Bioactive Compound Synthetic Capacity and Ecological Significance of Marine Bacterial Genus Pseudoalteromonas
Abstract
:1. Introduction
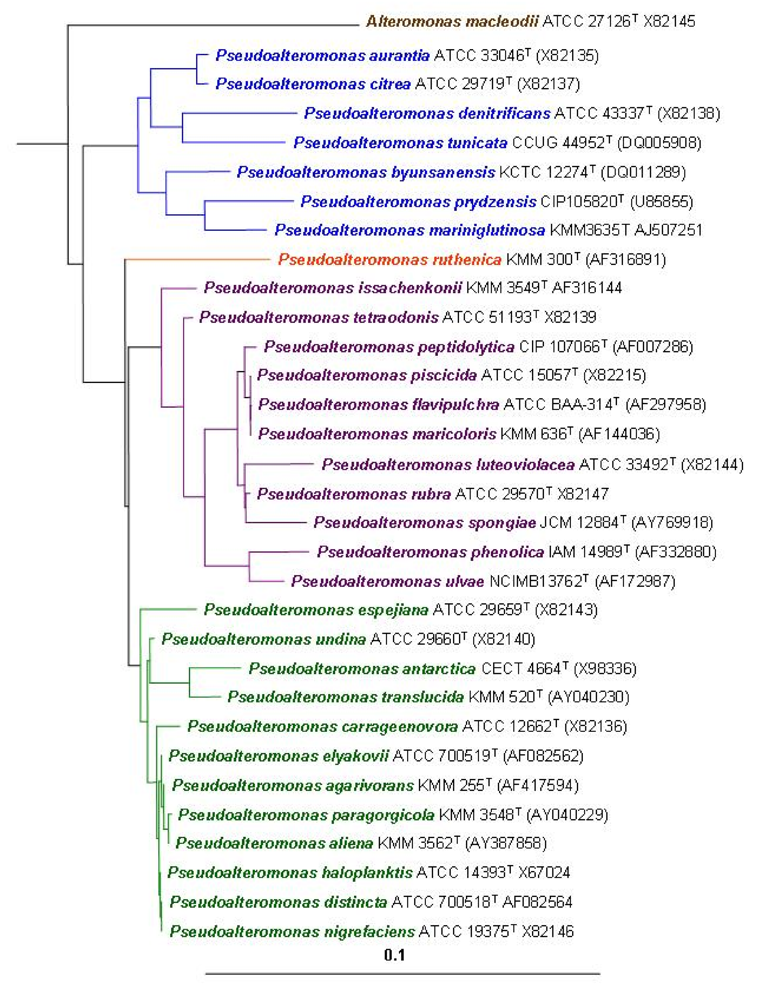
2. Ecological Significance
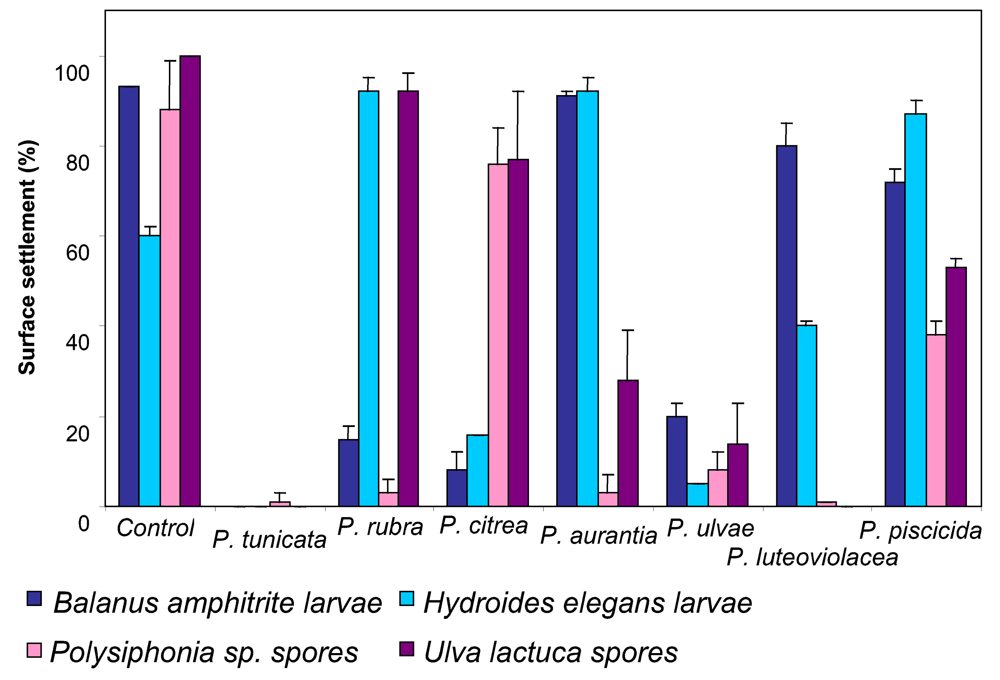
3 . P. tunicata as a model to study the mechanisms and ecology of natural antibiotic production, anti-fouling activity and biofilm formation
4. Natural product compound production by Pseudoalteromonas spp
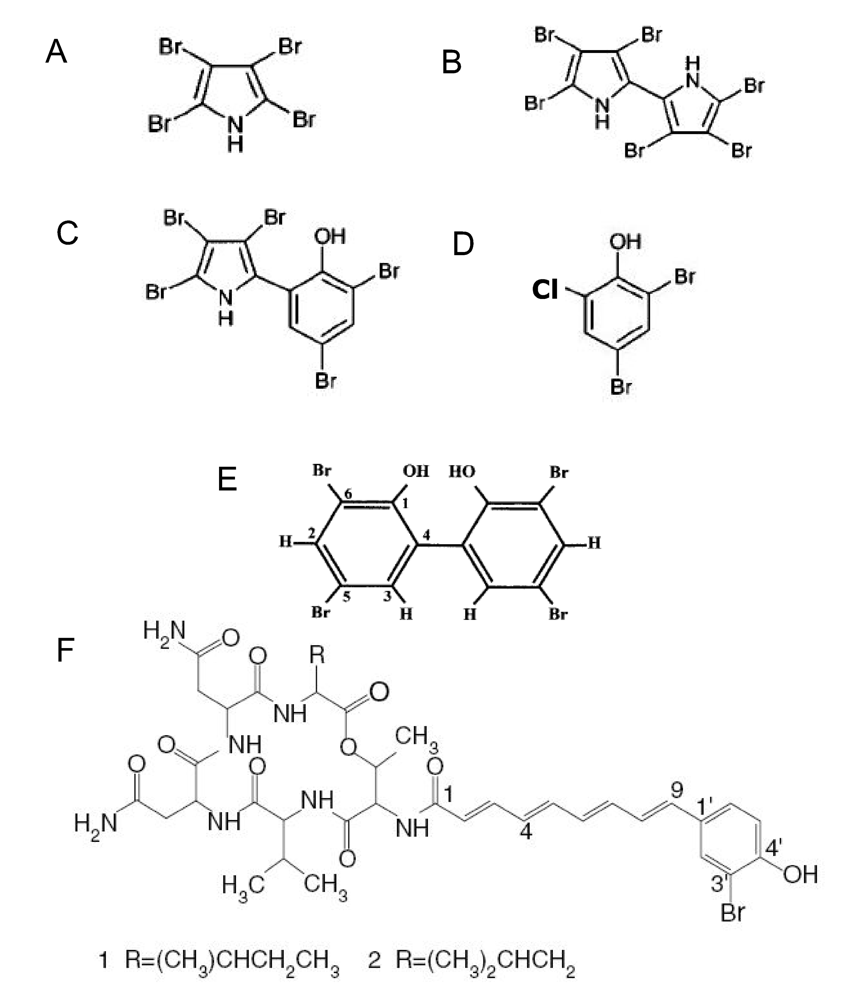
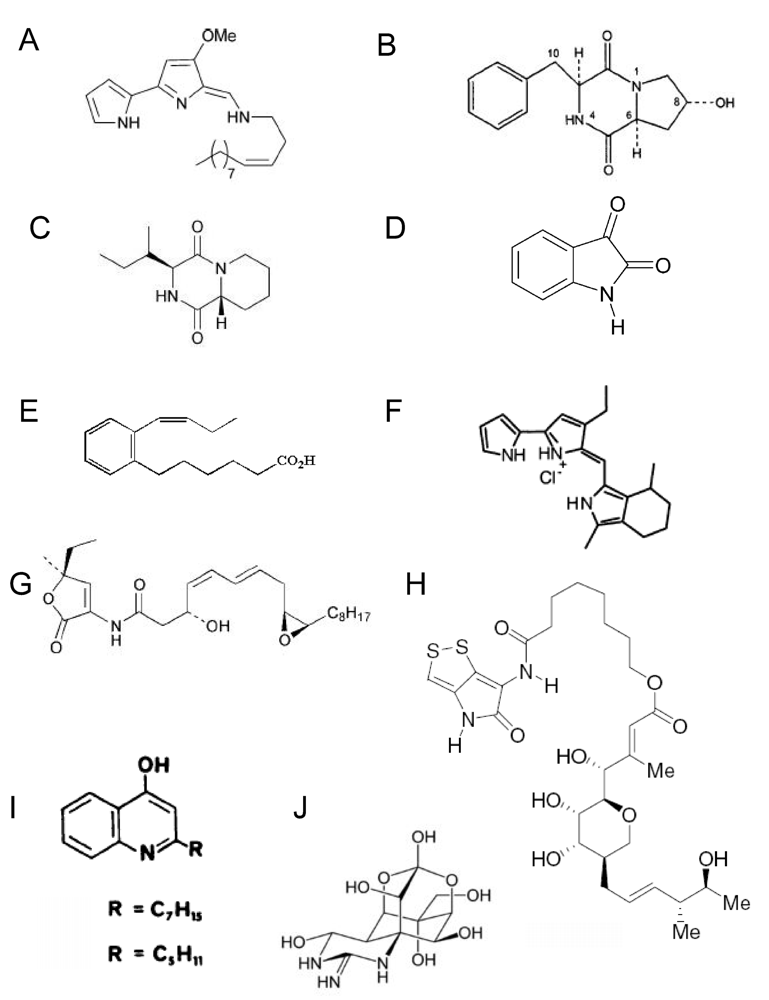
4.1. Low molecular weight substances
4.2 High molecular weight substances
5. Concluding Remarks
- Sample Availability: Available from the author.
References and Notes
- Abraham, TJ. Antibacterial marine bacterium deters luminous vibriosis in shrimp larvae. NAGA WorldFish Cent Quart 2004, 27, 28–31. [Google Scholar]
- Anderson, RJ; Wolfe, MS; Faulkner, DJ. Autotoxic antibiotic production by a marine Chromobacterium. Mar Biol 1974, 27, 281–285. [Google Scholar]
- Ballester, M; Ballester, JM; Belaich, JP. Isolation and characterization of a high molecular weight antibiotic produced by a marine bacterium. Microb Ecol 1977, 3, 289–303. [Google Scholar]
- Barja, JL; Lemos, ML; Toranzo, AE. Purification and characterization of an antibacterial substance produced by a marine Alteromonas species. Antimicrob Agents Chemother 1989, 33, 1674–1679. [Google Scholar]
- Baumann, L; Baumann, P; Mandel, M; Allen, RD. Taxonomy of aerobic marine eubacteria. J Bacteriol 1972, 110, 402–429. [Google Scholar]
- Bayles, KW. The biological role of death and lysis in biofilm development. Nature Rev Microbiol 2007, 5, 721–726. [Google Scholar]
- Bein, SJ. A study of certain chromogenic bacteria isolated from “Red Tide” water with a description of a new species. Bull Mar Sci Gulf Caribb 1954, 4, 110–119. [Google Scholar]
- Bowman, JP; McMeekin, TA. Brenner, DJ, Krieg, NR, Staley, JT, Garrity, GM, Eds.; Bergey’s Manual of Bacteriology, 2Springer: New York, 2005; Volume 2, pp. 467–478. [Google Scholar]
- Buck, JD; Meyers, SP. In vitro inhibition of Rhodotorula minuta by a variant of marine bacterium Pseudomonas piscicida. Helgol Wiss Meeresunters 1966, 13, 171. [Google Scholar]
- Burke, C; Thomas, T; Egan, S; Kjelleberg, S. The use of functional genomics for the identification of a gene cluster encoding for the biosynthesis of an antifungal tambjamine in the marine bacterium Pseudoalteromonas tunicata. Env Microbiol 2007, 9, 814–818. [Google Scholar]
- Burkholder, PR; Pfister, RM; Leitz, LH. Production of a pyrrole antibiotic by a marine bacterium. Appl Microbiol 1966, 14, 649–653. [Google Scholar]
- Burmølle, M; Webb, JS; Rao, D; Hansen, LH; Sorensen, SJ; Kjelleberg, S. Enhanced biofilm formation and increased resistance to antimicrobial agents and bacterial invasion are caused by synergistic interactions in multispecies biofilms. Appl Env Microbiol 2006, 72, 3916–3923. [Google Scholar]
- Campàs, C; Dalmau, M; Montaner, B; Barragán, M; Bellosillo, B; Colomer, D; Pons, G; Pérez-Tomás, R; Gil, J. Prodigiosin induces apoptosis of B and T cells from B-cell chronic lymphocytic leukemia. Leukemia 2003, 17, 746–750. [Google Scholar]
- Colwell, RR; Sparks, AK. Properties of Pseudomonas enalia, a marine bacterium pathogenic for the invertebrate Crassostrea gigas (Thunberg). Appl Microbiol 1967, 15, 980–986. [Google Scholar]
- Costa-Ramos, C; Rowley, AF. Effect of extracellular products of Pseudoalteromonas atlantica on the edible crab Cancer pagurus. Appl Env Microbiol 2004, 70, 729–735. [Google Scholar]
- Demina, NS; Veslopolova, EF; Gaenko, GP. Marine-bacteria Alteromonas piscicida produces thrombolytic enzymes. Izv Akad Nauk SSSR Ser Biologich 1990, 3, 415–419. [Google Scholar]
- Dobretsov, S; Dahms, H-W; Qian, P-Y. Inhibition of biofouling by marine microorganisms and their metabolites. Biofouling 2006, 22, 43–54. [Google Scholar]
- Dobretsov, S; Qian, PY. Effect of bacteria associated with the green alga Ulva reticulata on marine micro- and macrofouling. Biofouling 2002, 18, 217–228. [Google Scholar]
- Dobretsov, S; Xiong, HR; Xu, Y; Levin, LA; Qian, PY. Novel antifoulants: Inhibition of larval attachment by proteases. Mar Biotechnol 2007, 9, 388–397. [Google Scholar]
- Egan, S; Thomas, T; Holmström, C; Kjelleberg, S. Phylogenetic relationship and antifouling activity of bacterial epiphytes from the marine alga Ulva lactuca. Env Microbiol 2000, 2, 343–347. [Google Scholar]
- Egan, S; Holmström, C; Kjelleberg, S. Pseudoalteromonas ulvae sp. nov., a bacterium with antifouling activities isolated from the surface of a marine alga. Int J Syst Evol Microbiol 2001, 51 , 1499–1504. [Google Scholar]
- Egan, S; James, S; Holmström, C; Kjelleberg, S. Correlation between pigmentation and antifouling compounds produced by Pseudoalteromonas tunicata. Env Microbiol 2002, 4, 433–442. [Google Scholar]
- Egan, S; James, S; Kjelleberg, S. Identification and characterization of a putative transcriptional regulator controlling the expression of fouling inhibitors in Pseudoalteromonas tunicata. Appl Env Microbiol 2002, 68, 372–378. [Google Scholar]
- Faulkner, DJ. Sammes, EG, Ed.; In Topics in Antibiotic Chemistry; John Wiley: New York, 1978; Chapter 2. pp. 9–58. [Google Scholar]
- Franks, A; Haywood, P; Holmström, C; Egan, S; Kjelleberg, S; Kumar, N. Isolation and structure elucidation of a novel yellow pigment from the marine bacterium Pseudoalteromonas tunicata. Molecules 2005, 10, 1286–1291. [Google Scholar]
- Franks, A; Egan, S; Holmström, C; James, S; Lappin-Scott, H; Kjelleberg, S. Inhibition of fungal colonization by Pseudoalteromonas tunicata provides a competitive advantage during surface colonization. Appl Env Microbiol 2006, 72, 6079–6087. [Google Scholar]
- Gallacher, S; Birkbeck, TH. A tissue-culture assay for direct detection of sodium-channel blocking toxins in bacterial culture supernates. FEMS Microbiol Lett 1992, 92, 101–108. [Google Scholar]
- Gallacher, S; Birkbeck, TH. Effect of phosphate concentration on production of tetrodotoxin by Alteromonas tetraodonis. Appl Env Microbiol 1993, 59, 3981–3983. [Google Scholar]
- Gauthier, MJ. Alteromonas rubra sp. nov., a new marine antibiotic-producing bacterium. Int J Syst Bacteriol 1976, 26, 459–466. [Google Scholar]
- Gauthier, MJ. Modification of bacterial respiration by a macromolecular polyanionic antibiotic produced by a marine Alteromonas. Antimicrob Agents Chemother 1976, 76, 361–366. [Google Scholar]
- Gauthier, MJ. Alteromonas citrea, a new Gram-negative, yellow-pigmented species from seawater. Int J Syst Bacteriol 1977, 27, 349–354. [Google Scholar]
- Gauthier, MJ. Validation of the name Alteromonas luteoviolacea. Int J Syst Bacteriol 1982, 32 , 82–86. [Google Scholar]
- Gauthier, MJ; Breittmayer, VA. A new antibiotic-producing bacterium from seawater: Alteromonas aurantia sp. nov. Int J Syst Bacteriol 1979, 29, 366–372. [Google Scholar]
- Gauthier, MJ; Flatau, GN. Antibacterial activity of marine violet-pigmented Alteromonas with special reference to the production of brominated compounds. Can J Microbiol 1976, 22, 1612–1619. [Google Scholar]
- Gauthier, G; Gauthier, M; Christen, R. Phylogenetic analysis of the genera Alteromonas, Shewanella and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (emended) and Pseudoalteromonas gen. nov., and proposal of twelve new species combinations. Int J Syst Bacteriol 1995, 45, 755–761. [Google Scholar]
- Gerber, NN; Gauthier, MJ. New prodigiosin-like pigment from Alteromonas rubra. Appl Env Microbiol 1979, 37, 1176–1179. [Google Scholar]
- Gribble, GW. The diversity of naturally occurring organobromine compounds. Chem Soc Rev 1999, 28, 335–348. [Google Scholar]
- Grossart, HP; Kiørboe, T; Tang, K; Ploug, H. Bacterial colonization of particles: growth and interactions. Appl Env Microbiol 2003, 69, 3500–3509. [Google Scholar]
- Hansen, AJ; Weeks, OB; Colwell, RR. Taxonomy of Pseudomonas piscicida (Bein) Buck, Meyers and Leifson. J Bacteriol 1965, 89, 752–761. [Google Scholar]
- Harder, T; Dobretsov, S; Qian, PY. Waterborne polar macromolecules act as algal antifoulants in the seaweed Ulva reticulata. Mar Ecol Prog Ser 2004, 274, 133–141. [Google Scholar]
- Holland, GS; Jamieson, DD; Reichelt, JR; Viset, G; Wells, RJ. Chem Ind (London) 1984, 850.
- Holmström, C; Egan, S; Franks, A; McCloy, S; Kjelleberg, S. Antifouling activities expressed by marine surface associated Pseudoalteromonas species. FEMS Microbiol Ecol 2002, 41, 47–58. [Google Scholar]
- Holmström, C; James, S; Egan, S; Kjelleberg, S. Inhibition of common fouling organisms by marine bacterial isolates with special reference to the role of pigmented bacteria. Biofouling 1996, 10 , 251–259. [Google Scholar]
- Holmström, C; Kjelleberg, S. Marine Pseudoalteromonas species are associated with higher organisms and produce active extracellular agents. FEMS Microbiol Ecol 1999, 30, 285–293. [Google Scholar]
- Holmström, C; James, S; Neilan, B; White, D; Kjelleberg, S. Pseudoalteromonas tunicata sp. nov., a bacterium that produces anti-fouling agents. Int J Syst Bacteriol 1998, 48, 1205–1212. [Google Scholar]
- Huang, YL; Dobretsov, S; Ki, JS; Yang, LH; Qian, PY. Presence of acylhomoserine lactone in subtidal biofilm and the implication in larval behavioral response in the polychaete Hydroides elegans. Microb Ecol 2007, 54, 384–392. [Google Scholar]
- Huang, YL; Dobretsov, S; Xiong, HR; Qian, PY. Effect of biofilm formation by Pseudoalteromonas spongiae on induction of larval settlement of the polychaete Hydroides elegans. Appl Env Microbiol 2007, 73, 6284–6288. [Google Scholar]
- Huggett, MJ; Williamson, JE; de Nys, R; Kjelleberg, S; Steinberg, P. Larval settlement of the common Australian sea urchin Heliocidaris erythrogramma in response to bacteria from the surface of coralline algae. Oecologia 2006, 149, 604–619. [Google Scholar]
- Isnanetyo, A; Kamei, Y. MC21-A, a bactericidal antibiotic produced by a new marine bacterium, Pseudoalteromonas phenolica sp. nov. O-BC30T against methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 2003, 47, 480–488. [Google Scholar]
- Ivanova, EP; Flavier, S; Christen, R. Phylogenetic relationships among marine Alteromonas-like proteobacteria: emended description of the family Alteromonadaceae and proposal of Pseudoalteromonadaceae fam. nov., Colwelliaceae fam. nov., Shewanellaceae fam. nov., Moritellaceae fam. nov., Ferrimonadaceae fam. nov., Idiomarinaceae fam. nov. and Psychromonadaceae fam. nov. Int J Syst Evol Microbiol 2004, 54, 1773–1788. [Google Scholar]
- Ivanova, EP; Gorshkova, NM; Zhukova, NV; Lysenko, AM; Zelepuga, EA; Prokof'eva, NG; Mikhailov, VV; Nicolau, DV; Christens, R. Characterization of Pseudoalteromonas distincta-like sea-water isolates and description of Pseudoalteromonas aliena sp nov. Int J Syst Evol Microbiol 2004, 54, 1431–1437. [Google Scholar]
- James, SG; Holmström, C; Kjelleberg, S. Purification and characterization of a novel antibacterial protein from the marine bacterium D2. Appl Env Microbiol 1996, 62, 2783–2788. [Google Scholar]
- Jiang, Z; Boyd, KG; Mearns-Spragg, A; Adams, DR; Wright, PC; Burgess, JG. Two diketopiperazines and halogenated phenol from cultures of the marine bacterium, Pseudoalteromonas luteoviolacea. Nat Prod Lett 2000, 14, 435–440. [Google Scholar]
- Kalinovskaya, NI; Ivanova, EP; Alexeeva, YV; Gorshkova, NM; Kuznetsova, TA; Dmitronek, AD; Nicolau, DV. Low-molecular-weight, biologically active compounds from marine Pseudoalteromonas species. Curr Microbiol 2004, 48, 441–446. [Google Scholar]
- Kamei, Y; McCarthy, SA; Kakimoto, D; Johnson, R. Inhibition of Paramecium caudatum by an Alteromonas luteoviolacea antibiotic. Antimicrob Agents Chemother 1986, 30, 301–303. [Google Scholar]
- Kawauchi, K; Shibutani, K; Yagisawa, H; Kamata, H; Nakatsuji, S; Anzai, H; Yokoyama, Y; Ikegami, Y; Moriyama, Y; Hirata, H. A possible immunosuppressant, cycloprodigiosin hydrochloride, obtained from Pseudoalteromonas denitrificans. Biochem Biophys Res Comm 1997, 237, 543–547. [Google Scholar]
- Kim, HS; Hayashi, M; Shibata, Y; Wataya, Y; Mitamura, T; Horii, T; Kawauchi, K; Hirata, H; Tsuboi, S; Moriyama, Y. Cycloprodigiosin hydrochloride obtained from Pseudoalteromonas denitrificans is a potent antimalarial agent. Biol Pharm Bull 1999, 22, 532–534. [Google Scholar]
- König, G; Kehraus, S; Seibert, S; Abdel-Lateff, A; Müller, D. Natural products from marine organisms and their associated microbes. Chembiochem 2006, 7, 229–238. [Google Scholar]
- Leitz, T; Wagner, T. The marine bacterium Alteromonas espejiana induces metamorphosis of the hydroid Hydractinia echinata. Mar Biol 1993, 115, 173–178. [Google Scholar]
- Leone, S; Silipo, A; Nazarenko, EL; Lanetta, R; Parrilli, M; Molinaro, A. Molecular structure of endotoxins from Gram-negative, marine bacteria: an update. Mar Drugs 2007, 5, 85–112. [Google Scholar]
- Lindquist, N; Fenical, W. New tambjamine class alkaloids from the marine ascidian Atapozoa sp. and its nudibranch predators – origins of the tambjamines in atapozoa. Experientia 1991, 47, 504–508. [Google Scholar]
- Long, RA; Qureshi, A; Faulkner, DJ; Azam, F. 2-n-pentyl-4-quinolinol produced by a marine Alteromonas sp. and its potential ecological and biogeochemical roles. Appl Environ Microbiol 2003, 69, 568–576. [Google Scholar]
- Longeon, A; Peduzzi, J; Barthelemy, M; Corre, S; Nicolas, JL; Guyot, M. Purification and partial identification of novel antimicrobial protein from marine bacterium Pseudoalteromonas species strain X153. Ma Biotech 2004, 6, 633–641. [Google Scholar]
- Lovejoy, C; Bowman, JP; Hallegraeff, GM. Algicidal effects of a novel marine Pseudoalteromonas isolate (class Proteobacteria, gamma subdivision) on harmful algal bloom species of the genera Chatonella: Gymnodinium: Heterosigma. Appl Environ Microbiol 1998, 64 , 2806–2813. [Google Scholar]
- Lowell, FM. The structure of a bromine rich antibiotic. J Amer Chem Soc 1966, 88, 4510–4511. [Google Scholar]
- Maeda, M; Nogami, K; Kanematsu, M; Hirayama, K. The concept of biological control methods in aquaculture. Hydrobiologia 1997, 358, 285–290. [Google Scholar]
- Maeshima, M; Nakayasu, T; Kawauchi, K; Hirata, H; Shimmen, T. Cycloprodigiosin uncouples H+-pyrophosphatase of plant vacuolar membranes in the presence of chloride ion. 1999. Plant Cell Physiol 1999, 40, 439–442. [Google Scholar]
- Magae, J; Miller, MW; Nagai, K; Shearer, GM. Effect of metacycloprodigiosin, an inhibitor of killer T cells on murine skill and heart transplants. J Antiobiot 1996, 48, 86–90. [Google Scholar]
- Mai-Prochnow, A; Evans, F; Dalisay-Saludes, D; Stelzer, S; Egan, S; James, S; Webb, JS; Kjelleberg, S. Biofilm development and cell death in the marine bacterium Pseudoalteromonas tunicata. Appl Env Microbiol 2004, 70, 3232–3238. [Google Scholar]
- Mai-Prochnow, A; Webb, JS; Ferrari, BC; Kjelleberg, S. Ecological advantages of autolysis during the development and dispersal of Pseudoalteromonas tunicata biofilms. Appl Env Microbiol 2006, 72, 5414–5420. [Google Scholar]
- McCarthy, SA; Johnson, RM; Kakimoto, D. Characterisation of an antibiotic produced by Alteromonas luteoviolacea Gauthier 1982, 85 isolated from Kinko Bay, Japan. J Appl Bacteriol 1994, 77, 426–432. [Google Scholar]
- Meyers, SP; Baslow, MH; Bein, SJ; Marks, CE. Studies of Flavobacterium piscicida Bein. I. Growth, toxicity, and ecological considerations. J Bacteriol 1950, 78, 225–230. [Google Scholar]
- Mitova, M; Tutino, ML; Infusini, G; Marino, G; De Rosa, S. Extracellular peptides from Antarctic psychrophile Pseudoalteromonas haloplanktis. Mar Biotechnol 2005, 7, 523–531. [Google Scholar]
- Nam, YD; Chang, HW; Park, JR; Kwon, HY; Quan, ZX; Park;, YH; Lee, JS; Yoon, JH; Bae, JW. Pseudoalteromonas marina sp. nov., a marine bacterium isolated from tidal flats of the Yellow Sea, and reclassification of Pseudoalteromonas sagamiensis as Algicola sagamiensis comb. nov. Int J Syst Evol Microbiol 2007, 57, 12–18. [Google Scholar]
- Negri, A; Webster, NS; Hill, RT; Heyward, AJ. Metamorphosis of broadcast spawning corals in response to bacteria isolated from crustose algae. Mar Ecol Prog Ser 2001, 223, 121–131. [Google Scholar]
- Nelson, EJ; Ghiorse, WC. Isolation and identification of Pseudoalteromonas piscicida strain Curad associated with diseased damselfish (Pomacentridae) eggs. J Fish Diseases 1999, 22, 253–260. [Google Scholar]
- Novick, NJ; Tyler, ME. Isolation and characterization of Alteromonas luteoviolacea strains with sheathed flagella. Int J Syst Bacteriol 1985, 35, 111–113. [Google Scholar]
- O’Toole, G; Kaplan, HB; Kolter, R. Biofilm formation as microbial development. Annu Rev Microbiol 2000, 54, 49–79. [Google Scholar]
- Patel, P; Callow, ME; Joint, I; Callow, JA. Specificity in larval settlement – modifying response of bacterial biofilms towards zoospores of the marine alga Enteromorpha. Env Microbiol 2004, 5, 338–349. [Google Scholar]
- Perez-Tomas, R; Montaner, B; Llagostera, R; Soto-Cerrato, V. The prodigiosins, proapoptotic drugs with anticancer properties. Biochem Pharmacol 2003, 66, 1447–1452. [Google Scholar]
- Pujalte, MJ; Sitja-Bobadilla, A; Macian, MC; Alvarez-Pellitero, P; Garay, E. Occurrence and virulence of Pseudoalteromonas spp. in cultured gilthead sea bream (Sparus aurata L.) and European sea bass (Dicentrarchus labrax L.). Molecular and phenotypic characterisation of P.undina strain U58. Aquaculture 2007, 271, 47–53. [Google Scholar]
- Qian, PY; Lau, SCK; Dahms, H-U; Dobretsov, S; Harder, T. Marine biofilms as mediators of colonization by marine macroorganisms: Implications for antifouling and aquaculture. Mar Biotechnol 2007, 9, 399–410. [Google Scholar]
- Rao, D; Webb, JS; Kjelleberg, S. Microbial colonization and competition on the marine alga Ulva australis. Appl Env Microbiol 2006, 72, 5547–5555. [Google Scholar]
- Riquelme, C; Hayashida, G; Araya, R; Uchida, A; Satomi, M; Ishida, Y. Isolation of a native bacterial strain from the scallop Argopecten purpuratus with inhibitory effects against pathogenic vibrios. J Shellfish Res 1996, 15, 369–374. [Google Scholar]
- Sakata, T; Sakaguchi, K; Kakimoto, D. Antiobiotic production by marine pigmented bacteria. I. Antibacterial effect of Alteromonas luteoviolaceus. Mem Fac Fish, Kagoshima Univ 1982, 31, 243–250. [Google Scholar]
- Sakata, T; Sakaguchi, K; Kakimoto, D. Antiobiotic production by marine pigmented bacteria. II. Purification and characterization of antibiotic substances of Alteromonas luteoviolacea. Mem Fac Fish, Kagoshima Univ 1986, 35, 29–37. [Google Scholar]
- Saravanan, P; Nancharaiah, YV; Venugopalan, VP; Subba Rao, T; Jayachandran, S. Biofilm formation by Pseudoalteromonas ruthenica and its removal by chlorine. Biofouling 2006, 22, 371–381. [Google Scholar]
- Seipp, S; Wittig, K; Stiening, B; Bottger, A; Leitz, T. Metamorphosis of Hydractinia echinata (Cnidaria) is caspase-dependent. Int J Dev Biol 2006, 50, 63–70. [Google Scholar]
- Shiozawa, H; Kagasaki, T; Kinoshita, T; Haruyama, H; Domon, H; Utsui, Y; Kodama, K; Takahashi, S. Thiomarinol, a new hybrid antimicrobial antibiotic produced by a marine bacterium fermentation, isolation, structure, and antimicrobial activity. J Antibiot 1993, 46, 1834–1842. [Google Scholar]
- Shiozawa, H; Shimada, A; Takahashi, S. Thiomarinol D, E, F and G, new hybrid antimicrobial antibiotics produced by a marine bacterium: isolation, structure and antimicrobial activity. J Antibiot 1997, 50, 449–452. [Google Scholar]
- Simidu, U; Kita-Tsukamoto, K; Yasumoto, T; Yotsu, M. Taxonomy of marine bacteria that produce tetrodotoxin. Int J Syst Bacteriol 1990, 40, 331–336. [Google Scholar]
- Skerratt, JH. Bacterial and algal interaction in a Tasmanian estuary. In PhD Thesis; University of Tasmania, Hobart: Tasmania, Australia, 2001; pp. 219–235. [Google Scholar]
- Skerratt, JH; Bowman, JP; Hallegraeff, G; James, S; Nichols, PD. Algicidal bacteria associated with blooms of a toxic dinoflagellate in a temperate Australian estuary. Mar Ecol Prog Ser 2002, 244, 1–15. [Google Scholar]
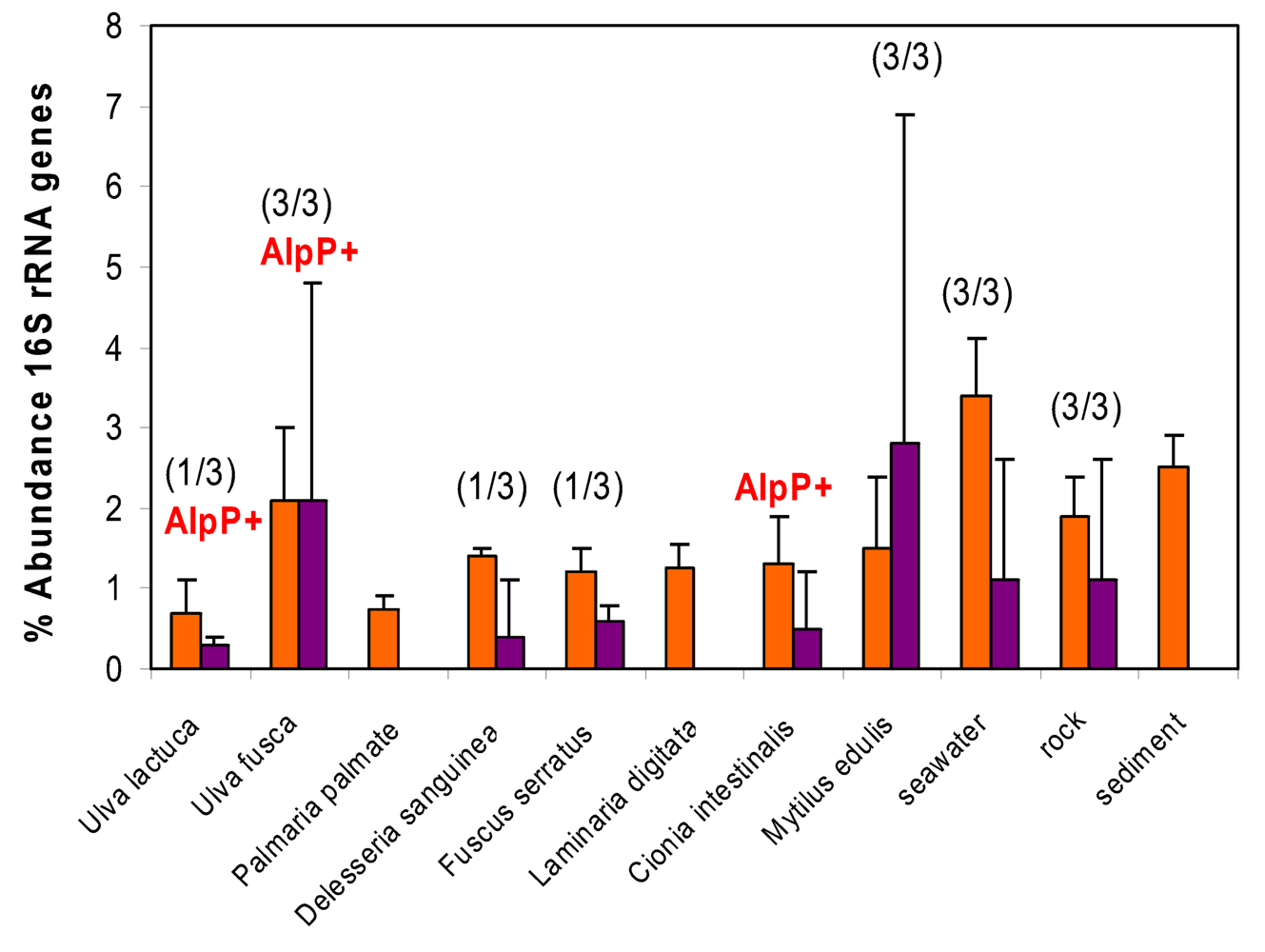
- Skovhus, T; Holmström, C; Kjelleberg, S; Dahllöf, I. Molecular investigation of the distribution, abundance and diversity of the genus Pseudoalteromonas in marine samples. FEMS Microbiol Ecol 2007, 61, 348–361. [Google Scholar]
- Sobolevskaya, MP; Smetanina, OF; Speitling, M; Shevchenko, LS; Dmitrenok, PS; Laatsch, H; Kuznetsova, TA; Ivanova, EP; Elyakov, GB. Controlling production of brominated cyclic depsipeptides by Pseudoalteromonas maricaloris KMM 636T. Lett Appl Microbiol 2005, 40, 243–248. [Google Scholar]
- Stelzer, S; Egan, S; Larsen, MR; Bartlett, DH; Kjelleberg, S. Unravelling the role of the ToxR-like transcriptional regulator WmpR in the marine antifouling bacterium Pseudoalteromonas tunicata. Microbiology 2006, 152, 1385–1394. [Google Scholar]
- Tanigaki, K; Sato, T; Tanaka, Y; Ochi, T; Nishikawa, A; Nagai, K; Kwashima, H; Ohkuma, S. BE-18591 as a new H+/Cl− symport ionophore that inhibits immunoproliferation and gastritis. FEBS Lett 2002, 524, 37–42. [Google Scholar]
- Wieczorek, SK; Todd, CD. Inhibition and facilitation of the settlement of epifaunal marine invertebrate larvae by microbial biofilm cues. Biofouling 1998, 12, 81–93. [Google Scholar]
- Wigglesworth-Cooksey, B; Cooksey, KE. Use of fluorophore-conjugated lectins to study cell-cell interactions in model marine biofilms. Appl Env Microbiol 2005, 71, 428–435. [Google Scholar]
- Williamson, N; Simonsen, H; Ahmed, R; Goldet, G; Slater, H; Woodley, L; Leeper, FJ; Salmond, GPC. Biosynthesis of the red antibiotic, prodigiosin, in Serratia: identification of a novel 2-methyl-n-amyl-pyrrole (MAP) assembly pathway, definition of the terminal condensing enzyme, and implications for undecylprodigiosin biosynthesis in Streptomyces. Mol Microbiol 2005, 56, 971–989. [Google Scholar]
- Wratten, SJ; Wolfe, MS; Andersen, RJ; Faulkner, DJ. Antibiotic metabolites from a marine pseudomonad. Antimicrob Agents Chemother 1977, 11, 411–414. [Google Scholar]
- Yee, LH; Holmström, C; Fuary, ET; Lewin, NC; Kjelleberg, S; Steinberg, PD. Inhibition of fouling by marine bacteria immobilised in kappa-carrageenan beads. Biofouling 2007, 23, 287–294. [Google Scholar]
- Yoshikawa, K; Nakayama, Y; Hayashi, M; Unemoto, T; Mochida, K. Korormicin, an antibiotic specific for Gram-negative marine bacteria, strongly inhibits the respiratory chain-linked Na+-translocating NADH: Quinone reductase from the marine Vibrio alginolyticus. J Antiobiot 1999, 52 , 182–185. [Google Scholar]
- Zapata, M; Silva, F; Luza, Y; Wilkens, M; Riquelme, C. The inhibitory effect of biofilms produced by wild bacterial isolates to the larval settlement of the fouling ascidia Cionia intestinalis and Pyura praeputialis. Electr J Biotechnol 2007, 10, 149–159. [Google Scholar]
| Species | Pigmented a | Source | Bioactive compoundsb | Inhibitory activities [other activities]b |
|---|---|---|---|---|
| P. aliena | + (melanin) | seawater | Unknown compound(s) formed | Anti-tumorigenic activity-Ehrlich ascites carcinoma cell line inhibitedc |
| P. agarivorans | - | seawater, ascidians | -d | -d [degrades algal polysaccharides] |
| P. antarctica | - | seawater, sea-ice, muddy soils, sediment | - | None observede,f [novel polysaccharides, cold-active enzymes] |
| P. atlantica | - | seawater, marine alga | - | May cause opportunistic disease in crabs [strong degrader of algal polysaccharides] |
| P. aurantia | + (yellow) | surface of Ulva lactuca, seawater | Unknown compound(s) formed | Antimicrobial activity; inhibits settlement of invertebrate larvae |
| P. byunsanensis | - | tidal flat sediment | - | - |
| P. carrageenovora | - | seawater, marine alga | None observede [strong degrader of algal polysaccharides] | |
| P. citrea | + (yellow, melanin) | seawater, mussels, ascidians, sponges | Unknown compound(s) formed | Inhibits settlement of invertebrate larvae; cytotoxic against sea urchin [algal polysaccharide degradation] |
| P. denitrificans | + (red) | seawater | high molecular weight polyanionic substance; cycloprodigiosin HCl | Anti-tumorigenic activity; inhibits T-cell/lymphocyte proliferation; anti-malarial activity; induces settlement of sea urchin Heliocidaris erythrogrammag |
| P. distincta | ± (melanin) | sponge | - | - |
| P. elyakovii | - | mussels, marine alga | - | None observede,f |
| P. espejiana | - | seawater | - | None observede,f |
| P. flavipulchra | + (orange) | seawater | - | - |
| P. haloplanktis | - | seawater | novel diketopiperazines | - [Probiotic benefits to shellfish; cold-active enzymes] |
| P. issachenkonii | - | marine alga | isatin; unknown reddish-brown compound | Anti-fungal activity; hemolytic |
| P. luteoviolacea | + (purple, yellow) | seawater, marine alga | toxic antimicrobial protein; brominated pyrrole-containing compounds, 4- benzaldehyde; n-propyl-4-hydroxybenzoate | Antimicrobial activity; inhibits algal spore settlement; cytotoxic against sea urchin Strongylocentrotus intermedius; induces settlement of sea urchin Heliocidaris erythrogrammag |
| P. maricoloris | + (yellow) | sponges | bromo-alterochromides A and B | Antibacterial activity; cytotoxicity against sea urchins |
| P. marina | - | tidal flat sediment | - | - |
| P. mariniglutinosa | - | diatoms | - | - |
| P. nigrifaciens | ± (melanin) | seawater, salted foods, mussels | - | - |
| P. paragorgicola | - | sponge | - | - |
| P. peptidolytica | +(yellow) | seawater | unknown compounds | Antimicrobial activity, hemolytice |
| P. phenolica | + (brown) | seawater | 3,3’,5,5’-tetra-bromo-2,2- biphenyldiol | Antimicrobial activity |
| P. piscicida (and related bacteria) | + (yellow) | estuarine waters, fish samples | toxic protein; possible yellow cyclic/acyclic brominated depsipeptide compounds; unknown anti-algal compound(s) | Antibacterial; algicidal activity; possible cytotoxicity [opportunistic fish pathogen; thrombolytic enzymes] |
| P. rubra | + (red) | seawater | high molecular weight polyanionic substance; cycloprodigiosin HCl; rubrenoic acids | Antimicrobial activity; anti- tumorigenic activity; inhibits T-cell/lymphocyte proliferation; anti-malarial; bronchodilatatorice,f |
| P. ruthenica | +(pale orange) | shellfish | unknown compounds | Antimicrobial activity |
| P. spongiae | + (pale orange) | sponge | - | Strongly induces settlement of Hydroides elegans |
| P. tetraodonis | - | puffer fish | tetrodotoxin | Neurotoxic effectse,f |
| P. translucida | - | seawater | - | - |
| P. tunicata | + (green, purple, yellow) | marine alga, tunicates | unknown purple pigment; tambjamine-like alkaloid YP1; toxic protein AlpP; other unknown sunstances | Anti-fungal, anti-algal, antimicrobial, inhibits settlement of invertebrate larvae and algal spores; inhibits protists |
| P. ulvae | + (purple) | marine alga | unknown substances | Inhibits invertebrate larval settlement and algal spore germination and settlement |
| P. undina | - | seawater, fish | - | hemolytic; [probiotic benefits; possible opportunistic fish pathogen] |
Share and Cite
Bowman, J.P. Bioactive Compound Synthetic Capacity and Ecological Significance of Marine Bacterial Genus Pseudoalteromonas. Mar. Drugs 2007, 5, 220-241. https://doi.org/10.3390/md504220
Bowman JP. Bioactive Compound Synthetic Capacity and Ecological Significance of Marine Bacterial Genus Pseudoalteromonas. Marine Drugs. 2007; 5(4):220-241. https://doi.org/10.3390/md504220
Chicago/Turabian StyleBowman, John P. 2007. "Bioactive Compound Synthetic Capacity and Ecological Significance of Marine Bacterial Genus Pseudoalteromonas" Marine Drugs 5, no. 4: 220-241. https://doi.org/10.3390/md504220




