Apparent Thixotropic Properties of Saline/Glycerol Drops with Biotinylated Antibodies on Streptavidin-Coated Glass Slides: Implications for Bacterial Capture on Antibody Microarrays
Abstract
:1. Introduction
2. Results and Discussion
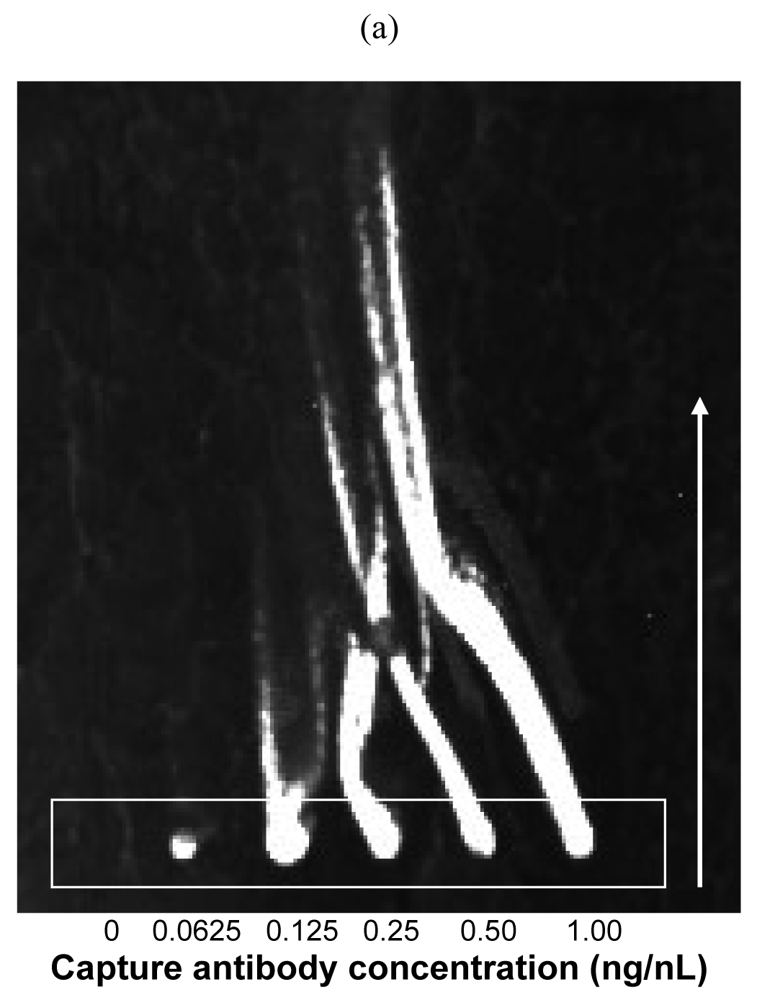
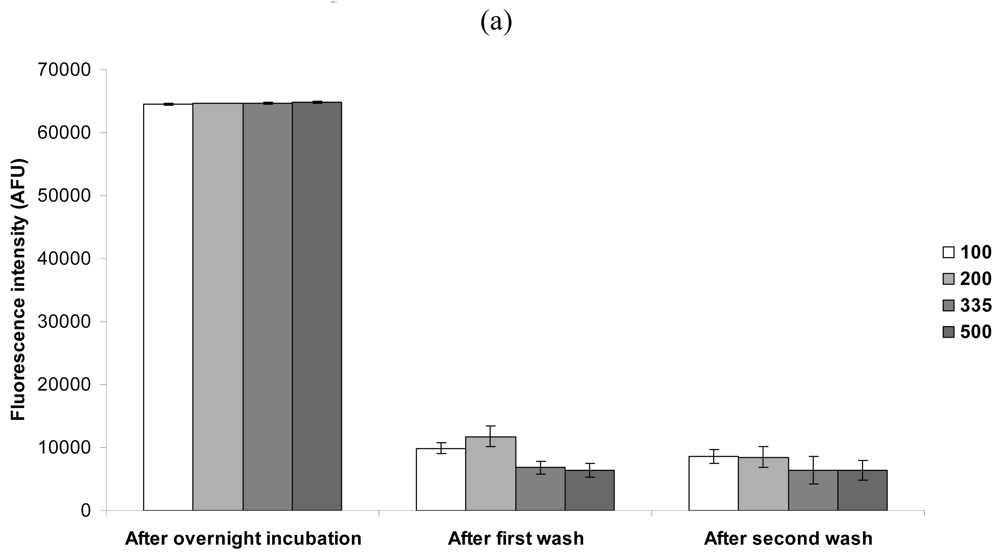
2.1. Influence of Lateral Shearing on Biotinylated Antibodies in PBS/Glycerol Spots
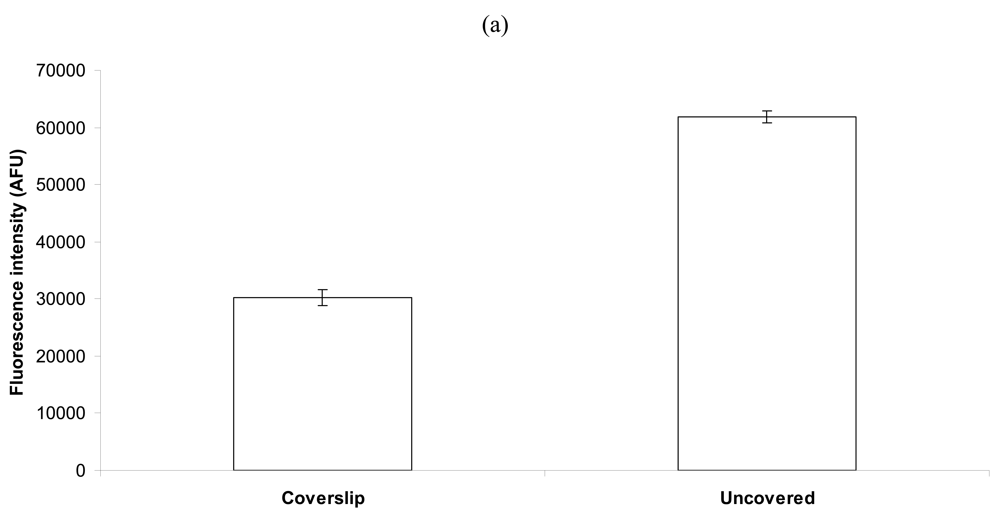
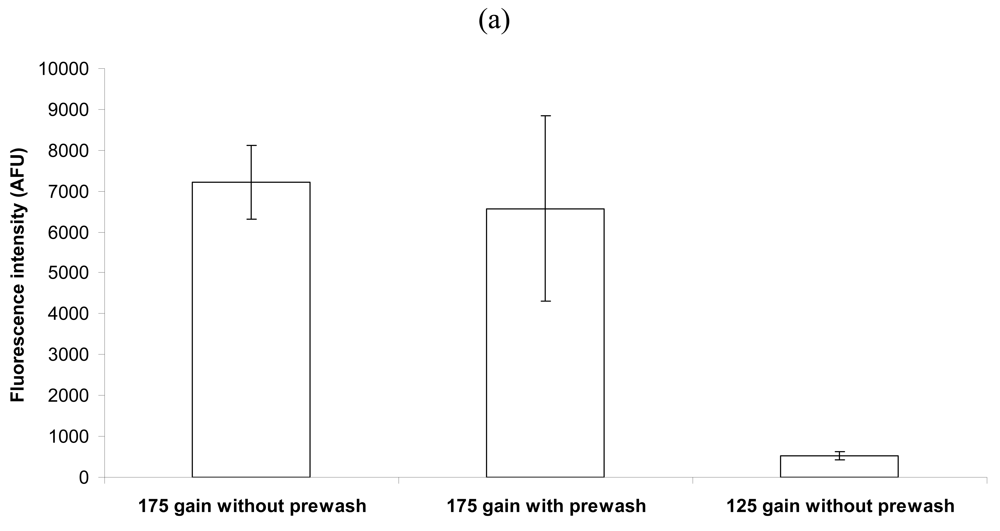
2.2. Effect of a Coverslip on Bacterial Capture and Detection
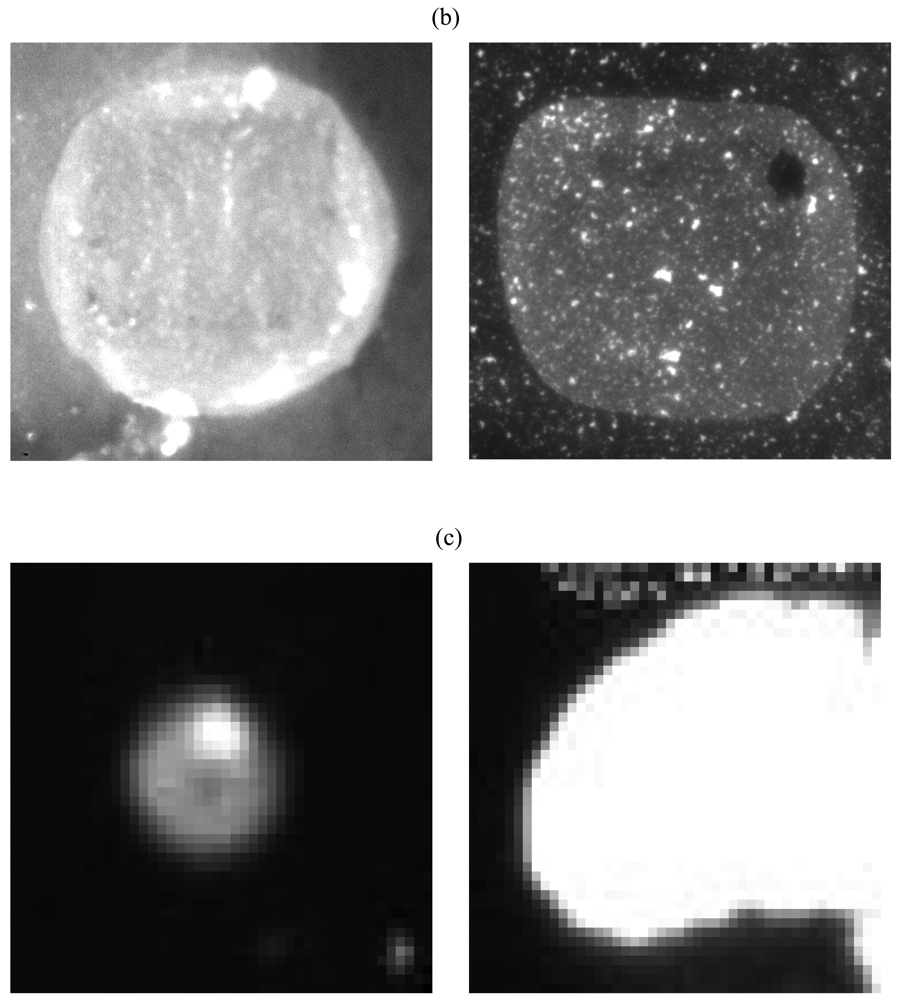
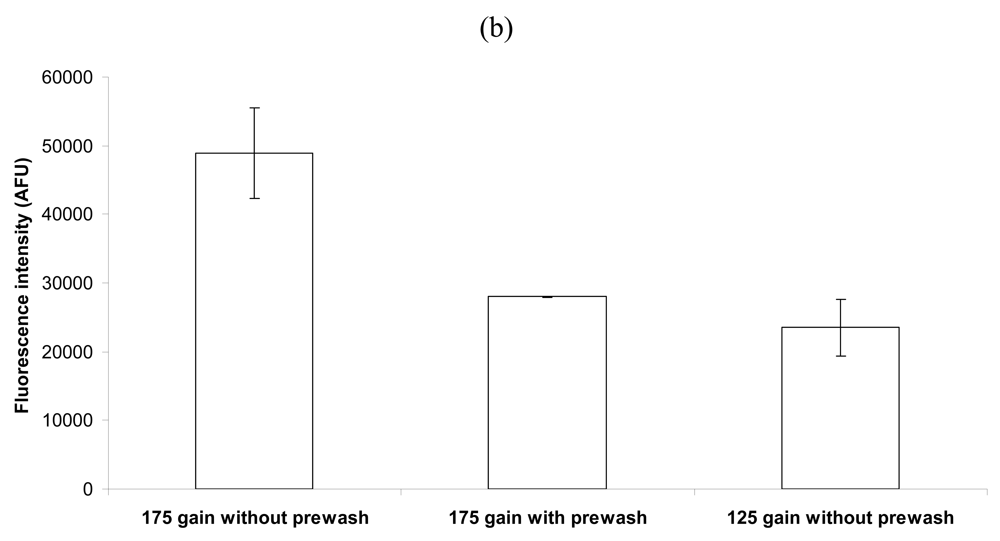
2.3. Effect of Removal of Unbound Capture Antibody Prior to Bacterial Capture
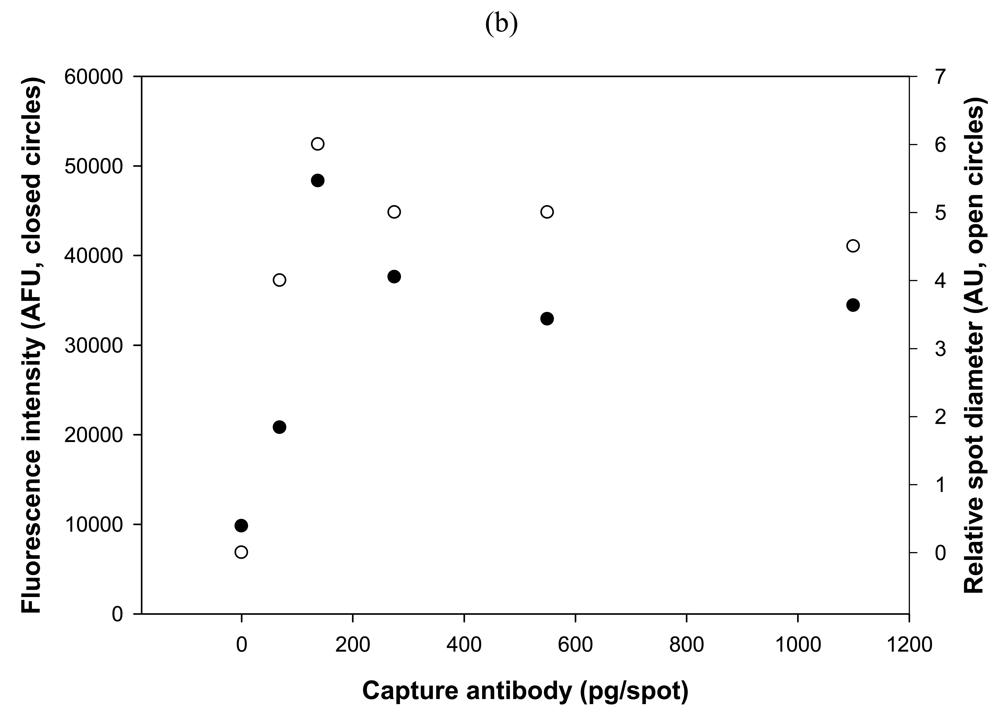
2.4. Effects Due to Spot Size
3. Experimental Section
3.1. Materials
3.2. Apparatus
3.3. Streptavidin-Coating of Microarray Slides
3.4. Antibody and Microarray Slide Preparation for Bacterial Capture
3.5. Growth and Enumeration of E. coli O157:H7
3.6. Antibody Microarray Detection of Bacteria
3.7. Biotinylated Fluorescein Experiments
3.8. Fluorescence Microscopy of Microarray Slides
3.9. Data Analysis
4. Conclusions
Acknowledgments
References
- Bartell, F.E.; Zuidema, H.H. Wetting characteristics of solids of low surface tension such as talc, waxes and resins. J. Am. Chem. Soc. 1936, 58, 1449–1454. [Google Scholar]
- Mack, G.L. The determination of contact angles from measurements of the dimensions of small bubbles and drops. 1. The spheroidal segment method for acute angles. J. Phys. Chem. 1936, 40, 159–167. [Google Scholar]
- Alteraifi, A.M.; Sherif, D.; Moet, A. Interfacial effects in the spreading kinetics of liquid droplets on solid substrates. J. Colloid Interface Sci. 2003, 264, 221–227. [Google Scholar]
- Dimov, N.K.; Ahmed, E.H.; Alargova, R.G.; Kralchevsky, P.A.; Durbut, P.; Broze, G.; Mehreteab, A. Deposition of oil drops on a glass substrate in relation to the process of washing. J. Colloid Interface Sci. 2000, 224, 116–125. [Google Scholar]
- Freundlich, H.; Bircumshaw, L.L. Ueber das thixotrope verhalten von aluminiumhydroxydgelen. Colloid Polym. Sci. 1926, 40, 19–22. [Google Scholar]
- Kerst, A.; Chmielewski, C.; Livesay, C.; Buxbaum, R.E.; Heidemann, S.R. Liquid crystal domains and thixotropy of filamentous actin suspensions. Proc. Natl. Acad. Sci. U. S. A. 1990, 87, 4241–4245. [Google Scholar]
- Akkoyun, A.; Bilitewski, U. Optimisation of glass surfaces for optical immunosensors. Biosens. Bioelectron. 2002, 17, 655–664. [Google Scholar]
- Olle, E.W.; Messamore, J.; Deogracias, M.P.; McClintock, S.D.; Anderson, T.D.; Johnson, K.J. Comparison of antibody array substrates and the use of glycerol to normalize spot morphology. Exp. Mol. Pathol. 2005, 79, 206–209. [Google Scholar]
- Wu, P.; Grainger, D.W. Comparison of hydroxylated print additives on antibody microarray performance. J. Proteome Res. 2006, 5, 2956–2965. [Google Scholar]
- Schena, M.; Shalon, D.; Davis, R.W.; Brown, P.O. Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science (Washington, DC, U. S.) 1995, 270, 467–470. [Google Scholar]
- Macbeath, G.; Schreiber, S.L. Printing proteins as microarrays for high-throughput function determination. Science (Washington, DC, U. S.) 2000, 289, 1760–1763. [Google Scholar]
- Rao, R.S.; Visuri, S.R.; McBride, M.T.; Albala, J.S.; Matthews, D.L.; Coleman, M.A. Comparison of multiplexed techniques for detection of bacterial and viral proteins. J. Proteome Res. 2004, 3, 736–742. [Google Scholar]
- Sapsford, K.E.; Shubin, Y.S.; Delehanty, J.B.; Golden, J.P.; Taitt, C.R.; Shriver-Lake, L.C.; Ligler, F.S. Fluorescence-based array biosensors for detection of biohazards. J. Appl. Microbiol. 2004, 96, 47–58. [Google Scholar]
- Vora, G.J.; Meador, C.E.; Stenger, D.A.; Andreadis, J.D. Nucleic acid amplification strategies for DNA microarray-based pathogen detection. Appl. Environ. Microbiol. 2004, 70, 3047–3054. [Google Scholar]
- Nielsen, U.B.; Geierstanger, B.H. Multiplexed sandwich assays in microarray format. J. Immunol. Methods. 2004, 290, 107–120. [Google Scholar]
- Peng, Z.; Bang-Ce, Y. Small molecule microarrays for drug residue detection in foodstuffs. J. Agric. Food Chem. 2006, 54, 6978–6983. [Google Scholar]
- Gehring, A.G.; Albin, D.M.; Bhunia, A.K.; Reed, S.A.; Tu, S.-I.; Uknalis, J. Antibody microarray detection of Escherichia coli O157:H7: quantification, assay limitations, and capture efficiency. Anal. Chem. (Washington, DC, U. S.) 2006, 78, 6601–6607. [Google Scholar]
- Yang, Y.; Zhang, C.; Tang, Z.; Zhang, X.; Lu, Z. Experimental studies on capture human leukocytes with cell immuno-chip. Clin. Chem. (Washington, DC, U. S.) 2005, 51, 910–911. [Google Scholar]
- Sapsford, K.E.; Liron, Z.; Shubin, Y.S.; Ligler, F.S. Kinetics of antigen binding to arrays of antibodies in different sized spots. Anal. Chem. (Washington, DC, U. S.) 2001, 73, 5518–5524. [Google Scholar]
- Gehring, A.G.; Patterson, D.L.; Tu, S.-I. Use of a light-addressable potentiometric sensor for the detection of Escherichia coli O157:H7. Anal. Biochem. 1998, 258, 293–298. [Google Scholar]
- Tu, S.-I.; Uknalis, J.; Patterson, D.L.; Gehring, A.G. Detection of immunomagnetically captured, 4′,6-diamidino-2-phenylindole (DAPI)-labeled E. coli O157:H7 by fluorescent microscope imaging. J.Rapid Methods Autom. Microbiol. 1998, 6, 259–276. [Google Scholar]

© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Albin, D.M.; Gehring, A.G.; Reed, S.A.; Tu, S.-I. Apparent Thixotropic Properties of Saline/Glycerol Drops with Biotinylated Antibodies on Streptavidin-Coated Glass Slides: Implications for Bacterial Capture on Antibody Microarrays. Sensors 2009, 9, 995-1011. https://doi.org/10.3390/s90200995
Albin DM, Gehring AG, Reed SA, Tu S-I. Apparent Thixotropic Properties of Saline/Glycerol Drops with Biotinylated Antibodies on Streptavidin-Coated Glass Slides: Implications for Bacterial Capture on Antibody Microarrays. Sensors. 2009; 9(2):995-1011. https://doi.org/10.3390/s90200995
Chicago/Turabian StyleAlbin, David M., Andrew G. Gehring, Sue A. Reed, and Shu-I Tu. 2009. "Apparent Thixotropic Properties of Saline/Glycerol Drops with Biotinylated Antibodies on Streptavidin-Coated Glass Slides: Implications for Bacterial Capture on Antibody Microarrays" Sensors 9, no. 2: 995-1011. https://doi.org/10.3390/s90200995



