira, NM; Shalabi, F. Identification of naturally occurring trans, trans Δ-5,9 fatty acids from the sponge Plakortis halichondroides. Lipids 1990, 25, 835–840. [Google Scholar]
- Dembitsky, VM; Srebnik, M. Natural halogenated fatty acids: their analogues and derivatives. Prog. Lipid Res 2002, 41, 315–367. [Google Scholar]
- Carballeira, NM; Medina, JR. New Δ5,9 fatty acids in the phospholipids of the sea anemone Stoichactis helianthus. J. Nat. Prod 1994, 57, 1688–1695. [Google Scholar]
- Carballeira, NM; Reyes, M. Identification of a new 6-bromo-5,9-eicosadienoic acid from the anemone Condylactis gigantea and the Zoanthid Palythoa caribaeorum. J. Nat. Prod 1995, 58, 1689–1694. [Google Scholar]
- Lam, WK; Hahn, S; Ayanoglu, E; Djerassi, C. Phospholipid studies of marine organisms. 22. Structure and biosynthesis of a novel brominated fatty acid from a Hymeniacidonid sponge. J. Org. Chem 1989, 54, 3428–3432. [Google Scholar]
- Schlenk, H. Odd numbered polyunsaturated fatty acids. Prog. Chem. Fats Other Lipids 1970, IX(Part 5), 589–605. [Google Scholar]
- Kawashima, H; Ohnishi, M. Occurrence of novel nonmethylene-interrupted C24 polyenoic fatty acids in female gonad lipids of the limpet Cellana grata. Biosci. Biotechnol. Biochem 2006, 70, 2575–2578. [Google Scholar]
- Kawashima, H. Unusual minor nonmethylene-interrupted di-, tri- and tetraenoic fatty acids in limpet gonads. Lipids 2005, 40, 627–630. [Google Scholar]
- Joseph, JD. Ackman, RG, Ed.; Distribution and composition of lipids in marine invertebrates. In Marine Biogenic Lipids, Fats and Oils; CRC Press: Boca Raton, FL, USA, 1989; Volume II, pp. 49–143. [Google Scholar]
- Asset, G; Leroy, A; Bauge, E; Wolff, RL; Fruchant, J-C; Dallongeville, J. Effects of dietary maritime pine (Pinus pinaster)-seed oil on high-density lipoprotein levels and in vitro cholesterol efflux in mice expressing human apolipoprotein A-I. British J. Nutrition 2000, 84, 353–360. [Google Scholar]
- Sayanova, O; Haslam, R; Venegas Caleron, M; Napier, JA. Cloning and characterization of unusual fatty acid desaturases from Anemone leveillei: Identification of an acyl-coenzyme A C20 Δ5-desaturase responsible for the synthesis of sciadonic acid. Plant Physiol 2007, 144, 455–467. [Google Scholar]
- Treschow, AP; Hodges, LD; Wright, PFA; Wynne, PM; Kalafatis, N; Macrides, T. Novel anti-inflammatory⌉-3 PUFAs from the New Zealand green-lipped mussel, Perna canaliculus. Comp. Biochem. Physiol 2007, 147B, 645–656. [Google Scholar]
- Carballeira, NM; Cruz, C. 5,9-Nonadecadienoic acids in Malvaviscus arboreus and Allamanda cathartica. Phytochemistry 1998, 49, 1253–1256. [Google Scholar]
- Pearce, RE; Stillway, LW. Non-methylene-interrupted and ω4 dienoic fatty acids of the white shrimp Penaeus setiferus. Lipids 1977, 12, 544–549. [Google Scholar]
- Zhukova, NV. Lipid classes and fatty acid composition of the tropical nudibranch mollusks Chromodoris sp. and Phyllidia coelestis. Lipids 2007, 42, 1169–1175. [Google Scholar]
- Imbs, AB; Demidkova, DA; Dautova, TN; Latyshev, NA. Fatty acid biomarkers of symbionts and unusual inhibition of tetracosapolyenoic acid biosynthesis in corals (Octocorallia). Lipids 2009, 44, 325–335. [Google Scholar]
- Thiel, V; Blumenberg, M; Hefter, J; Pape, T; Pomponi, S; Reed, J; Reitner, J; Wörheide, G; Michaelis, W. A chemical view of the most ancient metazoa: biomarker chemotaxonomy of hexactinellid sponges. Naturwissenschaften 2002, 89, 60–66, Erratum: Naturwissenschaften 2002, 89, 233–234. [Google Scholar]
- Sato, D; Ando, Y. Distribution of novel nonmethylene-interrupted fatty acids over neutral and polar lipids of Ophiuroidea (Brittle Star). J. Oleo Sci 2002, 51, 563–567. [Google Scholar]
- Ayanoglu, E; Walkup, RD; Sica, D; Djerassi, C. Phospholipid studies of marine organisms: III. New phospholipid fatty acids from Petrosia ficiformis. Lipids 1982, 17, 617–625. [Google Scholar]
- Christie, WW; Brechany, EY; Stefanov, K; Popov, S. The fatty acids of the sponge Dysidea fragilis from the Black Sea. Lipids 1992, 27, 640–644. [Google Scholar]
- Hahn, S; Lam, WK; Wu, I; Silva, CJ; Djerassi, C. Unusual pattern of fatty acid biosynthesis. J. Biol. Chem 1989, 264, 21043–21046. [Google Scholar]
- Wallis, JG; Watts, JL; Browse, J. Polyunsaturated fatty acid synthesis: what will they think of next? Trends Biochem. Sci 2002, 27, 467–473. [Google Scholar]
- Buist, PH. Fatty acid desaturases: selecting the dehydrogenation channel. Nat. Prod. Rep 2004, 21, 249–262. [Google Scholar]
- Barnathan, G. Non-methylene-interrupted fatty acids from marine invertebrates: Occurrence, characterization and biological properties. Biochimie 2009, 91, 671–678. [Google Scholar]
- Hochmuth, T; Piel, J. Polyketide synthases of bacterial symbionts in sponges – Evolution-based applications in natural products research. Phytochemistry 2009, 70, 1841–1849. [Google Scholar]
| Acids | Genera/species | Kind of organisms | Ref. |
|---|---|---|---|
| 5,9-16:2 | Stoichactis helianthus | Cnidaria (Hexacorallia) | [22] |
| 5,9-17:2 | Dictyostelium discoideum | Microorganism, soil-living amoeba | [25] |
| 5,9-18:2 taxoleic acd | Condylactis gigantea, Palythoa caribaeorum, Stoichactis helianthus | Cnidaria (Hexacorallia) | [22,23] |
| Cellana grata, Collisella dorsuosa | Marine molluscs | [26,27] | |
| Tripneustes esculentus | Echinoderm | [28] | |
| Ginkgo biloba | Terrestrial plant | [7] | |
| 5,9,12-18:3 pinolenic acid (Z,Z,Z) and/or columbinic acid(E,Z,Z) | Abies sp., Cedrus sp., Cupressus sp., Juniperus sp., Laryx sp., Picea sp., Pinus sp., Sequoia sp., Thuya sp. | Terrestrial plants (conifers, gymnosperms) | [8,29] |
| Anemone leveillei (Ranunculaceae) | [30] | ||
| 5,9,12,15-18:4 | Perna canaliculus | Marine mollusc (Lamellibranchiata) | [31] |
| Abies sp., Cedrus sp., Cupressus sp., Juniperus sp., Laryx sp., Picea sp., Sequoia sp., Thuya sp. | Terrestrial plants (conifers) | [8,29] | |
| 5,9-19:2 | Allamanda cathartica (Apocynaceae) Malvaviscus arboreus (Malvaceae) | Terrestrial plants (angiosperms) | [32] |
| i-5,9-19:2 | Allamanda cathartica (Apocynaceae) Malvaviscus arboreus (Malvaceae) | Terrestrial plants (angiosperms) | [32] |
| ai-5,9-19:2 | Allamanda cathartica (Apocynaceae) Malvaviscus arboreus (Malvaceae) | Terrestrial plants (angiosperms) | [32] |
| 5,9,12,16-19:4 | Perna canaliculus | Marine mollusc (Lamellibranchiata) | [31] |
| 5,9-20:2 | Condylactis gigantea, Palythoa caribaeorum, Stoichactis helianthus | Cnidaria (Hexacorallia) | [22,23] |
| 6-Br,5,9-20:2 | Condylactis gigantea, Palythoa caribaeorum | Cnidaria (Hexacorallia) | [22,23] |
| 7,11-20:2 | Penaeus setiferus | Arthropod (shrimp) | [33] |
| 5,9-21:2 | Condylactis gigantea, Stoichactis helianthus | Cnidaria (Hexacorallia) | [22,23] |
| 6-Br,5,9-21:2 | Stoichactis helianthus | Cnidaria (Hexacorallia) | [22] |
| 5,9,12,15,18-21:5 | Perna canaliculus | Marine mollusc | [23] |
| 5,9-22:2 | Condylactis gigantea, Palythoa caribaeorum, Stoichactis helianthus | Cnidaria (Hexacorallia) | [22,23] |
| Cellana grata, Collisella dorsuosa | Marine molluscs | [26–27] | |
| 6-Br,5,9-22:2 | Stoichactis helianthus | Cnidaria (Hexacorallia) | [22] |
| 9,13-22:2 | Penaeus setiferus | Arthropod (shrimp) | [33] |
| 5,9,15-22:3 | Collisella dorsuosa | Marine molluscs | [27] |
| 5,9,19-22:3 | Stoichactis helianthus | Cnidaria (Hexacorallia) | [22] |
| 5,9-23:2 | Stoichactis helianthus | Cnidaria (Hexacorallia) | [22] |
| 5,9-24:2 | Condylactis gigantea, Palythoa caribaeorum | Cnidaria (Hexacorallia) | [23] |
| Cellana grata, Chromodoris sp., Collisella dorsuosa, Phyllidia coelesti | Marine molluscs | [26,27,34] | |
| 5,9,15-24:3 | Cellana grata, Collisella dorsuosa | Marine molluscs | [26,27] |
| 5,9,17-24:3 | Cellana grata, Collisella dorsuosa | Marine molluscs | [26,27] |
| 5,9,15,18-24:4 | Cellana grata | Marine mollusc | [26] |
| 5,9,15,18,21-24:5 | Cellana grata | Marine mollusc | [26] |
| 5,9-25:2 | Chromodoris sp., Phyllidia coelesti | Marine molluscs | [34] |
| Bebryce studeri | Cnidaria (Octocorallia) | [35] | |
| i-5,9-25:2 | Phyllidia coelesti | Marine molluscs | [33] |
| 5,9-26:2 | Heterochone sp. | Marine sponge, Hexactinellida Marine molluscs | [36] |
| Chromodoris sp., Phyllidia coelesti | Cnidaria (Octocorallia) | [34] | |
| Bebryce studeri | [35] | ||
| i-5,9-26:2 | Chromodoris sp., Phyllidia coelesti | Marine molluscs | [34] |
| 5,9,19-26:3 | Bebryce studeri | Cnidaria (Octocorallia) | [35] |
| 5,9-28:2 | Aulosaccus cf. mitsukuri, Heterochone sp., Rosella sp., Sympagella nux | Marine sponges, Hexactinellida Cnidaria (Octocorallia) | [36] |
| Bebryce studeri | [35] | ||
| 5,9,19-28:3 | Bebryce studeri | Cnidaria (Octocorallia) | [35] |
| 5,9,23-28:3 | Hyalonema sp. | Marine sponge, Hexactinellida | [36] |
| 5,9-29:2 | Hyalonema sp. | Marine sponge, Hexactinellida | [36] |
| 5,9,22-29:3 | Acanthascus sp., Aulosaccus cf. mitsukuri, Euplectella sp., Heterochone sp., Hyalonema sp. | Marine sponges, Hexactinellida | [36] |
| 5,9,21-30:3 | Acanthascus sp., Aulosaccus cf. mitsukuri, Euplectella sp., Hyalonema sp., Heterochone sp., Staurocalyptus sp., Sympagella nux | Marine sponges, Hexactinellida | [36] |
| 5,9,23-30:3 | Acanthascus sp., Aulosaccus cf. mitsukuri, Euplectella sp., Farrea sp., Heterochone sp., Hyalonema sp., Ipheteon panicea, Staurocalyptus sp., Sympagella nux | Marine sponges, Hexactinellida | [36] |
| 5,9,25-30:3 | Hyalonema sp. | Marine sponges, Hexactinellida | [36] |
| 5,9-31:2 | Hyalonema sp. | Marine sponge, Hexactinellida | [36] |
| 5,9,21-31:3 | Staurocalyptus sp. | Marine sponge, Hexactinellida | [36] |
| 5,9,22-31:3 | Acanthascus sp., Aulosaccus cf. mitsukuri, | Marine sponges, Hexactinellida | [36] |
| 5,9,23-32:3 | Ipheteon panicea, Staurocalyptus sp. | Marine sponges, Hexactinellida | [36] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kornprobst, J.-M.; Barnathan, G. Demospongic Acids Revisited. Mar. Drugs 2010, 8, 2569-2577. https://doi.org/10.3390/md8102569
Kornprobst J-M, Barnathan G. Demospongic Acids Revisited. Marine Drugs. 2010; 8(10):2569-2577. https://doi.org/10.3390/md8102569
Chicago/Turabian StyleKornprobst, Jean-Michel, and Gilles Barnathan. 2010. "Demospongic Acids Revisited" Marine Drugs 8, no. 10: 2569-2577. https://doi.org/10.3390/md8102569





