An Investigation on Micro-Raman Spectra and Wavelet Data Analysis for Pemphigus Vulgaris Follow-up Monitoring.
Abstract
:1. Introduction
2. Materials and methods
2.1 Sample preparation and spectra acquisition
2.2 Wavelet deconvolution
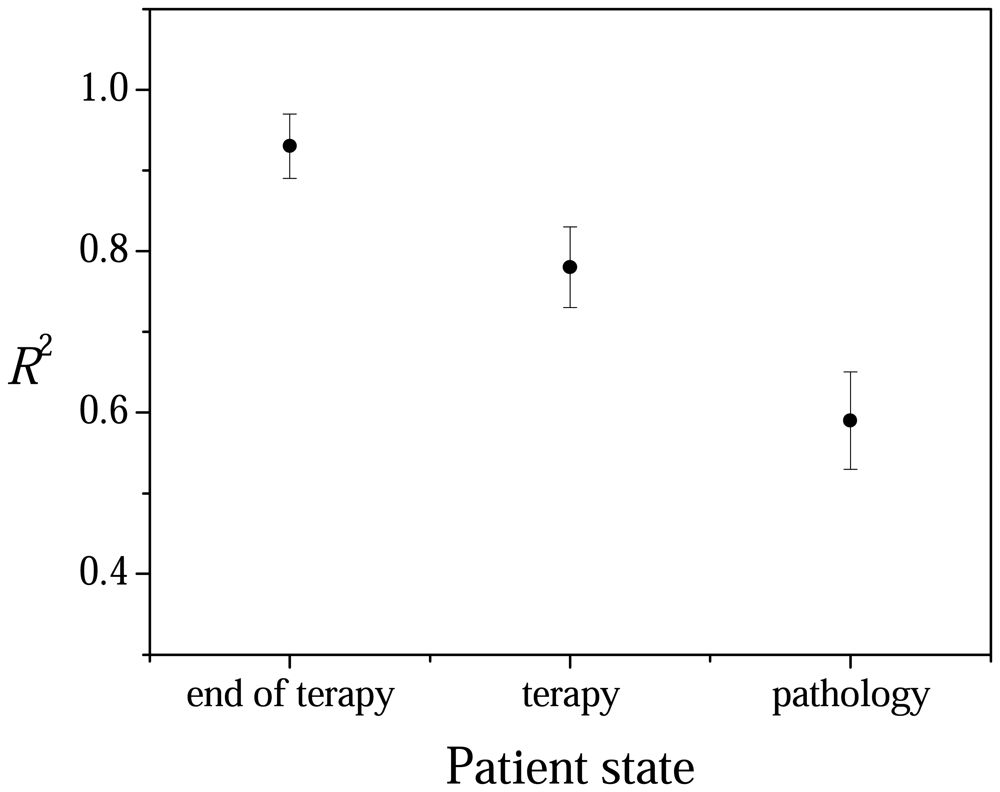
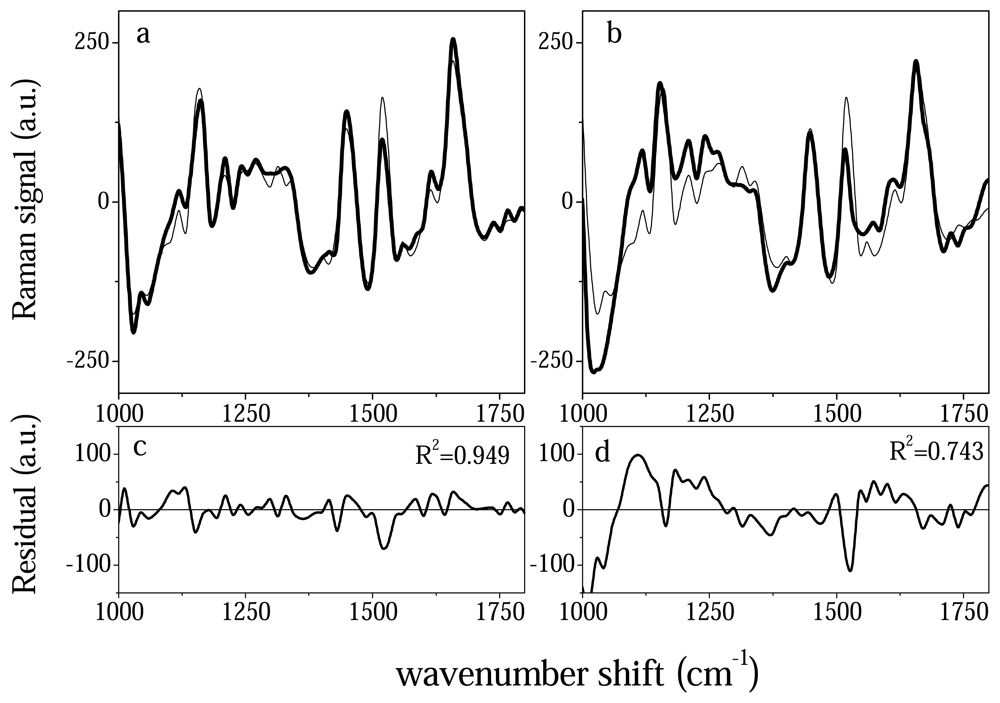
2.3 Linear regression analysis
3. Results and discussion
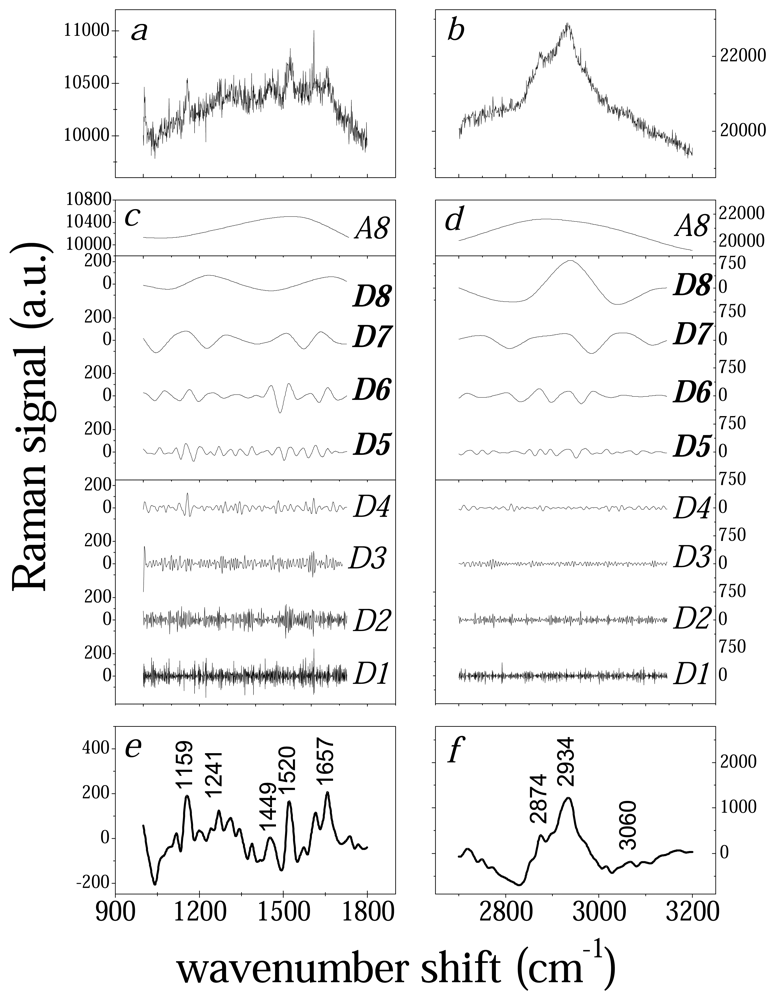
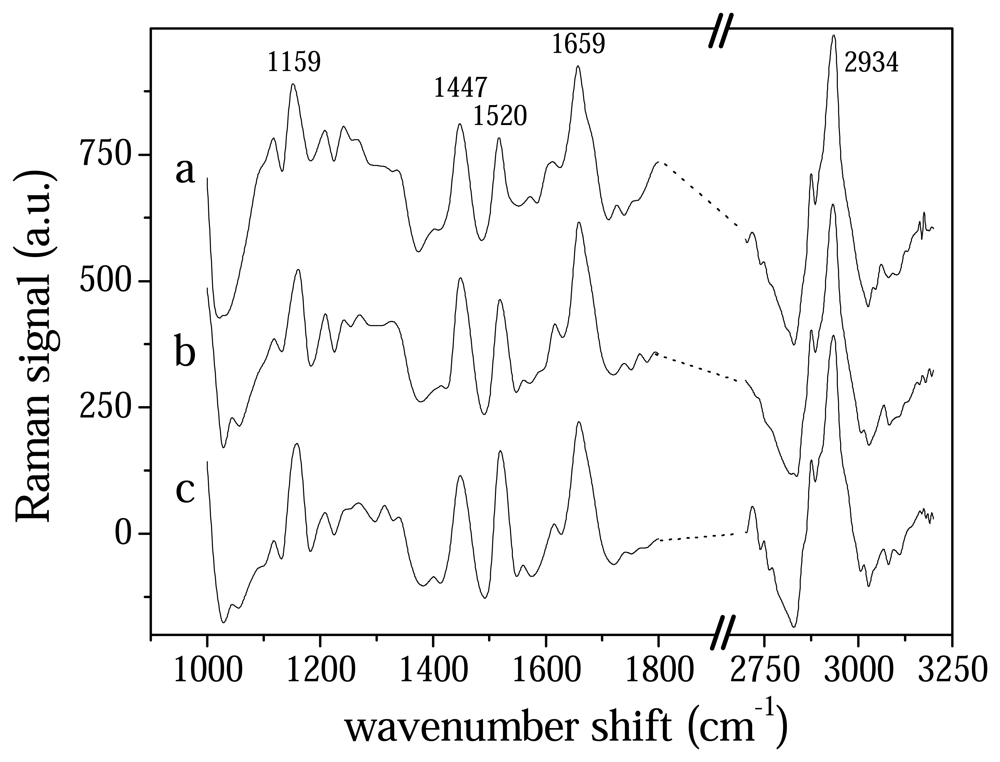
3.1 Wavelet analysis
4. Conclusions
References
- Lanza, A.; Cirillo, N.; Femiano, F.; Gombos, F. How does acantholysis occur in pemphigus vulgaris: a critical review. J Cutan Pathol. 2006, 33, 401–12. [Google Scholar]
- Scully, C.; Challacombe, S.J. Pemphygus vulgaris: update on ethiopatogenesis, oral manifestations, and managements. Crit. Rev, Oral Biol. Med. 2002, 13, 397–402. [Google Scholar]
- Cirillo, N.; Santoro, R.; Lanza, M.; Annese, P.; Gombos, F.; Lanza, A. Mucocutaneous pemphigus vulgaris carrying high-titre antidesmoglein 1 antibodies with skin lesions resembling pemphigus erythematosus. Clin Exp Dermatol. 2007, 33, 32–35. [Google Scholar]
- Manoharan, R.; Wang, Y.; Feld, M.S. Histochemical analysis of biological tissues using Raman spectroscopy. Spectrochimica Acta Part A 1996, 52, 215–249. [Google Scholar]
- Lau, D.P.; Huang, Z.; Lui, H.; Man, C.S.; Berean, K.; Morrison, M.D.; Zeng, H. Raman spectroscopy for optical diagnosis in normal and cancerous tissue of the nasopharynx-preliminary findings. Laser in Surgery and Medicine 2003, 32, 210–214. [Google Scholar]
- Rohleder, D.; Kocherscheidt, G.; Gerber, K.; Kiefer, W.; Kohler, W.; Mochs, J.; Petrich, W. Comparison of mid-infrared and Raman spectroscopy in the quantitative analysis of serum. J. Biom. Optics 2005, 10, 0301108. [Google Scholar]
- Pichardo-Molina, J.L.; Frausto-Reyes, C.; Barbosa-Garcia, O.; Huerta-Franco, R.; Gonzales- Trujillo, J.L.; Ramirez-Alvarado, C.A.; Gutierrez-Juarez, G.; Medina-Gutierrez, C. Raman spectroscopy and multivariate analysis of serum samples from breast cancer patients. Laser Med. Sci. 2007, 22, 229–236. [Google Scholar]
- Gaeta, G.M.; Zenone, F.; Camerlingo, C.; Riccio, R.; Moro, G.; Lepore, M.; Indovina, P.L. Data analysis in Raman measurements of biological tissues using wavelet technique. Proceedings of SPIE 2006, 5687, 170–178. [Google Scholar]
- Zenone, F.; Lepore, M.; Perna, G.; Carmone, P.; Delfino, I.; Gaeta, G.M.; Capozzi, V. Oral pathology diagnosis by means of micro-Raman spectroscopy on biopsies and blood serum. Proceedings of SPIE 2007, 6245, 62450V. [Google Scholar]
- Malini, R.; Venkatakrishna, K.; Kurien, J.; Pai, K.M.; Rao, L.; Kartha, V.B.; Murali Krishna, C. Discrimination of normal, inflammatory, premalignat and malignant oral tissue: a Raman spectroscopy study. Biopolymers 2006, 81, 179–193. [Google Scholar]
- Camerlingo, C.; Zenone, F.; Gaeta, G.M.; Riccio, R.; Lepore, M. Wavelet data processing of micro-Raman spectra of biological samples. Meas. Sci. Technol. 2006, 17, 298–303. [Google Scholar]
- Daubechies, I. Ten Lectures on Wavelets; (CBMS-NSF Series in Applied Mathematics vol 61); Philadelphia, ; PA; SIAM, 1992. [Google Scholar]
- Cohen, A.; Daubechies, I.; Feauveau, J. Biorthogonal bases of compactly supported wavelets. Commun. Pure Appl. Math. 1992, 45, 485–493. [Google Scholar]
- Ljunglöf, A.; Larsson, M.; Knuuttila, K.G.; Lindgren, J. Measurement of ligand distribution in individual adsorbent particles using confocal scanning laser microscopy and confocal micro- Raman spectroscopy. J. Chromatogr. A 2000, 893, 235–243. [Google Scholar]
- Ramanauskaite, R.B.; Seger-Nolten, I.G.M.J.; de Grauw, K.J.; Sijtsema, N.M.; Van der Maas, L.; Greve, J.; Otto, C.; Figdor, C.G. Carotenoid levels in human lymphocytes measured by Raman microspectroscopy. Pure and Appl. Chem. 1997, 69, 2131–2138. [Google Scholar]
- Rumi, G., Jr; Matus, Z.; Tóth, G.; Pár, A.; Nagy, Z.; Vinczi, A.; Rumi, G.; Mózsik, G. Changes of serum carotenoids in patients with esophageal, gastric, hepatocellular, pancreatic and colorectal cancer. J of Physiology-Paris 2001, 95, 239–245. [Google Scholar]
- Naziroğlu, M.; Kökçam, I.; Simşek, H.; Karakilçik, A.Z. Lipid peroxidation and antioxidants in plasma and red blood cells from patients with pemphigus vulgaris. J Basic Clin Physiol Pharmacol. 2003, 14, 31–42. [Google Scholar]
- Cirillo, N.; Gombos, F.; Lanza, A. Pemphigus vulgaris immunoglobulin G can recognize a 130 000 MW antigen other than desmoglein 3 on peripheral blood mononuclear cell surface. Immunology 2007, 121, 377–82. [Google Scholar]
- Ichimiya, M.; Yamamoto, K.; Muto, M. Successful treatment of pemphigus vegetans by addition of etretinate to systemic steroids. Clin Exp Dermatol. 1998, 23, 178–80. [Google Scholar]
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Camerlingo, C.; Zenone, F.; Perna, G.; Capozzi, V.; Cirillo, N.; Gaeta, G.M.; Lepore, M. An Investigation on Micro-Raman Spectra and Wavelet Data Analysis for Pemphigus Vulgaris Follow-up Monitoring. Sensors 2008, 8, 3656-3664. https://doi.org/10.3390/s8063656
Camerlingo C, Zenone F, Perna G, Capozzi V, Cirillo N, Gaeta GM, Lepore M. An Investigation on Micro-Raman Spectra and Wavelet Data Analysis for Pemphigus Vulgaris Follow-up Monitoring. Sensors. 2008; 8(6):3656-3664. https://doi.org/10.3390/s8063656
Chicago/Turabian StyleCamerlingo, Carlo, Flora Zenone, Giuseppe Perna, Vito Capozzi, Nicola Cirillo, Giovanni Maria Gaeta, and Maria Lepore. 2008. "An Investigation on Micro-Raman Spectra and Wavelet Data Analysis for Pemphigus Vulgaris Follow-up Monitoring." Sensors 8, no. 6: 3656-3664. https://doi.org/10.3390/s8063656



