Formation of a Volunteer Harmful Algal Bloom Network in British Columbia, Canada, Following an Outbreak of Diarrhetic Shellfish Poisoning
Abstract
:1. Introduction
1.1. Shellfish Poisoning
1.2. History of Shellfish Poisonings in British Columbia
1.3. Impact of Harmful Algal Blooms on the Shellfish Industry
1.4. Shellfish Marine Biotoxin and Phytoplankton Monitoring Programs
1.5. The Journey from a DSP Outbreak to a Volunteer Phytoplankton Monitoring Network
2. Methods

2.1. Symposium Structure for Garnering Feedback
2.2. Establishment of a Volunteer Harmful Algal Monitoring Network for Shellfish Growers in BC
3. Results
3.1. Symposium Evaluation
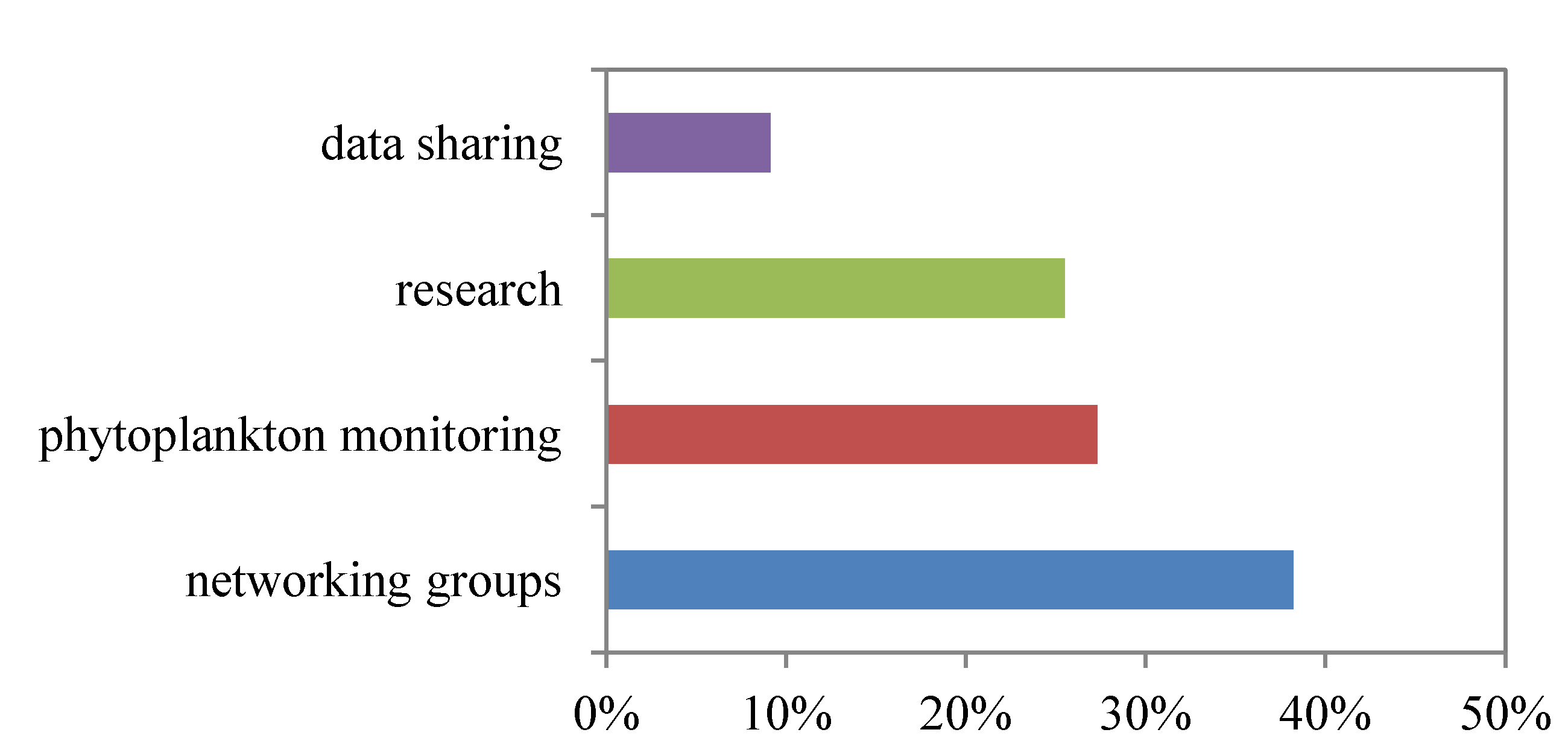
| Responses to evaluation survey | n (%) |
|---|---|
| Q. 2 Identify things learned (n = 106) | |
| Advanced knowledge | 36 (34.0) |
| Phytoplankton monitoring | 20 (31.1) |
| Basic knowledge | 14 (13.2) |
| Program roles for shellfish management | 13 (12.2) |
| Laboratory testing | 12 (11.3) |
| US collaborations and research | 6 (5.7) |
| Knowledge gaps | 5 (4.7) |
3.2. Formation and Structure of the Volunteer Harmful Algal Monitoring Network in BC

| Resource | Source |
|---|---|
| Plankton Identification Handbook for the Shellfish Growers on the West Coast of Canada | [38] |
| Harmful Algae Bloom Event Response Protocols for the Shellfish Grower of British Columbia 2013 | [39] |
| Phyto’ pedia—The Phytoplankton Encyclopaedia Project | [40] |
| List of On-line Resources Provided by BC Shellfish Grower’s Association | [41] |
4. Discussion and Conclusions
Acknowledgments
Conflicts of Interest
References
- British Columbia Ministry of Environment and British Columbia Seafood Industry, The 2011 British Columbia Seafood Industry Year in Review; British Columbia Ministry of Environment and British Columbia Seafood Industry: Victoria, BC, Canada, 2011; pp. 1–16.
- Van Dolah, F.M. Marine algal toxins: Origins, health effects, and their increased occurrence. Environ. Health Perspect 2000, 108, S133–S141. [Google Scholar] [CrossRef]
- Gerssen, A.; Pol-Hofstad, I.E.; Poelman, M.; Mulder, P.P.J.; Van den Top, H.J.; De Boer, J. Marine toxins: Chemistry, toxicity, occurrence and detection, with special reference to the Dutch situation. Toxins 2010, 2, 878–904. [Google Scholar] [CrossRef]
- Bricelj, V.M.; Connell, L.; Konoki, K.; Macquarrie, S.P.; Scheuer, T.; Catterall, W.A.; Trainer, V.L. Sodium channel mutation leading to saxitoxin resistance in clams increases risk of PSP. Nature 2005, 434, 763–767. [Google Scholar] [CrossRef]
- James, K.J.; Carey, B.; O’Halloran, J.; van Pelt, F.N.A.M.; Škrabáková, Z. Shellfish toxicity: Human health implications of marine algal toxins. Epidemiol. Infect. 2010, 138, 927–940. [Google Scholar] [CrossRef]
- Todd, E.C. Seafood-associated diseases and control in Canada. Rev. Sci. Tech. 1997, 16, 661–672. [Google Scholar]
- Fleming, L.E.; Broad, K.; Clement, A.; Dewailly, E.; Elmir, S.; Knap, A.; Pomponi, S.A.; Smith, S.; Solo Gabriele, H.; Walsh, P. Oceans and human health: Emerging public health risks in the marine environment. Mar. Pollut. Bull. 2006, 53, 545–560. [Google Scholar] [CrossRef]
- Taylor, M.; McIntyre, L.; Ritson, M.; Stone, J.; Bronson, R.; Bitzikos, O.; Rourke, W.; Galanis, E.; Team, O.I. Outbreak of diarrhetic shellfish poisoning associated with mussels, British Columbia, Canada. Mar. Drugs 2013, 11, 1669–1676. [Google Scholar] [CrossRef]
- Esenkulova, S.; Haigh, N. First report of Dinophysis species causing diarrhetic shellfish poisoning in British Columbia, Canada. Harmful Algae News 2012, 45, 16–17. [Google Scholar]
- Quayle, D.B. Paralytic Shellfish Poisoning—Safe Shellfish; Fisheries Research Board of Canada: Nanaimo, BC, Canada, 1966; pp. 1–11. [Google Scholar]
- Ngo, J. Assessing Environmental Data to Predict Shellfish Biotoxin Illness. University of British Columbia and BC Centre for Disease Control: Vancouver, BC, Canada, 2013; unpublished work. [Google Scholar]
- Jeffery, B.; Barlow, T.; Moizer, K.; Paul, S.; Boyle, C. Amnesic shellfish poison. Food Chem. Toxicol. 2004, 42, 545–557. [Google Scholar] [CrossRef]
- McIntyre, L. Participant Evaluations and Summary of Speaker Presentations. In Proceedings of Canadian Diarrhetic Shellfish Poisoning Symposium, North Vancouver, BC, Canada, 27 November 2012; pp. 1–17.
- Patel, M.M.; Hall, A.J.; Vinjé, J.; Parashar, U.D. Noroviruses: A comprehensive review. J. Clin. Virol. 2009, 44, 1–8. [Google Scholar] [CrossRef]
- Kelly, M.T.; Dan Stroh, E.M. Occurrence of Vibrionaceae in natural and cultivated oyster populations in the Pacific Northwest. Diagn. Microbiol. Infect. Dis. 1988, 9, 1–5. [Google Scholar] [CrossRef]
- Khaira, B.; Galanis, E. Descriptive epidemiology of Vibrio parahaemolyticus and other Vibrio species infections in British Columbia: 2001–2006. Can. Commun. Dis. Rep. 2007, 33, 12–22. [Google Scholar]
- David, S.; McIntyre, L.; MacDougall, L.; Kelly, D.; Liem, S.; Schallie´, K.; McNabb, A.; Houde, A.; Mueller, P.; Ward, P.; et al. An outbreak of norovirus caused by consumption of oysters from geographically dispersed harvest sites, British Columbia, Canada, 2004. Foodborne Pathog. Dis. 2007, 4, 349–358. [Google Scholar] [CrossRef]
- McIntyre, L.; Galanis, E.; Mattison, K.; Mykytczuk, O.; Buenaventura, E.; Wong, J.; Prystajecky, N.; Ritson, M.; Stone, J.; Moreau, D.; et al. Multiple clusters of norovirus among shellfish consumers linked to symptomatic oyster harvesters. J. Food Prot. 2012, 75, 1715–1720. [Google Scholar] [CrossRef]
- Landsberg, J.H. The effects of harmful algal blooms on aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Cassis, D.; Pearce, C.M.; Maldonado, M.T. Effects of the environment and culture depth on growth and mortality in juvenile Pacific oysters in the Strait of Georgia, British Columbia. Aquac. Environ. Interact. 2011, 1, 259–274. [Google Scholar]
- Taylor, F.J.R.; Harrison, P.J. Harmful Algal Blooms in Western Canadian Coastal Waters. In Harmful Algal Blooms in the PICES Region of the North Pacific; Taylor, F.J.R., Trainer, V.M., Eds.; PICES Scientific Reports: Vancouver, BC, Canada, 2002; Volumn 23, pp. 77–88. [Google Scholar]
- Keppler, C.J.; Hoguet, J.; Smith, K.; Ringwood, A.H.; Lewitus, A.J. Sublethal effects of the toxic alga Heterosigma akashiwo on the southeastern oyster (Crassostrea virginica). Harmful Algae 2005, 4, 275–285. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. Harmful algal blooms in the Australian region. Mar. Pollut. Bull. 1992, 25, 186–190. [Google Scholar] [CrossRef]
- Shumway, S.E.; Barter, J.; Sherman-Caswell, S. Auditing the impact of toxic algal blooms on oysters. Environ. Auditor 1990, 2, 41–56. [Google Scholar]
- Anderson, D.M.; Hoagland, P.; Kaoru, Y.; White, A.W. Estimated Annual Economic Impacts from Harmful Algal Blooms (HABs) in the United States; Woods Hole Oceanographic Institute: Woods Hole, MA, USA, 2000; pp. 1–96. [Google Scholar]
- Canadian Food Inspection Agency Canadian Shellfish Sanitation Program. Chapter 11—Control of Marine Biotoxins. Available online: http://www.inspection.gc.ca/english/fssa/fispoi/man/cssppccsm/chap11e.shtml (accessed on 4 April 2013).
- Havforskingsinstituttet Algaeinfo. Available online: http://algeinfo.imr.no/eng/ (accessed on 12 June 2013).
- Anderson, D.M.; Andersen, P.; Bricelj, V.M.; Cullen, J.J.; Rensel, J.E. Monitoring and Management Strategies for Harmful Algal Blooms in Coastal Waters; APEC # 201-MR-01.1; Asia Pacific Economic Program, Singapore and Intergovernmental Oceanographic Commission: Paris, France, 2001; pp. 1–274. [Google Scholar]
- Rhodes, L.L.; Mackenzie, A.L.; Kaspar, H.F.; Todd, K.E. Harmful algae and mariculture in New Zealand. ICES J. Mar. Sci. 2001, 58, 398–403. [Google Scholar]
- National Centers for Coastal Ocean Science Phytoplankton Monitoring Network (PMN). Available online: http://products.coastalscience.noaa.gov/pmn/default.aspx (accessed on 14 August 2013).
- The Scottish Association for Marine Science Monitoring programme for the presence of toxin producing plankton in shellfish production areas in Scotland. Available online: http://www.sams.ac.uk/keith-davidson/toxic-plankton-monitoring/?searchterm=phytoplankton%20monitoring (accessed on 15 August 2013).
- Fuica, N.M.; Rojas, X.; Clément, A.; Bittner, V.; Silva, M.T.; Uribe, C. HABs Occurrence and Impact on Salmon Farming in South of Chile: Analysis of the Monitoring Program from Intesal, SalmonChile. Available online: http://www.salmonchile.cl/salmociencia/002/Paper3.pdf (accessed on 14 August 2013).
- Government of Canada Bedford Institute of Oceanography. Available online: http://www.bio.gc.ca/index-eng.php (accessed on 13 June 2013).
- NOAA Northwest Fisheries Science Center and UW School of Environmental and Forest Sciences. ORHAB Partnership. Available online: http://www.orhab.org/index.html (accessed on 31 May 2013).
- Jack Rensel, J.E.; Haigh, N.; Tynan, T.J. Fraser river sockeye salmon marine survival decline and harmful blooms of Heterosigma akashiwo. Harmful Algae 2010, 10, 98–115. [Google Scholar]
- Haigh, N. HAMP 2012 Harmful Plankton Handbook; Vancouver Island University: Nanaimo, BC, Canada, 2012; pp. 1–55. [Google Scholar]
- Canadian Institute of Public Health Inspectors. Continuing Education Series Canadian Diarrhetic Shellfish Poisoning Symposium. Available online: http://www.ciphi.bc.ca/webinar/ (accessed on 31 May 2013).
- Haigh, N. Plankton Identification Handbook for Shellfish Growers on the West Coast of Canada; Microthalassia Consultants Inc.: Nanaimo, BC, Canada, 2013; pp. 1–59. [Google Scholar]
- Cassis, D. Harmful Algae Bloom Event Response Protocols for the Shellfish Growers of British Columbia 2013; AquaBC Consulting: Vancouver, BC, Canada, 2013; pp. 1–17. [Google Scholar]
- Phyto’ Pedia—The Phytoplankton Encyclopaedia Project. Available online: http://www.eos.ubc.ca/research/phytoplankton/index.html (accessed on 30 April 2013).
- BC Shellfish Growers Association Harmful Algae Monitoring Network. Available online: http://bcsga.ca/2013/05/harmful-algae-monitoring-network/ (accessed on 10 June 2013).
- Fisheries and Oceans Canada Biotoxin (PSP, ASP, DSP) & Sanitary Contamination Closures—Pacific Region. Available online: http://www.pac.dfo-mpo.gc.ca/fm-gp/contamination/biotox/index-eng.htm (accessed on 31 May 2013).
- Lewitus, A.J.; Horner, R.A.; Caron, D.A.; Garcia-Mendoza, E.; Hickey, B.M.; Hunter, M.; Huppert, D.D.; Kudela, R.M.; Langlois, G.W.; Largier, J.L.; et al. Harmful algal blooms along the North American west coast region: History, trends, causes, and impacts. Harmful Algae 2012, 19, 133–159. [Google Scholar] [Green Version]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
McIntyre, L.; Cassis, D.; Haigh, N. Formation of a Volunteer Harmful Algal Bloom Network in British Columbia, Canada, Following an Outbreak of Diarrhetic Shellfish Poisoning. Mar. Drugs 2013, 11, 4144-4157. https://doi.org/10.3390/md11114144
McIntyre L, Cassis D, Haigh N. Formation of a Volunteer Harmful Algal Bloom Network in British Columbia, Canada, Following an Outbreak of Diarrhetic Shellfish Poisoning. Marine Drugs. 2013; 11(11):4144-4157. https://doi.org/10.3390/md11114144
Chicago/Turabian StyleMcIntyre, Lorraine, David Cassis, and Nicola Haigh. 2013. "Formation of a Volunteer Harmful Algal Bloom Network in British Columbia, Canada, Following an Outbreak of Diarrhetic Shellfish Poisoning" Marine Drugs 11, no. 11: 4144-4157. https://doi.org/10.3390/md11114144




