Bioactive Cembranoids, Sarcocrassocolides P–R, from the Dongsha Atoll Soft Coral Sarcophyton crassocaule
Abstract
:1. Introduction

2. Results and Discussion
| Sarcrocrassocolide P (1) | Sarcrocrassocolide Q (2) | Sarcrocrassocolide R (3) | ||||
|---|---|---|---|---|---|---|
| position | δC, mult. a | δH (J in Hz) b | δC, mult. a | δH (J in Hz) b | δC, mult. c | δH (J in Hz) d |
| 1 | 38.5, CH | 3.11, brd (9.5) c | 37.7, CH | 3.06, brs | 40.5, CH | 3.02, d (11.0) |
| 2 | 37.3, CH2 | 1.80, m | 35.7, CH2 | 2.05, t (5.0) | 39.3, CH2 | 2.14, m |
| 1.32, ddd (14.5, 10.5, 9.5) | 1.80, m | 1.82, ddd (19.0, 5.5, 1.5) | ||||
| 3 | 73.1, CH | 3.73, t (10.0) | 75.8, CH | 5.04, dd (6.5, 5.0) | 71.6, CH | 4.25, d (5.0) |
| 4 | 89.9, C | 74.7, C | 150.5, C | |||
| 5 | 36.4, CH2 | 1.94, t (11.5) | 37.9, CH2 | 1.68, m | 31.2, CH2 | 2.16, m |
| 1.81, m | 2.12, m | |||||
| 6 | 23.1, CH2 | 2.26, m | 23.1, CH2 | 2.18, m | 23.4, CH2 | 2.59, m |
| 2.15, m | 2.21, m | |||||
| 7 | 125.7, CH | 5.08, t (7.0) | 123.3, CH | 5.13, t (7.0) | 126.3, CH | 5.07, d (10.5) |
| 8 | 135.8, C | 136.5, C | 133.9, C | |||
| 9 | 39.4, CH2 | 2.28, m | 37.6, CH2 | 2.21, m | 36.7, CH2 | 2.29, d (13.0) |
| 2.09, m | 2.09, m | |||||
| 10 | 24.7, CH2 | 2.44, qd (10.0, 2.5) | 24.6, CH2 | 2.34, m | 24.4, CH2 | 2.17, m |
| 2.11, m | 2.24, m | 1.31, m | ||||
| 11 | 127.3, CH | 5.32, dd (10.0, 3.5) | 129.3, CH | 5.32, brt (7.0) | 61.7, CH | 2.56, dd (11.0, 4.0) |
| 12 | 130.3, C | 129.2, C | 59.7, C | |||
| 13 | 77.5, CH | 5.40, s | 76.5, CH | 5.37, s | 46.5, CH2 | 2.00, dd (14.0, 11.5) |
| 1.24, d (14.0) | ||||||
| 14 | 79.1, CH | 5.28, brs | 82.7, CH | 4.43, dd (5.0, 2.0) | 81.1, CH | 4.32, d (11.5) |
| 15 | 140.5, C | 140.5, C | 139.5, C | |||
| 16 | 170.1, C | 169.8, C | 170.1, C | |||
| 17 | 120.9, CH2 | 6.24, d (2.0) | 122.2, CH2 | 6.23, d (2.5) | 123.2, CH2 | 6.29, d (1.5) |
| 5.65, d (2.0) | 5.78, d (2.5) | 5.69, d (1.5) | ||||
| 18 | 19.7, CH3 | 1.44, s | 24.4, CH3 | 1.44, s | 107.3, CH2 | 5.17, s |
| 4.78, s | ||||||
| 19 | 16.0, CH3 | 1.67, s | 16.8, CH3 | 1.65, s | 14.9, CH3 | 1.76, s |
| 20 | 14.5, CH3 | 1.72, s | 14.6, CH3 | 1.72, s | 17.5, CH3 | 1.38, s |
| 4-OAc | 22.1, CH3 | 2.04, s | ||||
| 172.1, C | ||||||
| 3-OAc | 21.2, CH3 | 2.08, s | ||||
| 170.4, C | ||||||
| 13-OAc | 20.8, CH3 | 1.99, s | 20.8, CH3 | 2.03, s | ||
| 169.3, C | 169.5, C | |||||
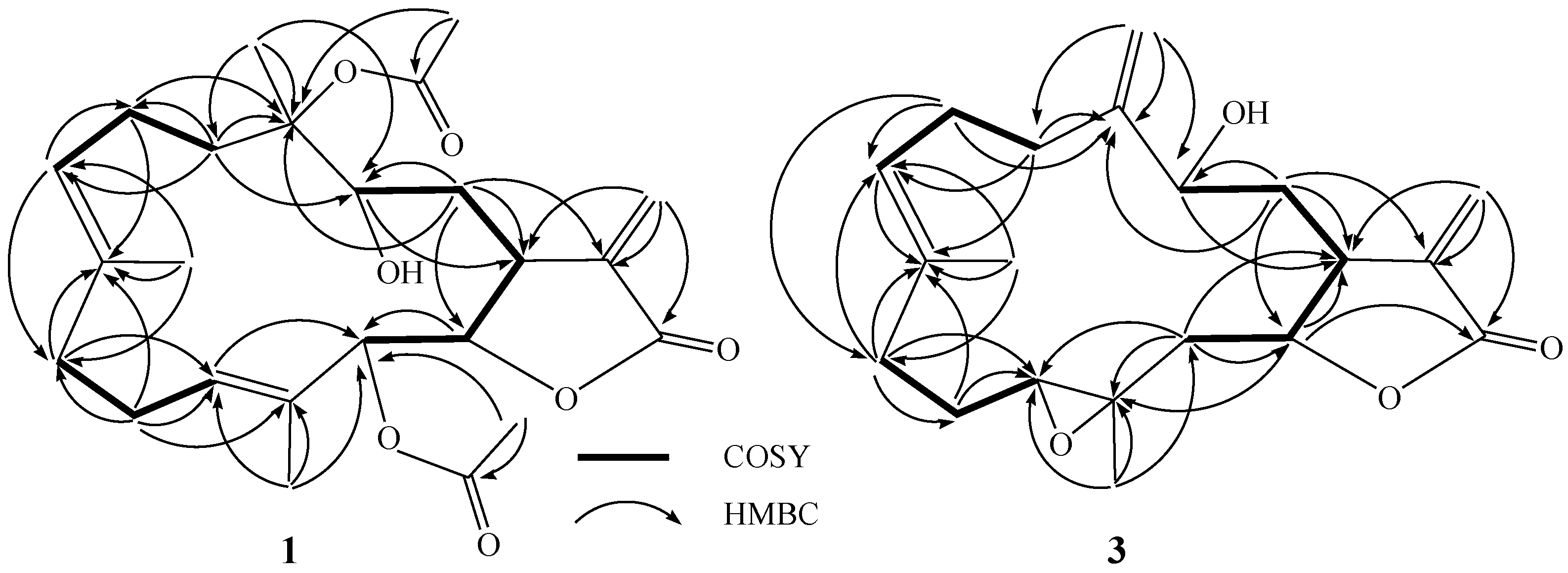

 +7.0, +31.6, +21.9 and +108.9, respectively), were found to have identical spectroscopic data and close specific optical rotations with those of previously discovered compounds, crassocolide A (
+7.0, +31.6, +21.9 and +108.9, respectively), were found to have identical spectroscopic data and close specific optical rotations with those of previously discovered compounds, crassocolide A (  +6.5), B (
+6.5), B (  +26.5), D (
+26.5), D (  +16.8) and E (
+16.8) and E (  +99.6), respectively [23]. Thus, the structures of compounds 4–7 were confirmed.
+99.6), respectively [23]. Thus, the structures of compounds 4–7 were confirmed.| Compound | DLD-1 a | CCRF-CEM b | HL-60 c |
|---|---|---|---|
| 1 | 21.8 | 48.8 | 24.9 |
| 2 | 35.8 | 73.1 | 18.6 |
| 3 | 10.0 | 28.1 | 8.7 |
| 4 | 5.7 | 6.3 | (–) d |
| 5 | 3.8 | 8.7 | 7.3 |
| 6 | 27.7 | 41.9 | 34.6 |
| 7 | 7.9 | 11.1 | 8.4 |
| Doxorubicin | 0.77 | 1.16 | 0.046 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Separation
 −76 (c 0.4, CHCl3); IR (neat) νmax 3445, 2924, 2851, 1767, 1733, 1652, 1435, 1371, and 1229 cm−1; UV (MeOH) λmax 205 (log ε = 3.5); 13C and 1H NMR data, see Table 1; ESIMS m/z 457 [M + Na]+; HRESIMS m/z 457.2199 [M + Na]+ (calcd. for C24H34O7Na, 457.2202).
−76 (c 0.4, CHCl3); IR (neat) νmax 3445, 2924, 2851, 1767, 1733, 1652, 1435, 1371, and 1229 cm−1; UV (MeOH) λmax 205 (log ε = 3.5); 13C and 1H NMR data, see Table 1; ESIMS m/z 457 [M + Na]+; HRESIMS m/z 457.2199 [M + Na]+ (calcd. for C24H34O7Na, 457.2202). −84 (c 0.1, CHCl3); IR (neat) νmax 3445, 2917, 2849, 1750, 1733, 1653, 1434, 1372, and 1236 cm−1; UV (MeOH) λmax 214 (log ε = 3.8); 13C and 1H NMR data, see Table 1; ESIMS m/z 457 [M + Na]+; HRESIMS m/z 457.2201 [M + Na]+ (calcd. for C24H34O7Na, 457.2202).
−84 (c 0.1, CHCl3); IR (neat) νmax 3445, 2917, 2849, 1750, 1733, 1653, 1434, 1372, and 1236 cm−1; UV (MeOH) λmax 214 (log ε = 3.8); 13C and 1H NMR data, see Table 1; ESIMS m/z 457 [M + Na]+; HRESIMS m/z 457.2201 [M + Na]+ (calcd. for C24H34O7Na, 457.2202). −178 (c 0.1, CHCl3); IR (neat) νmax 3420, 2931, 1751, 1654, 1450, 1375, and 1270 cm−1; UV (MeOH) λmax 213 (log ε = 3.7); 13C and 1H NMR data, see Table 1; ESIMS m/z 355 [M + Na]+; HRESIMS m/z 355.1888 [M + Na]+ (calcd. for C20H28O4Na, 355.1885).
−178 (c 0.1, CHCl3); IR (neat) νmax 3420, 2931, 1751, 1654, 1450, 1375, and 1270 cm−1; UV (MeOH) λmax 213 (log ε = 3.7); 13C and 1H NMR data, see Table 1; ESIMS m/z 355 [M + Na]+; HRESIMS m/z 355.1888 [M + Na]+ (calcd. for C20H28O4Na, 355.1885).3.4. Cytotoxicity Testing
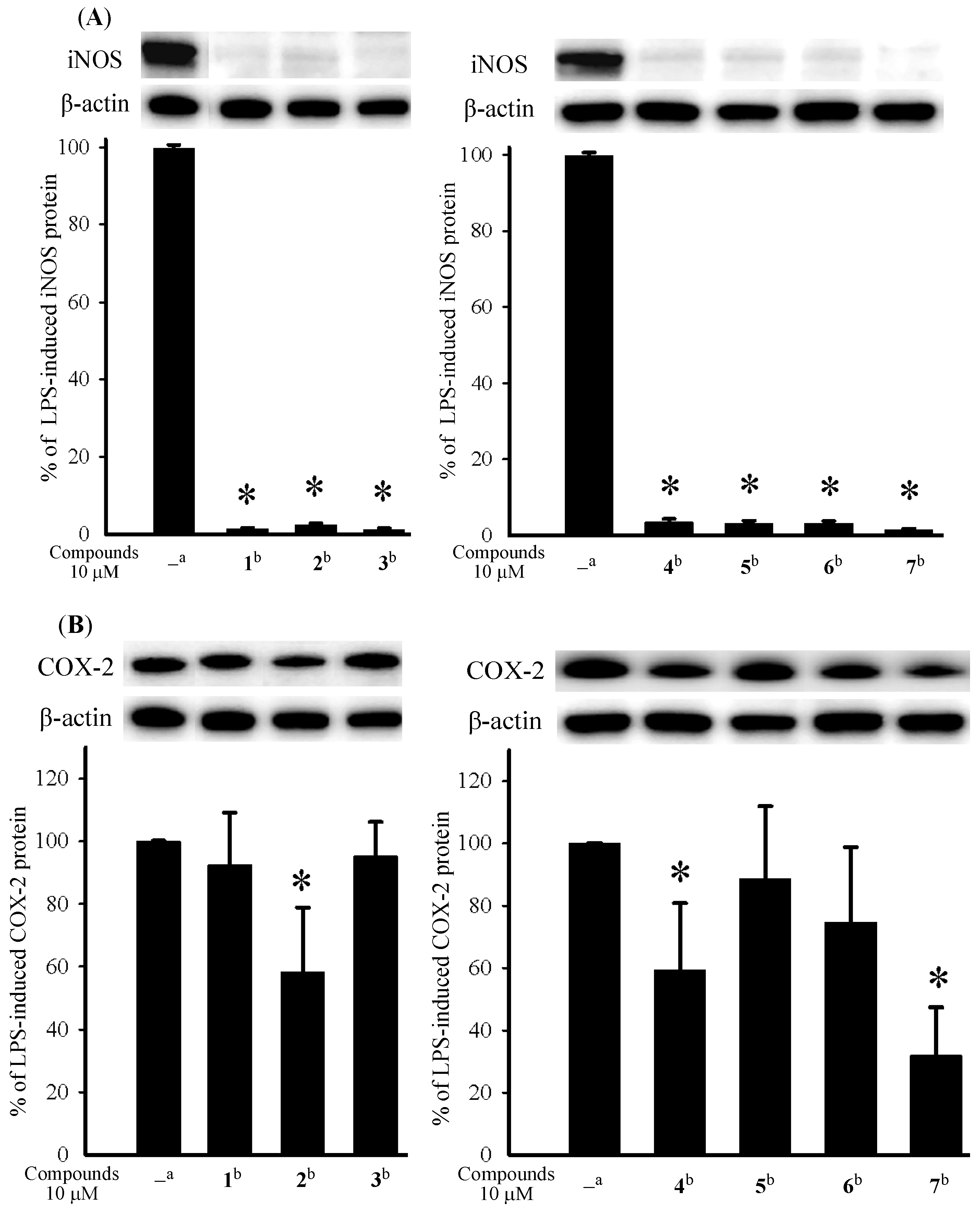
3.5. In Vitro Anti-Inflammatory Assay
4. Conclusions
Supplementary Files
Acknowledgments
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef]
- Bishara, A.; Rudi, A.; Benayahu, Y.; Kashman, Y. Three biscembranoids and their monomeric counterpart cembranoid, a biogenetic diels-alder precursor, from the soft coral Sarcophyton elegans. J. Nat. Prod. 2007, 70, 1951–1954. [Google Scholar] [CrossRef]
- Bensemhoun, J.; Rudi, A.; Bombarda, I.; Gaydou, E.M.; Kashman, Y.; Aknin, M. Flexusines A and B and epimukulol from the soft coral Sarcophyton flexuosum. J. Nat. Prod. 2008, 71, 1262–1264. [Google Scholar] [CrossRef]
- Marrero, J.; Benítez, J.; Rodríguez, A.D.; Zhao, H.; Raptis, R.G. Bipinnatins K–Q, Minor cembrane-type diterpenes from the west Indian gorgonian Pseudopterogorgia kallos: Isolation, structure assignment, and evaluation of biological activities. J. Nat. Prod. 2008, 71, 381–389. [Google Scholar] [CrossRef]
- Shi, Y.-P.; Rodríguez, A.D.; Barnes, C.L.; Sánchez, J.A.; Raptis, R.G.; Baran, P. New terpenoid constituents from Eunicea pinta. J. Nat. Prod. 2002, 65, 1232–1241. [Google Scholar] [CrossRef]
- Rashid, M.A.; Gustafson, K.R.; Boyd, M.R. HIV-Inhibitory cembrane derivatives from a Philippines collection of the soft coral Lobophytum species. J. Nat. Prod. 2000, 63, 531–533. [Google Scholar] [CrossRef]
- König, G.M.; Wright, A.D. New cembranoid diterpenes from the soft coral Sarcophyton ehrenbergi. J. Nat. Prod. 1998, 61, 494–496. [Google Scholar] [CrossRef]
- Wright, A.D.; Nielson, J.L.; Tapiolas, D.M.; Liptrot, C.H.; Motti, C.A. A great barrier reef Sinularia sp. yields two new cytotoxic diterpenes. Mar. Drugs 2012, 10, 1619–1630. [Google Scholar] [CrossRef]
- Yang, B.; Zhou, X.; Huang, H.; Yang, X.-W.; Liu, J.; Lin, X.; Li, X.; Peng, Y.; Liu, Y. New cembrane diterpenoids from a Hainan soft coral Sinularia sp. Mar. Drugs 2012, 10, 2023–2032. [Google Scholar] [CrossRef]
- Chao, C.-H.; Chou, K.-J.; Huang, C.-Y.; Wen, Z.-H.; Hsu, C.-H.; Wu, Y.-C.; Dai, C.-F.; Sheu, J.-H. Bioactive cembranoids from the soft coral Sinularia crassa. Mar. Drugs 2011, 9, 1955–1968. [Google Scholar] [CrossRef]
- Shih, H.-J.; Tseng, Y.-J.; Huang, C.-Y.; Wen, Z.-H.; Dai, C-F.; Sheu, J.-H. Cytotoxic and anti-inflammatory diterpenoids from the Dongsha Atoll soft coral Sinularia flexibilis. Tetrahedron 2012, 68, 244–249. [Google Scholar] [CrossRef]
- Su, J.-H.; Lin, Y.-F.; Lu, Y.; Yeh, H.-C.; Wang, W.-H.; Fan, T.-Y.; Sheu, J.-H. Oxygenated cembranoids from the cultured and wild-type soft corals Sinularia flexibilis. Chem. Pharm. Bull. 2009, 57, 1189–1192. [Google Scholar] [CrossRef]
- Su, J.-H.; Ahmed, A.F.; Sung, P.-J.; Chao, C.-H.; Kuo, Y.-H.; Sheu, J.-H. Manaarenolides A–I, new diterpenoids from the soft coral Sinularia manaarensis. J. Nat. Prod. 2006, 69, 1134–1139. [Google Scholar] [CrossRef]
- Lu, Y.; Huang, C.-Y.; Lin, Y.-F.; Wen, Z.-H.; Su, J.-H.; Kuo, Y.-H.; Chiang, M.Y.; Sheu, J.-H. Anti-inflammatory cembranoids from the soft corals Sinularia querciformis and Sinularia granosa. J. Nat. Prod. 2008, 71, 1754–1759. [Google Scholar] [CrossRef]
- Ahmed, A.F.; Tai, S.-H.; Wen, Z.-H.; Su, J.-H.; Wu, Y.-C.; Hu, W.-P.; Sheu, J.-H. A C-3 methylated isocembranoid and 10-oxocembranoids from a Formosan soft coral Sinularia grandilobata. J. Nat. Prod. 2008, 71, 946–951. [Google Scholar]
- Chen, B.-W.; Chao, C.-H.; Su, J.-H.; Huang, C.-Y.; Dai, C.-F.; Wen, Z.-H.; Sheu, J.-H. A novel symmetric sulfur-containing biscembranoid from the Formosan soft coral Sinularia flexibilis. Tetrahedron Lett. 2010, 44, 5764–5766. [Google Scholar]
- Su, J.-H.; Wen, Z.-H. Bioactive cembrane-based diterpenoids from the soft coral Sinularia triangular. Mar. Drugs 2011, 9, 944–951. [Google Scholar] [CrossRef]
- Cheng, S.-Y.; Wen, Z.-H.; Wang, S.-K.; Chiou, S.-F.; Hsu, C.-H.; Dai, C.-F.; Chiang, M.Y.; Duh, C.-Y. Unprecedented hemiketal cembranolides with anti-inflammatory activity from the soft coral Lobophytum durum. J. Nat. Prod. 2009, 72, 152–155. [Google Scholar] [CrossRef]
- Chao, C.-H.; Wen, Z.-H.; Wu, Y.-C.; Yeh, H.-C.; Sheu, J.-H. Cytotoxic and anti-inflammatory cembranoids from the soft coral Lobophytum crassum. J. Nat. Prod. 2008, 71, 1819–1824. [Google Scholar] [CrossRef]
- Wang, S.-K.; Duh, C.-Y. New Cytotoxic cembranolides from the soft coral Lobophytum michaelae. Mar. Drugs 2012, 10, 306–318. [Google Scholar] [CrossRef]
- Lin, S.-T.; Wang, S.-K.; Duh, C.-Y. Cembranoids from the Dongsha Atoll soft coral Lobophytum crassum. Mar. Drugs 2011, 9, 2705–2716. [Google Scholar] [CrossRef]
- Wang, S.-K.; Hsieh, M.-K.; Duh, C.-Y. Three new cembranoids from the Taiwanese soft coral Sarcophyton ehrenbergi. Mar. Drugs 2012, 10, 1433–1444. [Google Scholar] [CrossRef]
- Huang, H.-C.; Ahmed, A.F.; Su, J.-H.; Wu, Y.-C.; Chiang, M.Y.; Sheu, J.-H. Crassocolides A–F, new cembranoids with a trans-fused lactone from the soft coral Sarcophyton crassocaule. J. Nat. Prod. 2006, 69, 1554–1559. [Google Scholar] [CrossRef]
- Huang, H.-C.; Chao, C.-H.; Kuo, Y.-H.; Sheu, J.-H. Crassocolides G–M, cembranoids from a Formosan soft coral Sarcophyton crassocaule. Chem. Biodiv. 2009, 6, 1232–1242. [Google Scholar] [CrossRef]
- Cheng, Y.-B.; Shen, Y.-C.; Kuo, Y.-H.; Khalil, A.T. Cembrane diterpenoids from the Taiwanese soft coral Sarcophyton stolidotum. J. Nat. Prod. 2008, 71, 1141–1145. [Google Scholar] [CrossRef]
- Cheng, S.-Y.; Wang, S.-K.; Chiou, S.-F.; Hsu, C.-H.; Dai, C.-F.; Chiang, M.Y.; Duh, C.-Y. Cembranoids from the octocoral Sarcophyton ehrenbergi. J. Nat. Prod. 2010, 73, 197–203. [Google Scholar] [CrossRef]
- Su, J.-H.; Lu, Y.; Lin, W.-Y.; Wang, W.-H.; Sung, P.-J.; Sheu, J.-H. A cembranoid, trocheliophorol, from the cultured soft coral Sarcophyton trocheliophorum. Chem. Lett. 2010, 39, 172–173. [Google Scholar] [CrossRef]
- Wang, G.-H.; Huang, H.-C.; Su, J.-H.; Huang, C.-Y.; Hsu, C.-H.; Kuo, Y.-H.; Sheu, J.-H. Crassocolides N–P, three cembranoids from the Formosan soft coral Sarcophyton crassocaule Bioorg. Med. Chem. Lett. 2011, 21, 7201–7204. [Google Scholar]
- Sheu, J.-H.; Wang, G.-H.; Duh, C.-Y.; Soong, K. Pachyclavulariolides M-R, six novel diterpenoids from a Taiwanese soft coral Pachyclavularia violacea. J. Nat. Prod. 2003, 66, 662–666. [Google Scholar] [CrossRef]
- Lin, W.-Y.; Su, J.-H.; Lu, Y.; Wen, Z.-H.; Dai, C.-F.; Kuo, Y.-H.; Sheu, J.-H. Cytotoxic and anti-inflammatory cembranoids from the Dongsha Atoll soft coral Sarcophyton crassocaule. Bioorg. Med. Chem. 2010, 18, 1936–1941. [Google Scholar] [CrossRef]
- Lin, W.-Y.; Lu, Y.; Su, J.-H.; Wen, Z.-H.; Dai, C.-F.; Kuo, Y.-H.; Sheu, J.-H. Bioactive cembranoids from the Dongsha Atoll soft coral Sarcophyton crassocaule. Mar. Drugs 2011, 9, 994–1006. [Google Scholar] [CrossRef]
- Lin, W.-Y.; Lu, Y.; Chen, B.-W.; Huang, C.-Y.; Su, J.-H.; Wen, Z.-H.; Dai, C.-F.; Kuo, Y.-H.; Sheu, J.-H. Sarcocrassocolides M–O, bioactive cembranoids from the Dongsha Atoll soft coral Sarcophyton crassocaule. Mar. Drugs 2012, 10, 617–626. [Google Scholar] [CrossRef]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar]
- Wen, Z.-H.; Chao, C.-H.; Wu, M.-H.; Sheu, J.-H. A neuroprotective sulfone of marine origin and the in vivo anti-inflammatory activity of an analogue. Eur. J. Med. Chem. 2010, 45, 5998–6004. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, W.-Y.; Chen, B.-W.; Huang, C.-Y.; Wen, Z.-H.; Sung, P.-J.; Su, J.-H.; Dai, C.-F.; Sheu, J.-H. Bioactive Cembranoids, Sarcocrassocolides P–R, from the Dongsha Atoll Soft Coral Sarcophyton crassocaule. Mar. Drugs 2014, 12, 840-850. https://doi.org/10.3390/md12020840
Lin W-Y, Chen B-W, Huang C-Y, Wen Z-H, Sung P-J, Su J-H, Dai C-F, Sheu J-H. Bioactive Cembranoids, Sarcocrassocolides P–R, from the Dongsha Atoll Soft Coral Sarcophyton crassocaule. Marine Drugs. 2014; 12(2):840-850. https://doi.org/10.3390/md12020840
Chicago/Turabian StyleLin, Wan-Yu, Bo-Wei Chen, Chiung-Yao Huang, Zhi-Hong Wen, Ping-Jyun Sung, Jui-Hsin Su, Chang-Feng Dai, and Jyh-Horng Sheu. 2014. "Bioactive Cembranoids, Sarcocrassocolides P–R, from the Dongsha Atoll Soft Coral Sarcophyton crassocaule" Marine Drugs 12, no. 2: 840-850. https://doi.org/10.3390/md12020840






