The Effect of Low-Dose Marine n-3 Fatty Acids on Plasma Levels of sCD36 in Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Trial
Abstract
:1. Introduction
2. Results and Discussion
2.1. Subject Characteristics
| Control (n = 25) | n-3 PUFA (n = 25) | ||||
|---|---|---|---|---|---|
| Gender | |||||
| Male | 12 (48%) | 12 (48%) | |||
| Female | 13 (52%) | 13 (52%) | |||
| Age (years) | 55.4 ± 9.5 | 58.0 ± 7.4 | |||
| BMI (kg/m2) | 29.5 ± 3.3 | 30.8 ± 4.2 | |||
| Waist circumference (cm) | |||||
| Male | 106.4 ± 7.7 | 107.7 ± 7.7 | |||
| Female | 94.6 ± 6.9 | 99.8 ± 12.8 | |||
| Smoking | |||||
| Smokers | 3 (12%) | 5 (20%) | |||
| Non-smokers | 22 (88%) | 20 (80%) | |||
| Medication | |||||
| ASA | 3 (12%) | 15 (60%) | |||
| Lipid-lowering drugs | 19 (68%) | 23 (76%) | |||
| Insulin | 0 (0%) | 1 (4%) | |||
| Oral anti-diabetic drugs | 0 (0%) | 3 (12%) | |||
| β-blockers | 3 (12%) | 13 (52%) | |||
| Other anti-hypertensive drugs | 19 (48%) | 17 (44%) | |||
| Disease | |||||
| Dyslipidemia | 17 (68%) | 19 (76%) | |||
| Hypertension | 13 (52%) | 10 (40%) | |||
| DM2 | 0 (0%) | 4 (16%) | |||
| AMI | 1 (4%) | 3 (12%) | |||
| Baseline | 6 weeks | Change: Baseline—6 weeks | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | n-3 PUFA | P a | Control | n-3 PUFA | P b | Control | n-3 PUFA | P c | |||||||||||||
| Neutrophils (Wt%) | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||||||||
| 14:0 | 0.17 | ± | 0.04 | 0.16 | ± | 0.03 | 0.28 | 0.15 | ± | 0.05 | 0.16 | ± | 0.04 | 0.41 | −0.02 | ± | 0.05 | 0.00 | ± | 0.03 | 0.12 |
| 16:0 | 12.04 | ± | 0.60 | 11.81 | ± | 0.59 | 0.18 | 11.91 | ± | 0.62 | 11.83 | ± | 0.67 | 0.69 | −0.10 | ± | 0.47 | 0.03 | ± | 0.42 | 0.32 |
| 18:0 | 17.20 | ± | 0.59 | 16.98 | ± | 0.49 | 0.15 | 17.16 | ± | 0.55 | 17.00 | ± | 0.54 | 0.30 | −0.03 | ± | 0.50 | 0.02 | ± | 0.28 | 0.65 |
| 18:1n-9 | 31.53 | ± | 1.59 | 31.86 | ± | 1.69 | 0.41 | 31.76 | ± | 1.49 | 32.46 | ± | 1.56 | 0.47 | 0.37 | ± | 1.74 | 0.60 | ± | 1.07 | 0.58 |
| 18:2n-6 LA | 9.03 | ± | 0.93 | 8.64 | ± | 0.67 | 0.10 | 8.77 | ± | 0.73 | 8.88 | ± | 0.94 | 0.63 | −0.27 | ± | 0.87 | 0.25 | ± | 0.93 | 0.05 |
| 20:4n-6 AA | 13.27 | ± | 1.11 | 13.36 | ± | 0.85 | 0.75 | 13.43 | ± | 1.10 | 12.17 | ± | 0.93 | <0.01 | 0.10 | ± | 0.94 | −1.20 | ± | 0.73 | <0.01 |
| 20:5n-3 EPA | 0.58 | ± | 0.30 | 0.68 | ± | 0.33 | 0.25 | 0.57 | ± | 0.27 | 1.36 | ± | 0.35 | <0.01 | 0.04 | ± | 0.18 | 0.68 | ± | 0.31 | <0.01 |
| 22:5n-3 DPA | 1.62 | ± | 0.46 | 1.74 | ± | 0.44 | 0.34 | 1.61 | ± | 0.30 | 2.39 | ± | 0.79 | <0.01 | −0.03 | ± | 0.35 | 0.64 | ± | 0.53 | <0.01 |
| 22:6n-3 DHA | 1.34 | ± | 0.40 | 1.40 | ± | 0.51 | 0.61 | 1.33 | ± | 0.31 | 1.65 | ± | 0.58 | <0.01 | −0.00 | ± | 0.22 | 0.25 | ± | 0.34 | <0.01 |
| Adipose tissue (Wt%) | |||||||||||||||||||||
| 14:0 | 2.06 | ± | 0.38 | 2.04 | ± | 0.35 | 0.86 | ||||||||||||||
| 16:0 | 19.63 | ± | 1.91 | 19.96 | ± | 1.95 | 0.55 | ||||||||||||||
| 18:0 | 2.75 | ± | 0.61 | 2.78 | ± | 0.67 | 0.89 | ||||||||||||||
| 18:1n-9 | 48.10 | ± | 1.63 | 47.96 | ± | 1.63 | 0.76 | ||||||||||||||
| 18:2n-6 LA | 10.36 | ± | 1.08 | 10.25 | ± | 0.80 | 0.66 | ||||||||||||||
| 20:4n-6 AA | 0.46 | ± | 0.11 | 0.52 | ± | 0.13 | 0.09 | ||||||||||||||
| 20:5n-3 EPA | 0.11 | ± | 0.06 | 0.12 | ± | 0.05 | 0.80 | ||||||||||||||
| 22:5n-3 DPA | 0.35 | ± | 0.12 | 0.35 | ± | 0.10 | 0.98 | ||||||||||||||
| 22:6n-3 DHA | 0.29 | ± | 0.14 | 0.27 | ± | 0.15 | 0.68 | ||||||||||||||
| sCD36 baseline | sCD36 day 42 | Change in CD36 | P | |
|---|---|---|---|---|
| Controls | 0.50 (0.41, 0.59) | 0.47 (0.39, 0.56) | −0.01 (−0.11, 0.09) | |
| n-3 PUFA | 0.34 (0.27, 0.40) | 0.49 (0.25, 0.74) | 0.16 (−0.08, 0.39) | |
| Difference | 0.16 (0.05, 0.27) | −0.02 (−0.28, 0.24) | −0.17 (−0.42, 0.08) | 0.18 |
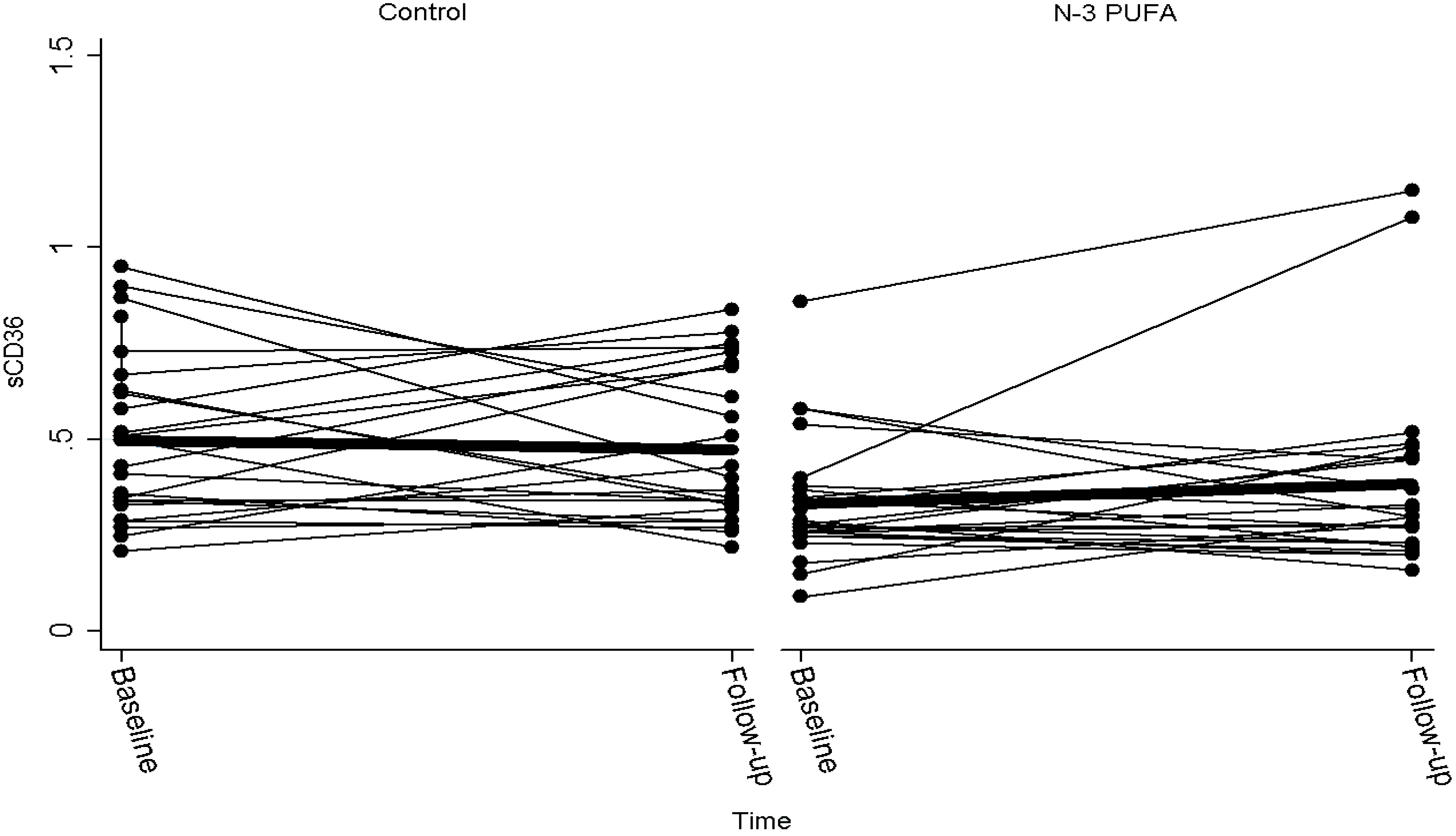
2.2. Discussion
3. Experimental Section
3.1. Study Subjects
3.2. Study Design
3.3. Blood and Adipose Tissue Sampling
3.4. Fatty Acid Composition of Neutrophils and Adipose Tissue
3.5. Measurement of sCD36
3.6. Food Questionnaire
3.7. Statistical Analyses
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- De Caterina, R. N-3 fatty acids in cardiovascular disease. New Engl. J. Med. 2011, 364, 2439–2450. [Google Scholar] [CrossRef]
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. n-3 Fatty acids from fish or fish-oil supplements, but not alpha-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar]
- Schmidt, E.B.; Arnesen, H.; de Caterina, R.; Rasmussen, L.H.; Kristensen, S.D. Marine n-3 polyunsaturated fatty acids and coronary heart disease. Part I. Background, epidemiology, animal data, effects on risk factors and safety. Thromb. Res. 2005, 115, 163–170. [Google Scholar] [CrossRef]
- Bjerregaard, L.J.; Joensen, A.M.; Dethlefsen, C.; Jensen, M.K.; Johnsen, S.P.; Tjonneland, A.; Rasmussen, L.H.; Overvad, K.; Schmidt, E.B. Fish intake and acute coronary syndrome. Eur. Heart J. 2010, 31, 29–34. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J. American heart association, nutrition committee, fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Rimm, E.B. Fish intake, contaminants, and human health: Evaluating the risks and the benefits. J. Am. Med. Assoc. 2006, 296, 1885–1899. [Google Scholar] [CrossRef]
- Burr, M.L.; Fehily, A.M.; Gilbert, J.F.; Rogers, S.; Holliday, R.M.; Sweetnam, P.M.; Elwood, P.C.; Deadman, N.M. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and reinfarction trial (DART). Lancet 1989, 2, 757–761. [Google Scholar]
- GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-prevenzione trial. Lancet 1999, 354, 447–455. [CrossRef]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M. n-3 Fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 Polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, S1505–S1519. [Google Scholar]
- Libby, P.; Ridker, P.M.; Hansson, G.K. Progress and challenges in translating the biology of atherosclerosis. Nature 2011, 473, 317–325. [Google Scholar] [CrossRef]
- Su, X.; Abumrad, N.A. Cellular fatty acid uptake: A pathway under construction. Trends Endocrinol. Metab. 2009, 20, 72–77. [Google Scholar] [CrossRef]
- Endemann, G.; Stanton, L.W.; Madden, K.S.; Bryant, C.M.; White, R.T.; Protter, A.A. CD36 is a receptor for oxidized low density lipoprotein. J. Biol. Chem. 1993, 268, 11811–11816. [Google Scholar]
- Handberg, A.; Levin, K.; Hojlund, K.; Beck-Nielsen, H. Identification of the oxidized low-density lipoprotein scavenger receptor CD36 in plasma: A novel marker of insulin resistance. Circulation 2006, 114, 1169–1176. [Google Scholar] [CrossRef]
- Handberg, A.; Skjelland, M.; Michelsen, A.E.; Sagen, E.L.; Krohg-Sorensen, K.; Russell, D.; Dahl, A.; Ueland, T.; Oie, E.; Aukrust, P.; et al. Doluble CD36 in plasma is increased in patients with symptomatic atherosclerotic carotid plaques and is related to plaque instability. Stroke 2008, 39, 3092–3095. [Google Scholar] [CrossRef]
- Koonen, D.P.; Jensen, M.K.; Handberg, A. Soluble CD36—A marker of the (pathophysiological) role of cd36 in the metabolic syndrome? Arch. Physiol. Biochem. 2011, 117, 57–63. [Google Scholar] [CrossRef]
- Trayhurn, P.; Wood, I.S. Adipokines: Inflammation and the pleiotropic role of white adipose tissue. Br. J. Nutr. 2004, 92, 347–355. [Google Scholar] [CrossRef]
- Hajer, G.R.; van Haeften, T.W.; Visseren, F.L. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur. Heart J. 2008, 29, 2959–2971. [Google Scholar] [CrossRef]
- Madonna, R.; Salerni, S.; Schiavone, D.; Glatz, J.F.; Geng, Y.J.; de Caterina, R. Omega-3 fatty acids attenuate constitutive and insulin-induced CD36 expression through a suppression of PPAR alpha/gamma activity in microvascular endothelial cells. Thromb. Haemost. 2011, 106, 500–510. [Google Scholar] [CrossRef]
- Wang, S.; Wu, D.; Matthan, N.R.; Lamon-Fava, S.; Lecker, J.L.; Lichtenstein, A.H. Reduction in Dietary omega-6 polyunsaturated fatty acids: Eicosapentaenoic acid plus docosahexaenoic acid ratio minimizes atherosclerotic lesion formation and inflammatory response in the LDL receptor null mouse. Atherosclerosis 2009, 204, 147–155. [Google Scholar] [CrossRef]
- Gammelmark, A.; Madsen, T.; Varming, K.; Lundbye-Christensen, S.; Schmidt, E.B. Low-Dose fish oil supplementation increases serum adiponectin without affecting inflammatory markers in overweight subjects. Nutr. Res. 2012, 32, 15–23. [Google Scholar] [CrossRef]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef]
- Serebruany, V.L.; Miller, M.; Pokov, A.N.; Lynch, D.; Jensen, J.K.; Hallen, J.; Atar, D. Early impact of prescription omega-3 fatty acids on platelet biomarkers in patients with coronary artery disease and hypertriglyceridemia. Cardiology 2011, 118, 187–194. [Google Scholar] [CrossRef]
- Van de Werf, F.; Ardissino, D.; Betriu, A.; Cokkinos, D.V.; Falk, E.; Fox, K.A.; Julian, D.; Lengyel, M.; Neumann, F.J.; Ruzyllo, W.; et al. Management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur. Heart J. 2003, 24, 28–66. [Google Scholar]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J. Omega-3 fatty acids and cardiovascular disease: New recommendations from the american heart association. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 151–152. [Google Scholar] [CrossRef]
- Bruni, F.; Pasqui, A.L.; Pastorelli, M.; Bova, G.; Cercignani, M.; Palazzuoli, A.; Sawamura, T.; Gioffre, W.R.; Auteri, A.; Puccetti, L. Different effect of statins on platelet oxidized-LDL receptor (CD36 and LOX-1) expression in hypercholesterolemic subjects. Clin. Appl. Thromb. Hemost. 2005, 11, 417–428. [Google Scholar] [CrossRef]
- Liani, R.; Halvorsen, B.; Sestili, S.; Handberg, A.; Santilli, F.; Vazzana, N.; Formoso, G.; Aukrust, P.; Davi, G. Plasma levels of soluble CD36, platelet activation, inflammation, and oxidative stress are increased in type 2 diabetic patients. Free Radic. Biol. Med. 2012, 52, 1318–1324. [Google Scholar] [CrossRef]
- Konstantinidou, V.; Khymenets, O.; Covas, M.I.; de la Torre, R.; Munoz-Aguayo, D.; Anglada, R.; Farre, M.; Fito, M. Time course of changes in the expression of insulin sensitivity-related genes after an acute load of virgin olive oil. OMICS 2009, 13, 431–438. [Google Scholar] [CrossRef]
- Eveleth, P.B. Physical status: The use and interpretation of anthropometry. Report of a WHO expert committee. Am. J. Hum. Biol. Organ. 1998, 8, 786–787. [Google Scholar] [CrossRef]
- Joensen, A.M.; Overvad, K.; Dethlefsen, C.; Johnsen, S.P.; Tjonneland, A.; Rasmussen, L.H.; Schmidt, E.B. Marine n-3 polyunsaturated fatty acids in adipose tissue and the risk of acute coronary syndrome. Circulation 2011, 124, 1232–1238. [Google Scholar] [CrossRef]
- Schmidt, E.B.; Varming, K.; Moller, J.M.; Pedersen, I.B.; Madsen, P.; Dyerberg, J. No effect of a very low dose of n-3 fatty acids on monocyte function in healthy humans. Scand.J. Clin. Lab. Invest. 1996, 56, 87–92. [Google Scholar] [CrossRef]
- Van Kuijk, F.J.; Thomas, D.W.; Stephens, R.J.; Dratz, E.A. Gas chromatography-mass spectrometry method for determination of phospholipid peroxides; I. Transesterification to form methyl esters. J. Free Radic. Biol. Med. 1985, 1, 215–225. [Google Scholar] [CrossRef]
- Andreasen, J.J.; Aardestrup, I.V.; Eschen, R.B.; Obel, T.; Lundbye-Christensen, S.; Schmidt, E.B. Fatty acid composition of the internal mammary artery in relation to dietary intake of marine n-3 polyunsaturated fatty acids and association with flow-mediated vasodilation. Cell. Mol. Biol. 2010, 56, 10–17. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Venø, S.K.; Nielsen, M.R.S.; Lundbye-Christensen, S.; Schmidt, E.B.; Handberg, A. The Effect of Low-Dose Marine n-3 Fatty Acids on Plasma Levels of sCD36 in Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Trial. Mar. Drugs 2013, 11, 3324-3334. https://doi.org/10.3390/md11093324
Venø SK, Nielsen MRS, Lundbye-Christensen S, Schmidt EB, Handberg A. The Effect of Low-Dose Marine n-3 Fatty Acids on Plasma Levels of sCD36 in Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Trial. Marine Drugs. 2013; 11(9):3324-3334. https://doi.org/10.3390/md11093324
Chicago/Turabian StyleVenø, Stine Krogh, Michael René Skjelbo Nielsen, Søren Lundbye-Christensen, Erik Berg Schmidt, and Aase Handberg. 2013. "The Effect of Low-Dose Marine n-3 Fatty Acids on Plasma Levels of sCD36 in Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Trial" Marine Drugs 11, no. 9: 3324-3334. https://doi.org/10.3390/md11093324
APA StyleVenø, S. K., Nielsen, M. R. S., Lundbye-Christensen, S., Schmidt, E. B., & Handberg, A. (2013). The Effect of Low-Dose Marine n-3 Fatty Acids on Plasma Levels of sCD36 in Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Trial. Marine Drugs, 11(9), 3324-3334. https://doi.org/10.3390/md11093324




