Extraction of Fucoxanthin from Raw Macroalgae excluding Drying and Cell Wall Disruption by Liquefied Dimethyl Ether
Abstract
:1. Introduction
2. Results and Discussion
2.1. Ethanol Soxhlet Extraction
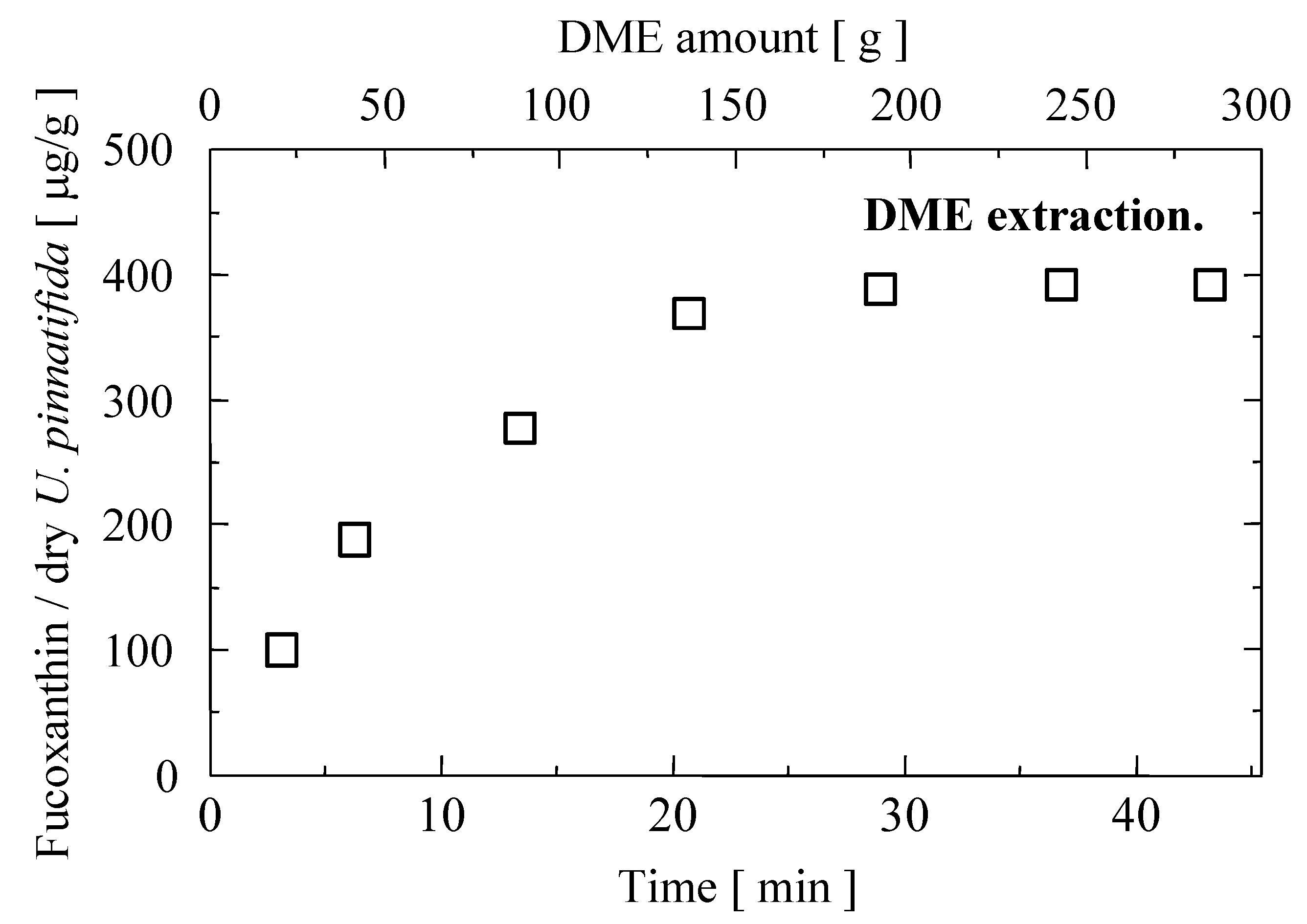
2.2. Liquefied DME Extraction
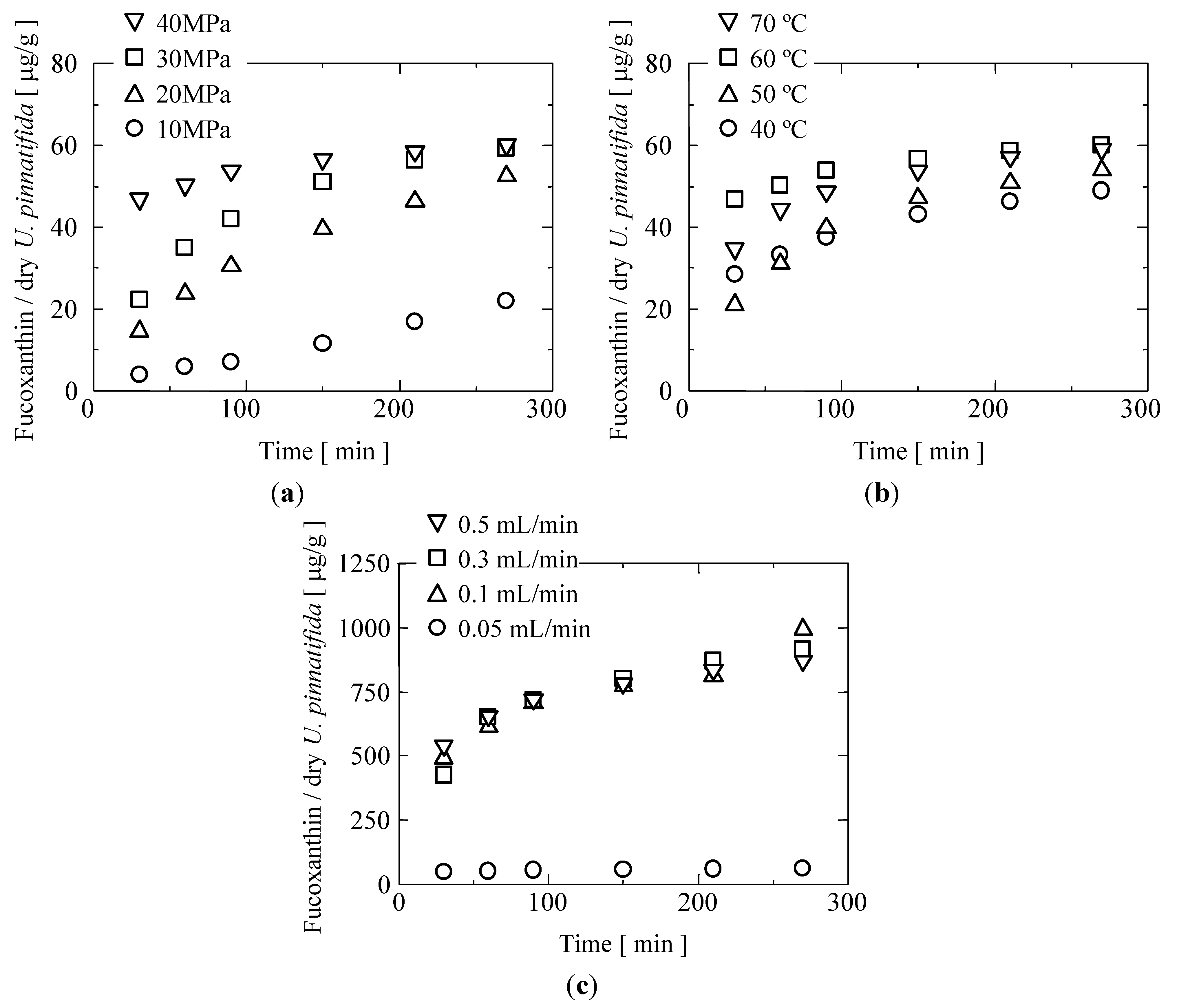
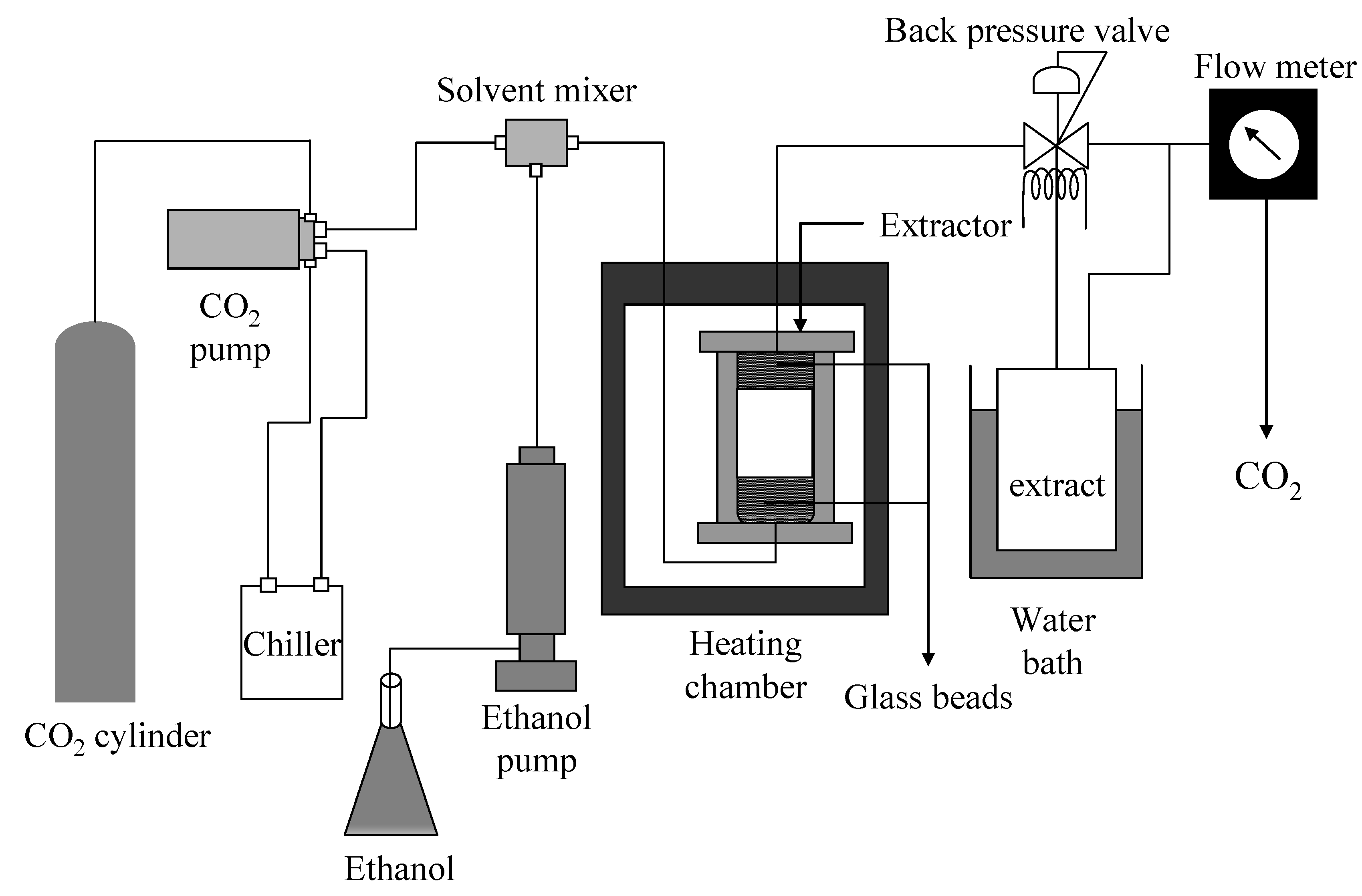
2.3. Supercritical CO2 Extraction
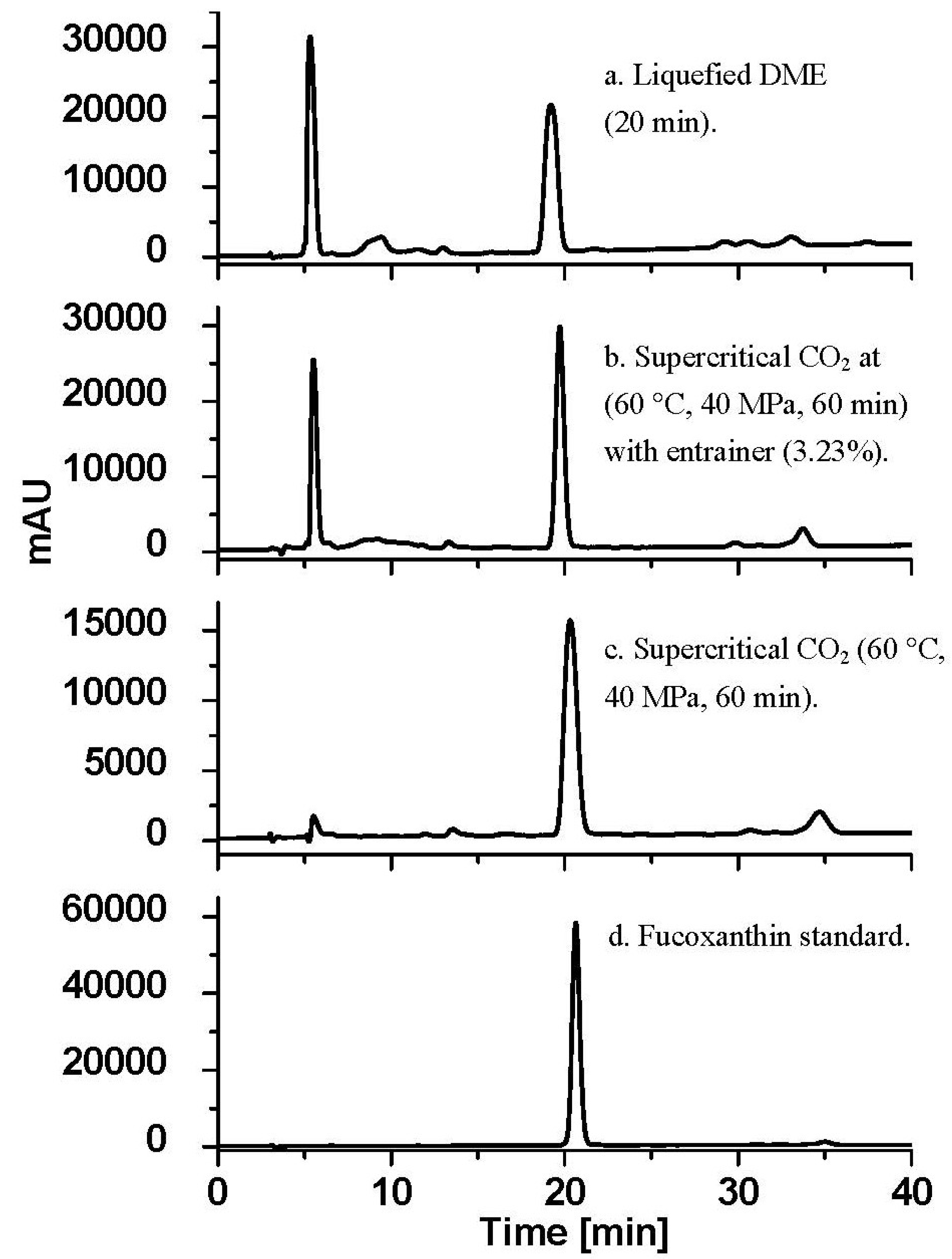
2.4. Comparison of the Results of Three Extraction Techniques
| Extraction Techniques | Time (h) | Temperature (°C) | Pressure (MPa) | Yield of Fucoxanthin (μg/g) |
|---|---|---|---|---|
| Ethanol soxhlet | 12 | 78 | * | 50 |
| Liquefied DME | 0.72 | 25 | * | 390 |
| Supercritical CO2 | 3 | 60 | 40 | 60.12 |
| 3 | 70 | 40 | 59.51 | |
| Supercritical CO2 with entrainer (3.23%) | 3 | 60 | 40 | 994.53 |
3. Experimental Section
3.1. Materials

| Materials | Ultimate Analysis (wet weight%, DAF) | |||
|---|---|---|---|---|
| C (±0.2) | H (±0.1) | N (±0.1) | O * (±0.3) | |
| Original algae | 33.0 | 5.5 | 3.5 | 58.0 |
| Residue | 36.1 | 5.9 | 4.7 | 53.3 |
| Extract | 55.6 | 8.6 | 1.4 | 34.4 |
3.2. Analytical Methods
3.3. Ethanol Soxhlet Extraction
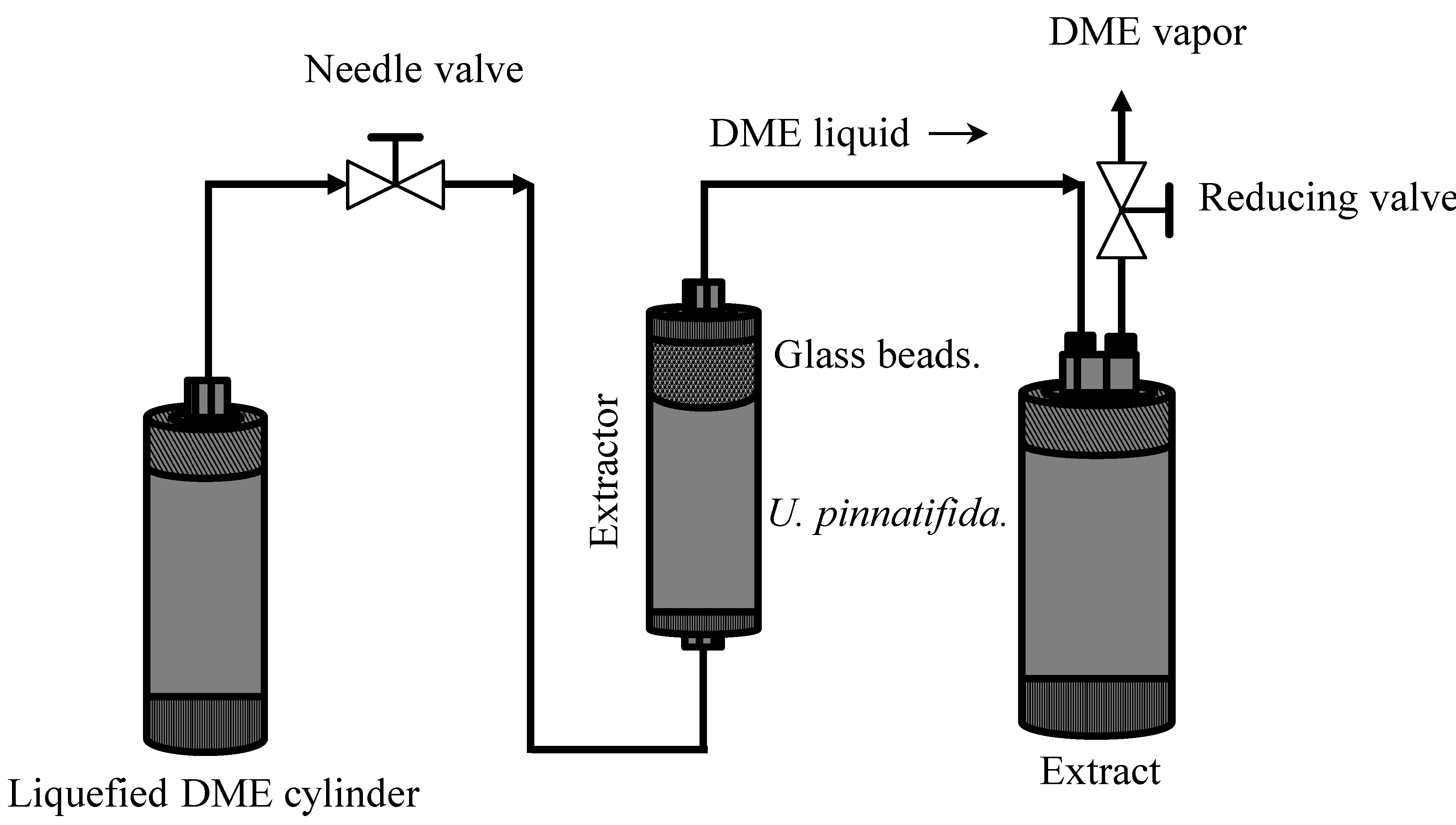
3.4. DME Extraction
3.5. Supercritical CO2 Extraction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maeda, H.; Hosokawa, M.; Sashima, T.; Funayama, K.; Miyashita, K. Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun. 2005, 332, 392–397. [Google Scholar] [CrossRef]
- Yan, X.; Chuda, Y.; Suzuki, M.; Nagata, T. Fucoxanthin as the major antioxidant in Hijikia fusiformis, a common edible seaweed. Biosci. Biotechnol. Biochem. 1999, 63, 605–607. [Google Scholar] [CrossRef]
- Jung, H.A.; Islam, M.N.; Lee, C.M.; Jeong, H.O.; Chung, H.Y.; Woo, H.C.; Choi, J.S. Promising antidiabetic potential of fucoxanthin isolated from the edible brown algae Eisenia bicyclis and Undaria pinnatifida. Fish. Sci. 2012, 78, 1321–1329. [Google Scholar] [CrossRef]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Miyashita, K. Dietary combination of fucoxanthin and fish oil attenuates the weight gain of white adipose tissue and decreases blood glucose in obese/diabetic KK-Ay mice. J. Agric. Food Chem. 2007, 55, 7701–7706. [Google Scholar] [CrossRef]
- Beppu, F.; Hosokawa, M.; Niwano, Y.; Miyashita, K. Effects of dietary fucoxanthin on cholesterol metabolism in diabetic/obese KK-Ay mice. Lipids Health Dis. 2012, 11, 112–119. [Google Scholar] [CrossRef]
- Hu, X.; Li, Y.; Li, C.; Fu, Y.; Cai, F.; Chen, Q.; Li, D. Combination of fucoxanthin and conjugated linoleic acid attenuates body weight gain and improves lipid metabolism in high-fat diet-induced obese rats. Arch. Biochem. Biophys. 2012, 519, 59–65. [Google Scholar] [CrossRef]
- Fung, A.; Hamid, N.; Lu, J. Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem. 2013, 136, 1055–1062. [Google Scholar] [CrossRef]
- Sachindra, N.M.; Sato, E.; Maeda, H.; Hosokawa, M.; Niwano, Y.; Kohno, M.; Miyashita, K. Radical scavenging and singlet oxygen quenching activity of marine carotenoid fucoxanthin and its metabolites. J. Agric. Food Chem. 2007, 55, 8516–8522. [Google Scholar] [CrossRef]
- Hosokawa, M.; Kudo, M.; Maeda, H.; Kohno, H.; Tanaka, T.; Miyashita, K. Fucoxanthin induces apoptosis and enhances the antiproliferative effect of the PPARγ ligand, troglitazone, on colon cancer cells. Biochim. Biophys. Acta 2004, 1675, 113–119. [Google Scholar]
- Kotake-Nara, E.; Kushiro, M.; Zhang, H.; Sugawara, T.; Miyashita, K.; Nagao, A. Carotenoids affect proliferation of human prostate cancer cells. J. Nutr. 2002, 131, 3303–3306. [Google Scholar]
- Shiratori, K.; Ohgami, K.; Ilieva, I.; Jin, X.-H.; Koyama, Y.; Miyashita, K.; Yoshida, K.; Kase, S.; Ohno, S. Effects of fucoxanthin on lipopolysaccharide-induced inflammation in vitro and in vivo. Exp. Eye Res. 2005, 81, 422–428. [Google Scholar] [CrossRef]
- Plaza, M.; Cifuentes, A.; Ibanez, E. In the search of new functional food ingredients from algae. Trends Food Sci. Technol. 2008, 19, 31–39. [Google Scholar] [CrossRef]
- Shang, Y.F.; Kim, S.M.; Lee, W.J.; Um, B.-H. Pressurized liquid method for fucoxanthin extraction from Eisenia bicyclis (Kjellman) Setchell. J. Biosci. Bioeng. 2011, 111, 237–241. [Google Scholar] [CrossRef]
- Plaza, M.; Santoyo, S.; Jaime, L.; Garcia-Blairsy, R.G.; Herrero, M.; Senorans, F.J.; Ibanez, E. Screening for bioactive compounds from algae. J. Pharm. Biomed. Anal. 2010, 51, 450–455. [Google Scholar] [CrossRef]
- Xiao, X.; Si, X.; Yuan, Z.; Xu, X.; Li, G. Isolation of fucoxanthin from edible brown algae by microwave-assisted extraction coupled with high-speed countercurrent chromatography. J. Sep. Sci. 2012, 35, 2313–2317. [Google Scholar] [CrossRef]
- Halim, R.; Harun, R.; Danquah, M.K.; Webley, P.A. Microalgal cell disruption for biofuel development. Appl. Energy 2012, 91, 116–121. [Google Scholar] [CrossRef]
- Halim, R.; Danquah, M.K.; Webley, P.A. Extraction of oil from microalgae for biodiesel production: A review. Biotechnol. Adv. 2012, 30, 709–732. [Google Scholar] [CrossRef]
- Halim, R.; Rupasinghe, T.W.T.; Tull, D.L.; Webley, P.A. Mechanical cell disruption for lipid extraction from microalgal biomass. Bioresour. Technol. 2013, 140, 53–63. [Google Scholar] [CrossRef]
- Hii, S.-L.; Choong, P.-Y.; Woo, K.-K.; Wong, C.-L. Stability studies of fucoxanthin from Sargassum Binderi. Aust. J. Basic Appl. Sci. 2010, 4, 4580–4584. [Google Scholar]
- Billakanti, J.M.; Catchpole, O.; Fenton, T.; Mitchell, K. Extraction of fucoxanthin from Undaria pinnatifida using enzymatic pre-treatment followed by DME and EtOH co-solvent extraction. In Proceedings of the 10th International Symposium on Supercritical Fluids, San Fransisco, CA, USA, 13–16 May 2012; King, J., Ed.; CASSS: Emeryville, CA, USA, 2012. [Google Scholar]
- Wu, J.; Zhou, Y.; Lemmon, E.W. An equation of state for the thermodynamic properties of dimethyl ether. J. Phys. Chem. Ref. Data 2011, 40, 023104. [Google Scholar] [CrossRef]
- Eltringham, W.; Catchpole, O.J. Relative permittivity measurements of gaseous, liquid, and supercritical dimethyl ether. J. Chem. Eng. Data 2007, 52, 363–367. [Google Scholar] [CrossRef]
- Holldorff, H.; Knapp, H. Binary vapor-liquid-liquid equilibrium of dimethyl ether-water and mutual solubilities of methyl chloride and water: Experimental results and data reduction. Fluid Phase Equilibria 1988, 44, 195–209. [Google Scholar] [CrossRef]
- European Food Safety Autorithy. Scientific opinion of the panel on food contact materials, enzymes, flavourings and processing aids (CEF) on dimethyl ether as an extraction solvent. EFSA J. 2009, 984, 1–13. [Google Scholar]
- Varlet, V.; Smith, F.; Augsburger, M. New trends in the kitchen: Propellants assessment of edible food aerosol sprays used on food. Food Chem. 2014, 142, 311–317. [Google Scholar] [CrossRef]
- Naito, M.; Radcliffe, C.; Wada, Y.; Hoshino, T.; Liu, X.; Arai, M.; Tamura, M. A comparative study on the autoxidation of dimethyl ether (DME) comparison with diethyl ether (DEE) and diisopropyl ether (DIPE). J Loss Prev. Process Ind. 2005, 18, 469–473. [Google Scholar] [CrossRef]
- Li, P.; Kanda, H.; Makino, H. Simultaneous production of bio-solid fuel and bio-crude from vegetal biomass using liquefied dimethyl ether. Fuel 2014, 116, 370–376. [Google Scholar] [CrossRef]
- Mendes, R.L.; Fernandes, H.L.; Coelho, J.P.; Reis, E.C.; Cabral, J.M.S.; Novais, J.M.; Palavra, A.F. Supercritical CO2 extraction of carotenoids and other lipids from Chlorella vulgaris. Food Chem. 1995, 53, 99–103. [Google Scholar] [CrossRef]
- Machmudah, S.; Shotipruk, A.; Goto, M.; Sasaki, M.; Hirose, T. Extraction of Astaxanthin from Haematococcus pluvialis using supercritical CO2 and ethanol as entrainer. Ind. Eng. Chem. Res. 2006, 45, 3652–3657. [Google Scholar] [CrossRef]
- Kitada, K.; Machmudah, S.; Sasaki, M.; Goto, M.; Nakashima, Y.; Kumamoto, S.; Hasegawa, T. Supercritical CO2 extraction of pigment components with pharmaceutical importance from Chlorella vulgaris. J. Chem. Technol. Biotechnol. 2009, 84, 657–661. [Google Scholar] [CrossRef]
- Roh, M.K.; Uddin, M.S.; Chun, B.S. Extraction of fucoxanthin and polyphenol from Undaria pinnatifida using supercritical carbon dioxide with co-solvent. Biotechnol. Bioprocess Eng. 2008, 13, 724–729. [Google Scholar] [CrossRef]
- Richter, B.E.; Jones, B.A.; Ezell, J.L.; Avdalovic, N.; Pohl, C. Accelerated solvent extraction: A technique for sample preparation. Anal. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- Kim, S.M.; Jung, Y.J.; Kwon, O.N.; Cha, K.H.; Um, B.H.; Chung, D.; Pan, C.H. A potential commercial source of fucoxanthin extracted from the microalga Phaeodactylum tricornutum. Appl. Biochem. Biotechnol. 2012, 166, 1843–1855. [Google Scholar] [CrossRef]
- Kanazawa, K.; Ozaki, Y.; Hashimoto, T.; Das, S.K.; Matsushita, S.; Hirano, M.; Okada, T.; Komoto, A.; Mori, N.; Nakatsuka, M. Commercial-scale preparation of biofunctional fucoxanthin from waste parts of brown sea algae Laminalia japonica. Food Sci. Technol. Res. 2008, 14, 573–582. [Google Scholar] [CrossRef]
- Bai, S.; Craig, M.V.; Liu, L.F.; Mayne, C.L.; Pugmire, R.J.; Grant, D.M. CO2 clustering of 1-decanol and methanol in supercritical fluids by 13C nuclear spin-lattice relaxation. J. Phys. Chem. 1997, 101, 2923–2928. [Google Scholar] [CrossRef]
- Bulgarevicg, D.S.; Sako, T.; Sujeta, T.; Otake, K.; Takebayashi, Y.; Kamizawa, C.; Horikawa, Y.; Kato, M. The role or general hydrogen-bonding interaction in the solvation process of organic compounds by supercritical CO2/n-alcohol mixtures. Ind. Eng. Chem. Res. 2002, 41, 2074–2081. [Google Scholar] [CrossRef]
- Nehari, A.A.; Kim, S.B.; Lee, Y.B.; Lee, H.T.; Chun, B.S. Characterization of oil including astaxanthin extracted from krill (Euphausia superba) using supercritical carbon dioxide and organic solvent as comparative method. Korean J. Chem. Eng. 2012, 29, 329–336. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kanda, H.; Kamo, Y.; Machmudah, S.; Wahyudiono; Goto, M. Extraction of Fucoxanthin from Raw Macroalgae excluding Drying and Cell Wall Disruption by Liquefied Dimethyl Ether. Mar. Drugs 2014, 12, 2383-2396. https://doi.org/10.3390/md12052383
Kanda H, Kamo Y, Machmudah S, Wahyudiono, Goto M. Extraction of Fucoxanthin from Raw Macroalgae excluding Drying and Cell Wall Disruption by Liquefied Dimethyl Ether. Marine Drugs. 2014; 12(5):2383-2396. https://doi.org/10.3390/md12052383
Chicago/Turabian StyleKanda, Hideki, Yuichi Kamo, Siti Machmudah, Wahyudiono, and Motonobu Goto. 2014. "Extraction of Fucoxanthin from Raw Macroalgae excluding Drying and Cell Wall Disruption by Liquefied Dimethyl Ether" Marine Drugs 12, no. 5: 2383-2396. https://doi.org/10.3390/md12052383
APA StyleKanda, H., Kamo, Y., Machmudah, S., Wahyudiono, & Goto, M. (2014). Extraction of Fucoxanthin from Raw Macroalgae excluding Drying and Cell Wall Disruption by Liquefied Dimethyl Ether. Marine Drugs, 12(5), 2383-2396. https://doi.org/10.3390/md12052383





