Flow Injection Chemiluminescent Immunoassay for Carcinoembryonic Antigen Using Boronic Immunoaffinity Column
Abstract
:1. Introduction
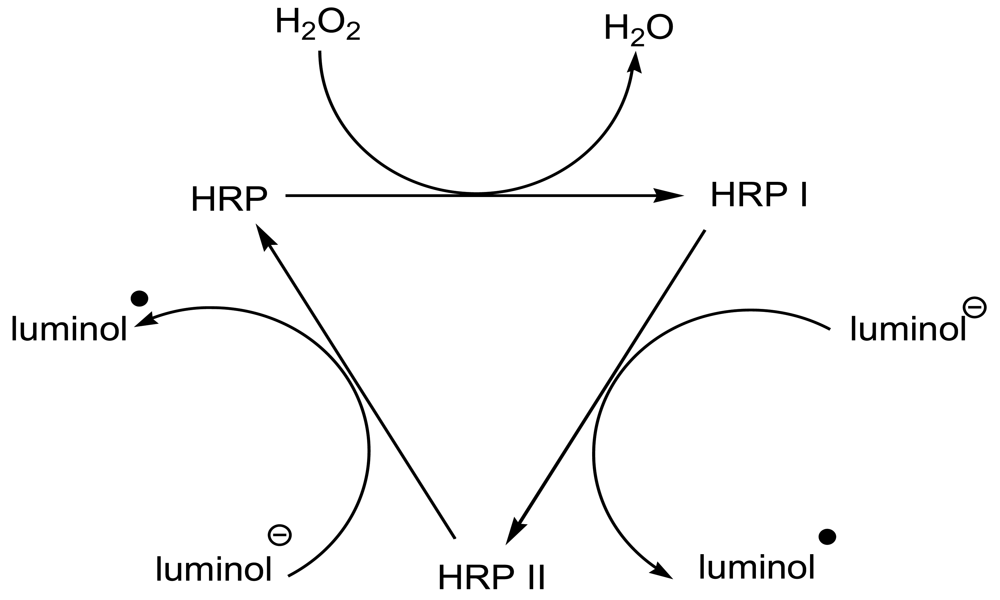
2. Results and Discussion
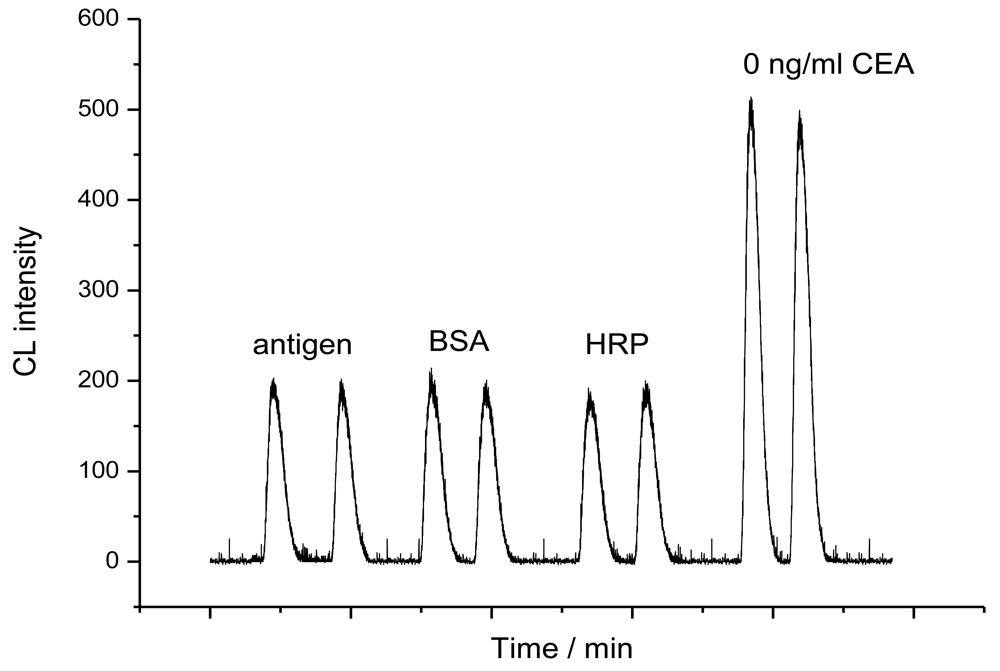
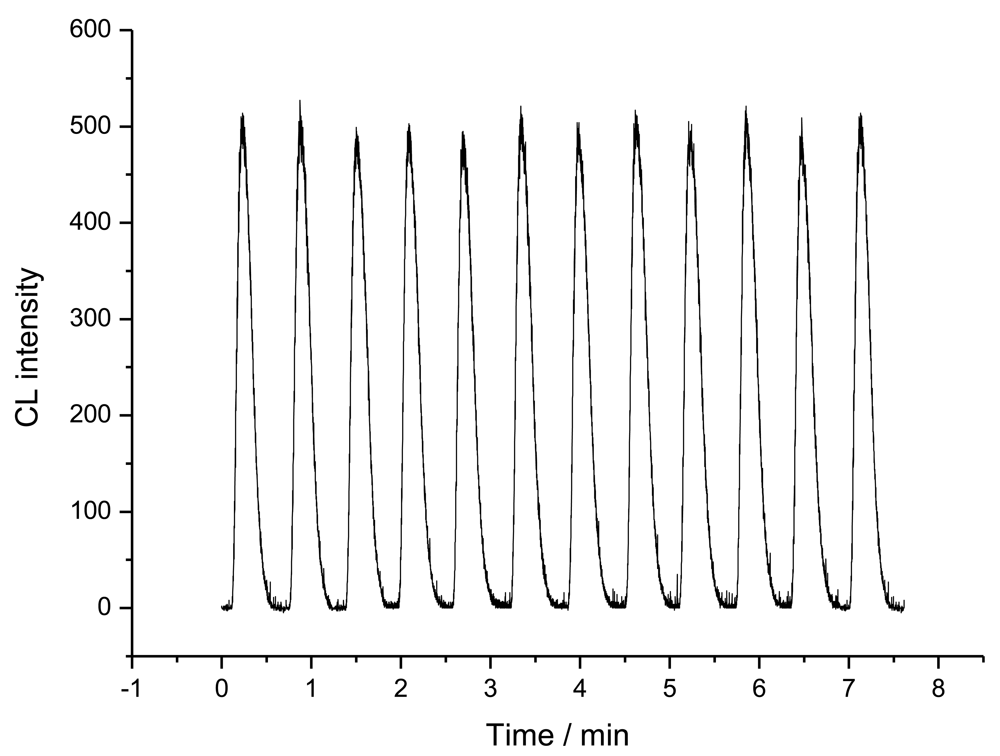
2.1. Binding performances of the immunoaffinity column
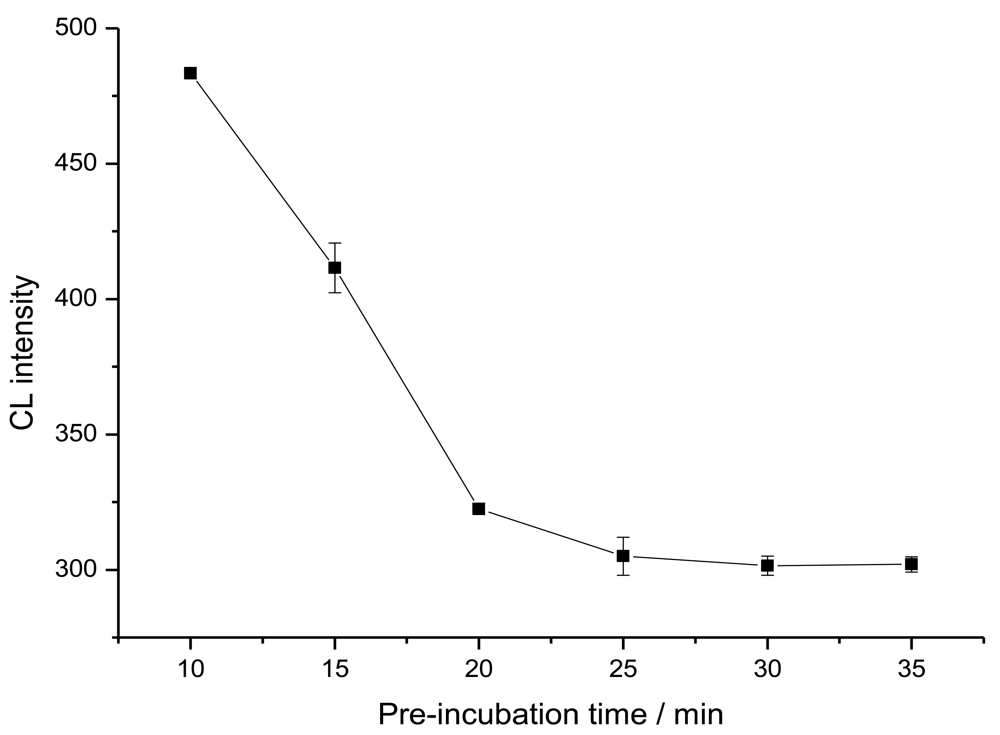
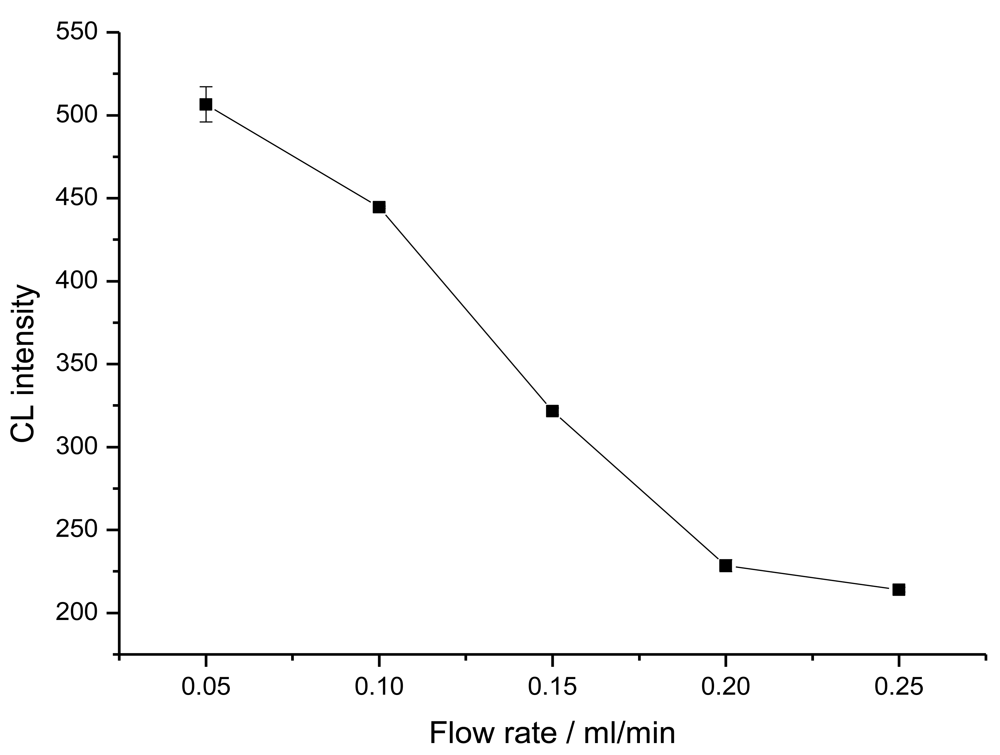
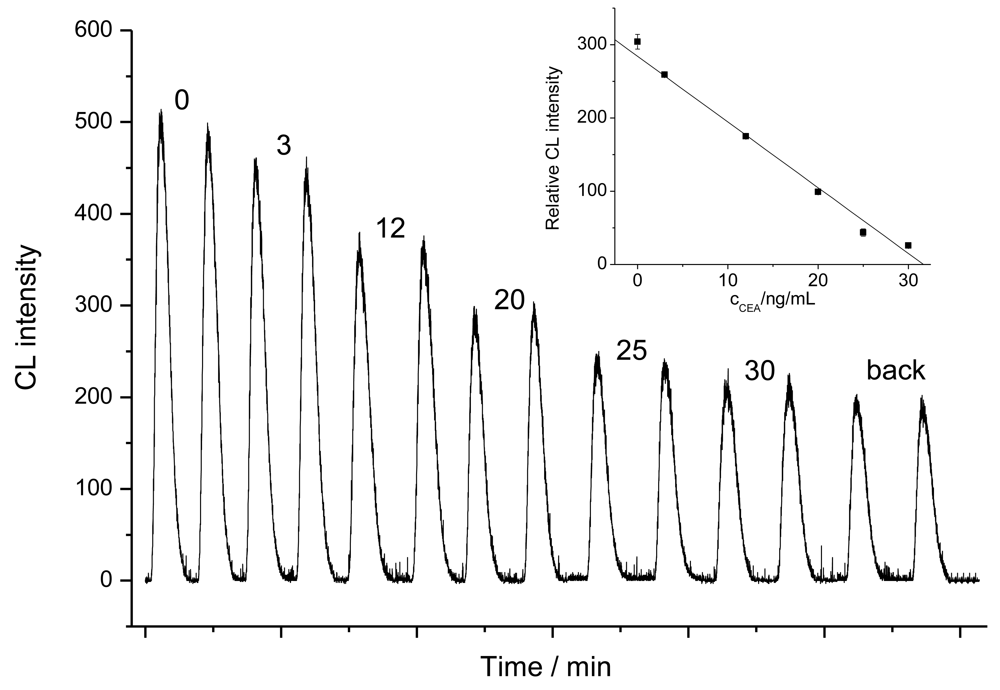
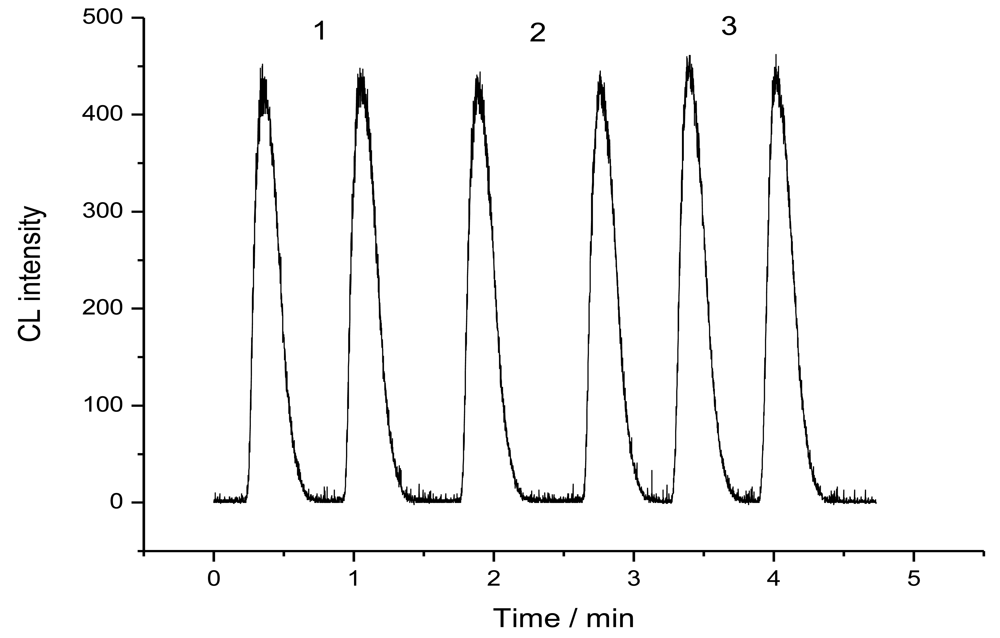
2.2. Non-competitive immunoassays for CEA detection
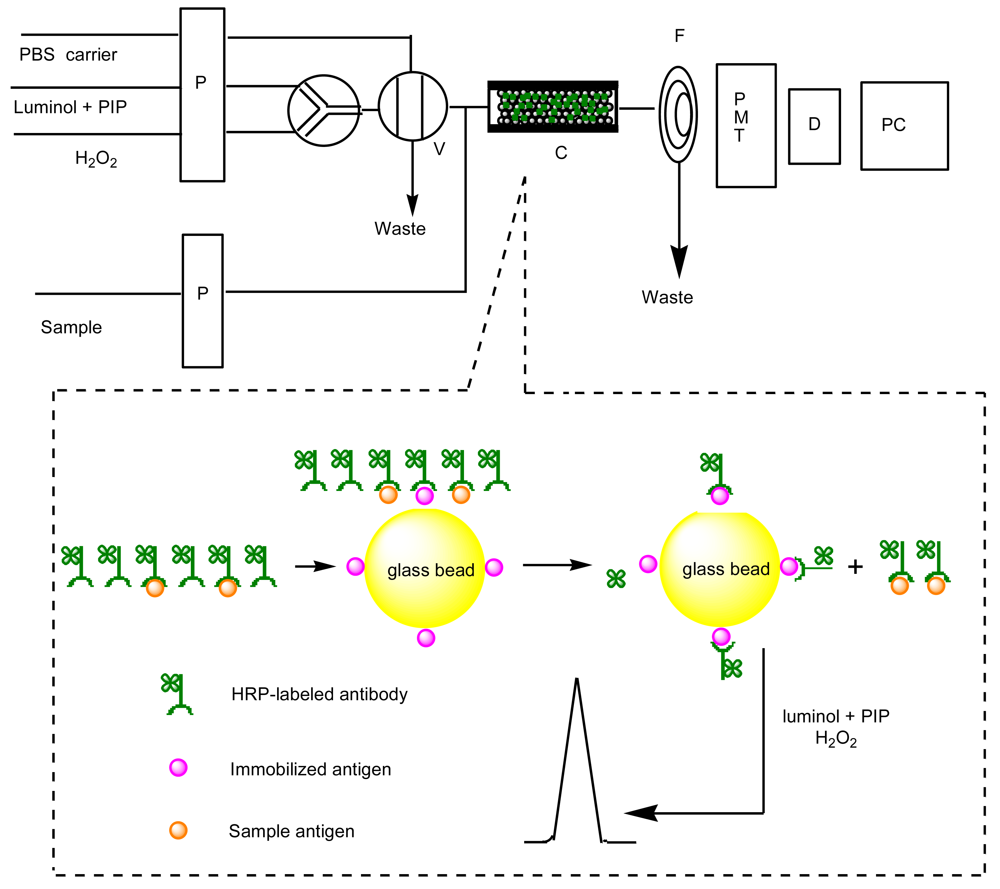
3. Materials and Methods
4. Conclusions
Acknowledgments
References and Notes
- Shively, J.E.; Paxton, R.; Nenmaier, M.; Hefta, S.; Hefta, L.; Hinoda, Y.; Beatty, J.D.; Beatty, B.; Williams, L.; Wagener, C. Carcinoembryonic antigen: molecular cloning and expression of CEA-related antigens, and the use of monoclonal antibodies to CEA in tumor imaging and therapy. Fresenius' J. Anal. Chem. 1988, 330, 310. [Google Scholar]
- Shively, J.E.; Beatty, J.D. CEA-mRNA expression in peripheral blood of patients with gastrointestinal carcinoma and its clinical significance. Crit. Rev. Oncol. Hematol. 1985, 2, 100–110. [Google Scholar]
- Christiansen, M.; Hogdall, C.K.; Andersen, J.R.; Norgaard-Pedersen, B. α-Fetoprotein in plasma and serum of healthy adults: preanalytical, analytical and biological sources of variation and construction of age-dependent reference intervals. Scand. J. Clin. Lab. Invest. 2001, 61, 205–215. [Google Scholar]
- Sun, W.; Jiao, K.; Zhang, S.; Zhang, C.; Zhang, Z. Electrochemical detection for horseradish peroxidase-based enzyme immunoassay using p-aminophenol as substrate and its application in detection of plant virus. Anal. Chim. Acta 2001, 434, 43–50. [Google Scholar]
- Ci, Y.X.; Qin, Y.; Chang, W.B.; Li, Y.Z. Application of a mimetic enzyme for the enzyme immunoassay for α-1-fetoprotein. Anal. Chim. Acta 1995, 300, 273–276. [Google Scholar]
- Matsuya, T.; Tashiro, S.; Hoshino, N.; Shibata, N.; Nagasaki, Y.; Kataoka, K. Acore-shell-type fluorescent nanosphere possessing reactive poly (ethylene glycol) tethered chains on the surface for zeptomole detection of protein in time-resolved fluorometric immunoassay. Anal. Chem. 2003, 75, 6124–6132. [Google Scholar]
- Song, S.P.; Li, B.; Hu, J.; Li, M.Q. Simultaneous multianalysis for tumor markers by antibody fragments microarray system. Anal. Chim. Acta 2004, 510, 147–152. [Google Scholar]
- Xue, M.D.; Haruyama, T.; Kobatake, E.; Aizawa, M. Electrochemical luminescence immunosensor for alpha-fetoprotein. Sens. Actuat. B. 1996, 36, 458–462. [Google Scholar]
- Zhuo, Y.; Yuan, R.; Chai, Y.Q.; Tang, D.P; Zhang, Y.; Wang, N.; Li, X.L.; Zhu, Q. A reagentless amperometric immunosensor based on gold nanoparticles/thionine/Nafion-membrane-modified gold electrode for determination of α-1-fetoprotein. Electrochem. Commun. 2005, 7, 355–360. [Google Scholar]
- Palmer, D.A.; Miller, J.N. Thiophilic gels: applications in flow injection immunoassay for macromolecules and haptens. Anal. Chim. Acta 1995, 303, 223–230. [Google Scholar]
- Nilsson, M.; Hakason, H.; Mattiasson, B. Process monitoring by flow-injection immunoassay evaluation of a sequential competitive binding assay. J. Chromatogr. A 1992, 597, 383–389. [Google Scholar]
- Liu, S.Q.; Bakovic, L.; Chen, A.C. Specific binding of glycoproteins with poly (aniline boronic acid) thin film. J. Electroanal. Chem. 2006, 591, 210–216. [Google Scholar]
- Zhang, X.T.; Wu, Y.F.; Tu, Y.F.; Liu, S.Q. A reusable electrochemical immunosensor for carcinoembryonic antigen via molecular recognition of glycoprotein antibody by phenylboronic acid self-assembly layer on gold. Analyst 2008, 133, 485–492. [Google Scholar]
- Wang, S.H.; Du, L.Y.; Lin, S.L.; Zhuang, H.S. Flow Injection Chemiluminescence for the Determination of Estriol via a Noncompetitive Enzyme Immunoassay. Microchim. Acta 2006, 155, 421–426. [Google Scholar]
- Liu, S.Q.; Miller, B.; Chen, A.C. Phenylboronic acid self-assembled layer on glassy carbon electrode for recognition of glycoprotein peroxidase. Electrochem. Commun. 2005, 7, 1232–1236. [Google Scholar]
- Dai, Z.; Yan, F.; Yu, H.; Hu, X.; Ju, H.X. Novel amperometric immunosensor for rapid separation- free immunoassay of carcinoembryonic antigen. J. Immunol. Method. 2004, 287, 13–20. [Google Scholar]
- Nilsson, M.; Hakason, H.; Mattiasson, B. Process monitoring by flow-injection immunoassay: evaluation of a sequential competitive binding assay. J. Chromatogr. 1992, 597, 383. [Google Scholar]
- Palmer, D.A.; Miller, J.N. Thiophilic gels: applications in flow-injection immunoassay for macromolecules and haptens. Anal. Chim. Acta 1995, 303, 223. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, X.; Li, H.; Wu, M.; Sun, C.; Li, L.; Yang, X. Flow Injection Chemiluminescent Immunoassay for Carcinoembryonic Antigen Using Boronic Immunoaffinity Column. Sensors 2009, 9, 10389-10399. https://doi.org/10.3390/s91210389
Ma X, Li H, Wu M, Sun C, Li L, Yang X. Flow Injection Chemiluminescent Immunoassay for Carcinoembryonic Antigen Using Boronic Immunoaffinity Column. Sensors. 2009; 9(12):10389-10399. https://doi.org/10.3390/s91210389
Chicago/Turabian StyleMa, Xiaoling, Huihui Li, Min Wu, Chong Sun, Laifa Li, and Xiaodi Yang. 2009. "Flow Injection Chemiluminescent Immunoassay for Carcinoembryonic Antigen Using Boronic Immunoaffinity Column" Sensors 9, no. 12: 10389-10399. https://doi.org/10.3390/s91210389



