Anti HSV-1 Activity of Halistanol Sulfate and Halistanol Sulfate C Isolated from Brazilian Marine Sponge Petromica citrina (Demospongiae)
Abstract
:1. Introduction
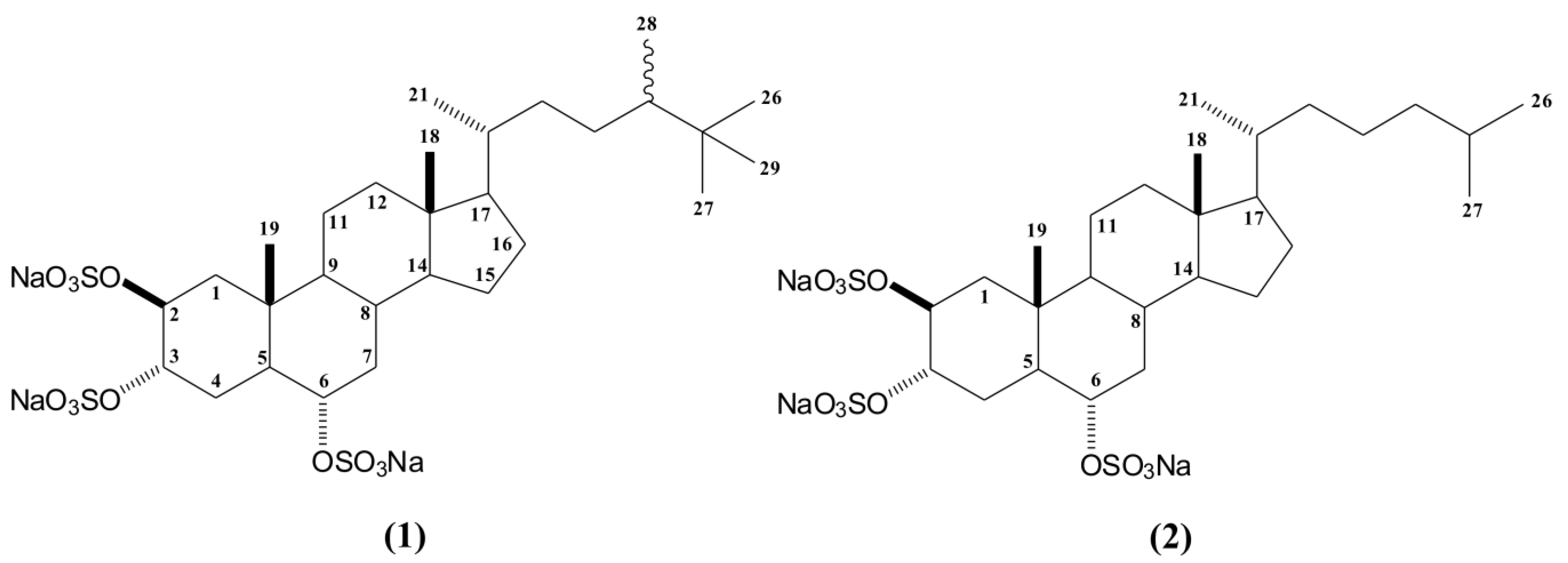
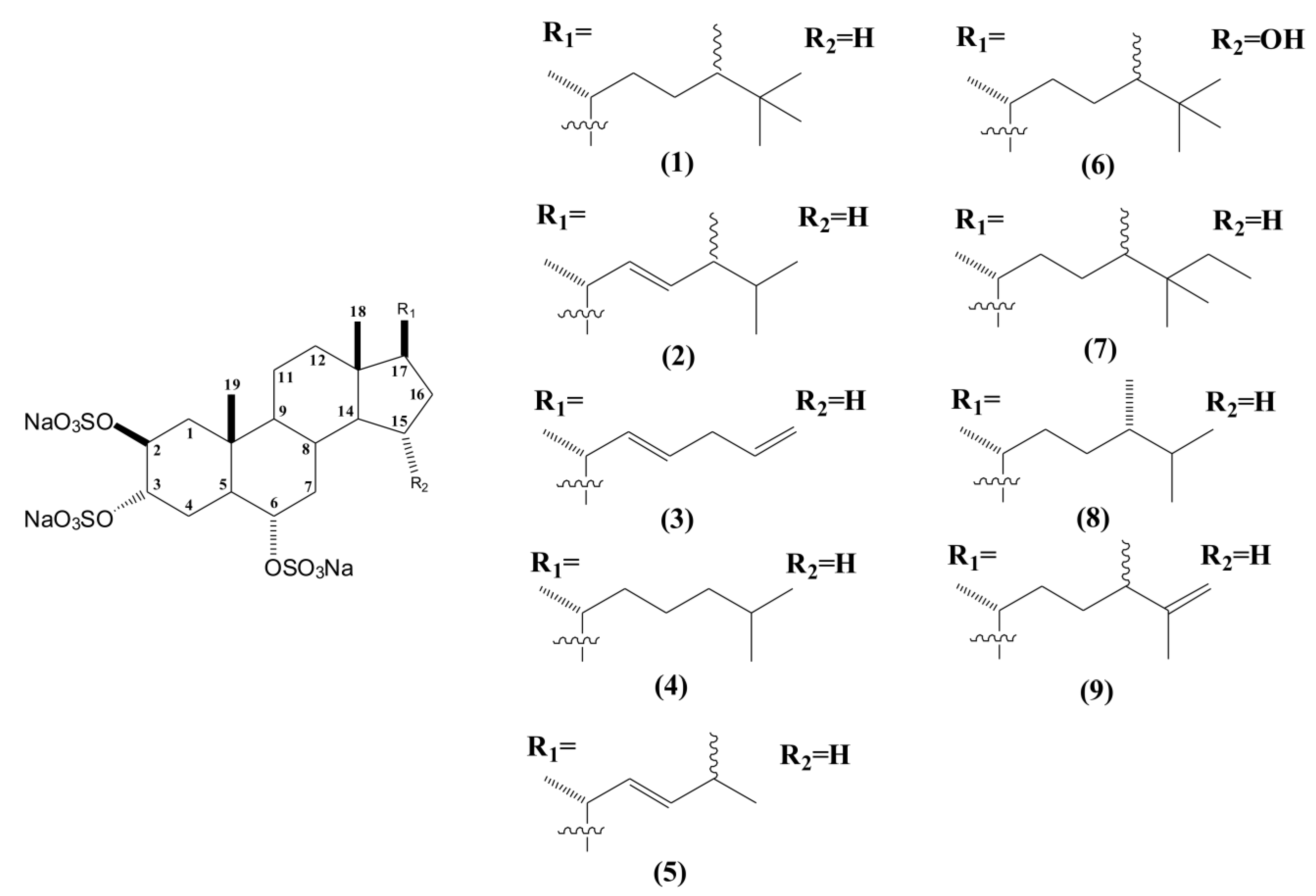
2. Results and Discussion
2.1. Bioguided Fractionation of the n-Butanol Fraction of P. citrina
3. Experimental Section
3.1. General Experimental Procedures
3.2. Sponge Collection
3.3. Extraction and Isolation of Compounds 1 and 2

3.4. Anti-HSV-1 Activity
3.4.1. Virus and Cell Line
3.4.2. Cytotoxicity Assay
4. Conclusions
Acknowledgments
Abbreviations
| CMC | Carboxymethylcellulose |
| COSY | Correlation Spectroscopy |
| ESI | Electrospray ionization |
| HMBC | Heteronuclear Multiple Bond Correlation |
| HSQC | Heteronuclear Single Quantum Correlation |
| HSV | Herpes Simplex Virus |
| MEM | Minimal Essential Medium |
| NMR | Nuclear Magnetic Resonance |
| PFU | Plaque Forming Units |
| TSH | Halistanol-Enriched Fraction |
Conflict of Interest
References
- Nakakawa, E.; Reynold, S.J. The management of herpes simplex virus infections in HIV infected patients: Current issues and the role of cidofovir. Virus Adapt. Treat. 2011, 3, 35–43. [Google Scholar]
- Sagar, S.; Kaur, M.; Minneman, K.P. Antiviral lead compounds from marine sponges. Mar. Drugs 2010, 8, 2619–2638. [Google Scholar] [CrossRef]
- Snoeck, R. Antiviral therapy of herpes simplex. Int. J. Antimicrob. Agents 2000, 16, 157–159. [Google Scholar] [CrossRef]
- De Clercq, E. Antivirals: Past, present and future. Biochem. Pharmacol. 2013, 85, 727–744. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Rodríguez, A.D.; Berlinck, R.G.S.; Fusetani, N. Marine pharmacology in 2007–8: Marine compounds with antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous system, and other miscellaneous mechanisms of action. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2011, 153, 191–222. [Google Scholar] [CrossRef]
- Moura, L.A.; Ortiz-Ramirez, F.; Cavalcanti, D.N.; Ribeiro, S.M.; Muricy, G.; Teixeira, V.L.; Fuly, A.L. Evaluation of marine brown algae and sponges from Brazil as anticoagulant and antiplatelet products. Mar. Drugs 2011, 9, 1346–1358. [Google Scholar] [CrossRef]
- Sepčić, K.; Kauferstein, S.; Mebs, D.; Tur, T. Biological activities of aqueous and organic extracts from tropical marine sponges. Mar. Drugs 2010, 8, 1550–1566. [Google Scholar] [CrossRef]
- Uzair, B.; Mahmmod, Z.; Tabassum, S. Antiviral activity of natural products extracted from marine organisms. BioImpacts 2011, 1, 203–211. [Google Scholar]
- Abbas, S.; Kelly, M.; Bowling, J.; Sims, J.; Waters, A.; Hamann, M. Advancement into the arctic region for bioactive sponge secondary metabolites. Mar. Drugs 2011, 9, 2423–2437. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine pharmacology. Nat. Prod. Rep. 2000, 77, 135–140. [Google Scholar]
- Gordaliza, M. Cytotoxic terpene quinones from marine sponges. Mar. Drugs 2010, 8, 2849–2870. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Ivanchina, N.V.; Krasokhin, V.B.; Makarieva, T.N.; Stonik, V.A. Glycosides from marine sponges (Porifera, Demospongiae): Structures, taxonomical distribution, biological activities and biological roles. Mar. Drugs 2012, 10, 1671–1710. [Google Scholar] [CrossRef]
- Noro, J.C.; Kalaitzis, J.A.; Neilan, B.A. Papua new guinea marine sponges. Chem. Biodivers. 2012, 9, 2077–2095. [Google Scholar]
- Donia, M.; Hamann, M.T. Marine natural products and their potential applications as anti-infective agents. Lancet Infect. Dis. 2003, 3, 338–348. [Google Scholar] [CrossRef]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug. Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef]
- Yasuhara-Bell, J.; Lu, Y. Marine compounds and their antiviral activities. Antivir. Res. 2010, 86, 231–240. [Google Scholar] [CrossRef]
- Berlinck, R.G.S.; Hadju, E.; Rocha, R.M.; Oliveira, J.H.H.L.; Hernandez, I.L.C.; Seleghim, M.H.R.; Granato, A.C.; Almeida, E.V.R.; Nunez, C.V.; Muricy, G.; et al. Challenges and rewards of research in marine natural products chemistry in Brazil. J. Nat. Prod. 2004, 67, 510–522. [Google Scholar] [CrossRef]
- Chairman, K.; Jeyamala, M.; Sankar, S.; Murugan, A.; Ranjit Singh, A.J.A. Immunomodulating Properties of Bioactive Compounds Present in Aurora globostellata. Int. J. Mar. Sci. 2013, 3, 151–157. [Google Scholar]
- Mayer, A.M.; Hamann, M.T. Marine pharmacology in 2000: Marine compounds with antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiplatelet, antituberculosis, and antiviral activities; affecting the cardiovascular, immune, and nervous systems and other miscellaneous mechanisms of action. Mar. Biotechnol. 2004, 6, 37–52. [Google Scholar] [CrossRef]
- Tziveleka, L.A.; Vagias, C.; Roussis, V. Natural products with anti-HIV activity from marineorganisms. Curr. Top. Med. Chem. 2003, 3, 1512–1535. [Google Scholar] [CrossRef]
- Zhou, X.; Lin, X.; Guo, X.; Yang, B.; Yang, X.W.; Liu, Y. Chemical constituents of the sponge Mycale species from south China sea. Rec. Nat. Prod. 2013, 7, 119–123. [Google Scholar]
- Munro, M.H.; Blunt, J.W.; Dumdei, E.J.; Hickford, S.J.; Lill, R.E.; Li, S.; Battershill, C.N.; Duckworth, A.R. The discovery and development of marine compounds with pharmaceutical potential. J. Biotechnol. 1999, 70, 15–25. [Google Scholar] [CrossRef]
- Sawadogo, W.R.; Schumacher, M.; Teiten, M.H.; Cerella, C.; Dicato, M.; Diederich, M. A Survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2011. Molecules 2013, 18, 3641–3673. [Google Scholar] [CrossRef]
- Sipkema, D.; Franssen, M.C.; Osinga, R.; Tramper, J.; Wijffels, R.H. Marine sponges as pharmacy. Mar. Biotechnol. 2005, 7, 142–162. [Google Scholar] [CrossRef]
- Muricy, G.; Hajdu, E.; Minervino, J.V.; Madeira, A.V.; Peixinho, S. Systematic revision of the genus Petromica Topsent (Demospongiae:Halichondrida), with a new species from the southwestern Atlantic. Hydrobiologia 2001, 443, 103–128. [Google Scholar] [CrossRef]
- Marinho, P.R.; Muricy, G.R.S.; Silva, M.F.L.; Marval, M.G.; Laport, M.S. Antibiotic-resistant bacteria inhibited b extracts and fractions from Brazilian marine sponges. Braz. J. Pharmacogn. 2010, 20, 267–275. [Google Scholar]
- Silva, A.C.; Kratz, J.M.; Farias, F.M.; Henriques, A.T.; Santos, J.; Leonel, R.M.; Lerner, C.; Mothes, B.; Barardi, C.R.M.; Simões, C.M.O. In vitro antiviral activity of marine sponges collected off Brazilian Coast. Biol. Pharm. Bull. 2006, 29, 135–140. [Google Scholar] [CrossRef]
- Bianco, E.M.; Oliveira, S.Q.; Rigotto, C.; Tonini, M.; Guimaraes, T.R.; Bittencourt, F.; Gouvêa, L.; Aresi, C.; Almeida, M.T.R.; Moritz, M.I.G.; et al. Anti-infective potential of marine invertebrates and seaweeds from the Brazilian coast. Molecules 2013, 18, 5761–5778. [Google Scholar] [CrossRef]
- Kossuga, M.H.; Lira, S.P.; Nascimento, A.P.; Gambardella, M.T.P.; Berlinck, G.S.; Torres, Y.R.; Nascimento, G.G.F.; Pimenta, E.F.; Silva, M.; Thiemann, O.H.; et al. Isolantion and biological activities of secondary metabolites from the sponges monanchora aff. arbuscula, aplysina sp. petromica ciocalyptoides and topsentia ophiraphidies, from the ascidian didemnum ligulum and from the octocoral carijoa riisei. Quim. Nova 2007, 5, 1194–1202. [Google Scholar]
- Marinho, P.R.; Simas, N.K.; Kuster, R.M.; Duarte, R.S.; Fracalanzza, S.E.L.; Ferreira, D.F.; Romanos, M.T.V.; Muricy, G.; Demarval, M.G.; Laport, M.S. Antibacterial activity and cytotoxicity analysis of halistanol trisulfate from marine sponge Petromica citrina. J. Antimicrob. Chemother. 2012, 67, 2396–2400. [Google Scholar] [CrossRef]
- Fusetani, N.; Matsunaga, S.; Konosu, S. Bioactive marine metabolites II. Halistanol sulfate, as antimicrobial novel steroid sulfate from the marine sponge Halichondria cf. moorei Bergquist. Tetrahedron Lett. 1981, 22, 1985–1988. [Google Scholar] [CrossRef]
- Kanazawa, S.; Fusetani, N.; Matsunaga, S. Halistanol sulfates A–E. New steroids sulfates, from a marine sponge, Epipolasis sp. Tetrahedron 1992, 48, 5467–5472. [Google Scholar] [CrossRef]
- Sperry, S.; Crews, P. Haliclostanone sulfate and halistanol sulfate from an indo-pacific Haliclona sponge. J. Nat. Prod. 1997, 60, 29–32. [Google Scholar] [CrossRef]
- Bifulco, I.B.; Minale, L.; Riccio, R. Novel HIV-inhibitory halistanol sulfates F–H from a marine sponge, Pseudoaxinissa digitata. J. Nat. Prod. 1994, 57, 164–167. [Google Scholar] [CrossRef]
- Aiello, A.; Fattorusso, E.; Menna, M. Steroids from sponges: Recent reports. Steroids 1999, 64, 687–714. [Google Scholar] [CrossRef]
- Kicha, A.A.; Ivanchina, N.V.; Kalinovskii, A.I.; Dmitrenok, P.S.; Sokolova, E.V.; Agafonova, I.G. Sulfated steroid glycosides from the viet namese starfish Linckia laevigata. Chem. Nat. Comp. 2007, 43, 76–80. [Google Scholar] [CrossRef]
- Levina, E.V.; Aminin, D.L.; Kovalchuk, S.N.; Kozhemyako, V.B.; Dyshlovoi, S.A.; Kalinovskii, A.I.; Dmitrenok, P.S. Polar steroids from Solaster endeca starfish and the physiological activity of polar steroids from three starfish species. Russ. J. Bioorg. Chem. 2010, 36, 233–239. [Google Scholar] [CrossRef]
- McKee, T.C.; Cardellina, J.H.; Riccio, R.; D’auria, M.V.; Iorizzi, M.; Minale, L.; Moran, R.A.; Gulakowski, R.J.; Mcmahon, J.B.; Buckheit, R.W.; et al. HIV-inhibitory natural products, 11. Comparative studies of sulfated sterols from marine invertebrates. J. Med. Chem. 1994, 37, 793–797. [Google Scholar] [CrossRef]
- Whitson, E.L.; Bugni, T.S.; Chockalingam, P.S.; Concepcion, G.P.; Harper, M.K.; He, M.; Hooper, J.N.A.; Mangalindan, G.C.; Ritacco, F.; Ireland, C.M. Spheciosterol sulfates, PKCζ inhibitors from a philippine sponge Spheciospongia sp. J. Nat. Prod. 2008, 71, 1213–1217. [Google Scholar] [CrossRef]
- Jin, Y.; Fotso, S.; Yongtang, Z.; Sevvana, M.; Laatsch, H.; Zhang, W. Halichondria sulfonic acid, a new HIV-1 inhibitory guanidino-sulfonic acid, and halistanol sulfate isolated from the marine sponge Halichondria rugosa Ridley & Dendy. Nat. Prod. Res. 2006, 20, 1129–1135. [Google Scholar] [CrossRef]
- McKee, T.C.; Cardellina, J.H.; Tischler, M.; Snader, K.M.; Boyd, M.R. Ibisterol sulfate, a novel HIV-inhibitory sulfated sterol from the deep water sponge Topsentia sp. Tetrahedron Lett. 1993, 34, 389–392. [Google Scholar] [CrossRef]
- Sun, H.H.; Cross, S.S.; Gunasekera, M.; Koehn, F.E. Weinbersterol disulfates A and B, antiviral steroid sulfates from the sponge Petrosia weinbergi. Tetrahedron 1991, 47, 1185–1190. [Google Scholar] [CrossRef]
- Bouhlal, R.; Haslin, C.; Chermann, J.C.; Colliec-Jouault, S.; Sinquin, C.; Simon, G.; Cerantola, S.; Riadi, H.; Bourgougnon, N. Antiviral activities of sulfated polysaccharides isolated from Sphaerococcus coronopifolius (Rhodophytha, Gigartinales) and Boergeseniella thuyoides (Rhodophyta, Ceramiales). Mar. Drugs 2011, 9, 1187–1209. [Google Scholar] [CrossRef]
- Cardozo, F.T.G.S.; Camelini, C.M.; Mascarello, A.; Rossi, M.J.; Nunes, R.J.; Barardi, C.R.M.; Mendonça, M.M.; Simões, C.M.O. Antiherpetic activity of a sulfated polysaccharide from Agaricus brasiliensis mycelia. Antivir. Res. 2011, 92, 108–114. [Google Scholar] [CrossRef]
- Cardozo, F.T.G.S.; Camelini, C.M.; Larsen, I.; Carballo, E.; Jose, G.; Stern, R.; Brummel, R.; Rossi, M.; Simões, C.M.O.; Brandt, C. In vivo anti-HSV activity of a sulfated derivative of Agaricus brasiliensis mycelial polysaccharide. Antimicrob. Agents Chemother. 2013, 57, 2541–2549. [Google Scholar] [CrossRef]
- Zhu, W.; Chiu, L.C.M.; Ooi, V.E.C.; Chan, P.K.S.; Ang, P.O., Jr. Antiviral property and mode of action of a sulphated polysaccharide from Sargassum patens against herpes simplex virus type 2. Int. J. Antimicrob. Agents 2004, 24, 81–85. [Google Scholar]
- Witvrouw, M.; De Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen. Pharmacol. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Ghosh, T.; Chattopadhyay, K.; Marschall, M.; Karnakar, P.; Mandal, P.; Ray, B. Focus on antivirally active sulfated polysaccharides: From structure-activity analysis to clinical evaluation. Glycobiology 2009, 19, 2–15. [Google Scholar]
- Fontaine-Rodrigues, E.C.; Knipe, D.M. Herpes simplex virus ICP27 increases translation of a subset of viral late mRNAs. J. Virol. 2008, 28, 3538–3545. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Marcelletti, J.F. Synergistic inhibition of herpesvirus replication by docosanol and antiviral nucleoside analogs. Antivir. Res. 2002, 56, 153–166. [Google Scholar] [CrossRef]
- Chuanasa, T.; Phromjai, J.; Lipipun, V.; Likhitwitayawuid, K.; Suzuki, M.; Pramyothin, P.; Hattori, M.; Shiraki, K. Anti-Herpes Simplex Virus (HSV-1) activity of oxyresveratrol derived from Thai medicinal plant: Mechanism of action and therapeutic efficacy on cutaneous HSV-1 infection in mice. Antivir. Res. 2008, 80, 62–70. [Google Scholar] [CrossRef]
- Burleson, F.G.; Chamberts, T.M.; Wiedbrauk, D.L. Virology: A Laboratory Manual; Academic Press: San Diego, CA, USA, 1992; p. 250. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Silva, I.T.; Costa, G.M.; Stoco, P.H.; Schenkel, E.P.; Reginatto, F.H.; Simões, C.M.O. In vitro antiherpes effects of a c-glycosylflavonoid enriched fraction of Cecropia glaziovii Sneth. Lett. Appl. Microbiol. 2010, 51, 143–148. [Google Scholar]
- Ekblad, M.; Bergstrom, T.; Banwell, M.G.; Bonnet, M.; Renner, J.; Ferro, V.; Trybala, E. Anti-herpes simplex virus activities of two novel disulphated cyclitols. Antivir. Chem. Chemother. 2006, 17, 97–106. [Google Scholar]
- Bettega, J.M.R.; Teixeira, H.; Bassani, V.L.; Barardi, C.R.M.; Simões, C.M.O. Evaluation of the antiherpetic activity of standardized extracts of Achyrocline satureioides. Phytother. Res. 2004, 18, 819–823. [Google Scholar] [CrossRef]
- Bertol, J.W.; Rigotto, C.; De Pádua, R.M.; Kreis, W.; Barardi, C.R.M.; Braga, F.C.; Simões, C.M.O. Antiherpes activity of glucoevatromonoside, a cardenolide isolated from a Brazilian cultivar of Digitalis lanata. Antivir. Res. 2011, 92, 73–80. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Da Rosa Guimarães, T.; Quiroz, C.G.; Rigotto, C.; De Oliveira, S.Q.; De Almeida, M.T.R.; Bianco, É.M.; Moritz, M.I.G.; Carraro, J.L.; Palermo, J.A.; Cabrera, G.; et al. Anti HSV-1 Activity of Halistanol Sulfate and Halistanol Sulfate C Isolated from Brazilian Marine Sponge Petromica citrina (Demospongiae). Mar. Drugs 2013, 11, 4176-4192. https://doi.org/10.3390/md11114176
Da Rosa Guimarães T, Quiroz CG, Rigotto C, De Oliveira SQ, De Almeida MTR, Bianco ÉM, Moritz MIG, Carraro JL, Palermo JA, Cabrera G, et al. Anti HSV-1 Activity of Halistanol Sulfate and Halistanol Sulfate C Isolated from Brazilian Marine Sponge Petromica citrina (Demospongiae). Marine Drugs. 2013; 11(11):4176-4192. https://doi.org/10.3390/md11114176
Chicago/Turabian StyleDa Rosa Guimarães, Tatiana, Carlos Guillermo Quiroz, Caroline Rigotto, Simone Quintana De Oliveira, Maria Tereza Rojo De Almeida, Éverson Miguel Bianco, Maria Izabel Goulart Moritz, João Luís Carraro, Jorge Alejandro Palermo, Gabriela Cabrera, and et al. 2013. "Anti HSV-1 Activity of Halistanol Sulfate and Halistanol Sulfate C Isolated from Brazilian Marine Sponge Petromica citrina (Demospongiae)" Marine Drugs 11, no. 11: 4176-4192. https://doi.org/10.3390/md11114176