1. Introduction
Transgenic plants, engineered with promoters driving the production of conveniently detectable protein products, have proven to be remarkably potent and robust tools for the dissection of gene activities. Such transgenic reporter gene constructions have been used broadly across multiple biological disciplines, in studies ranging from large scale genomic studies of expression patterns of unknown genes and gene networks, to the fine scale mapping of the expression patterns and characteristics of individual specific genes. Reporter gene constructions have employed many detectable protein products, including antibiotic or herbicide resistance proteins that govern growth, enzymatic activities such as
β-glucuronidase (GUS) that produce visible precipitates in plant tissues when presented with appropriate substrates, and light emitting proteins such as luciferase and the green fluorescent protein (GFP). While all these classes of reporter gene construction have proven valuable for a variety of applications, constructions using GFP are being utilized ever more widely since the assay procedure does not affect plant growth and development, whereas other methods require the addition of a substrate and usually sacrifice of the plant. To visualize GFP the plant need only be illuminated with a specific wavelength of light and imaged at another wavelength, and the plant is not damaged in the process [
1,
2].
As an increasing number of genes become extremely well characterized in terms of the environmental conditions that affect their expression, the promoters of these genes can in turn be deployed as effective in situ and in vivo sensors of plant reactions to their environment. For examples, genes induced by cold, heat, drought, flooding, touch, heavy metals, nutrients, light quality and atmospheric gas content have all been well characterized [e.g.:
3,
4-
8]. When the promoters for these genes are coupled with GFP in reporter gene constructions within transgenic plants, those plants become incredibly effective monitors of those particular environmental conditions, identifying both quantitatively and qualitatively the cells, tissues and organs of a plant responding to the condition. Once well characterized in controlled environmental conditions, transgenic reporter gene plants can be deployed to unknown or poorly characterized environments as useful biological monitors to examine the biological response to those environments.
Extraterrestrial environments present unique challenges to terrestrial organisms, presenting conditions that have not been faced during biological evolution on Earth. Altered gravity, hard radiation levels, atmospheric compositions and light qualities are all likely to be encountered in orbital, lunar and Martian habitats – habitats most likely to be encountered by humans and the organisms that will accompany humans. Plants certainly have been and will be a part of human spaceflight and extraterrestrial habitation efforts as parts of bioregenerative life support systems [
9,
10]. Therefore a clear understanding of plant responses to extraterrestrial environments is not only an interesting study of the limits of biology in the solar system, but also a strategic human life support imperative.
Transgenic
Arabidopsis thaliana (Arabidopsis) plants employing GUS as part of the reporter construction have been used in orbital space shuttle experiments to examine specific gene responses to the spaceflight environment [
11,
12]. Astronauts harvested the plants on orbit and conducted the staining experiments necessary to see the GUS reporter gene activity. The plants were returned to Earth and the results from that study have helped characterize plant responses to spaceflight. However, future spaceflights will have limited access to crew time for experimentation. In addition, early biological experiments in High Earth Orbit, on the moon and on Mars are very likely to be robotic or autonomous missions requiring that gene expression information be returned to Earth via telemetry. Further, tradeoffs between internal pressure, structure, crew time, etc. will dictate whether human tending of future bio-regenerative life support systems will be practical. Thus transgenic plants expressing environment-specific GFP are being developed, as is the technology to obtain and transmit GFP fluorescence images telemetrically [
2,
13].
This paper focuses on the deployment of a prototype of a spaceflight-capable plant GFP imaging system within an extraterrestrial habitat analog, the Arthur Clarke Mars Greenhouse at the Haughton Mars Project Research Station on Devon Island in the Canadian High Arctic. This project was devised and conducted in order to take a laboratory proven imaging concept and develop the requisite software, hardware, systems and communications capability to move that concept to a high level of practical technology readiness within a challenging operational environment.
2. Materials, Equipment and Context
2.1. Transgenic Plants
The species chosen for these experiments is Arabidopsis thaliana (Arabidopsis), a model plant that has a rich genetic and molecular biology background and a strong spaceflight heritage. Arabidopsis is also among the most understood of plants with regards to reactions and adaptation to environmental stresses, and when transformed with reporter gene constructions, Arabidopsis is a proven platform for development of phytosensors based on gene expression.
The green fluorescence protein used in these plants is referred to as GFP S65T, a “red shifted” version of the original jellyfish GFP [
14-
16]. This GFP was engineered to have a peak excitation at 488 nm and peak fluorescence at 512 nm, and has been deployed widely in the laboratory environment as a reporter for plants [
1,
17,
18]. Similar engineered versions of GFP have been deployed in other species [
19]. Plants containing a GFP reporter construct driven by the constitutive CaMV 35S promoter, as well as control plants without any GFP, were chosen for comparison in this demonstration project. The construction of these plants, as well as the use of these plants and those with environmentally inducible GFP constructions in a variety of spaceflight relevant experiments, has been detailed previously [
2,
8,
13,
20].
The growth format for the plants in this study is taken from the STS-93 space shuttle experiment [
11,
21]. Square format Petri plates containing a semisolid nutrient substrate were planted with sterile seeds, with germination and growth occurring in the plate-vertical orientation. This allows a simple and clearly organized presentation of both roots and shoots for images from the nadir position relative to the surface of the plates. The growth medium contains nutrients and water such that light and a temperature above 4°C are the only exogenous requirement for
Arabidopsis growth. The plants are grown under the ambient light conditions of the greenhouse until they are inserted into the imager. Plants on nutrient agar plates can remain viable for weeks with no exogenous light (note the date stamps in
Figure 4).
2.2. GFP Imager
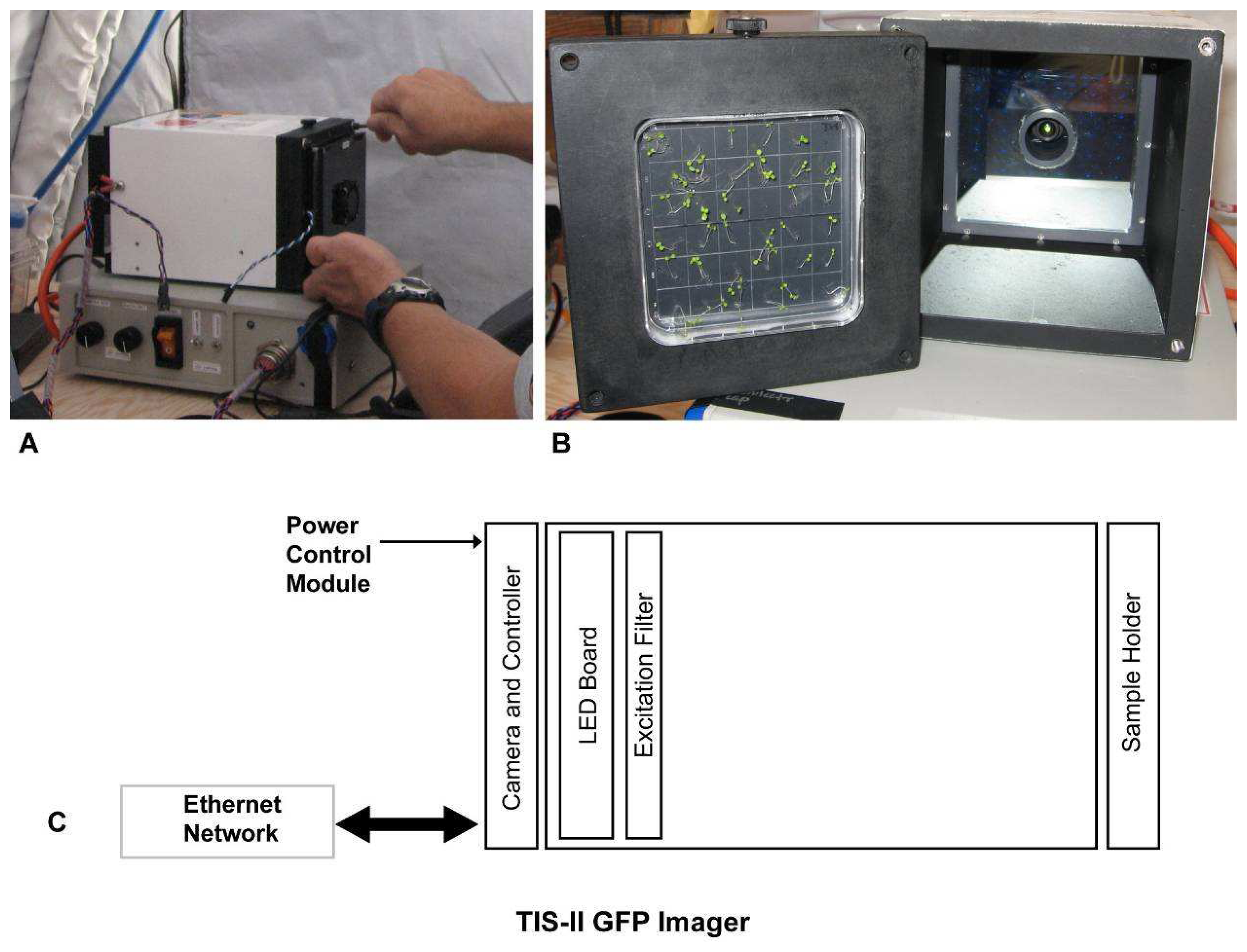
Telemetric data collection of GFP required the development and construction of prototype hardware that is compatible with the GFP gene expression system, with the biosensor plants, and with the power and communications environment of spaceflight-related system. The TAGES Imaging System-II (TIS-II) represents the second generation of hardware designed to collect GFP expression data in real-time aboard a spaceflight experiment (
Figure 1). Both TAGES GFP imagers use a system of blue-light light emitting diodes (LEDs) (Lumileds, a division of Philips semiconductor) for excitation of GFP and a specially designed narrow band pass filter with a spectrum spread of λ 470nm to 488nm, with a spectral mid point λ of 475nm. A longpass filter (500nm +) was designed for the optical lens cell for the imager sensor. The primary differences between TIS-I and TIS-II reside in the configuration of the camera and sophistication of the optics [
13].
Imaging is accomplished with a 5 mega pixel CMOS imager that can capture the square Petri growth plate in a 1280 × 1280 pixel image and transfer that image into on-board 3 mega pixel video FIFO Ram chips before processing and storage on a local MMC/SD card. An on-board Microchip PIC embedded processor is used to control all the imager and lighting functions, with communications through a local area network (LAN). The imager camera settings can be modified by uploading commands through the LAN. In addition commands can be issued to perform all the necessary functions of taking and downloading images from the TIS-II Imager to the local controlling computer.
The Lumiled LED emitters are used to provide illumination because these devices consume less power and produce more light than earlier models such as the T1-3/4 emitters. The LEDs are packaged with a slug extruding from the rear to remove heat and are surface mounted on a custom designed circuit board using 48 of the Lumileds. An equivalent board using older LEDs would have to be populated with over of the 500 T1-3/4 devices. For image capture, the LEDs are flashed on for a period of two video frames while the image is being taken. Also during the ‘on’ period the LEDs are controlled with a PWM (pulse width modulation) allowing the intensity to be changed to meet imaging requirements.
2.3. Arthur Clarke Mars Greenhouse
Several Canadian research programs involve the development of bioregenerative life support technologies for future long duration missions to the moon and Mars. One example is the Micro-Ecological Life Support Alternative (MELiSSA) Project's Higher Plant Compartment [
22] and relevant to the GFP imaging project in discussion, the Arthur Clarke Mars Greenhouse (ACMG) [
23]. The ACMG (
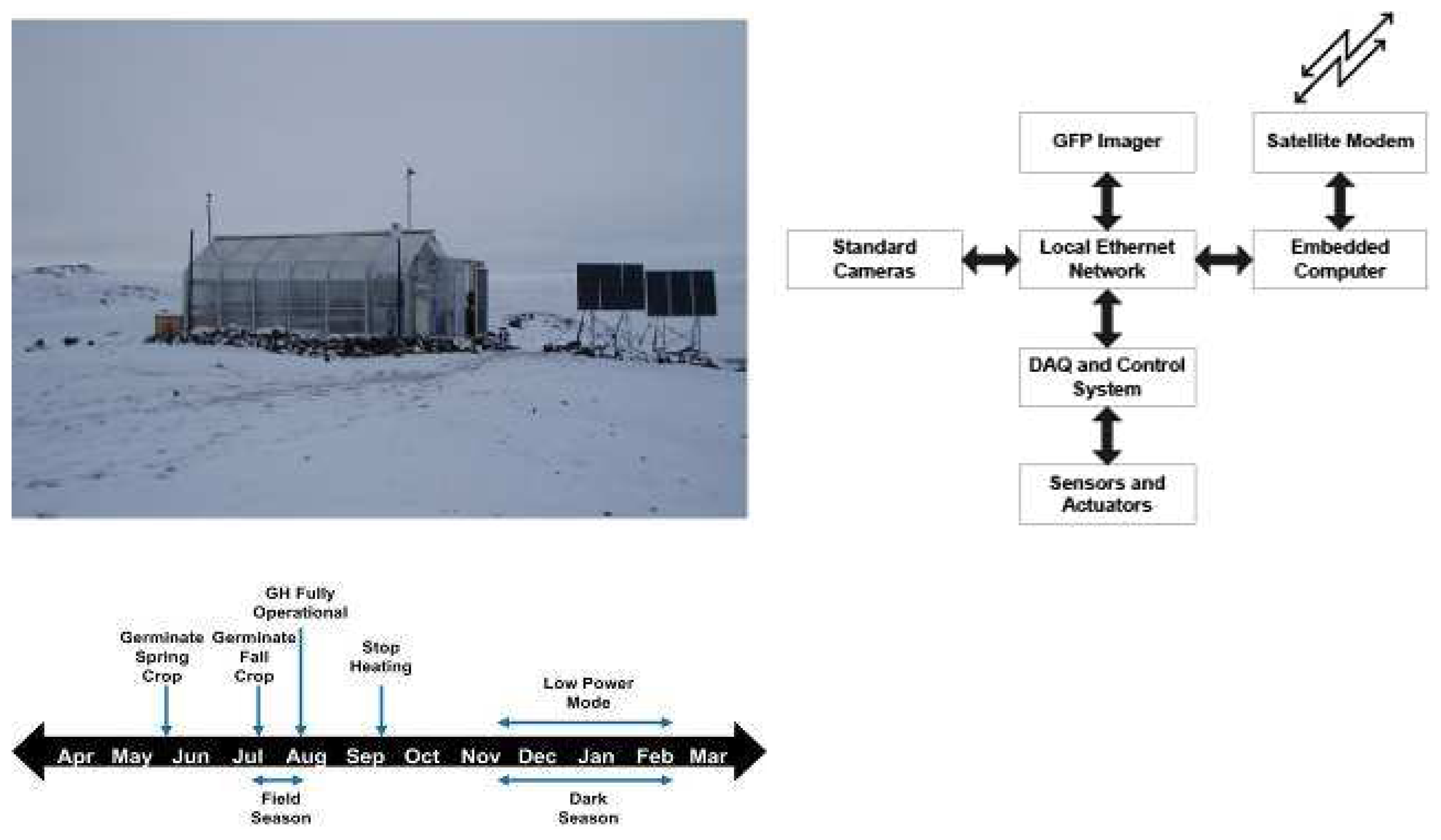
Figure 2) is located on the rim of Haughton Crater on Devon Island in the Canadian High Arctic and its fundamental purpose is to serve as a development analog for systems that would reside on the surface of the moon or Mars. The extreme environment of the Arctic is itself a challenge to the systems development process and procedure. All of the ACMG physical sensors, including the external weather station, are designed to collect data to −40°C, and have been successfully monitored in a variety of extreme conditions. In addition, the remote nature of the location places these systems development efforts within operational constraints that further mimic extraterrestrial deployments. For example, communications must be managed via highly specific, satellite-based transceivers and power must be developed and managed in situ. Researchers are typically only on-site during the month of July and thus human intervention is minimal, and non-existent for most of the year.
Figure 2 also shows a timeline representing the basic annual operational strategy and duration of on-site ACMG activity. Thus the ACMG is a very relevant testbed for developing system integration and data handling of the TIS-II GFP Imager as an advanced, gene expression sensor subsystem.
The ACMG greenhouse was established in 2002. A detailed description of the greenhouse systems and operational strategy is available in [
23]. In general the ACMG is not a full-featured, high fidelity simulation of a greenhouse to be established on Mars. It does, though, support scientific and operations research in an environment which is operationally relevant to Mars in unique ways – each at a specific level of fidelity and complexity. The main greenhouse systems include the plant growth system, environmental control system, power system, communication system and local network and the data acquisition and control system. The TIS-II GFP Imager and greenhouse interfaces are presented in
Figure 2. Data from the TIS-II GFP Imager is passed by the local Ethernet network through an Ethernet switch (EISB16-100T, CTRLink) to the data acquisition (DAQ) and control system (cFP-2120, National Instruments) where it is stored and later transferred to the embedded communications, command and control computer (Linux-based single board computer). From this point, data can be transferred through the local high-speed optical fiber network to local users, or onwards via a highspeed human planetary exploration mission-equivalent wireless and satellite communications network, when both are available during full operation. At other times, the communications, command and control computer can receive commands to initiate transfer of images and data via a year-round low-speed satellite modem (SDX-1100, Infosat) for eventual transmission by satellite. GFP images, along with all greenhouse data, are received by an operations center at Simon Fraser University in Vancouver, British Columbia, through a second SDX-1100 system operated via an autonomous command and control system operating on a Unix (Sun Microsystems Solaris-based) network-distributed mission critical server architecture, where it is made available to researchers through the Internet. To minimize technology deployment risk to the Arctic greenhouse any system destined for ACMG is first trialed in a duplicate greenhouse located at the CSA headquarters in Longueuil, Quebec. This duplicate greenhouse was constructed after several years of ACMG operations when it was realized that confidence in new hardware and software could be gained prior to integration into the ACMG. This duplicate greenhouse also provides an opportunity for greenhouse researchers to increase their work efficiency during the Arctic field season by first carrying out simulated deployments in the south.
2.4. Haughton Mars Project on Devon Island
The Haughton-Mars Project (HMP) is an interdisciplinary and international field research project making use of the Haughton crater impact structure and surrounding terrain as a terrestrial environmental analog for Mars and the moon. The Haughton Mars Project Research Station (HMP RS) is located in a remote polar desert on Devon Island, an uninhabited island in the territory of Nunavut in the Canadian High Arctic. The geologic features and biological attributes of Devon Island in general, and the HMP RS site in particular, offer a unique research and operations environment appropriate to the development of approaches, technologies, and field-based operational methodologies that might be critical to successful extraterrestrial human missions [
24]. The HMP RS is managed and operated jointly by the Mars Institute and the SETI Institute with support from the Canadian Space Agency and NASA.
3. Results and Discussion
The TIS-II GFP Imager was transported to the HMP RS in the 2006 summer field season for the primary purposes of defining the operational parameters of including the TIS-II GFP Imager as part of the sensor suite in the ACMG. Before installation in the ACMG the imager was bench tested at HMP with a laptop computer for passing control and data, operating via the local one (1) gigabit-per second switched fiber network. During the bench testing phase, the imager was enabled for remote internet access and data exchange control originating with the Experiments Monitoring Area at Kennedy Space Center (KSC). These communications were managed by the high-speed network infrastructure of the HMP, designed to emulate planetary exploration networks, and demonstrated the processing independence of the TIS-II GFP Imager, while also ground-testing the fundamental communications protocols for sending commands and receiving data from the imager, regardless of distance. Commands to take images, store images, process images and download images from KSC proved effective. This test established that the native high-speed HMP network infrastructure, satellite communications, and protocols were sufficiently robust to handle the command and data flow, and that the size and structure of the imager commands and image data files were appropriate for stable deployment within the HMP system.
3.1. Installation of the TIS-II GFP Imager into the ACMG
The successful bench tests connecting KSC to the TIS-II Imager at HMP RS were followed by physical installation of the TIS-II into the ACMG and electronic connection to the ACMG control systems. The TIS-II GFP Imager then passed from active control by internet connection to automated control within the greenhouse sensor and data acquisition systems. Command and data packets were controlled by the embedded computer of the greenhouse. Images were processed and stored locally in the DAQ and control system before satellite transfer as part of the ACMG data stream controlled by the Linux single board computer.
Figure 3 shows the imager set up against the northern wall of the greenhouse (left) and a closer view of the system as it is connected to its power supply and through a network cable to the ACMG systems.
3.1. Telemetric Data Collection
One goal of the integration of the TIS-II into the greenhouse systems is to enable future automated monitoring of growth and development of plants during periods without human intervention. The pictures in the top panel of
Figure 4 show a subset of the data collected under an autonomous scenario in 2006. Images were collected twice daily by the ACMG systems and downloaded via satellite link to a remote location as part of the normal flow of commands and data from and to the ACMG. The imager/greenhouse collected and telemetrically transferred these data for several weeks before decommissioning at the end of the field season. For additional image referencing, the bottom panel shows a bench-top germination experiment, a portion of a time-course experiment which monitored biosensor plant germination and initial growth over a two week period. In both cases, close inspection of the images reveals two phenotypes of plants, one that glows brightly green and one that can be seen only as a much less intense fluorescence in the red wavelengths due to the auto-fluorescence of chlorophyll. These two genotypes provide the range of GFP image responses that can be expected in an actual deployment of GFP biosensor plants.
The images in
Figure 4 indicate that the TIS-II Imager is fundamentally capable of capturing differences in the expression range of GFP fluorescence. Moreover, these images demonstrate the integration of GFP biosensor plants and imaging technology into, and as a part of, a distantly deployed and autonomous greenhouse data collection system. Over the coming field seasons the ACMG TIS-II imager team will continue to integrate the TIS-II Imager into the greenhouse environmental monitoring system allowing for the use of TIS-II Imager data in greenhouse control strategy just as current greenhouse thermocouples, RH sensors, light sensors are presently used for remote operations.
4. Conclusions
Telemetric data collection has been widely used in applications where human participation is cumbersome, dangerous or unfeasible. Many examples exist in the arena of spaceflight and planetary exploration, but the application to biological payloads has thus far been restricted to the collection of standard white-light photography that is limited to reporting the surface characteristics of the specimens involved. The incorporation of imaging technologies that enables the excitation and collection of specialized wavelengths greatly expands the range of scientific information that can be returned from a payload telemetrically. Organisms engineered with GFP reporter genes used in conjunction with fluorescent imaging systems can create a system of biological sensors that telemetrically report on the patterns of gene expression occurring in real time while adapting to a novel environment. The insights obtained with the integration of TIS-II into the ACMG have provided initial proof of concept for telemetric collection of fluorescent biosensor images. While the ultimate goal is to create a fully integrated system that can be deployed in any environment to collect real-time data on the physiological and molecular biological responses, intermediate development areas include improvement of fluorescent image data processing, reducing the form and power requirements for imaging, and producing engineering designs that address specific monitoring needs.
Acknowledgments
The authors specifically acknowledge the Arthur Clarke Mars Greenhouse (ACMG) and the Haughton Mars Project (HMP) for fundamentally making this study possible. The ACMG was established and is operated under the auspices of the HMP. Initial sponsorship support for the establishment of the ACMG was provided by NASA, SpaceRef Interactive Inc., SETI Institute (SI), Simon Fraser University (SFU), University of Guelph (UG), and the Canadian Space Agency (CSA). Research in the ACMG is currently led by principal investigator Dr. Alain Berinstain of the CSA and UG, with funding support from the Ontario Centres of Excellence (OCE), and in partnership with other researchers from CSA, UG, SFU, and SI and the University of Florida. The ACMG is located at the Haughton-Mars Project Research Station (HMP RS) on Devon Island, Canadian High Arctic. The HMP RS is managed and operated jointly by the Mars Institute and the SETI Institute with support from the Canadian Space Agency and NASA. The research of Paul and Ferl is also supported by NASA grant NNX07AH270.
References and Notes
- Chiu, W.; Niwa, Y.; Zeng, W.; Hirano, T.; Kobayashi, H.; Sheen, J. Engineered GFP as a vital reporter in plants. Curr. Biol. 1996, 6, 325–330. [Google Scholar]
- Manak, M.S.; Paul, A.L.; Sehnke, P.C.; Ferl, R.J. Remote sensing of gene expression in Planta: transgenic plants as monitors of exogenous stress perception in extraterrestrial environments. Life Support Biosph. Sci. 2002, 8, 83–91. [Google Scholar]
- Schaffer, R.; Landgraf, J.; Perez-Amador, M.; Wisman, E. Monitoring genome-wide expression in plants. Curr. Opin. Biotechnol. 2000, 11, 162–167. [Google Scholar]
- Seki, M.; Narusaka, M.; Abe, H.; Kasuga, M.; Yamaguchi-Shinozaki, K.; Carninci, P.; Hayashizaki, Y.; Shinozaki, K. Monitoring the expression pattern of 1300 Arabidopsis genes under drought and cold stresses by using a full-length cDNA microarray. Plant Cell 2001, 13, 61–72. [Google Scholar]
- Seki, M.; Narusaka, M.; Ishida, J.; Nanjo, T.; Fujita, M.; Oono, Y.; Kamiya, A.; Nakajima, M.; Enju, A.; Sakurai, T.; Satou, M.; Akiyama, K.; Taji, T.; Yamaguchi-Shinoza…, K.; Carninci, P.; Kawai, J.; Hayashizaki, Y.; Shinozaki, K. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J 2002, 31, 279–292. [Google Scholar]
- Klok, E.J.; Wilson, I.W.; Wilson, D.; Chapman, S.C.; Ewing, R.M.; Somerville, S.C.; Peacock, W.J.; Dolferus, R.; Dennis, E.S. Expression profile analysis of the low-oxygen response in Arabidopsis root cultures. Plant Cell 2002, 14, 2481–2494. [Google Scholar]
- Ozturk, Z.N.; Talame, V.; Deyholos, M.; Michalowski, C.B.; Galbraith, D.W.; Gozukirmizi, N.; Tuberosa, R.; Bohnert, H.J. Monitoring large-scale changes in transcript abundance in drought- and salt-stressed barley. Plant Mol Biol 2002, 48, 551–573. [Google Scholar]
- Paul, A.-L.; Schuerger, A.C.; Popp, M.P.; Richards, J.T.; Manak, M.S.; Ferl, R.J. Hypobaric biology: Arabidopsis gene expression at low atmospheric pressure. Plant Physiol 2004, 134, 215–223. [Google Scholar]
- Salisbury, F.B. Growing crops for space explorers on the moon, Mars, or in space. Adv Space Biol Med 1999, 7, 131–162. [Google Scholar]
- Ferl, R.; Wheeler, R.; Levine, H.G.; Paul, A.-L. Plants in space. Curr Opin Plant Biol 2002, 5, 258–263. [Google Scholar]
- Paul, A.-L.; Daugherty, C.J.; Bihn, E.A.; Chapman, D.K.; Norwood, K.L.; Ferl, R.J. Transgene expression patterns indicate that spaceflight affects stress signal perception and transduction in arabidopsis. Plant Physiol 2001, 126, 613–621. [Google Scholar]
- Paul, A.-L.; Ferl, R.J. Molecular aspects of stress-gene regulation during spaceflight. J Plant Growth Regul 2002, 21, 166–176. [Google Scholar]
- Paul, A.-L.; Murdoch, T.; Ferl, E.; Levine, H.G.; Ferl, R. The TAGES Imaging System: Optimizing a green fluorescent protein imaging system for plants. SAE Technical Paper 2003, 2003-01-2477. [Google Scholar]
- Prasher, D.C.; Eckenrode, V.K.; Ward, W.W.; Prendergast, F.G.; Cormier, M.J. Primary structure of the Aequorea victoria green-fluorescent protein. Gene 1992, 111, 229–233. [Google Scholar]
- Ormo, M.; Cubitt, A.B.; Kallio, K.; Gross, L.A.; Tsien, R.Y.; Remington, S.J. Crystal structure of the Aequorea victoria green fluorescent protein. Science 1996, 273, 1392–1395. [Google Scholar]
- Heim, R.; Cubitt, A.B.; Tsien, R.Y. Improved green fluorescence. Nature 1995, 373, 663–664. [Google Scholar]
- Sheen, J.; Hwang, S.; Niwa, Y.; Kobayashi, H.; Galbraith, D.W. Green-fluorescent protein as a new vital marker in plant cells. Plant J 1995, 8, 777–784. [Google Scholar]
- Haseloff, J.; Amos, B. GFP in plants. Trends Genet 1995, 11, 328–329. [Google Scholar]
- Hutter, H. Fluorescent reporter methods. In Methods in molecular biology; Clifton, N.J, 2006; Volume 351, pp. 155–173. [Google Scholar]
- Paul, A.-L.; Ferl, R.J. Plant biology in low atmospheric pressure - Implications for exploration mission design and advanced life support. Gravitational and Space Biology 2006, 19, 3–17. [Google Scholar]
- Paul, A.-L.; Popp, M.P.; Gurley, W.B.; Guy, C.; Norwood, K.L.; Ferl, R. Arabidopsis gene expression patterns are altered during spaceflight. J Adv Space Res 2005, 36, 1175–1181. [Google Scholar]
- Lasseur, C.; Paillé, C.; Lamaze, B.; Rebeyre, P.; Rodriguez, A.; Ordonez, L.; Marty, F. MELISSA: overview of the project and perspective. Proceedings of the 35th International Conference on Environmental Systems. SAE Technical paper 2005, 2005-01–3066. [Google Scholar]
- Giroux, R.; Berinstain, A.; Braham, S.; Graham, T.; Bamsey, M.; Boyd, K.; Silver, M.; Lussier-Desbiens, A.; Lee, P.; Boucher, M.; Cowing, K.; Dixon, M. Greenhouses in extreme environments: The Arthur Clarke Mars Greenhouse design and operation overview. Advances in Space Research 2006, 38, 1248–1259. [Google Scholar]
- Lee, P. Haughton-Mars Project: preparing for Mars in a unique public/private - and international - setting. Ad Astra 2002. [Google Scholar]
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.