Antiviral Sulfoquinovosyldiacylglycerols (SQDGs) from the Brazilian Brown Seaweed Sargassum vulgare
Abstract
:1. Introduction
2. Results and Discussion
2.1. Lipid Fractionation
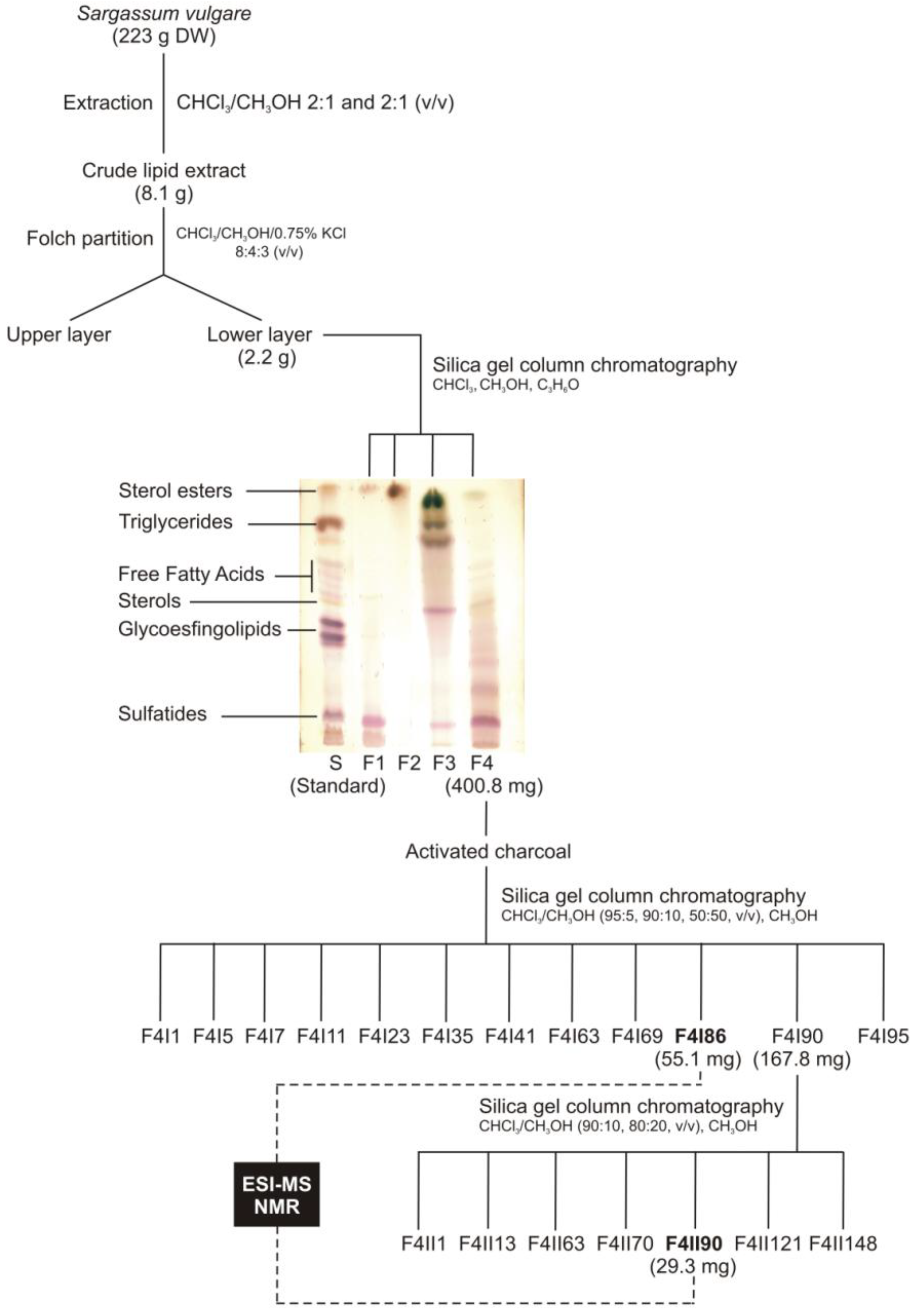
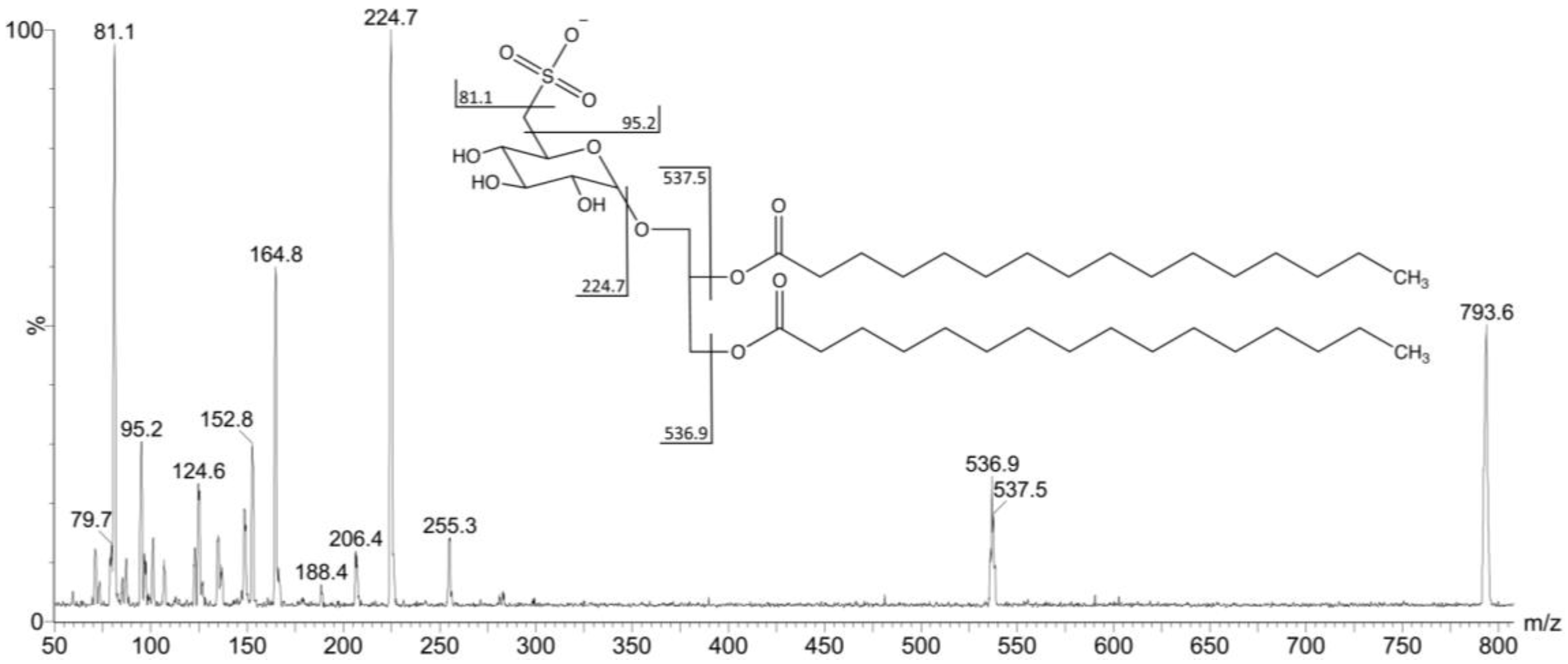
2.2. Mass Spectrometry of Sulfolipids
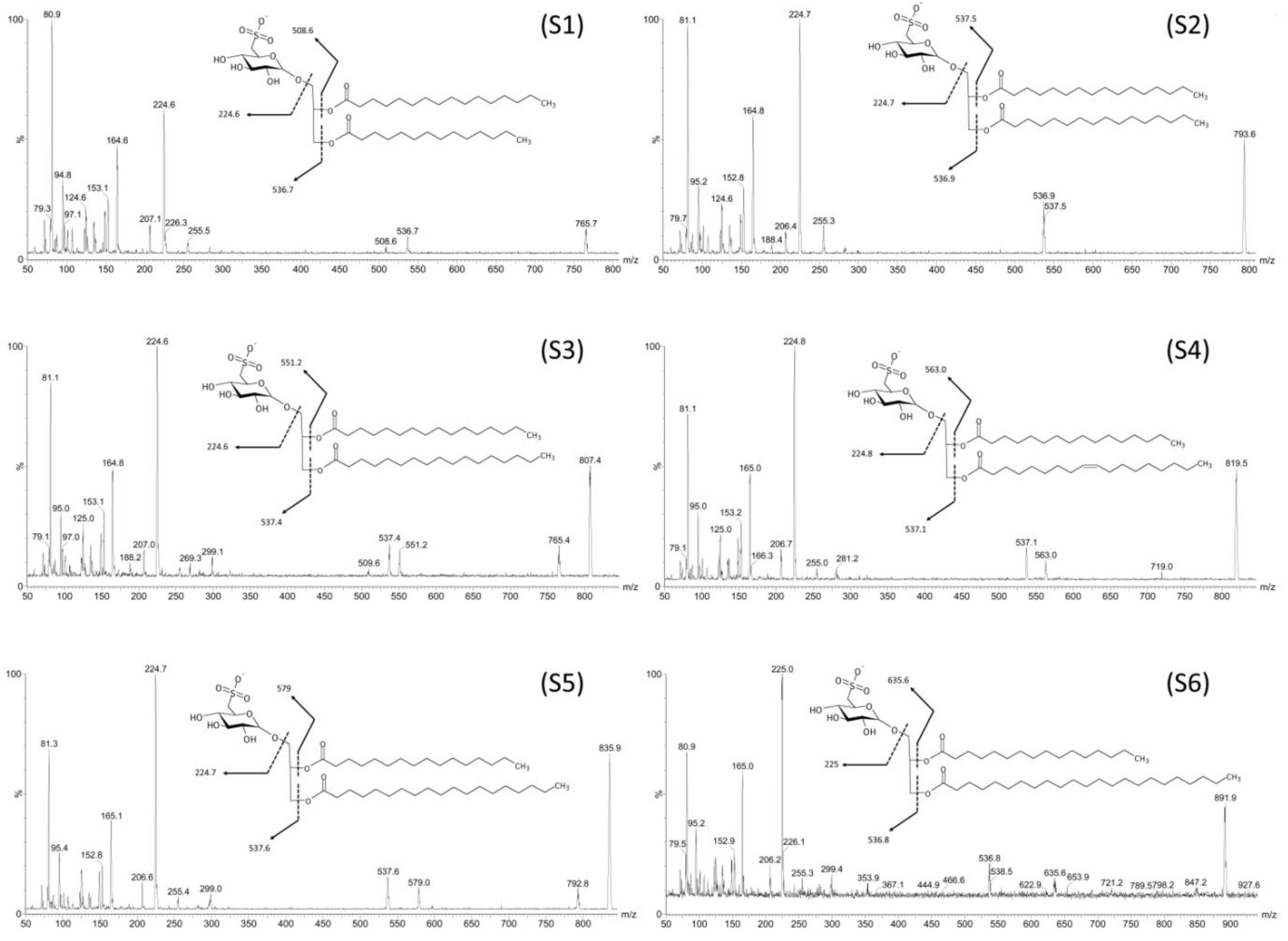
| Fraction | Compound | R1/R2 | [M − H]− m/z | [M − R1]− m/z | [M − R2]− m/z |
|---|---|---|---|---|---|
| F4I86, F4II90 | S1 | C14:0/C16:0 | 765.7 | 536.7 | 508.6 |
| F4I86, F4II90 | S2 | C16:0/C16:0 | 793.6 | 537 | 537 |
| F4I86, F4II90 | S3 | C17:0/C16:0 | 807.4 | 537.4 | 551.2 |
| F4I86 | S4 | C18:1/C16:0 | 819.5 | 537.1 | 563 |
| F4I86, F4II90 | S5 | C19:0/C16:0 | 835.9 | 537.6 | 579 |
| F4I86 | S6 | C23:0/C17:0 | 891.9 | 536.8 | 635.6 |
2.3. NMR Spectroscopy of Sulfolipids

3. Experimental Section
3.1. Biological Material
3.2. Extraction and Fractionation of Lipids
3.3. Mass Spectrometry
3.4. Nuclear Magnetic Resonance
3.5. Cells and Viruses
3.6. Cytotoxicity Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Vo, T.; Ngo, D.; Ta, Q.V.; Kim, S. Marine organisms as a therapeutic source against herpes simplex virus infection. Eur. J. Pharm. Sci. 2011, 44, 11–20. [Google Scholar] [CrossRef]
- Brady, R.C.; Bernstein, D.I. Treatment of herpes simplex virus infections. Antiviral. Res. 2004, 61, 73–81. [Google Scholar] [CrossRef]
- Celum, C.L. The interaction between herpes simplex virus and human immunodeficiency virus. Herpes 2004, 11, 36A–45A. [Google Scholar]
- Bacon, T.H.; Levin, M.J.; Leary, J.J.; Sarisky, R.T.; Sutton, D. Herpes simplex virus resistance to acyclovir and penciclovir after two decades of antiviral therapy. Clin. Microbiol. Rev. 2003, 16, 114–128. [Google Scholar] [CrossRef]
- Morfin, F.; Thouvenot, D. Herpes simplex virus resistance to antiviral drugs. J. Clin. Virol. 2003, 26, 29–37. [Google Scholar] [CrossRef]
- Maschek, J.A.; Baker, B.J. The chemistry of algal secondary metabolism. In Algal Chemical Ecology; Amsler, C.D., Ed.; Springer-Verlag: Berlin, Germany, 2008; pp. 1–24. [Google Scholar]
- Chakraborty, S.; Ghosh, U. Oceans: A store house of drugs—a review. J. Pharm. Res. 2010, 3, 1293–1296. [Google Scholar]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar] [CrossRef]
- Kim, S.K.; Karadeniz, F. Anti-HIV activity of extracts and compounds from marine algae. Adv. Food. Nutr. Res. 2011, 64, 255–265. [Google Scholar] [CrossRef]
- Saha, S.; Navid, M.H.; Bandyopadhyay, S.S.; Schnitzler, P.; Ray, B. Sulfated polysaccharides from Laminaria angustata: Structural features and in vitro antiviral activities. Carbohydr. Polym. 2012, 87, 123–130. [Google Scholar] [CrossRef]
- Bandyopadhyay, S.S.; Navid, M.H.; Ghosh, T.; Schnitzler, P.; Ray, B. Structural features and in vitro antiviral activities of sulfated polysaccharides from Sphacelaria indica. Phytochemistry 2011, 72, 276–283. [Google Scholar] [CrossRef]
- Cardozo, F.T.; Camelini, C.M.; Mascarello, A.; Rossi, M.J.; Nunes, R.J.; Barardi, C.R.; de Mendonça, M.M.; Simões, C.M. Antiherpetic activity of a sulfated polysaccharide from Agaricus brasiliensis mycelia. Antivir. Res. 2011, 92, 108–114. [Google Scholar] [CrossRef]
- Adhikari, U.; Mateu, C.G.; Chattopadhyay, K.; Pujol, C.A.; Damonte, E.B.; Ray, B. Structure and antiviral activity of sulfated fucans from Stoechospermum marginatum. Phytochemistry 2006, 67, 2474–2482. [Google Scholar] [CrossRef]
- de Souza, L.M.; Sassaki, G.L.; Romanos, M.T.; Barreto-Bergter, E. Structural characterization and anti-HSV-1 and HSV-2 activity of glycolipids from the marine algae Osmundaria obtusiloba isolated from Southeastern Brazilian coast. Mar. Drugs 2012, 10, 918–931. [Google Scholar] [CrossRef]
- Kind, T.; Meissen, J.K.; Yang, D.W.; Nocito, F.; Vaniya, A.; Cheng, Y.S.; VanderGheynst, J.S.; Fiehn, O. Qualitative analysis of algal secretions with multiple mass spectrometric platforms. J. Chromagraph. A 2012, 1244, 139–147. [Google Scholar]
- Packter, N.M. Lipids in plants and microbes: By J L Harwood and N J Russel. pp 162. George Allen & Unwin, London. 1984. Biochem. Educ. 1985, 13, 94–94. [Google Scholar] [CrossRef]
- Morimoto, T.; Murakami, N.; Nagatsu, A.; Sakakibara, J. Studies on glycolipids VII. Isolation of two new sulfoquinovosyl diacylglycerols from green alga Chlorella vulgaris. Chem. Pharm. Bull. 1993, 41, 1545–1548. [Google Scholar] [CrossRef]
- Gustafson, K.R.; Cardellina, J.H.; Fuller, R.W.; Weislow, O.S.; Kiser, R.F.; Snader, K.M.; Patterson, G.M.L.; Boyd, M.R. AIDS-antiviral sulfolipids from cyanobacteria (Blue-Green Algae). J. Natl. Cancer Inst. 1989, 81, 1254–1258. [Google Scholar] [CrossRef]
- Kikuchi, H.; Tsukitani, Y.; Manda, T.; Fujii, T.; Nakanishi, H.; Kobayashi, M.; Kitagawa, I. Marine Natural Products. X. Pharmacologically active glycolipids from the Okinawan marine sponge Phyllospongia foliascens (PALLAS). Chem. Pharm. Bull. 1982, 30, 3544–3547. [Google Scholar] [CrossRef]
- Chirasuwan, N.; Chaiklahan, R.; Kittakoop, P.; Chanasattru, W.; Ruengjitchatchawalya, M.; Tanticharoen, M.; Bunnag, B. Anti HSV-1 activity of sulphoquinovosyl diacylglycerol isolated from Spirulina platensis. Scienceasia 2009, 35, 137–141. [Google Scholar]
- Wang, H.; Li, Y.L.; Shen, W.Z.; Rui, W.; Ma, X.J.; Cen, Y.Z. Antiviral activity of a sulfoquinovosyldiacylglycerol (SQDG) compound isolated from the green alga Caulerpla racemosa. Bot. Mar. 2007, 50, 185–190. [Google Scholar]
- Khotimchenko, S.V. Distribution of glyceroglycolipids in marine algae and grasses. Chem. Nat. Compd. 2002, 38, 223–229. [Google Scholar] [CrossRef]
- Barreto-Bergter, E.; Sassaki, G.L.; Souza, L.M. Structural analysis of fungal cerebrosides. Front. Microbiol. 2011, 2, 1–11. [Google Scholar]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Zianni, R.; Bianco, G.; Lelario, F.; Losito, I.; Palmisano, F.; Cataldi, T.R.I. Fatty acid neutral losses observed in tandem mass spectrometry with collision-induced dissociation allows regiochemical assignment of sulfoquinovosyl-diacylglycerols. J. Mass Spectrom. 2013, 48, 205–215. [Google Scholar]
- Sassaki, G.L.; Gorin, P.A.J.; Tischer, C.A.; Iacomini, M. Sulfonoglycolipids from the lichenized basidiomycete Dictyonema glabratum: Isolation, NMR, and ESI-MS approaches. Glycobiology 2001, 11, 345–351. [Google Scholar] [CrossRef]
- Souza, L.M.; Iacomini, M.; Gorin, P.A.J.; Sari, R.S.; Haddad, M.A.; Sassaki, G.L. Glyco- and sphingophosphonolipids from the medusa Phyllorhiza punctata: NMR and ESI-MS/MS fingerprints. Chem. Phys. Lipids 2007, 145, 85–96. [Google Scholar] [CrossRef]
- Tsai, C.J.; Pan, B.S. Identification of sulfoglycolipid bioactivities and characteristic fatty acids of marine macroalgae. J. Agric. Food Chem. 2012, 60, 8404–8410. [Google Scholar] [CrossRef]
- Al-Fadhli, A.; Wahidulla, S.; D’Souza, L. Glycolipids from the red alga Chondria armata (Kutz.) Okamura. Glycobiology 2006, 16, 902–915. [Google Scholar] [CrossRef]
- Fusetani, N.; Hashimoto, Y. Structures of two water-soluble hemolysins isolated from green alga Ulva pertus. Agric. Biol. Chem. 1975, 39, 2021–2025. [Google Scholar] [CrossRef]
- Araki, S.; Sakurai, T.; Oohusa, T.; Kayama, M.; Sato, N. Characterization of sulphonoquinovosyl diacylglycerol from marine red alga. Plant Cell Physiol. 1989, 30, 775–781. [Google Scholar]
- Son, W.B. Glycolipids from Gracilaria verrucosa. Phytochemistry 1990, 29, 307–309. [Google Scholar] [CrossRef]
- Siddantha, A.K.; Ramvat, B.K.; Chauvan, V.D.; Achari, B.; Dutta, P.K.; Pakrashi, S.C. Sulphoglycolipid from the green alga Enteromorpha flexuosa (Wulf). J. Agric. Bot. Mar. 1991, 34, 365–367. [Google Scholar]
- Logvinov, S.V.; Denisenko, V.A.; Dmitrenok, P.S.; Moiseenko, O.P. Sulfoquinovosyldiacylglycerins from Scaphechinus mirabilis. Chem. Nat. Compd. 2012, 48, 175–179. [Google Scholar] [CrossRef]
- Harada, H.; Yamashita, U.; Kurihara, H.; Fukushi, E.; Kawabata, J.; Kamei, Y. Antitumor activity of palmitic acid found as a selective cytotoxic substance in a marine red alga. Anticancer Res. 2002, 22, 2587–2590. [Google Scholar]
- Kabara, J.J.; Swieczkowski, D.M.; Truant, J.P.; Conley, A.J.; Truant, J.P. Fatty-acids and derivatives as antimicrobial agents. Antimicrob. Agents Chemother. 1972, 2, 23–28. [Google Scholar] [CrossRef]
- Santoyo, S.; Jaime, L.; Plaza, M.; Herrero, M.; Rodriguez-Meizoso, I.; Ibañez, E.; Reglero, G. Antiviral compounds obtained from microalgae commonly used as carotenoid sources. J. Appl. Phycol. 2012, 24, 731–741. [Google Scholar] [CrossRef]
- Lee, D.Y.; Lin, X.; Paskaleva, E.E.; Liu, Y.; Puttamadappa, S.S.; Thornber, C.; Drake, J.R.; Habulin, M.; Shekhtman, A.; Canki, M. Palmitic acid is a novel CD4 fusion inhibitor that blocks HIV entry and infection. AIDS Res. Hum. Retrovir. 2009, 25, 1231–1241. [Google Scholar] [CrossRef]
- Spear, P.G. Herpes simplex virus: Receptors and ligands for cell entry. Cell Microbiol. 2004, 6, 401–410. [Google Scholar] [CrossRef]
- Wang, W.; Wang, S.X.; Guan, H.S. The antiviral activities and mechanisms of marine polysaccharides: An Overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar] [CrossRef]
- Neyts, J.; Snoeck, R.; Schols, D.; Balzarini, J.; Esko, J.D.; Van Schepdael, A.; De Clercq, E. Sulfated polymers inhibit the interaction of human cytomegalovirus with cell surface heparan sulfate. Virology 1992, 189, 48–58. [Google Scholar] [CrossRef]
- Skipski, V.P. Thin layer chromatography of neutral glycolipids. Methods Enzymol. 1975, 35, 396–425. [Google Scholar] [CrossRef]
- Markoulatos, P.; Georgopoulou, A.; Siafakas, N.; Plakokefalos, E.; Tzanakaki, G.; Kourea-Kremastinou, J. Laboratory diagnosis of common herpesvirus infections of the central nervous system by a multiplex PCR assay. J. Clin. Microbiol. 2001, 39, 4426–4432. [Google Scholar] [CrossRef]
- Borenfreund, E.; Puerner, J.A. Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol. Lett. 1985, 24, 119–124. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating 50 per cent end-points. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Nishimura, T.; Toku, K.; Fukuyasu, H. Antiviral compounds. XII. Antiviral activity aminohydrazones of alkoxyphenyl substituted carbonyl compounds against influenza virus in eggs and mice. Kitasato Arch. Exp. Med. 1977, 50, 39–46. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Plouguerné, E.; De Souza, L.M.; Sassaki, G.L.; Cavalcanti, J.F.; Villela Romanos, M.T.; Da Gama, B.A.P.; Pereira, R.C.; Barreto-Bergter, E. Antiviral Sulfoquinovosyldiacylglycerols (SQDGs) from the Brazilian Brown Seaweed Sargassum vulgare. Mar. Drugs 2013, 11, 4628-4640. https://doi.org/10.3390/md11114628
Plouguerné E, De Souza LM, Sassaki GL, Cavalcanti JF, Villela Romanos MT, Da Gama BAP, Pereira RC, Barreto-Bergter E. Antiviral Sulfoquinovosyldiacylglycerols (SQDGs) from the Brazilian Brown Seaweed Sargassum vulgare. Marine Drugs. 2013; 11(11):4628-4640. https://doi.org/10.3390/md11114628
Chicago/Turabian StylePlouguerné, Erwan, Lauro M. De Souza, Guilherme L. Sassaki, Jéssica Figueiredo Cavalcanti, Maria Teresa Villela Romanos, Bernardo A. P. Da Gama, Renato Crespo Pereira, and Eliana Barreto-Bergter. 2013. "Antiviral Sulfoquinovosyldiacylglycerols (SQDGs) from the Brazilian Brown Seaweed Sargassum vulgare" Marine Drugs 11, no. 11: 4628-4640. https://doi.org/10.3390/md11114628




