Chlorinated Didemnins from the Tunicate Trididemnum solidum
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bioassay-Guided Isolation
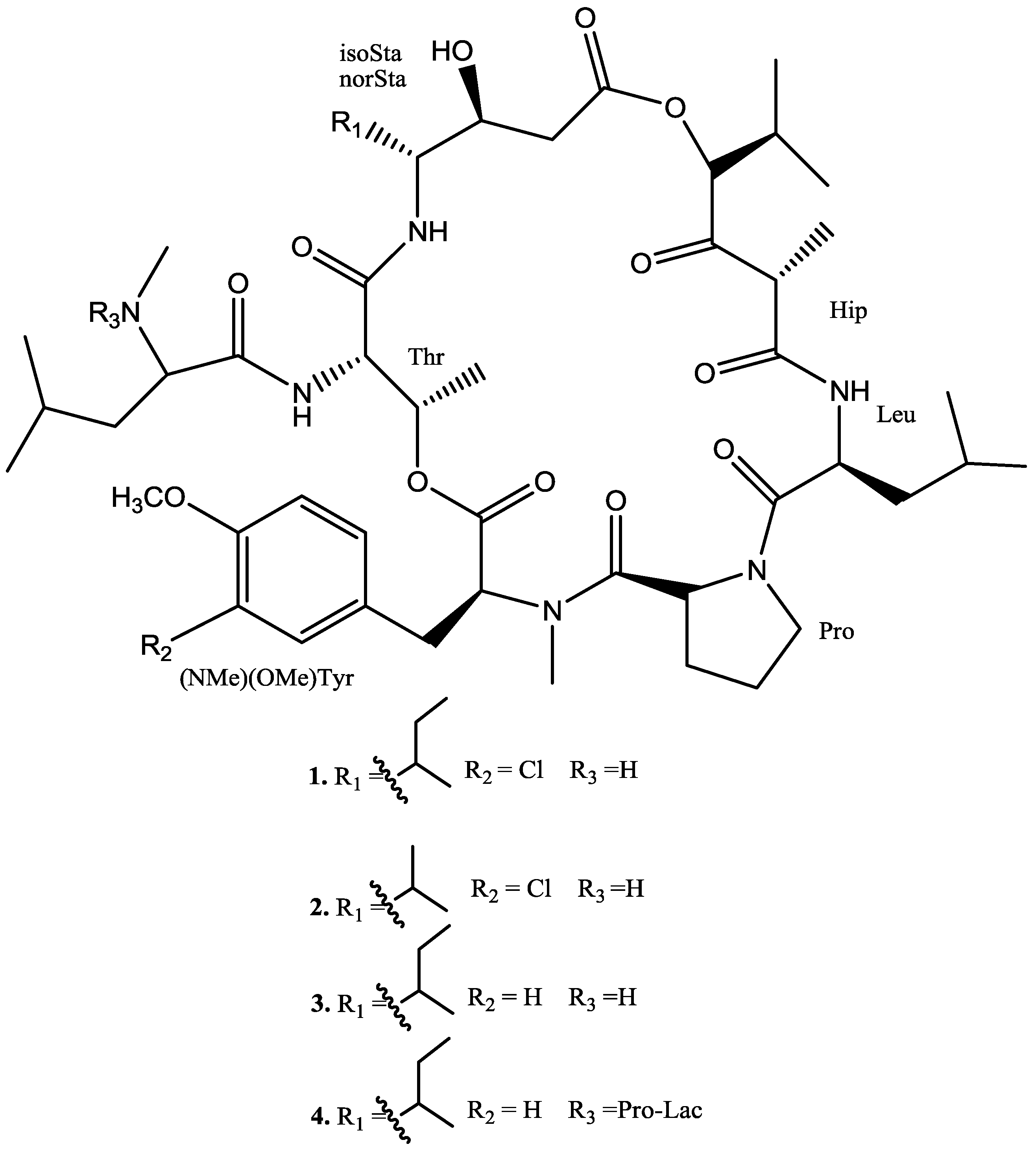
2.2. Structural Elucidation of the New Compounds
| 1 a | 2 b | ||||
|---|---|---|---|---|---|
| Subunit | C# | δC | δH (J in Hz) | δC | δH (J in Hz) |
| IsoSta or norSta | NH | - | 7.62 (d, 9.5) | - | 7.8 (d, 9.8) |
| CO | 169.1 | - | 169.7 | - | |
| C2H2 | 40.7 | 2.9 (m)/4.06 (d, 17.0) | 40.8 | 3.02 (m)/4.03 (d, 18.0) | |
| C3H | 67.1 | 4.68 (t, 10) | 67.6 | 4.69 (t, 10.2) | |
| C4H | 56.3 | 4.66 (brt, 9.5) | 57.7 | 4.51 (brt, 9.7) | |
| C5H | 34.5 | 2.51 (m) | 27.7 | 2.5 (m) | |
| C6H2/C6H3 | 28.1 | 1.45/1.73 (m) | 17.2 | 1.16 (d, 6.6) | |
| C7H3 | 12.1 | 1.07 (t, 7.5) | - | - | |
| C5CH3 | 14.6 | 1.18 (d, 7.0) | 22.1 | 1.22 (d, 6.3) | |
| Hip | CO | 172.2 | - | 172.5 | - |
| C2H | 49.4 | 4.27 * | 49.9 | 4.34 (q, 6.9) | |
| C3 | 204.6 | - | 208.8 | - | |
| C4H | 80.2 | 5.63 (d, 4.0) | 80.4 | 5.61 (d, 4.0) | |
| C5H | 29.6 | 2.39 m | 29.8 | 2.43 (m) | |
| C6H3 | 18.9 | 0.86 (d, 6.0) | 19.2 | 0.83 * | |
| C5CH3 | 16.9 | 0.84 (d, 6.0) | 17.1 | 0.85 * | |
| C2CH3 | 14.8 | 1.49 (d, 7.0) | 15.2 | 1.53 (d, 6.7) | |
| Leu | NH | - | 8.31 (d, 9.0) | - | 8.35 (d, 9.0) |
| CO | 171.2 | - | 171.4 | - | |
| CαH | 49.4 | 5.21 (brt, 9.5) | 49.8 | 5.22 (brt, 9.4) | |
| CβH2 | 41.8 | 1.96/1.68 (m) | 41.9 | 1.79/1.48 (m) | |
| CγH | 24.7 | 1.70 (m) * | 24.9 | 1.9 (m) | |
| CδH3 | 20.9 | 0.83 (d, 6.0) | 21.3 | 0.94 * | |
| Cδ′H3 | 22.0 | 1.03 (d, 6.0) | 21.5 | 1.03 * | |
| Pro | CO | 170.6 | - | 169.3 | - |
| CαH | 57.8 | 4.83 (m) | 58.8 | 4.83 (m) | |
| CβH2 | 27.7 | 1.96 (m),1.71 (m) | 27.9 | 2.04 (m), 1.78 (m) | |
| CγH2 | 24.8 | 1.63 (m), 1.91 (m) | 24.9 | 1.58 (m), 1.90 (m) | |
| CδH2 | 47.3 | 3.50 (m), 3.60 (m) | 47.5 | 3.50 (m), 3.58 (m) | |
| Cl-N,O-Me2Tyr | CO | 170.6 | - | 170.9 | - |
| NCH3 | 38.4 | 2.67 (s) | 38.8 | 2.66 (s) | |
| CαH | 65.3 | 4.28 * | 65.7 | 4.26 (dd, 9.5, 5.2) | |
| CβH2 | 33.9 | 3.50 (m) | 33.9 | 3.55 (m) | |
| Cγ | 131.6 | - | 131.2 | - | |
| CσH | 131.5 | 7.43 (s) | 131.8 | 7.44 (s) | |
| Cε | 122.1 | - | 122.4 | - | |
| Cξ | 154.1 | - | 154.5 | - | |
| CρH | 112.7 | 7.02 (d, 8.0) | 113.2 | 7.02 (d, 8.0) | |
| CψH | 129.3 | 7.26 (d, 8.0) | 129.7 | 7.28 (d, 8.0) | |
| OCH3 | 55.9 | 3.78 (s) | 56.3 | 3.80 (s) | |
| Thr | CO | 168.9 | - | 169.3 | - |
| NH | - | 10.3 (d, 6.0) | - | 10.3 (d, 6.1) | |
| CαH | 57.8 | 5.37 (brd, 6.5) | 58.1 | 5.38 (brd, 6.5) | |
| CβH | 70.4 | 5.74 (brd, 5.5) | 70.5 | 5.77 (brd, 6.3) | |
| Cβ-CH3 | 15.9 | 1.64 (d, 6.0) | 16.2 | 1.69 (d, 6.0) | |
| MeLeu | CO | 169.6 | - | 170.6 | - |
| NCH3 | 31.2 | 3.14 (s) | 31.8 | 3.15(s) | |
| CαH | 59.6 | 4.8 * | 59.1 | 4.84 (m) | |
| CβH2 | 39.5 | 2.15 (m) | 39.5 | 2.6 (m) | |
| CγH | 25.0 | 1.68 (m) | 25.2 | 1.8 (m) | |
| CδH3 | 23.4 | 0.84 (d, 6.0) | 23.7 | 0.94 (d, 5.6) | |
| Cδ′H3 | 21.8 | 0.92 (d, 6.0) | 22.9 | 1.03 * | |
| Compound | iNOS | NF-κB |
|---|---|---|
| 1 | 0.4 | 0.26 |
| 2 | 0.42 | 0.62 |
| 3 | 0.19 | 0.19 |
| 4 | 0.002 | 0.03 |
| Parthenolide α | 2.8 | 2.8 |
| Compound | SK-MEL | KB | BT-549 | SK-OV-3 | VERO |
|---|---|---|---|---|---|
| 1 | 0.12 | 0.26 | 0.16 | 0.26 | 4.8 |
| 2 | 0.06 | 0.42 | 0.16 | 0.38 | 2.08 |
| 3 | 0.055 | 0.16 | 0.07 | 0.16 | 4.78 |
| 4 | 0.022 | 0.09 | 0.02 | 0.1 | 0.15 |
| Doxorubicin α | 1.1 | 1.66 | 1.01 | 1.66 | 14 |
2.3. Acid Hydrolysis and Marfey Analysis
3. Experimental Section
3.1. General
3.2. Tunicate Material
3.3. Extraction and Isolation
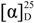 : −99.3 (c, 0.35, CHCl3); UV (MeOH) λmax (log ε) 208 (2.45) 232 (2.63); 1H- and 13C-NMR data, see Table 1; HRESIMS m/z [M + H]+ 977.53505/979.53655 (actual 977.53662) (calcd for C49H78ClN6O12+ 977.53662).
: −99.3 (c, 0.35, CHCl3); UV (MeOH) λmax (log ε) 208 (2.45) 232 (2.63); 1H- and 13C-NMR data, see Table 1; HRESIMS m/z [M + H]+ 977.53505/979.53655 (actual 977.53662) (calcd for C49H78ClN6O12+ 977.53662).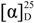 : −86.3 (c, 0.40, CHCl3); UV (MeOH) λmax (log ε) 206 (2.39) 230 (2.49); 1H- and 13C-NMR data, see Table 1; HERSIMS at m/z [M + H]+ 963.52073/965.52101 (calcd for C48H77 ClN6O12+ 963.52097).
: −86.3 (c, 0.40, CHCl3); UV (MeOH) λmax (log ε) 206 (2.39) 230 (2.49); 1H- and 13C-NMR data, see Table 1; HERSIMS at m/z [M + H]+ 963.52073/965.52101 (calcd for C48H77 ClN6O12+ 963.52097).3.4. Biological Assays
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Rinehart, K.L., Jr.; Gloer, J.B.; Hughes, R.G., Jr.; Renis, H.E.; McGovren, J.P.; Swynenberg, E.B.; Stringfellow, D.A.; Kuentzel, S.L.; Liv, L.H. Didemnins: Antiviral and antitumor depsipeptides from a Caribbean tunicate. Science 1981, 212, 933–935. [Google Scholar]
- Rinehart, K.L., Jr.; Gloer, J.B.; Cook, J.C., Jr.; Mizsak, S.A.; Scahill, T.A. Structures of the didemnins, antiviral and cytotoxic depsipeptides from a Caribbean tunicate. J. Am. Chem. Soc. 1981, 103, 1857–1859. [Google Scholar] [CrossRef]
- Rinehart, K.L., Jr.; Gloer, J.B.; Wilson, G.R.; Hughes, R.G., Jr.; Li, L.H.; Renis, H.E.; McGovren, J.P. Antiviral and antitumor compounds from tunicates. Fed. Proc. 1983, 42, 87–90. [Google Scholar]
- Rinehart, K.L., Jr. Didemnin and Its Biological Properties. In Peptides, Chemistry and Biology; ESCOM: Leiden, The Netherlands, 1988; pp. 626–631. [Google Scholar]
- Jouin, P.; Poncet, J.; Dufour, M.N.; Aumelas, A.; Pantaloni, A.; Cros, S.; Francois, G. Antineoplastic activity of didemnin congeners: Nordidemnin and modified chain analogues. J. Med. Chem. 1991, 34, 486–491. [Google Scholar] [CrossRef]
- Rinehart, K.L.; Kishore, V.; Bible, K.C.; Sakai, R.; Sullins, D.W.; Li, K.M. Didemnins and tunichlorin: Novel natural products from the marine tunicate Trididemnum solidum. J. Nat. Prod. 1988, 51, 1–21. [Google Scholar] [CrossRef]
- Ishida, K.; Matsuda, H.; Murakami, M.; Yamaguchi, K. Micropeptins 478-A and -B, plasmin inhibitors from the cyanobacterium Microcystis aeruginaosa. J. Nat. Prod. 1997, 60, 184–187. [Google Scholar] [CrossRef]
- Apter, S.G.; Carmeli, S. Protease inhibitors from a water bloom of the cyanobacterium Microcystis aeruginosa. J. Nat. Prod. 2009, 72, 1429–1436. [Google Scholar] [CrossRef]
- Marfey, P. Determination of d-amino acids. II. Use of a bifunctional reagent, 1,5-difluoro-2,4-dinitrobenzene. Carlsberg Re. Commun. 1984, 49, 591–596. [Google Scholar] [CrossRef]
- Gnaim, J.M.; Sheldon, R.A. Selective ortho-chlorination of phenol using sulfuryl chloride in the presence of t-butylaminomethyl polystyrene as a heterogenous amine catalyst. Tetrahedron Lett. 2004, 45, 8471–8473. [Google Scholar] [CrossRef]
- Sakai, R.; Stroh, J.G.; Sullins, D.W.; Rinehart, K. Seven new didemnins from the marine tunicate Trididemnum solidum. J. Am. Chem. Soc. 1995, 117, 3734–3748. [Google Scholar] [CrossRef]
- Li, W.R.; Ewing, W.R.; Harris, B.D.; Joullie, M.M. Total synthesis and structural investigations of Didemnins A, B and C. J. Am. Chem. Soc. 1990, 112, 7569–7672. [Google Scholar]
- Tsukimoto, M.; Nagaoka, M.; Shishido, Y.; Fujimoto, J.; Nishisaka, F.; Matsumoto, S.; Harunari, E.; Imada, C.; Matsuzaki, T. Bacterial production of the tunicate-derived antitumor cyclic depsipeptides didemnin B. J. Nat. Prod. 2011, 74, 2329–2331. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sethi, G.; Nair, A.; Ichikawa, H. Nuclear factor-κB: A holy grail in cancer prevention and therapy. Curr. Signal Transduct. Ther. 2006, 1, 25–52. [Google Scholar] [CrossRef]
- Melisi, D.; Chiao, P.J. NF-kappa B as a target for cancer therapy. Expert Opin. Ther. Targets 2007, 11, 133–144. [Google Scholar] [CrossRef]
- Brasier, A.R. The NF-kappaB regulatory network. Cardiovasc. Toxicol. 2006, 6, 111–130. [Google Scholar] [CrossRef]
- Navab, M.; Gharavi, N.; Watson, A.D. Inflammation and metabolic disorders. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 459–464. [Google Scholar] [CrossRef]
- Yoo, H.J.; Kang, H.J.; Jung, H.J.; Kim, K.; Lim, C.J.; Park, E.H. Anti-Inflammatory, anti-angiogenic and anti-nociceptive activities of Saururus chinensis extract. J. Ethnopharmacol. 2008, 120, 282–286. [Google Scholar] [CrossRef]
- Lirk, P.; Hoffmann, G.; Rieder, J. Inducible nitric oxide synthase—Time for reappraisal. Curr. Drug Targets Inflamm. Allergy 2002, 1, 89–108. [Google Scholar] [CrossRef]
- Kroncke, K.D.; Fehsel, K.; Kolb-Bachofen, V. Inducible nitric oxide synthase in human diseases. Clin. Exp. Immunol 1998, 113, 147–156. [Google Scholar] [CrossRef]
- Ma, G.; Khan, S.I.; Benavides, G.; Schuhly, W.; Fischer, N.H.; Khan, I.A.; Pasco, D.S. Inhibition of NF-kappaB-mediated transcription and induction of apoptosis by melampolides and repandolides. Cancer Chemother. Pharmacol. 2007, 60, 35–43. [Google Scholar] [CrossRef]
- Quang, D.N.; Harinantenaina, L.; Nishizawa, T.; Hashimoto, T.; Kohchi, C.; Soma, G.; Asakawa, Y. Inhibition of nitric oxide production in RAW 264.7 cells by azaphilones from xylariaceous fungi. Biol. Pharm. Bull. 2006, 29, 34–37. [Google Scholar] [CrossRef]
- Mustafa, J.; Khan, S.I.; Ma, G.; Walker, L.A.; Khan, I.A. Synthesis and anticancer activities of fatty acid analogs of podophyllotoxin. Lipids 2004, 39, 167–172. [Google Scholar]
- Xu, Y.; Kerten, R.D.; Nam, S.-J.; Lu, L.; Al-Wuwailem, A.M.; Zheng, H.; Fenical, W.; Dorrestein, P.C.; Moore, S.B. Bacterial biosynthesis and maturation of the didemnin anti-cancer agents. J. Am. Chem. Soc. 2012, 134, 8625–8632. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ankisetty, S.; Khan, S.I.; Avula, B.; Gochfeld, D.; Khan, I.A.; Slattery, M. Chlorinated Didemnins from the Tunicate Trididemnum solidum. Mar. Drugs 2013, 11, 4478-4486. https://doi.org/10.3390/md11114478
Ankisetty S, Khan SI, Avula B, Gochfeld D, Khan IA, Slattery M. Chlorinated Didemnins from the Tunicate Trididemnum solidum. Marine Drugs. 2013; 11(11):4478-4486. https://doi.org/10.3390/md11114478
Chicago/Turabian StyleAnkisetty, Sridevi, Shabana I. Khan, Bharathi Avula, Deborah Gochfeld, Ikhlas A. Khan, and Marc Slattery. 2013. "Chlorinated Didemnins from the Tunicate Trididemnum solidum" Marine Drugs 11, no. 11: 4478-4486. https://doi.org/10.3390/md11114478
APA StyleAnkisetty, S., Khan, S. I., Avula, B., Gochfeld, D., Khan, I. A., & Slattery, M. (2013). Chlorinated Didemnins from the Tunicate Trididemnum solidum. Marine Drugs, 11(11), 4478-4486. https://doi.org/10.3390/md11114478




