Dependence of Impedance of Embedded Single Cells on Cellular Behaviour
Abstract
:1. Introduction
2. Materials and Methods
2.1. Analytical Equation for Resistance of Embedded Single Cell
2.2. Impedance Measurement of Single Cells Embedded in Capillary
3. Results and Discussion
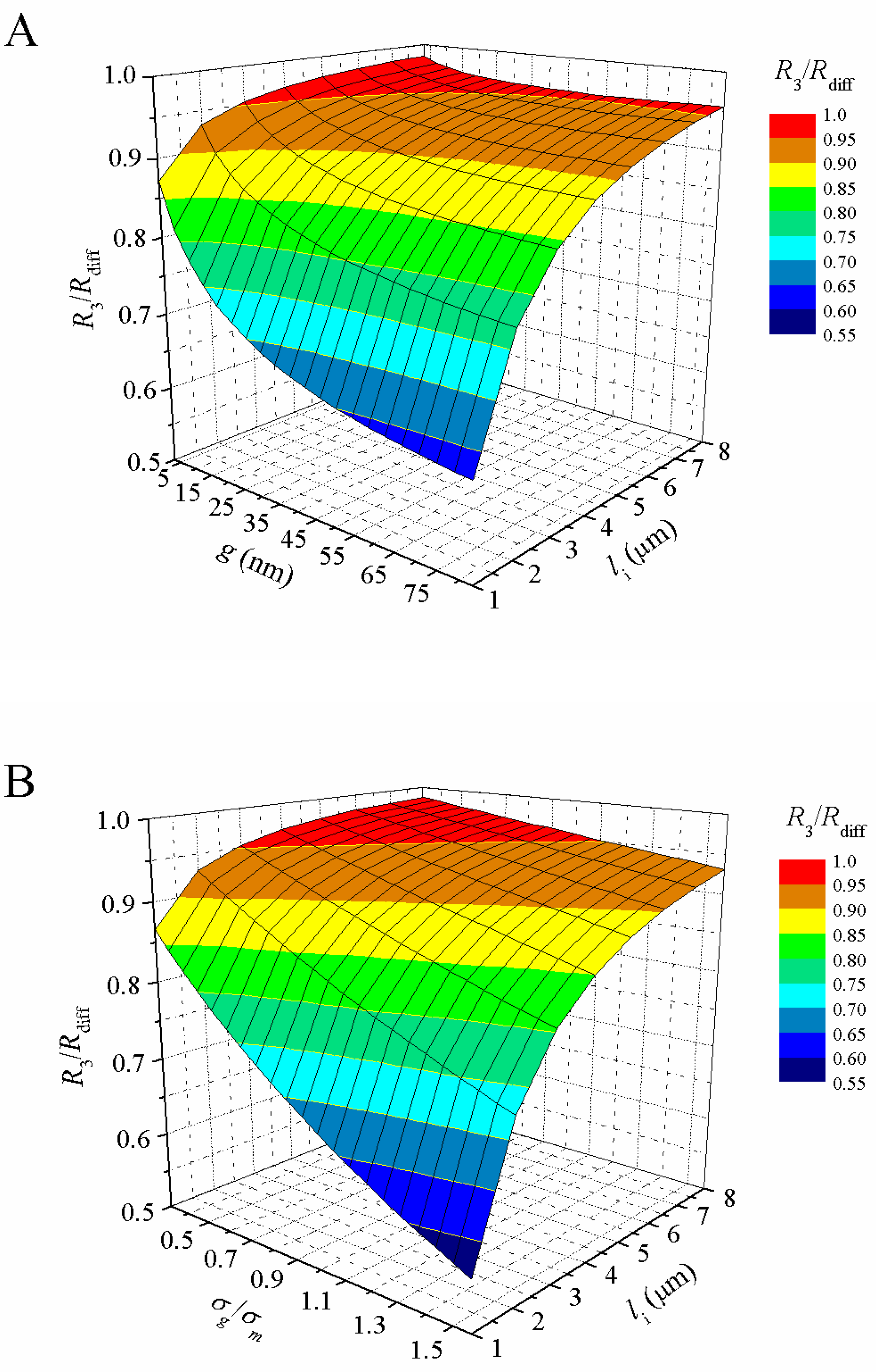
3.1. Theoretical Resistance of Embedded Single Cell
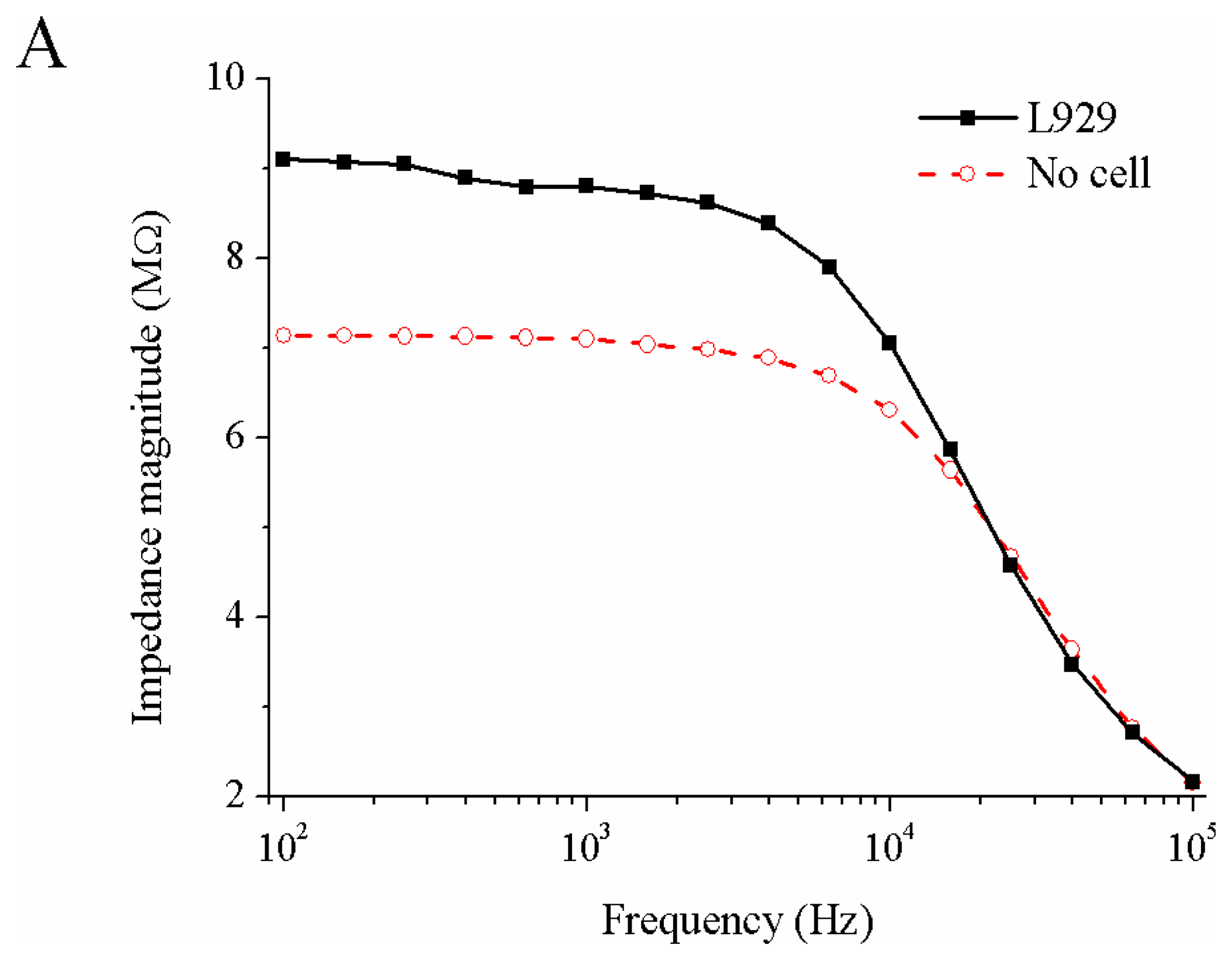
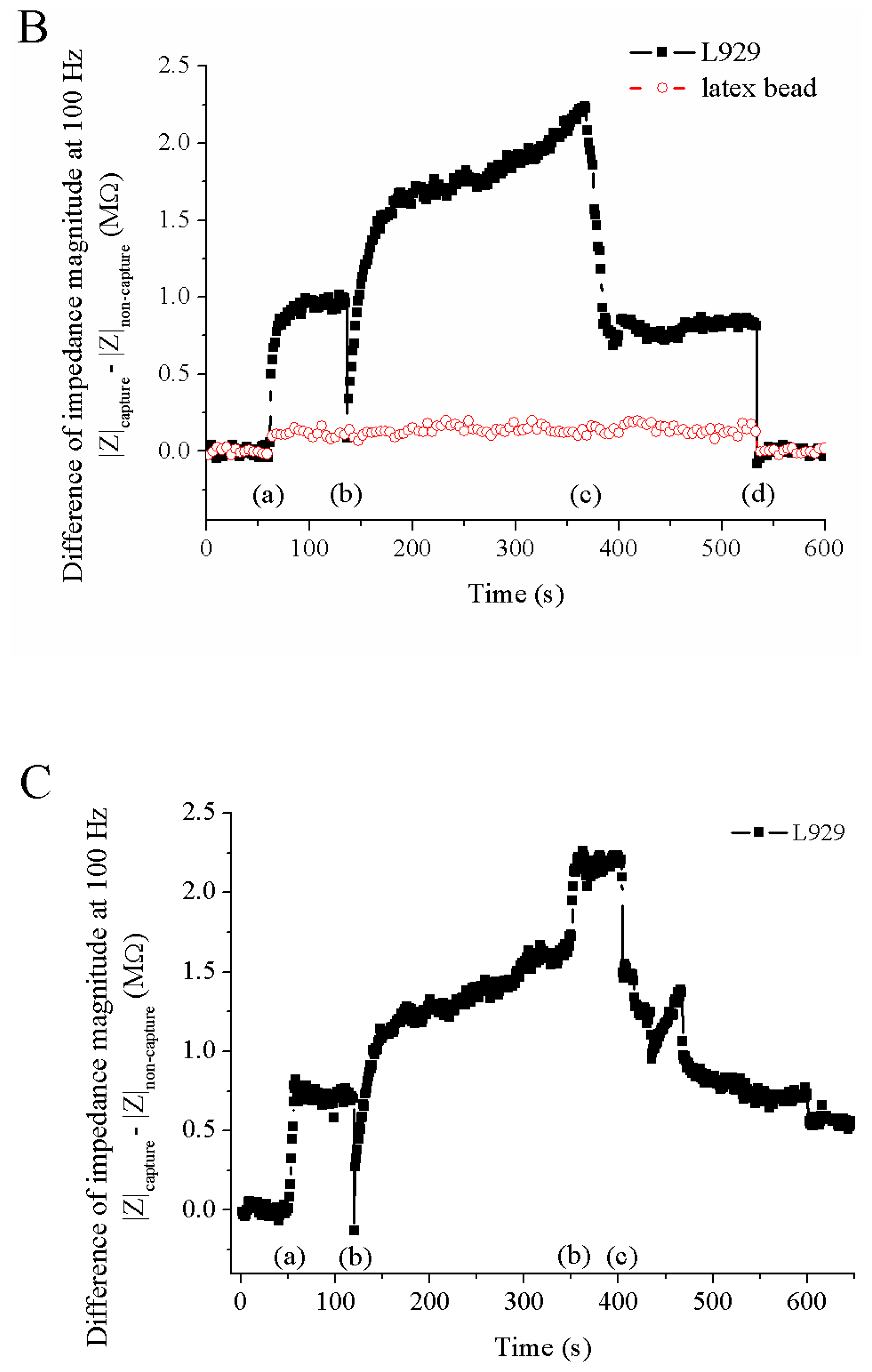
3.2. Measured Impedance of Single Cells Embedded in Capillary
- a)
- capture of a particle (cell or bead)
- b)
- application of DMSO (100 μl of culture medium including 5% DMSO)
- c)
- breakdown of cell membrane
- d)
- release of a particle (cell or bead)
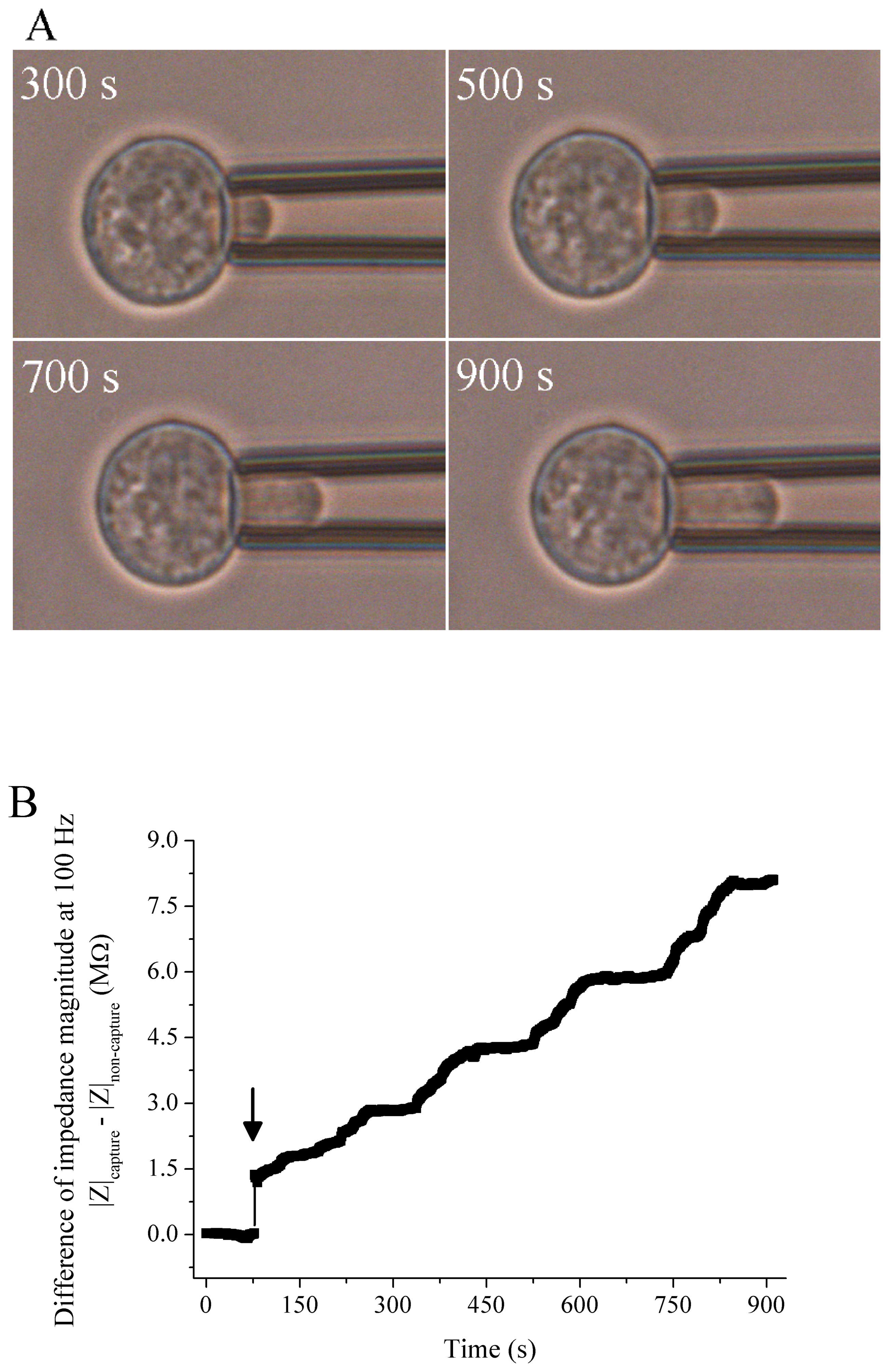
3.3. Interpretation of Measured Impedance of Embedded Single Cell
4. Conclusions
References
- Carlo, D.D.; Lee, L. P. Dynamic single-cell analysis for quantitative biology. Analytical Chemistry 2006, 6, 7918–7925. [Google Scholar]
- Mack, A.; Thielecke, H.; Robitzki, A. 3D-biohybrid systems: applications in drug screening. TRENDS in Biotechnology 2001, 20, 56–61. [Google Scholar]
- Vestergaard, M.; Kerman, K.; Tamiya, E. An overview of label-free electrochemical protein sensors. Sensors 2007, 7, 3442–3458. [Google Scholar]
- Giaever, I.; Keese, C.R. A morphological biosensor for mammalian cells. Nature 1993, 366, 591–592. [Google Scholar]
- Cho, S.; Becker, S.; Briesen, H.; Thielecke, H. Impedance monitoring of herpes simplex virus-induced cytopathic effect in Vero cells. Sensors and Actuators B: Chemical 2007, 123, 978–982. [Google Scholar]
- Schwan, H. P. Advances in Biological and Medical Physics; Lawrence, J. H., Tobias, C. A., Eds.; Academic Press: New York, 1957; pp. 147–209. [Google Scholar]
- Schwan, H. P. Physical technique in biological research; Nastuk, W. L., Ed.; Academic Press: New York, 1965; pp. 323–407. [Google Scholar]
- Bao, J.Z.; Davis, C. C.; Schmukler, R. E. Frequency domain impedance measurements of erythrocytes-Constant phase angle impedance characteristics and a phase transition. Biophysical Journal 1992, 61, 1427–1434. [Google Scholar]
- Bao, J.Z.; Davis, C. C.; Schmukler, R. E. Impedance spectroscopy of human erythrocytes: System calibration and nonlinear modeling. IEEE Transactions on biomedical engineering 1993, 40, 364–378. [Google Scholar]
- Takashima, S.; Asami, K.; Takahashi, Y. Frequency domain studies of impedance characteristics of biological cells using micropipette technique. Biophysical Journal 1988, 54, 995–1000. [Google Scholar]
- Asami, K.; Takahashi, Y.; Takashima, S. Frequency domain analysis of membrane capacitance of cultured cells (HeLa and myeloma) using the micropipette technique. Biophysical Journal 1990, 58, 143–148. [Google Scholar]
- Ayliffe, H.E.; Frazier, A. B.; Rabbitt, R. D. Electric impedance spectroscopy using microchannels with integrated metal electrodes. Journal of Microelectromechanical Systems 1999, 8, 50–57. [Google Scholar]
- Han, A.; Frazier, A.B. Ion channel characterization using single cell impedance spectroscopy. Lab on a Chip 2006, 6, 1412–1414. [Google Scholar]
- Cho, Y.H.; Yamamoto, T.; Sakai, Y.; Fujii, T.; Kim, B. Development of microfluidic device for electrical/physical characterization of single cell. Journal of Microelectromechanical Systems 2006, 15, 287–295. [Google Scholar]
- Diaz-Rivera, R.E.; Rubinsky, B. Electrical and thermal characterization of nanochannels between a cell and a silicon based micro-pore. Biomedical Microdevices 2006, 8, 25–34. [Google Scholar]
- Cho, S.; Thielecke, H. Micro hole-based cell chip with impedance spectroscopy. Biosensors and Bioelectronics 2007, 22, 1764–1768. [Google Scholar]
- Evans, E.A.; Waugh, R.; Melnik, L. Elastic area compressibility modulus of red cell membrane. Biophysical Journal 1976, 16, 585–595. [Google Scholar]
- Needham, D.; Hochmuth, R.M. A sensitive measure of surface stress in the resting neutrophil. Biophysical Journal 1992, 61, 1664–1670. [Google Scholar]
- Newman, J. Resistance for flow of current to a disk. Journal of Electrochemical Society 1966, 113, 501–502. [Google Scholar]
- Fang, Y. Non-invasive optical biosensor for probing cell. Sensors 2007, 7, 2316–2329. [Google Scholar]
- Tokuhiro, T.; Menafra, L.; Szmant, H. H. Contribution of relaxation and chemical shift results to the elucidation of the structure of the water-DMSO liquid system. Journal of Chemical Physics 1974, 61, 2275–2282. [Google Scholar]
- Vessey, D.A.; Cunningham, J. M.; Selden, A. C.; Woodman, A. C.; Hodgson, H. J. F. Dimethylsulphoxide induces a reduced growth rate, altered cell morphology and increased epidermal-growth-factor binding in Hep G2 cells. Biochemical Journal 1991, 277, 773–777. [Google Scholar]



© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Cho, S.; Castellarnau, M.; Samitier, J.; Thielecke, H. Dependence of Impedance of Embedded Single Cells on Cellular Behaviour. Sensors 2008, 8, 1198-1211. https://doi.org/10.3390/s8021198
Cho S, Castellarnau M, Samitier J, Thielecke H. Dependence of Impedance of Embedded Single Cells on Cellular Behaviour. Sensors. 2008; 8(2):1198-1211. https://doi.org/10.3390/s8021198
Chicago/Turabian StyleCho, Sungbo, Marc Castellarnau, Josep Samitier, and Hagen Thielecke. 2008. "Dependence of Impedance of Embedded Single Cells on Cellular Behaviour" Sensors 8, no. 2: 1198-1211. https://doi.org/10.3390/s8021198



