Magnetic Nanoparticle Sensors
Abstract
:1. Introduction
2. Magnetic Relaxation Switches (MRSws)
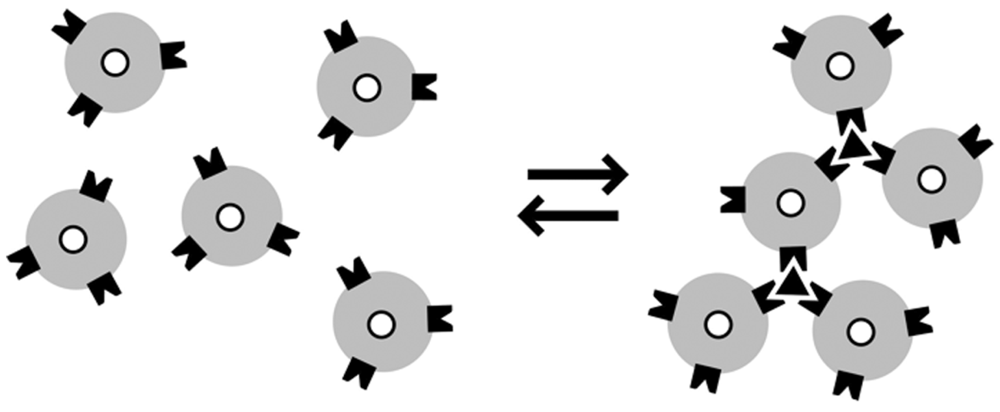
2.1. Mechanism of MRSws
2.2. Magnetic Particles
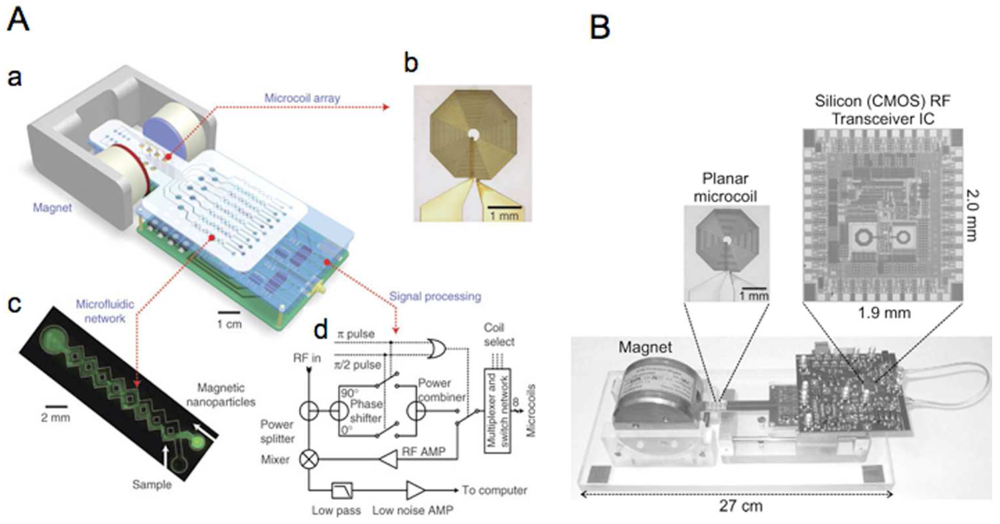
2.3. Instrumentation
2.4. Applications of Type I and Type II MRSw's
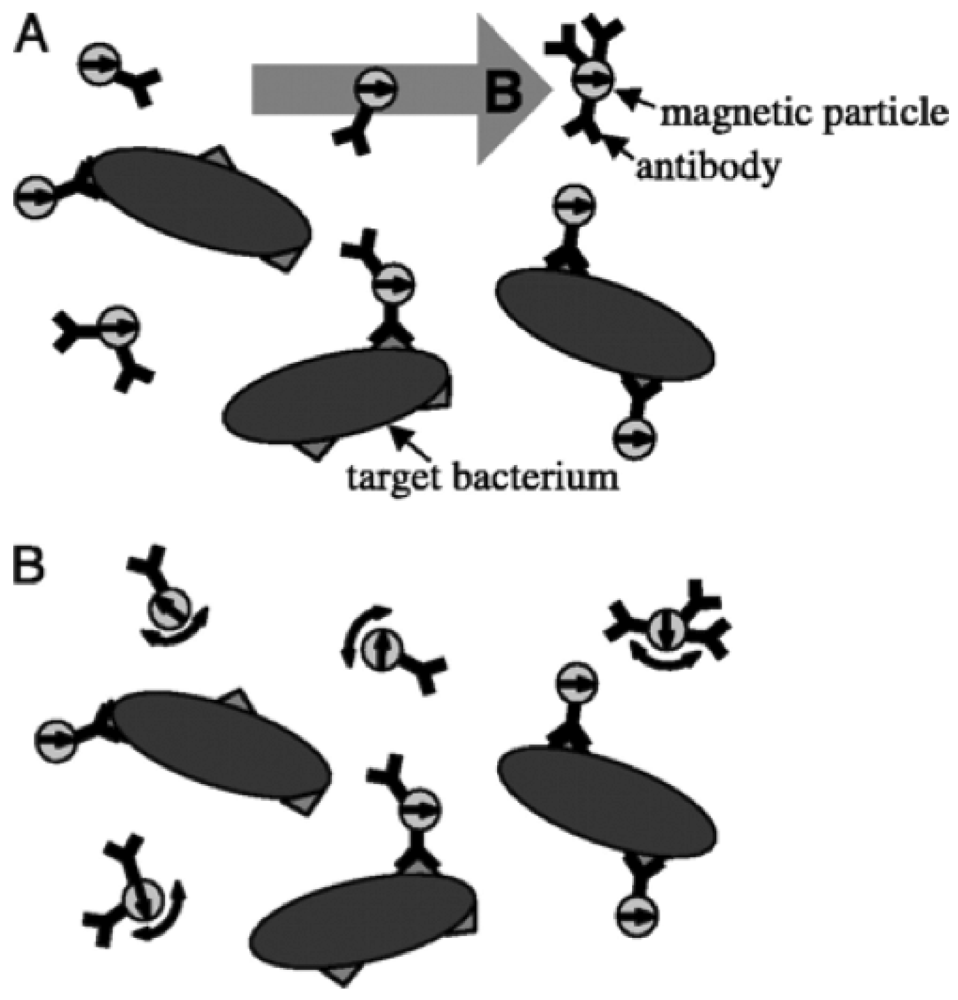
2.4.1. Type I MRSw
2.4.2. Type II MRSw
3. Magnetic Particle Relaxation-Based Sensors
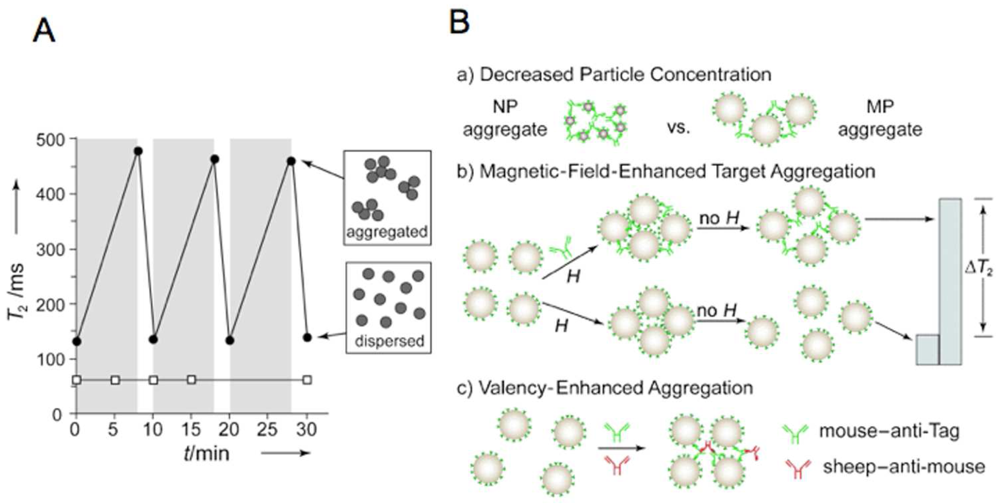
3.1. Theory
3.2. Assays
3.2.1. Néel Relaxation Sensors
3.2.2. Brownian Relaxation Sensors
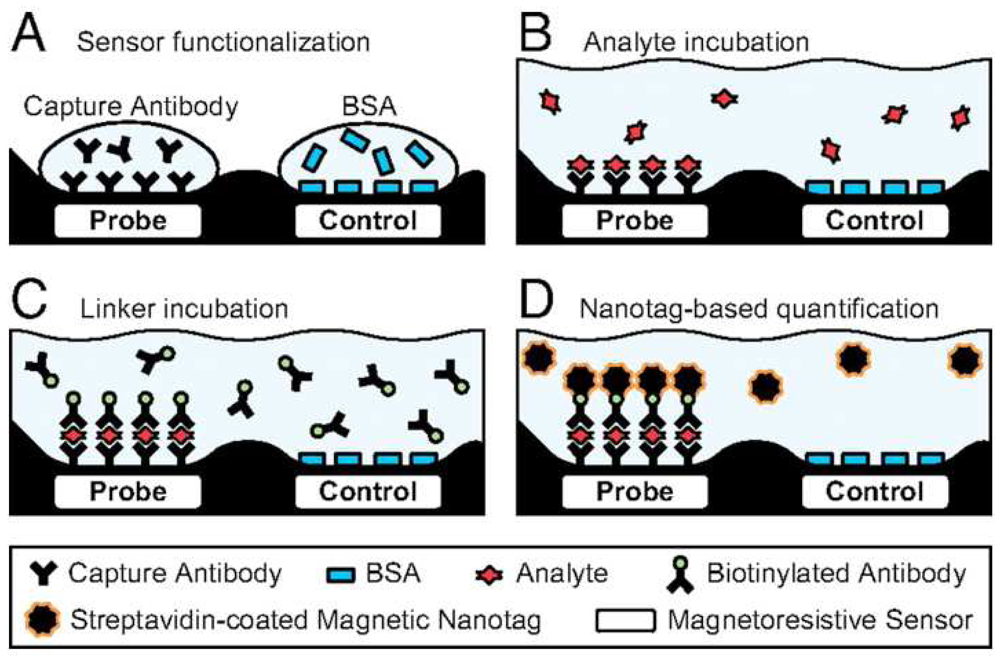
4. Magnetoresistive Sensors
5. Conclusions
Acknowledgments
References
- Lee, J.H.; Huh, Y.M.; Jun, Y.W.; Seo, J.W.; Jang, J.T.; Song, H.T.; Kim, S.; Cho, E.J.; Yoon, H.G.; Suh, J.S.; Cheon, J. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med. 2007, 13, 95–99. [Google Scholar]
- Weissleder, R.; Pittet, M.J. Imaging in the era of molecular oncology. Nature 2008, 452, 580–589. [Google Scholar]
- Weissleder, R. Molecular imaging in cancer. Science 2006, 312, 1168–1171. [Google Scholar]
- Josephson, L.; Perez, J.M.; Weissleder, R. Magnetic nanosensors for the detection of oligonucleotide sequences. Angew. Chem. Int. Ed. 2001, 40, 3204–3208. [Google Scholar]
- Perez, J.M.; Josephson, L.; O'Loughlin, T.; Hogemann, D.; Weissleder, R. Magnetic relaxation switches capable of sensing molecular interactions. Nat. Biotechnol. 2002, 20, 816–820. [Google Scholar]
- Koh, I.; Hong, R.; Weissleder, R.; Josephson, L. Nanoparticle-target interactions parallel antibody-protein interactions. Anal. Chem. 2009, 81, 3618–3622. [Google Scholar]
- Hong, R.; Cima, M.J.; Weissleder, R.; Josephson, L. Magnetic microparticle aggregation for viscosity determination by MR. Magn. Reson. Med. 2008, 59, 515–520. [Google Scholar]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar]
- Kim, G.Y.; Josephson, L.; Langer, R.; Cima, M.J. Magnetic relaxation switch detection of human chorionic gonadotrophin. Bioconjug. Chem. 2007, 18, 2024–2028. [Google Scholar]
- Lee, H.; Sun, E.; Ham, D.; Weissleder, R. Chip-NMR biosensor for detection and molecular analysis of cells. Nat. Med. 2008, 14, 869–874. [Google Scholar]
- Perez, J.M.; Simeone, F.J.; Saeki, Y.; Josephson, L.; Weissleder, R. Viral-induced self-assembly of magnetic nanoparticles allows the detection of viral particles in biological media. J. Am. Chem. Soc. 2003, 125, 10192–10193. [Google Scholar]
- Taktak, S.; Weissleder, R.; Josephson, L. Electrode chemistry yields a nanoparticle-based nmr sensor for Calcium. Langmuir 2008, 24, 7596–7598. [Google Scholar]
- Harisinghani, M.G.; Barentsz, J.; Hahn, P.F.; Deserno, W.M.; Tabatabaei, S.; van de Kaa, C.H.; de la Rosette, J.; Weissleder, R. Noninvasive detection of clinically occult lymph-node metastases in prostate cancer. N. Engl. J. Med. 2003, 348, 2491–2499. [Google Scholar]
- Jun, Y.W.; Huh, Y.M.; Choi, J.S.; Lee, J.H.; Song, H.T.; Kim, S.; Yoon, S.; Kim, K.S.; Shin, J.S.; Suh, J.S.; Cheon, J. Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging. J. Am. Chem. Soc. 2005, 127, 5732–5733. [Google Scholar]
- Park, J.; An, K.J.; Hwang, Y.S.; Park, J.G.; Noh, H.J.; Kim, J.Y.; Park, J.H.; Hwang, N.M.; Hyeon, T. Ultra-large-scale syntheses of monodisperse nanocrystals. Nat. Mater. 2004, 3, 891–895. [Google Scholar]
- Lee, H.; Yoon, T.J.; Figueiredo, J.L.; Swirski, F.K.; Weissleder, R. Rapid detection and profiling of cancer cells in fine-needle aspirates. Proc. Natl. Acad. Sci. USA 2009. [Google Scholar] [CrossRef]
- Lee, H.; Yoon, T.J.; Weissleder, R. Ultrasensitive detection of bacteria using core-shell nanoparticles and an NMR-filter system. Angew.Chem. Int. Ed. 2009, 48, 5657–5660. [Google Scholar]
- Park, J.H.; von Maltzahn, G.; Zhang, L.L.; Schwartz, M.P.; Ruoslahti, E.; Bhatia, S.N.; Sailor, M.J. Magnetic iron oxide nanoworms for tumor targeting and imaging. Adv. Mater. 2008, 20, 1630–1635. [Google Scholar]
- Koh, I.; Hong, R.; Weissleder, R.; Josephson, L. Sensitive NMR sensors detect antibodies to influenza. Angew. Chem. Int. Ed. 2008, 47, 4119–4121. [Google Scholar]
- Perez, J.M.; Josephson, L.; Weissleder, R. Use of magnetic nanoparticles as nanosensors to probe for molecular interactions. Chembiochem 2004, 5, 261–264. [Google Scholar]
- Sun, N.; Liu, Y.; Lee, H.; Weissleder, R.; Ham, D. CMOS RF biosensor utilizing nuclear magnetic resonance. IEEE J. Solid-State Circuits 2009, 44, 1629–1643. [Google Scholar]
- Grimm, J.; Perez, J.M.; Josephson, L.; Weissleder, R. Novel nanosensors for rapid analysis of telomerase activity. Cancer Res. 2004, 64, 639–643. [Google Scholar]
- Tsourkas, A.; Hofstetter, O.; Hofstetter, H.; Weissleder, R.; Josephson, L. Magnetic relaxation switch immunosensors detect enantiomeric impurities. Angew. Chem. Int. Ed. 2004, 43, 2395–2399. [Google Scholar]
- Sun, E.Y.; Josephson, L.; Weissleder, R. “Clickable” nanoparticles for targeted imaging. Mol. Imaging 2006, 5, 122–128. [Google Scholar]
- Sun, E.Y.; Weissleder, R.; Josephson, L. Continuous analyte sensing with magnetic nanoswitches. Small 2006, 2, 1144–1147. [Google Scholar]
- Weissleder, R.; Kelly, K.; Sun, E.Y.; Shtatland, T.; Josephson, L. Cell-specific targeting of nanoparticles by multivalent attachment of small molecules. Nat. Biotechnol. 2005, 23, 1418–1423. [Google Scholar]
- Perez, J.M.; O'Loughin, T.; Simeone, F.J.; Weissleder, R.; Josephson, L. DNA-based magnetic nanoparticle assembly acts as a magnetic relaxation nanoswitch allowing screening of DNA-cleaving agents. J. Am. Chem. Soc. 2002, 124, 2856–2857. [Google Scholar]
- Daniel, K.D.; Kim, G.Y.; Vassiliou, C.C.; Galindo, M.; Guimaraes, A.R.; Weissleder, R.; Charest, A.; Langer, R.; Cima, M.J. Implantable diagnostic device for cancer monitoring. Biosens. Bioelectron. 2009, 24, 3252–3257. [Google Scholar]
- Daniel, K.D.; Kim, G.Y.; Vassiliou, C.C.; Jalali-Yazdi, F.; Langer, R.; Cima, M.J. Multi-reservoir device for detecting a soluble cancer biomarker. Lab. Chip. 2007, 7, 1288–1293. [Google Scholar]
- Doyle, P.S.; Bibette, J.; Bancaud, A.; Viovy, J.L. Self-assembled magnetic matrices for DNA separation chips. Science 2002, 295, 2237. [Google Scholar]
- Baudry, J.; Rouzeau, C.; Goubault, C.; Robic, C.; Cohen-Tannoudji, L.; Koenig, A.; Bertrand, E.; Bibette, J. Acceleration of the recognition rate between grafted ligands and receptors with magnetic forces. Proc. Natl. Acad. Sci.USA 2006, 103, 16076–16078. [Google Scholar]
- Singh, H.; Laibinis, P.E.; Hatton, T.A. Rigid, superparamagnetic chains of permanently linked beads coated with magnetic nanoparticles. Synthesis and rotational dynamics under applied magnetic fields. Langmuir 2005, 21, 11500–11509. [Google Scholar]
- Zerrouki, D.; Baudry, J.; Pine, D.; Chaikin, P.; Bibette, J. Chiral colloidal clusters. Nature 2008, 455, 380–382. [Google Scholar]
- Cohen-Tannoudji, L.; Bertrand, E.; Baudry, J.; Robic, C.; Goubault, C.; Pellissier, M.; Johner, A.; Thalmann, F.; Lee, N.K.; Marques, C.M.; Bibette, J. Measuring the kinetics of biomolecular recognition with magnetic colloids. Phys. Rev. Lett. 2008, 100, 108301:1–108301:4. [Google Scholar]
- Chemla, Y.R.; Grossman, H.L.; Poon, Y.; McDermott, R.; Stevens, R.; Alper, M.D.; Clarke, J. Ultrasensitive magnetic biosensor for homogeneous immunoassay. Proc. Natl. Acad. Sci. USA 2000, 97, 14268–14272. [Google Scholar]
- Grossman, H.L.; Myers, W.R.; Vreeland, V.J.; Bruehl, R.; Alper, M.D.; Bertozzi, C.R.; Clarke, J. Detection of bacteria in suspension by using a superconducting quantum interference device. Proc. Natl. Acad. Sci. USA 2004, 101, 129–134. [Google Scholar]
- Lee, S.; Myers, W.R.; Grossman, H.L.; Cho, H.M.; Chemla, Y.R.; Clarke, J. Magnetic gradiometer based on a high-transition temperature superconducting quantum interference device for improved sensitivity of a biosensor. Appl. Phys. Lett. 2002, 81, 3094–3096. [Google Scholar]
- Hong, C.Y.; Wu, C.C.; Chiu, Y.C.; Yang, S.Y.; Horng, H.E.; Yang, H.C. Magnetic susceptibility reduction method for magnetically labeled immunoassay. Appl. Phys. Lett. 2006, 88, 212512:1–212512:3. [Google Scholar]
- Hong, C.Y.; Chen, W.S.; Jian, Z.F.; Yang, S.Y.; Horng, H.E.; Yang, L.C.; Yang, H.C. Wash-free immunomagnetic detection for serum through magnetic susceptibility reduction. Appl. Phys. Lett. 2007, 90, 074105:1–074105:3. [Google Scholar]
- Connolly, J.; St Pierre, T.G. Proposed biosensors based on time-dependent properties of magnetic fluids. J. Magn. Magn. Mater. 2001, 225, 156–160. [Google Scholar]
- Chung, S.H.; Hoffmann, A.; Bader, S.D.; Liu, C.; Kay, B.; Makowski, L.; Chen, L. Biological sensors based on Brownian relaxation of magnetic nanoparticles. Appl. Phys. Lett. 2004, 85, 2971–2973. [Google Scholar]
- Fornara, A.; Johansson, P.; Petersson, K.; Gustafsson, S.; Qin, J.; Olsson, E.; Ilver, D.; Krozer, A.; Muhammed, M.; Johansson, C. Tailored magnetic nanoparticles for direct and sensitive detection of biomolecules in biological samples. Nano Lett. 2008, 8, 3423–3428. [Google Scholar]
- Stromberg, M.; Goransson, J.; Gunnarsson, K.; Nilsson, M.; Svedlindh, P.; Stromme, M. Sensitive molecular diagnostics using volume-amplified magnetic nanobeads. Nano Lett. 2008, 8, 816–821. [Google Scholar]
- Stromberg, M.; Zardan Gomez de la Torre, T.; Goransson, J.; Gunnarsson, K.; Nilsson, M.; Svedlindh, P.; Stromme, M. Multiplex detection of DNA sequences using the volume-amplified magnetic nanobead detection assay. Anal. Chem. 2009, 81, 3398–3406. [Google Scholar]
- Wang, S.X.; Li, G. Advances in giant magnetoresistance biosensors with magnetic nanoparticle tags: Review and outlook. IEEE Trans. Magn. 2008, 44, 1687–1702. [Google Scholar]
- Graham, D.L.; Ferreira, H.A.; Freitas, P.P. Magnetoresistive-based biosensors and biochips. Trends Biotechnol. 2004, 22, 455–462. [Google Scholar]
- Fu, A.; Hu, W.; Xu, L.; Wilson, R.J.; Yu, H.; Osterfeld, S.J.; Gambhir, S.S.; Wang, S.X. Protein-functionalized synthetic antiferromagnetic nanoparticles for biomolecule detection and magnetic manipulation. Angew. Chem. Int. Ed. 2009, 48, 1620–1624. [Google Scholar]
- Osterfeld, S.J.; Yu, H.; Gaster, R.S.; Caramuta, S.; Xu, L.; Han, S.J.; Hall, D.A.; Wilson, R.J.; Sun, S.; White, R.L.; Davis, R.W.; Pourmand, N.; Wang, S.X. Multiplex protein assays based on real-time magnetic nanotag sensing. Proc. Natl. Acad. Sci. USA 2008, 105, 20637–20640. [Google Scholar]
- Srinivasan, B.; Li, Y.; Jing, Y.; Xu, Y.; Yao, X.; Xing, C.; Wang, J.P. A detection system based on giant magnetoresistive sensors and high-moment magnetic nanoparticles demonstrates zeptomole sensitivity: potential for personalized medicine. Angew. Chem. Int. Ed. 2009, 48, 2764–2767. [Google Scholar]
- De Palma, R.; Reekmans, G.; Liu, C.; Wirix-Speetjens, R.; Laureyn, W.; Nilsson, O.; Lagae, L. Magnetic bead sensing platform for the detection of proteins. Anal. Chem. 2007, 79, 8669–8677. [Google Scholar]
- Hu, W.; Wilson, C.R.J.; Koh, A.; Fu, A.H.; Faranesh, A.Z.; Earhart, C.M.; Osterfeld, S.J.; Han, S.J.; Xu, L.; Guccione, S.; Sinclair, R.; Wang, S.X. High-moment antiferromagnetic nanoparticles with tunable magnetic properties. Adv. Mater. 2008, 20, 1479–1483. [Google Scholar]
- Lowery, T.J.; Palazzolo, R.; Wong, S.M.; Prado, P.J.; Taktak, S. Single-coil, multisample, proton relaxation method for magnetic relaxation switch assays. Anal. Chem. 2008, 80, 1118–1123. [Google Scholar]
| Particle | Size | Composition | Characteristics | Reference |
|---|---|---|---|---|
| CLIO | ∼30 nm | 5 nm core, 10 nm dextran coating | MRSw, R2 = 50 (s·mM Fe)-1 | [5] |
| Core/shell | 16 nm | Fe core, iron oxide shell, 2.5 nm shell thickness | MRSw, R2 = 260 (s·mM Fe)-1 | [17] |
| Mn-MNPa | 16 nm | Mn-doped iron oxide | MRSw, R2 = 420 (s·mM metal)-1 | [16] |
| MP | 1000 nm | Commercial (Dynabeads) | MRSw, R2 = 43 (s·mM Fe)-1 | [19] |
| Iron oxide | 56 nm | Commercial (Quantum Magnetics, Miltenyi Biotech) | SQUID | [35,36] |
| Iron oxide | 19.5 nm | AC susceptometer | [42] | |
| Cubic FeCo | 12.8 nm | 1.5 nm oxidized shell | GMR | [49] |
| SAFb | 100 nm | Multilayers of ferromagnetic, interlayer of nonmagnetic material | GMR, disk shape | [47] |
| Magnetic bead | 130, 250 nm | Commercial (Micromod Partikeltechnologie) | SQUID | [43] |
| Analyte | Magnetic particle/instrumentation | Sensitivity | Sample volume | Reference | |
|---|---|---|---|---|---|
| MRSw type I | nucleotide | CLIO, bench top relaxometer | Low nM∼pM | 300 μL | [4,5] |
| proteins | CLIO, bench top relaxometer | Low nM | 300 μL | [5,9] | |
| virus | CLIO, MRI | 50 viruses/100 μL | 100 μL | [11] | |
| bacteria | core/shell, DMRa | 20 CFUb/100 μL (membrane filetered) | 5 μL | [17] | |
| Cancer cell | Mn-MNP, DMR | 2 cells/1 μL | 5 μL | [16] | |
| MRSw Type II | antibody | MP, bench top relaxometer | <1 pM | 300 μL | [19] |
| AC susceptometer | antibody | Iron oxide NP | <1 nM | [42] | |
| SQUID | bacteria | Iron oxide NP | 1.1 × 105 bacteria/20 μL | [36] | |
| DNA | Magnetic bead | 3∼10 pM (signal amplification) | [43] | ||
| GMR | Protein | Cubic FeCo NP | 2 × 106 proteins | 2 μL | [49] |
| DNA | Antiferromagnetic NP | 10 pM | [47] | ||
| Protein | Iron oxide NP | 2.4 pM | [48] |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Koh, I.; Josephson, L. Magnetic Nanoparticle Sensors. Sensors 2009, 9, 8130-8145. https://doi.org/10.3390/s91008130
Koh I, Josephson L. Magnetic Nanoparticle Sensors. Sensors. 2009; 9(10):8130-8145. https://doi.org/10.3390/s91008130
Chicago/Turabian StyleKoh, Isaac, and Lee Josephson. 2009. "Magnetic Nanoparticle Sensors" Sensors 9, no. 10: 8130-8145. https://doi.org/10.3390/s91008130




