Transcriptional Changes Caused by Bisphenol A in Oryzias javanicus, a Fish Species Highly Adaptable to Environmental Salinity
Abstract
:1. Introduction
2. Results and Discussion
| COG Groups * | Description | SW | FW | ||
|---|---|---|---|---|---|
| Gene Count | (%) | Gene Count | (%) | ||
| Information storage and processing | Translation, ribosomal structure and biogenesis | 56 | 15 | 16 | 11 |
| RNA processing and modification | 11 | 2 | |||
| Transcription | 8 | 4 | |||
| Replication, recombination and repair | 3 | 0 | |||
| Chromatin structure and dynamics | 1 | 1 | |||
| Cellular processes and signaling | Cell cycle control, cell division, chromosome partitioning | 14 | 34 | 3 | 31 |
| Defense mechanisms | 19 | 11 | |||
| Signal transduction mechanisms | 69 | 21 | |||
| Cell wall/membrane/envelope biogenesis | 2 | 0 | |||
| Cell motility | 0 | 1 | |||
| Cytoskeleton | 14 | 10 | |||
| Extracellular structures | 3 | 1 | |||
| Intracellular trafficking, secretion, and vesicular transport | 12 | 6 | |||
| Posttranslational modification, protein turnover, chaperones | 46 | 14 | |||
| Metabolism | Energy production and conversion | 35 | 28 | 15 | 31 |
| Carbohydrate transport and metabolism | 11 | 7 | |||
| Amino acid transport and metabolism | 25 | 9 | |||
| Nucleotide transport and metabolism | 7 | 2 | |||
| Coenzyme transport and metabolism | 0 | 1 | |||
| Lipid transport and metabolism | 38 | 17 | |||
| Inorganic ion transport and metabolism | 9 | 6 | |||
| Secondary metabolites biosynthesis, transport and catabolism | 22 | 9 | |||
| Poorly characterized | General function prediction only | 20 | 23 | 17 | 27 |
| Function unknown | 108 | 42 | |||
| Total | - | 533 | 100 | 215 | 100 |
| Functions | Best Hit Description | SW | FW |
|---|---|---|---|
| Information storage and processing | Crocodylus porosus 28S ribosomal RNA | +1.84 ± 0.07 | +1.71 ± 0.03 |
| Oncorhynchus mykiss 28S ribosomal RNA | +1.82 ± 0.05 | +1.83 ± 0.04 | |
| Solea senegalensis ribosomal protein S25 | −1.84 ± 0.07 | −1.72 ± 0.07 | |
| Cellular processes and signaling | Danio rerio fibronectin 1b | −1.84 ± 0.07 | −2.05 ± 0.03 |
| Danio rerio nardilysin | −1.91 ± 0.08 | −1.62 ± 0.03 | |
| Danio rerio signal recognition particle 72 | −1.63 ± 0.01 | −1.52 ± 0.04 | |
| Oncorhynchus mykiss leukocyte elastase inhibitor | −2.03 ± 0.04 | −1.63 ± 0.05 | |
| Oreochromis niloticus decorin | +2.82 ± 0.07 | +6.74 ± 0.04 | |
| Oryzias javanicus complement C5 precursor | +1.51 ± 0.09 | +2.05 ± 0.03 | |
| Oryzias javanicus peptidylprolyl isomerase domain and WD repeat containing 1 | −1.55 ± 0.06 | −1.54 ± 0.02 | |
| Paralichthys olivaceus complement component C8 beta | +2.01 ± 0.04 | +1.52 ± 0.01 | |
| Paralichthys olivaceus fibrinogen beta chain precursor | +2.22 ± 0.07 | +1.52 ± 0.07 | |
| Salmo salar homocysteine-responsive endoplasmic reticulum-resident ubiquitin-like domain member 1 protein | −1.51 ± 0.04 | −1.55 ± 0.06 | |
| Salmo salar transforming growth factor-beta-induced protein ig-h3 | +2.02 ± 0.03 | +1.54 ± 0.07 | |
| Metabolism | Anoplopoma fimbria ATP synthase lipid-binding protein, mitochondrial precursor | +1.55 ± 0.05 | +1.56 ± 0.08 |
| Chaetodon mertensii cytochrome P450 CYP2N | +1.61 ± 0.01 | +1.54 ± 0.07 | |
| Danio rerio 5,10-methylenetetrahydrofolate reductase-like | −2.04 ± 0.06 | −2.33 ± 0.04 | |
| Homo sapiens ATP citrate lyase, transcript variant 2 | +2.82 ± 0.04 | +1.52 ± 0.07 | |
| Oryzias javanicus angiotensin I converting enzyme (peptidyl-dipeptidase A) 1 | +1.51 ± 0.05 | +2.01 ± 0.07 | |
| Oryzias javanicus arylamine N-acetyl transferase | −1.82 ± 0.02 | −2.11 ± 0.08 | |
| Osmerus mordax glycerol-3-phosphate dehydrogenase, cytoplasmic | −3.04 ± 0.03 | −1.84 ± 0.04 | |
| Paralichthys olivaceus fibrinogen beta chain precursor | +2.21 ± 0.07 | +1.54 ± 0.07 | |
| Paralichthys olivaceus ornithine decarboxylase antizyme large isoform | −1.91 ± 0.05 | −1.53 ± 0.08 | |
| Takifugu rubripes apolipoprotein A-IV4 | +2.92 ± 0.04 | +3.92 ± 0.09 | |
| Poorly characterized | Anoplopoma fimbria nuclear protein 1 | −2.94 ± 0.01 | −1.52 ± 0.05 |
| Fundulus heteroclitus TBT-binding protein | +4.21 ± 0.04 | +1.81 ± 0.04 | |
| Homo sapiens genomic sequence surrounding NotI site, clone NL6-EJ23R | +1.62 ± 0.01 | +1.54 ± 0.08 | |
| Oryzias javanicus BCSC-1 isoform 1 | −1.83 ± 0.08 | −1.74 ± 0.02 | |
| Oryzias javanicus choriogenin H | −1.82 ± 0.01 | −2.04 ± 0.03 | |
| Oryzias javanicus tumor suppressor | −1.51 ± 0.05 | −1.54 ± 0.03 | |
| Tetraodon nigroviridis full-length cDNA | −1.71 ± 0.02 | −2.04 ± 0.05 | |
| Tetraodon nigroviridis full-length cDNA | −1.62 ± 0.03 | −1.63 ± 0.04 | |
| Tetraodon nigroviridis full-length cDNA | +3.82 ± 0.08 | +1.54 ± 0.01 |
| Functions | Best Hit Description | SW | FW |
|---|---|---|---|
| Information storage and processing | Oryzias javanicus 40S ribosomal protein S18 | −2.41 ± 0.03 | +1.81 ± 0.08 |
| Oryzias javanicus 60S ribosomal protein L23 | −1.92 ± 0.07 | +1.82 ± 0.04 | |
| Pimephales promelas BTEB transcription factor | −1.93 ± 0.01 | +1.81 ± 0.01 | |
| Salmo salar ribosomal protein L13a | −1.82 ± 0.05 | +1.82 ± 0.06 | |
| Scophthalmus maximus 60S ribosomal protein L14 | −1.84 ± 0.09 | +1.92 ± 0.03 | |
| Solea senegalensis ribosomal protein L13 | −2.01 ± 0.09 | +1.83 ± 0.09 | |
| Cellular processes and signaling | Carassius auratus protein phosphatase 2 regulatory subunit B beta | −2.52 ± 0.08 | +1.84 ± 0.07 |
| Danio rerio RAB8A, member RAS oncogene family | −1.83 ± 0.07 | +1.83 ± 0.01 | |
| Danio rerio transient receptor potential cation channel, subfamily M, member 7 | −1.84 ± 0.06 | +1.73 ± 0.09 | |
| Monodelphis domestica heat shock protein 84b | +5.51 ± 0.03 | −1.82 ± 0.00 | |
| Mus musculus WAPL protein | −1.83 ± 0.07 | +2.14 ± 0.08 | |
| Oryzias javanicus alpha-2-macroglobulin | +2.03 ± 0.03 | −1.93 ± 0.03 | |
| Oryzias javanicus complement C3-S | +1.93 ± 0.04 | −1.94 ± 0.01 | |
| Oryzias javanicus complement factor H-related protein | +1.94 ± 0.02 | −2.36 ± 0.05 | |
| Oryzias javanicus estrogen receptor alpha | +1.65 ± 0.09 | −1.95 ± 0.05 | |
| Oryzias javanicus protein kinase C and casein kinase substrate in neurons 2 | +2.15 ± 0.02 | −1.86 ± 0.03 | |
| Oryzias javanicus translocase of inner mitochondrial membrane 13 homolog | +1.75 ± 0.06 | −1.86 ± 0.06 | |
| Oryzias latipes hox gene cluster | +1.65 ± 0.06 | −1.67 ± 0.01 | |
| Oryzias latipes translationally-controlled tumor protein | −1.72 ± 0.04 | +1.83 ± 0.05 | |
| Solea senegalensis glutathione S transferase Rho-class | +6.94 ± 0.06 | −1.84 ± 0.09 | |
| Metabolism | Danio rerio importin 7 (ipo7), mRNA | +1.73 ± 0.07 | −1.84 ± 0.03 |
| Danio rerio phospholipase A2, group XIIB | −1.53 ± 0.08 | +1.52 ± 0.07 | |
| Danio rerio uridine phosphorylase 2 | −3.72 ± 0.07 | +2.91 ± 0.08 | |
| Oryzias javanicus apolipoprotein A1 precursor | +1.85 ± 0.04 | −1.92 ± 0.07 | |
| Oryzias javanicus phosphatidylinositol transfer protein beta isoform | +1.91 ± 0.05 | −1.82 ± 0.03 | |
| Oryzias javanicus phytanoyl-CoA hydroxylase | −1.94 ± 0.05 | +1.83 ± 0.03 | |
| Oryzias javanicus potassium large conductance calcium-activated channel, subfamily M, alpha member 1 | +1.95 ± 0.08 | −2.34 ± 0.04 | |
| Pseudopleuronectes americanus apolipoprotein A1 precursor | +2.02 ± 0.04 | −1.82 ± 0.02 | |
| Salmo salar acetyl-CoA acetyltransferase | +1.84 ± 0.06 | −1.94 ± 0.08 | |
| Spisula solidissima mRNA for nerve hemoglobin (nHb gene) | −1.91 ± 0.08 | +2.11 ± 0.02 | |
| Poorly characterized | Danio rerio arno protein (ARF exchange factor) | +1.83 ± 0.08 | −1.84 ± 0.01 |
| Danio rerio DNA-directed RNA polymerase II subunit RPB2 | −1.82 ± 0.08 | +1.94 ± 0.08 | |
| Mus musculus BAC clone RP24-324J2 from chromosome 10 | −1.84 ± 0.04 | +1.96 ± 0.07 | |
| Mus musculus clone RP23-105K11 | +1.92 ± 0.08 | −1.83 ± 0.04 | |
| Oryzias javanicus alanine-glyoxylate aminotransferase | +1.81 ± 0.02 | −1.92 ± 0.02 | |
| Oryzias javanicus TAR DNA binding protein | +1.82 ± 0.09 | −2.01 ± 0.04 | |
| Oryzias javanicus type II iodothyronine deiodinase | +1.73 ± 0.09 | −1.91 ± 0.04 | |
| Oryzias melastigma telomerase reverse transcriptase | −4.31 ± 0.01 | +1.85 ± 0.05 | |
| Paralichthys olivaceus heparin cofactor II | −1.81 ± 0.04 | +2.44 ± 0.08 | |
| Sparus aurata delta 6-desaturase | +1.92 ± 0.06 | −6.21 ± 0.09 | |
| Tetraodon nigroviridis full-length cDNA | −1.85 ± 0.04 | +1.83 ± 0.08 | |
| Tetraodon nigroviridis full-length cDNA | −1.84 ± 0.08 | +2.12 ± 0.04 | |
| Tetraodon nigroviridis full-length cDNA | +1.96 ± 0.08 | −1.82 ± 0.07 | |
| Oryzias latipes HN1-like protein | +1.92 ± 0.01 | −2.01 ± 0.04 |

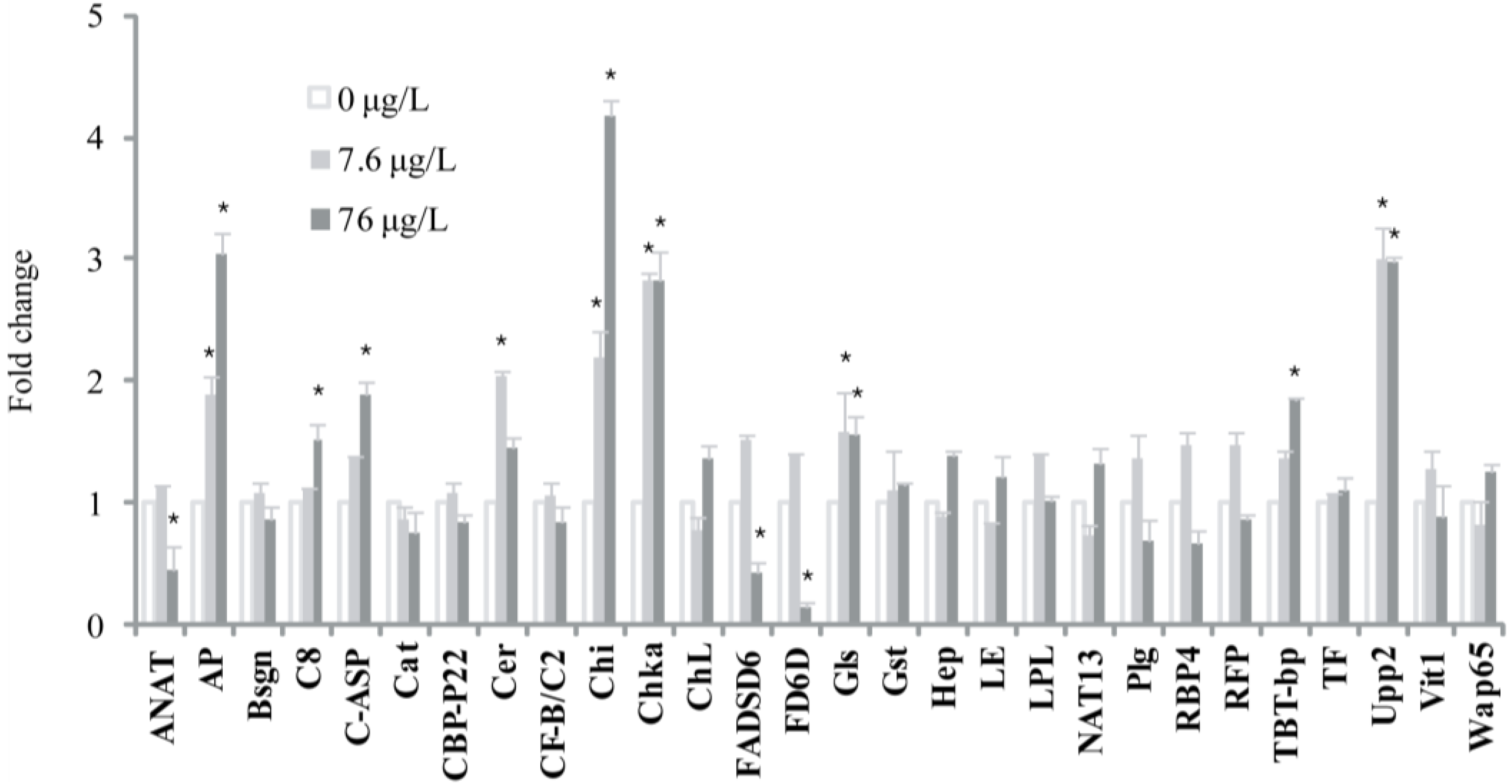
3. Materials and Methods
3.1. Animals, Exposure to BPA, and RNA Preparation
3.2. Development of cDNA Libraries
3.3. Preparation of cDNA Probes and Microarray Hybridization
3.4. Data Acquisition and Identification of Differentially Expressed Genes
3.5. Gene Ontology Clustering
3.6. Quantitative Real-Time Reverse-Transcription PCR and Statistical Analysis
4. Conclusions
Supplementary Files
Acknowledgments
Conflicts of Interest
References
- Crews, D.; Willingham, E.; Skipper, J.K. Endocrine disruptors: Present issues, future directions. Q. Rev. Biol. 2000, 75, 243–260. [Google Scholar]
- Kazeto, Y.; Place, A.R.; Trant, J.M. Effects of endocrine disrupting chemicals on the expression of CYP19 genes in zebrafish (Danio rerio) juveniles. Aquat. Toxicol. 2004, 69, 25–34. [Google Scholar] [CrossRef]
- Tabb, M.M.; Blumberg, B. New modes of action for endocrine-disrupting chemicals. Mol. Endocrinol. 2006, 20, 475–482. [Google Scholar] [CrossRef]
- Hansen, H.J.; Olsen, A.G.; Willumsen, N.J. The influence of ambient salinity and temperature on lipid metabolism in toad (Bufo bufo) skin. Is phosphatidylethanolamine an endogenous regulator of ion channels? Comp. Biochem. Physiol. Comp. Physiol. 1994, 108, 599–608. [Google Scholar]
- Stueckle, T.A.; Shock, B.; Foran, C.M. Multiple stressor effects of methoprene, permethrin, and salinity on limb regeneration and molting in the mud fiddler crab (Uca pugnax). Environ. Toxicol. Chem. 2009, 28, 2348–2359. [Google Scholar] [CrossRef]
- DeLorenzo, M.E.; Danese, L.E.; Baird, T.D. Influence of increasing temperature and salinity on herbicide toxicity in estuarine phytoplankton. Environ. Toxicol. 2011, 28, 359–371. [Google Scholar]
- Lavado, R.; Maryoung, L.A.; Schlenk, D. Hypersalinity acclimation increases the toxicity of the insecticide phorate in Coho salmon (Oncorhynchus kisutch). Environ. Sci. Technol. 2011, 45, 4623–4629. [Google Scholar] [CrossRef]
- Roberts, T.R. Systemic observations on tropical Asian medakas or ricefish of the genus Oryzias with descriptions of four new species. Icthyol. Res. 1998, 45, 213–224. [Google Scholar] [CrossRef]
- Inoue, K.; Takei, Y. Diverse adaptability in Oryzias species to high environmental salinity. Zool. Sci. 2002, 19, 727–734. [Google Scholar] [CrossRef]
- Kishida, M.; McLellan, M.; Miranda, J.A.; Callard, G.V. Estrogen and xenoestrogens upregulate the brain aromatase isoform (P450aromB) and perturb markers of early development in zebrafish (Danio rerio). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 129, 261–268. [Google Scholar] [CrossRef]
- Kundakovic, M.; Champagne, F.A. Epigenetic perspective on the developmental effects of bisphenol A. Brain Behav. Immun. 2011, 25, 1084–1093. [Google Scholar] [CrossRef]
- Mills, L.J.; Chichester, C. Review of evidence: Are endocrine-disrupting chemicals in the aquatic environment impacting fish populations? Sci. Total Environ. 2005, 343, 1–34. [Google Scholar] [CrossRef]
- Cheshenko, K.; Pakdel, F.; Segner, H.; Kah, O.; Eggen, R.I. Interference of endocrine disrupting chemicals with aromatase CYP19 expression or activity, and consequences for reproduction of teleost fish. Gen. Comp. Endocrinol. 2008, 155, 31–62. [Google Scholar] [CrossRef]
- Woo, S.; Jeon, H.; Lee, T.K.; Kim, S.R.; Lee, S.; Yum, S. Expression profiling of liver in Java medaka fish exposed to 17β-estradiol. Mol. Cell. Toxicol. 2011, 7, 271–281. [Google Scholar] [CrossRef]
- Hwang, D.S.; Lee, J.S.; Lee, K.W.; Rhee, J.S.; Han, J.; Lee, J.; Park, G.S.; Lee, Y.M. Cloning and expression of ecdysone receptor (EcR) from the intertidal copepod, Tigriopus japonicus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 151, 303–312. [Google Scholar] [CrossRef]
- Rhee, J.S.; Kim, R.O.; Seo, J.S.; Lee, J.; Lee, Y.M.; Lee, J.S. Effects of salinity and endocrine-disrupting chemicals on expression of prolactin and prolactin receptor genes in the euryhaline hermaphroditic fish, Kryptolebias marmoratus. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 152, 413–423. [Google Scholar] [CrossRef]
- Hall, W.H.; Anderson, R.D. The influence of salinity on the toxicity of various classes of chemicals to aquatic biota. Crit. Rev. Toxicol. 1995, 25, 281–346. [Google Scholar] [CrossRef]
- Verslycke, T.; Vangheluwe, M.; Heijerick, D.; de Schamphelaere, K.; van Sprang, P.; Janssen, C.R. The toxicity of metal mixtures to the estuarine mysid Neomysis integer (Crustacea: Mysidacea) under changing salinity. Aquat. Toxicol. 2003, 64, 307–315. [Google Scholar] [CrossRef]
- Paul, I.; Savage, K.I.; Blayney, J.K.; Lamers, E.; Gately, K.; Kerr, K.; Sheaff, M.; Arthur, K.; Richard, D.J.; Hamilton, P.W.; et al. PARP inhibition induces BAX/BAK-independent synthetic lethality of BRCA1-deficient non-small cell lung cancer. J. Pathol. 2011, 224, 564–574. [Google Scholar] [CrossRef]
- Wei, L.; Lan, L.; Yasui, A.; Tanaka, K.; Saijo, M.; Matsuzawa, A.; Kashiwagi, R.; Maseki, E.; Hu, Y.; Parvin, J.D.; et al. BRCA1 contributes to transcription-coupled repair of DNA damage through polyubiquitination and degradation of Cockayne syndrome B protein. Cancer Sci. 2011, 102, 1840–1847. [Google Scholar] [CrossRef]
- Pretto, D.; Barco, R.; Riviera, J.; Neel, N.; Gustavson, M.D.; Eid, J.E. The synovial sarcoma translocation protein SYT-SSX2 recruits beta-catenin to the nucleus and associates with it in an active complex. Oncogene 2006, 25, 3661–3669. [Google Scholar] [CrossRef]
- Hildebrand, A.; Romaris, M.; Rasmussen, L.M.; Heinegard, D.; Twardzik, D.R.; Border, W.A.; Ruoslahti, E. Interaction of the small interstitial proteoglycans biglycan, decorin and fibromodulin with transforming growth factor beta. Biochem. J. 1994, 302, 527–534. [Google Scholar]
- Santra, M.; Reed, C.C.; Iozzo, R.V. Decorin binds to a narrow region of the epidermal growth factor (EGF) receptor, partially overlapping but distinct from the EGF-binding epitope. J. Biol. Chem. 2002, 277, 35671–35681. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Kodama, Y.; Matsumoto, T. Bone matrix decorin binds transforming growth factor-beta and enhances its bioactivity. J. Biol. Chem. 1994, 269, 32634–32638. [Google Scholar]
- Jahanyar, J.; Joyce, D.L.; Southard, R.E.; Loebe, M.; Noon, G.P.; Koerner, M.M.; Torre-Amione, G.; Youker, K.A. Decorin-mediated transforming growth factor-beta inhibition ameliorates adverse cardiac remodeling. J. Heart Lung Transpl. 2007, 26, 34–40. [Google Scholar] [CrossRef]
- Yan, W.; Wang, P.; Zhao, C.X.; Tang, J.; Xiao, X.; Wang, D.W. Decorin gene delivery inhibits cardiac fibrosis in spontaneously hypertensive rats by modulation of transforming growth factor-beta/Smad and p38 mitogen-activated protein kinase signaling pathways. Hum. Gene Ther. 2009, 20, 1190–1200. [Google Scholar] [CrossRef]
- Woo, S.; Yum, S.; Kim, D.W.; Park, H.S. Transcripts level responses in a marine medaka (Oryzias javanicus) exposed to organophosphorus pesticide. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 149, 427–432. [Google Scholar] [CrossRef]
- Woo, S.; Yum, S.; Park, H.S.; Lee, T.K.; Ryu, J.C. Effects of heavy metals on antioxidants and stress-responsive gene expression in Javanese medaka (Oryzias javanicus). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 149, 289–299. [Google Scholar] [CrossRef]
- Zanette, J.; de Almeida, E.A.; da Silva, A.Z.; Guzenski, J.; Ferreira, J.F.; di Mascio, P.; Marques, M.R.; Bainy, A.C. Salinity influences glutathione S-transferase activity and lipid peroxidation responses in the Crassostrea gigas oyster exposed to diesel oil. Sci. Total Environ. 2011, 409, 1976–1983. [Google Scholar] [CrossRef]
- Woo, S.; Son, S.; Park, H.S.; Vulpe, C.; Ryu, J.C.; Yum, S. Differentially expressed genes in marine medaka fish (Oryzias javanicus) exposed cadmium. Mol. Cell. Toxicol. 2008, 4, 293–299. [Google Scholar]
- Woo, S.; Won, H.; Ryu, J.C.; Yum, S. Differential gene expression profiling in Iprobenfos-exposed marine medaka by heterologous microarray hybridization. Toxicol. Environ. Health Sci. 2010, 2, 231–237. [Google Scholar] [CrossRef]
- Zhou, C.; Liu, J. Inhibition of human telomerase reverse transcriptase gene expression by BRCA1 in human ovarian cancer cells. Biochem. Biophys. Res. Commun. 2003, 303, 130–136. [Google Scholar] [CrossRef]
- Senthilkumar, P.K.; Robertson, L.W.; Ludewig, G. PCB153 reduces telomerase activity and telomere length in immortalized human skin keratinocytes (HaCaT) but not in human foreskin keratinocytes (NFK). Toxicol. Appl. Pharmacol. 2012, 259, 115–123. [Google Scholar] [CrossRef]
- Yamashita, S.; Ogawa, K.; Ikei, T.; Udono, M.; Fujiki, T.; Katakura, Y. SIRT1 prevents replicative senescence of normal human umbilical cord fibroblast through potentiating the transcription of human telomerase reverse transcriptase gene. Biochem. Biophys. Res. Commun. 2012, 417, 630–634. [Google Scholar] [CrossRef]
- Kashiwada, S.; Ishikawa, H.; Miyamoto, N.; Ohnishi, Y.; Magara, Y. Fish test for endocrine-disruption and estimation of water quality of Japanese rivers. Water Res. 2002, 36, 2161–2166. [Google Scholar] [CrossRef]
- Rocke, D.; Durbin, B. A model for measurement error for gene expression arrays. J. Comput. Biol. 2001, 8, 557–569. [Google Scholar] [CrossRef]
- Woo, S.; Yum, S.; Jung, J.H.; Shim, W.J.; Lee, C.H.; Lee, T.K. Heavy metal-induced differential gene expression of metallothionein in Javanese medaka, Oryzias javanicus. Mar. Biotechnol. 2006, 8, 654–662. [Google Scholar] [CrossRef]
- Won, H.; Yum, S.; Woo, S. Identification of differentially expressed genes in liver of marine medaka fish exposed to benzo(a)pyrene. Toxicol. Environ. Health Sci. 2011, 3, 39–45. [Google Scholar] [CrossRef]
- Stevens, D.P.; McLaughlin, M.J.; Heinrich, T. Determining toxicity of lead and zinc runoff in soils: Salinity effects on metal partitioning and on phytotoxicity. Environ. Toxicol. Chem. 2003, 22, 3017–3024. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Woo, S.; Denis, V.; Yum, S. Transcriptional Changes Caused by Bisphenol A in Oryzias javanicus, a Fish Species Highly Adaptable to Environmental Salinity. Mar. Drugs 2014, 12, 983-998. https://doi.org/10.3390/md12020983
Woo S, Denis V, Yum S. Transcriptional Changes Caused by Bisphenol A in Oryzias javanicus, a Fish Species Highly Adaptable to Environmental Salinity. Marine Drugs. 2014; 12(2):983-998. https://doi.org/10.3390/md12020983
Chicago/Turabian StyleWoo, Seonock, Vianney Denis, and Seungshic Yum. 2014. "Transcriptional Changes Caused by Bisphenol A in Oryzias javanicus, a Fish Species Highly Adaptable to Environmental Salinity" Marine Drugs 12, no. 2: 983-998. https://doi.org/10.3390/md12020983
APA StyleWoo, S., Denis, V., & Yum, S. (2014). Transcriptional Changes Caused by Bisphenol A in Oryzias javanicus, a Fish Species Highly Adaptable to Environmental Salinity. Marine Drugs, 12(2), 983-998. https://doi.org/10.3390/md12020983





