Liraglutide Therapy for Type 2 Diabetes: Overcoming Unmet Needs
Abstract
:1. Introduction
- improving glycaemic control without compromising safety, e.g., hypoglycaemia
- preserving β-cell function
- providing clinically meaningful weight loss
- addressing cardiovascular risk factors accompanying diabetes
- offering a simple and flexible regimen

2. Background to the Incretin Effect
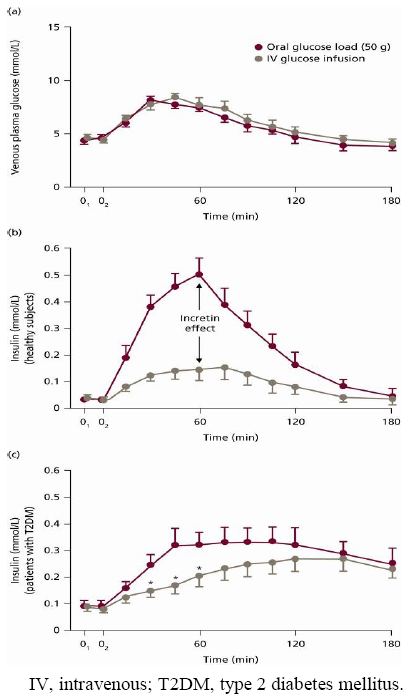
3. GLP-1 and its Relevance to Treating Unmet Needs in Type 2 Diabetes
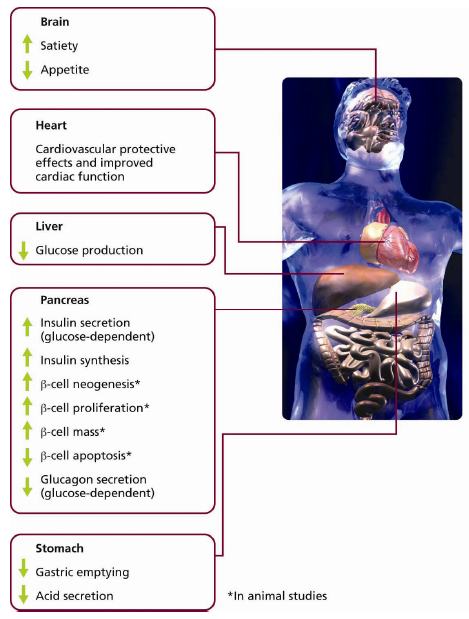
4. Clinical Limitations of GLP-1 Due to the Presence of DPP-4
5. Development of Liraglutide
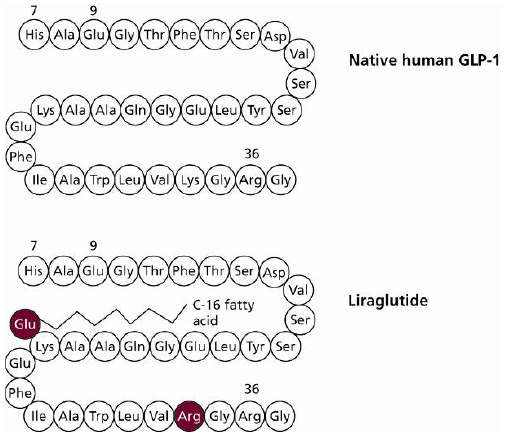
6. Clinical Effects of Liraglutide

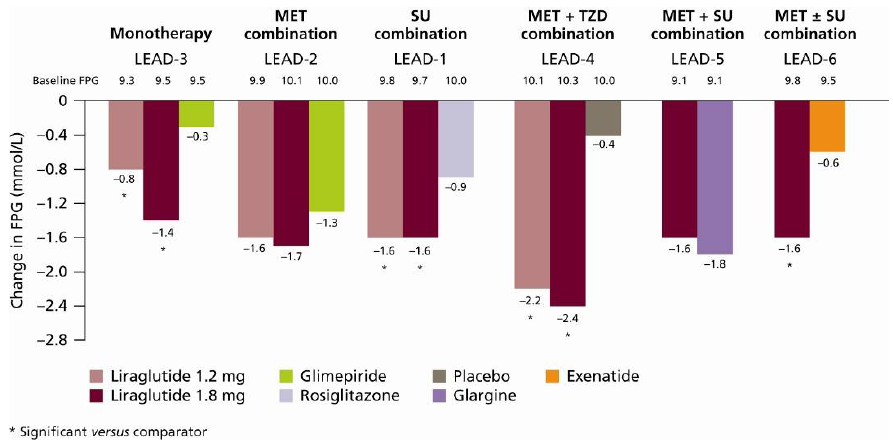
6.1. HbA1c, FPG and PPG
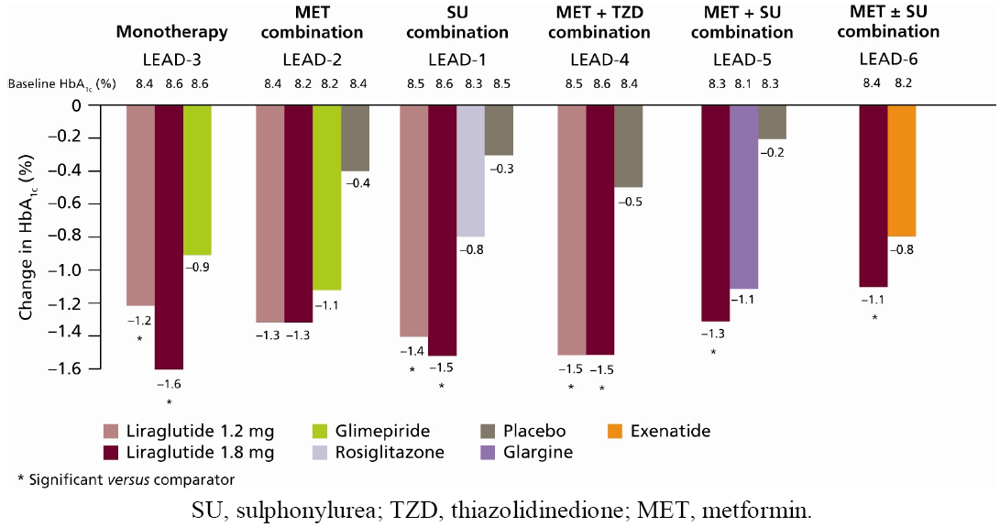

6.2. β-cell Function
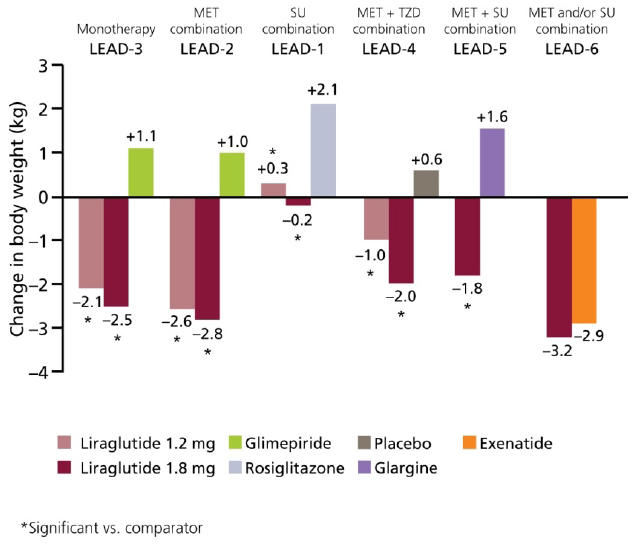
6.3. Weight
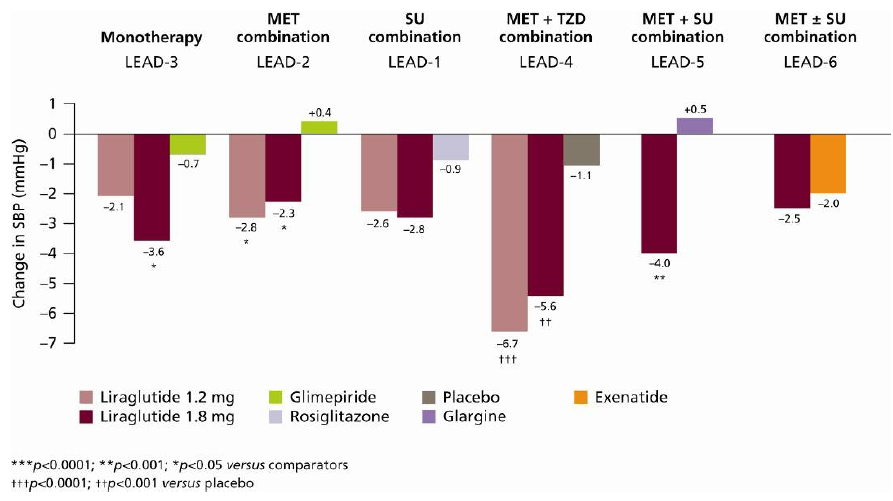
6.4. Systolic Blood Pressure
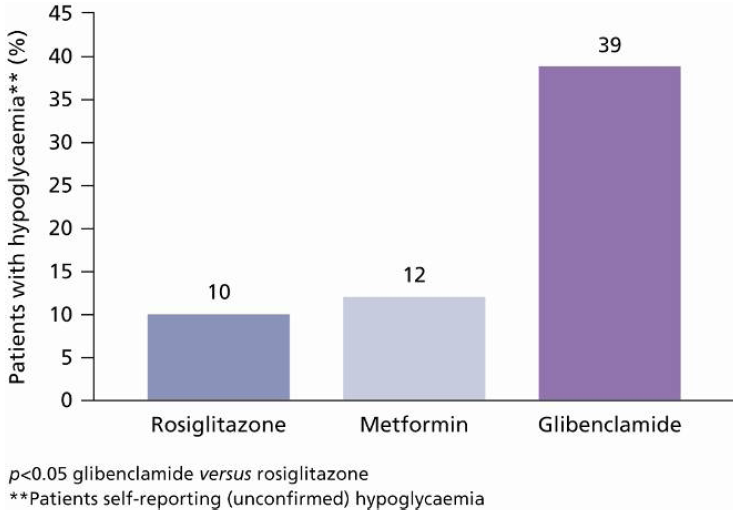
6.5. Safety and Tolerability

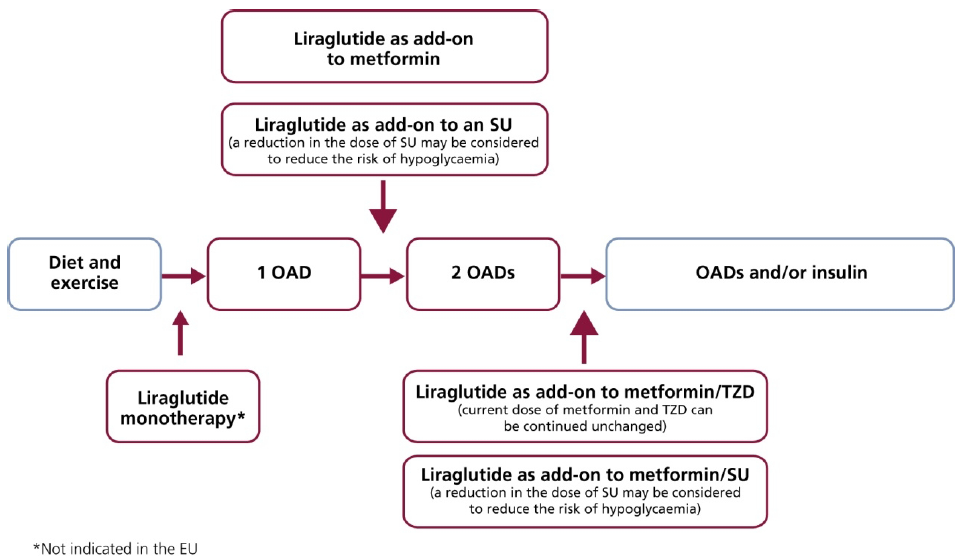
7. How Liraglutide Can Be Used in Clinical Practice
8. When Should Liraglutide Be Used in the Treatment Paradigm?
9. Conclusions
Acknowledgements
References
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998, 352, 837–853. [PubMed]
- Kahn, S.E.; Haffner, S.M.; Heise, M.A.; Herman, W.H.; Holman, R.R.; Jones, N.P.; Kravitz, B.G.; Lachin, J.M.; O'Neill, M.C.; Zinman, B.; Viberti, G. for the ADOPT Study Group. Glycemic durability of rosiglitazone, metformin or glyburide monotherapy. N. Engl. J. Med. 2006, 355, 2427–2443. [Google Scholar] [PubMed]
- Hubert, H.B.; Feinleib, M.; McNamara, P.M.; Castelli, W.P. Obesity as an independent risk factor for cardiovascular disease: A 26-year follow-up of participants in the Framingham Heart Study. Circulation 1983, 67, 968–977. [Google Scholar]
- Han, T.S.; Tijhuis, M.A.; Lean, M.E.; Seidell, J.C. Quality of life in relation to overweight and body fat distribution. Am. J. Public Health 1998, 88, 1814–1820. [Google Scholar]
- Odegard, P.S.; Capoccia, K. Medication taking and diabetes: A systematic review of the literature. Diabetes Educ. 2007, 33, 1014–1029. [Google Scholar]
- Baggio, L.; Drucker, J. Biology of incretins: GLP-1 and GIP. Gastroenterol. 2007, 132, 2131–2157. [Google Scholar]
- Nauck, M.; Stöckmann, F.; Ebert, R.; Creutzfeldt, W. Reduced incretin effect in type 2 (non-insulin dependent) diabetes. Diabetologia 1986, 29, 46–52. [Google Scholar]
- Vilsbøll, T.; Krarup, T.; Madsbad, S.; Holst, J.J. Defective amplification of the late phase insulin response to glucose by GIP in obese Type II diabetic patients. Diabetologia 2002, 45, 1111–1119. [Google Scholar]
- Højberg, P.V.; Vilsbøll, T.; Rabøl, R.; Knop, F.K.; Bache, M.; Krarup, T.; Holst, J.J.; Madsbad, S. Four weeks of near-normalisation of blood glucose improves the insulin response to glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide in patients with type 2 diabetes. Diabetologia 2009, 52, 199–207. [Google Scholar]
- Nauck, M.A.; Heimesaat, M.M.; Behle, K.; Holst, J.J.; Nauck, M.S.; Ritzel, R.; Hüfner, M.; Schmiegel, W.H. Effects of glucagon-like peptide 1 on counterregulatory hormone responses, cognitive functions, and insulin secretion during hyperinsulinemic, stepped hypoglycemic clamp experiments in healthy volunteers. J. Clin. Endocrinol. Metab. 2002, 87, 1239–1246. [Google Scholar] [CrossRef] [PubMed]
- Bulotta, A.; Hui, H.; Anastasi, E.; Bertolotto, C.; Boros, L.G.; Di Mario, U.; Perfetti, R. Cultured pancreatic ductal cells undergo cell cycle re-distribution and beta-cell-like differentiation in response to glucagon-like peptide-1. J. Mol. Endocrinol. 2002, 29, 347–360. [Google Scholar]
- Farilla, L.; Bulotta, A.; Hirshberg, B.; Li Calzi, S.; Khoury, N.; Noushmehr, H.; Bertolotto, C.; Di Mario, U.; Harlan, D.M.; Perfetti, R. Glucagon-like peptide 1 inhibits cell apoptosis and improves glucose responsiveness of freshly isolated human islets. Endocrinology 2003, 144, 5149–5158. [Google Scholar]
- Zander, M.; Madsbad, S.; Madsen, J.L.; Holst, J.J. Effect of 6-week course of glucagon-like peptide 1 on glycaemic control, insulin sensitivity, and beta-cell function in type 2 diabetes: A parallel-group study. Lancet 2002, 359, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.A.; Després, J.P.; Richard, A.J.; Brette, S.; Deanfield, J.E. the IDEA Steering Committee and National Co-ordinators. Does abdominal obesity have a similar impact on cardiovascular disease and diabetes? A study of 91,246 ambulant patients in 27 European countries. Eur. Heart J. 2009, 30, 3055–3063. [Google Scholar] [CrossRef] [PubMed]
- Nyström, T. The potential beneficial role of glucagon-like peptide-1 in endothelial dysfunction and heart failure associated with insulin resistance. Horm. Metab. Res. 2008, 40, 593–606. [Google Scholar]
- Bose, A.K.; Mocanu, M.M.; Carr, R.D.; Brand, C.L.; Yellon, D.M. Glucagon-like peptide 1 can directly protect the heart against ischemia/reperfusion injury. Diabetes 2005, 54, 146–151. [Google Scholar]
- Thrainsdottir, I.; Malmberg, K.; Olsson, A.; Gutniak, M.; Rydén, L. Initial experience with GLP-1 treatment on metabolic control and myocardial function in patients with type 2 diabetes mellitus and heart failure. Diab. Vasc. Dis. Res. 2004, 1, 40–43. [Google Scholar]
- Nikolaidis, L.A.; Mankad, S.; Sokos, G.G.; Miske, G.; Shah, A.; Elahi, D.; Shannon, R.P. Effects of glucagon-like peptide-1 in patients with acute myocardial infarction and left ventricular dysfunction after successful reperfusion. Circulation 2004, 109, 962–965. [Google Scholar]
- Nyström, T.; Gutniak, M.K.; Zhang, Q.; Zhang, F.; Holst, J.J.; Ahrén, B.; Sjöholm, A. Effects of glucagon-like peptide-1 on endothelial function in type 2 diabetes patients with stable coronary artery disease. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E1209–E1215. [Google Scholar]
- Patel, A. ADVANCE Collaborative Group. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): A randomised controlled trial. Lancet 2007, 370, 829–840. [Google Scholar]
- Knudsen, L.B.; Nielsen, P.F.; Huusfeldt, P.O.; Johansen, N.L.; Madsen, K.; Pedersen, F.Z.; Thøgersen, H.; Wilken, M.; Agersø, H. Potent derivatives of glucagon-like peptide-1 with pharmacokinetic properties suitable for once daily administration. J Med Chem 2000, 43, 1664–1669. [Google Scholar]
- Elbrønd, B.; Jakobsen, G.; Larsen, S.; Agersø, H.; Jensen, L.B.; Rolan, P.; Sturis, J.; Hatorp, V.; Zdravkovic, M. Pharmacokinetics, pharmacodynamics, safety, and tolerability of a single-dose of NN2211, a long-acting glucagon-like peptide 1 derivative, in healthy male subjects. Diab. Care 2002, 25, 1398–1404. [Google Scholar]
- Steensgaard, D.B.; Thomsen, J.K.; Olsen, H.B.; Knudsen, L.B. The molecular basis for the delayed absorption of the once-daily human GLP-1 analogue, liraglutide. Diabetes 2008, 57 Suppl. 1, A164 (552-P). [Google Scholar]
- Agersø, H.; Jensen, L.B.; Elbrønd, B,; Rolan, P.; Zdravkovic, M. The pharmacokinetics, pharmacodynamics, safety and tolerability of NN2211, a new long-acting GLP-1 derivative, in healthy men. Diabetologia 2002, 45, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Marre, M.; Shaw, J.; Brändle, M.; Bebakar, W.M.; Kamaruddin, N.A.; Strand, J.; Zdravkovic, M.; Le Thi, T.D.; Colagiuri, S. LEAD-1 SU Study Group. Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU). Diabetic Med. 2009, 26, 268–278. [Google Scholar] [CrossRef]
- Nauck, M.; Frid, A.; Hermansen, K.; Shah, N.S.; Tankova, T.; Mitha, I.H.; Zdravkovic, M.; Düring, M.; Matthews, D.R. for the LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin in type 2 diabetes. Diab. Care 2009, 32, 84–90. [Google Scholar]
- Garber, A.; Henry, R.; Ratner, R.; Garcia-Hernadez, P.A.; Rodriguez-Pattzi, H.; Olvera-Alvarez, I.; Hale, P.M.; Zdravkovic, M.; Bode, B. LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): Randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009, 373, 473–481. [Google Scholar] [PubMed]
- Zinman, B.; Gerich, J.; Buse, J.B.; Lewin, A.; Schwartz, S.; Raskin, P.; Hale, P.M.; Zdravkovic, M.; Blonde, L. Efficacy and safety of the human GLP-1 analog liraglutide in combination with metformin and TZD in patients with type 2 diabetes mellitus (LEAD-4 Met+TZD). Diab. Care 2009, 32, 1224–1230. [Google Scholar]
- Russell-Jones, D.; Vaag, A.; Schmitz, O.; Sethi, B.K.; Lalic, N.; Antic, S.; Zdravkovic, M.; Ravn, G.M.; Simó, R. Liraglutide vs insulin glargine and placebo in combination with metformin and sulfonylurea therapy in type 2 diabetes mellitus (LEAD-5 met+SU): A randomised controlled trial. Diabetologia 2009, 52, 2046–2055. [Google Scholar] [CrossRef] [PubMed]
- Buse, J.; Rosenstock, J.; Sesti, G.; Schmidt, W.E.; Montanya, E.; Brett, J.; Zychma, M.; Blonde, L. for the LEAD-6 Study Group. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: A 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet 2009, 374, 39–47. [Google Scholar] [PubMed]
- Buteau, J.; Foisy, S.; Joly, E.; Prentki, M. Glucagon-like peptide 1 induces pancreatic beta-cell proliferation via transactivation of the epidermal growth factor receptor. Diabetes 2003, 52, 124–132. [Google Scholar]
- Jendle, J.; Nauck, M.A.; Matthews, D.R.; Frid, A.; Hermansen, K.; Düring, M.; Zdravkovic, M.; Strauss, B.J.; Garber, A.J. Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes. Metab. 2009, 11, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Gallwitz, B. Preclinical and Clinical Data on Extraglycemic Effects of GLP-1 Receptor Agonists. Rev. Diabet. Stud. 2009, 6, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Liraglutide (Victoza) prescribing information. Novo Nordisk. 2009. Available online: http://www.glucagon.com/pdfs/VictozaPIEU.pdf accessed on 23 December 2009.
- Noel, R.A.; Braun, D.K.; Patterson, R.E.; Bloomgren, G.L. Increased risk of acute pancreatitis and biliary disease observed in patients with type 2 diabetes: A retrospective cohort study. Diabetes Care 2009, 32, 834–838. [Google Scholar]
- Koehler, J.A.; Baggio, L.L.; Lamont, B.J.; Ali, S.; Drucker, D.J. Glucagon-like peptide-1 receptor activation modulates pancreatitis-associated gene expression but does not modify the susceptibility to experimental pancreatitis in mice. Diabetes 2009, 58, 2148–2161. [Google Scholar]
- Gallwitz, B.; Vaag, A.; Falahati, A.; Madsbad, S. Adding liraglutide to oral antidiabetic drug therapy: Onset of treatment effects over time. Int. J. Clin. Pract. 2010, 64, 267–276. [Google Scholar]
- Korytkowski, M. When oral agents fail: Practical barriers to starting insulin. Int. J. Obes. Relat. Metab. Disord. 2002, 26, S18–S24. [Google Scholar] [CrossRef]
- Nathan, D.M.; Buse, J.B.; Davidson, M.B.; Ferrannini, E.; Holman, R.R.; Sherwin, R.; Zinman, B. Medical management of hyperglycemia in type 2 diabetes: A consensus algorithm for the initiation and adjustment of therapy: A consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2009, 32, 193–203. [Google Scholar] [PubMed]
- Rodbard, H.W.; Jellinger, P.S.; Davidson, J.A.; Einhorn, D.; Garber, A.J.; Grunberger, G.; Handelsman, Y.; Horton, E.S.; Lebovitz, H.; Levy, P.; Moghissi, E.S.; Schwartz, S.S. Statement by an American Association of Clinical Endocrinologists/American College of Endocrinology consensus panel on type 2 diabetes mellitus: An algorithm for glycemic control. Endocr. Pract. 2009, 15, 540–559. [Google Scholar]
- National Institute for Health and Clinical Excellence (NICE). Type 2 Diabetes: The Management of Type 2 Diabetes (update). Clinical Guidelines CG66. 2008. Available online: http://guidance.nice.org.uk/CG66 accessed on 24 December 2009.
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sjöholm, Å. Liraglutide Therapy for Type 2 Diabetes: Overcoming Unmet Needs. Pharmaceuticals 2010, 3, 764-781. https://doi.org/10.3390/ph3030764
Sjöholm Å. Liraglutide Therapy for Type 2 Diabetes: Overcoming Unmet Needs. Pharmaceuticals. 2010; 3(3):764-781. https://doi.org/10.3390/ph3030764
Chicago/Turabian StyleSjöholm, Åke. 2010. "Liraglutide Therapy for Type 2 Diabetes: Overcoming Unmet Needs" Pharmaceuticals 3, no. 3: 764-781. https://doi.org/10.3390/ph3030764