Review on Hydrogel-based pH Sensors and Microsensors
Abstract
:1. Introduction
2. Hydrogel Behaviour
2.1. Thermodynamics
a) Hydrogel behaviour in solvents
b) Behaviour of polyelectrolyte hydrogels
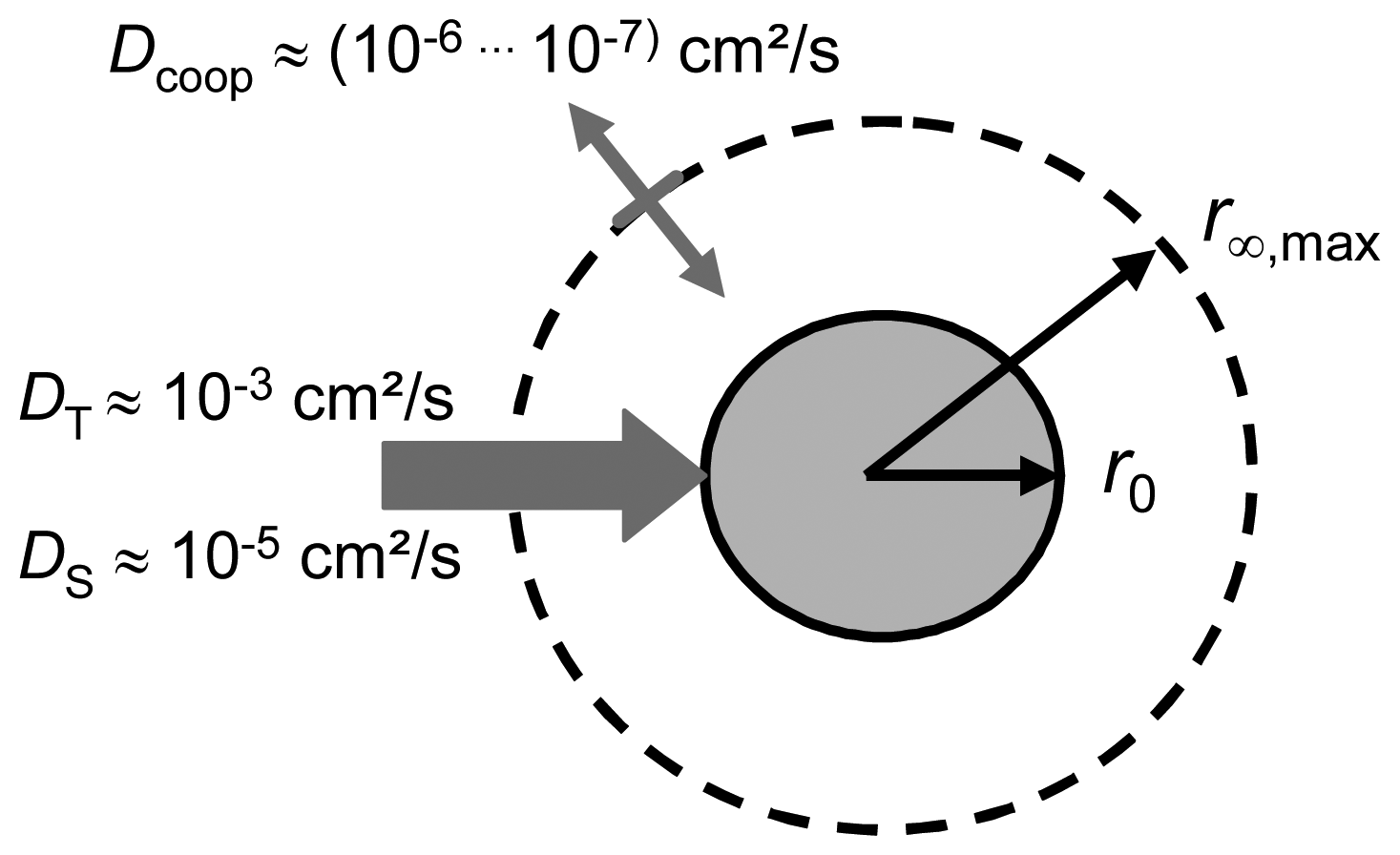
2.2. Swelling kinetics
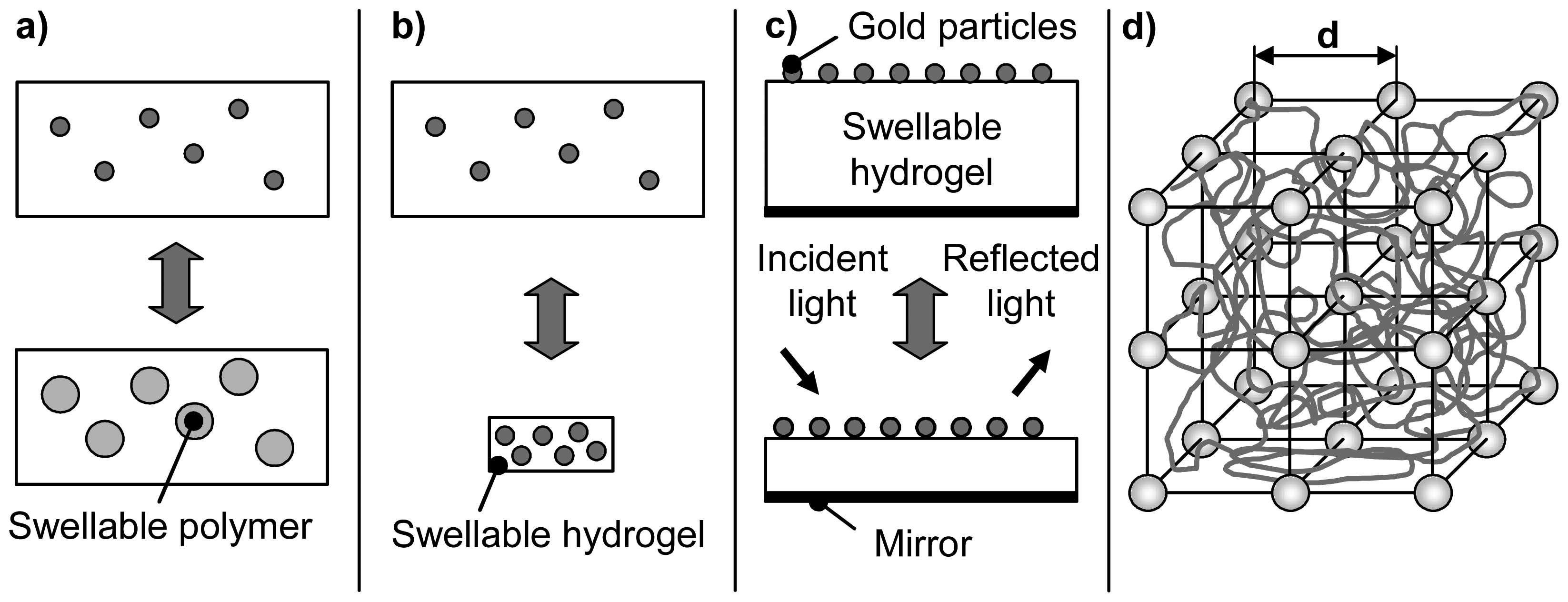
3. Sensor Transducers
- -
- transducers based on mechanical work performed by hydrogel swelling and shrinking, and
- -
- transducers observing changes in properties (e.g. densities, mass, volume, stiffness) of free swelling gels.
3.1. Transducers of free swelling gels
3.1.1 Optical transducers
a) Optical transmission
b) Refractive index
c) Reflection
d) Optical wavelength diffraction
e) Fluorescence intensity
3.1.2 Conductometric transducer
3.1.3 Oscillating transducers
a) Quartz crystal micro balance
b) Magnetoelastic sensors
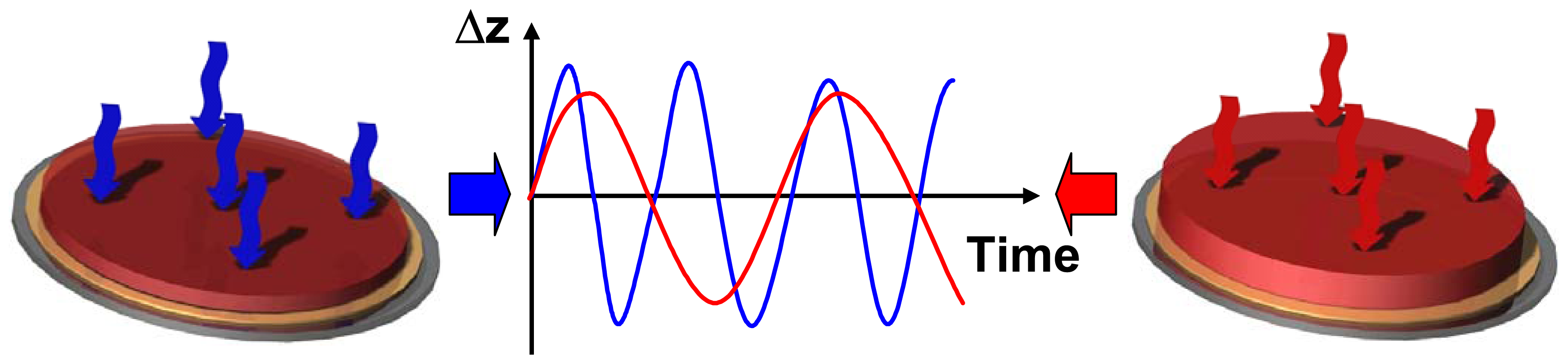
3.2. Transducers based on mechanical work of the hydrogel
3.2.1 Optical transducers
a) Reflective diaphragms
b) Fibre BRAGG grating sensors
3.2.2 Mechanical transducers
a) Microcantilevers
b) Bending plate transducers
4. Sensitive Hydrogel
5. Sensor Properties
5.1 Transducer resolution
5.2 Sensitivity and accuracy of measurement
a) Sensitivity
b) Accuracy of measurement
c) Working range
5.3 Response time
5.4 Calibration and offset correction
5.5 Long-term stability
6. Conclusions
Acknowledgments
References and Notes
- Kuhn, W. Reversible Dehnung und Kontraktion bei Änderung der Ionisation eines Netzwerks polyvalenter Fadenmolekülionen. Experientia 1949, 5(8), 318–319. [Google Scholar]
- Kuhn, W.; Hargitay, B.; Katchalsky, A.; Eisenberg, H. Reversible dilation and contraction by changing the state of ionization of high-polymer acid networks. Nature 1950, 165, 514–516. [Google Scholar]
- Katchalsky, A. Rapid swelling and deswelling of reversible gels of polymeric acids by ionization. Experientia 1949, 5(8), 319–320. [Google Scholar]
- Breitenbach, J.W.; Karlinger, H. Über Quellung von vernetzter Polymethacrylsäure. Monatsh. Chem. 1949, 80(2), 312–313. [Google Scholar]
- Tanaka, T. Collapse of gels and the critical endpoint. Phys. Rev. Lett. 1978, 40(12), 820–823. [Google Scholar]
- Tanaka, T.; Nishio, I.; Sun, S.-T.; Ueno-Nishio, S. Collapse of gels in an electric-field. Science 1982, 218, 467–469. [Google Scholar]
- Suzuki, A.; Tanaka, T. Phase-transition in polymer gels induced by visible-light. Nature 1990, 346, 345–347. [Google Scholar]
- Zhang, Y.; Ji, H.F.; Brown, G.M.; Thundat, T. Detection of CrO42- using a hydrogel swelling microcantilever sensor. Anal. Chem. 2003, 75(18), 4773–4777. [Google Scholar]
- Irie, M.; Misumi, Y.; Tanaka, T. Stimuli-responsive polymers: chemical induced reversible phase separation of an aqueous solution of poly(N-isopropylacrylamide) with pendent crown ether groups. Polymer 1993, 34(21), 4531–4535. [Google Scholar]
- Tanaka, T.; Wang, C.; Pande, V.; Grosberg, A.Y.; English, A.; Masamune, S.; Gold, H.; Levy, R.; King, K. Polymer gels that can recognize and recover molecules. Faraday Discuss. 1995, 101, 201–206. [Google Scholar]
- Li, W.; Zhao, H.; Teasdale, P.R.; John, R.; Zhang, S. Synthesis and characterisation of a polyacrylamide-polyacrylic acid copolymer hydrogel for environmental analysis of Cu and Cd. React. Funct. Polym. 2002, 52, 31–41. [Google Scholar]
- Hoffmann, J.; Plötner, M.; Kuckling, D.; Fischer, W.J. Photopatterning of thermally sensitive hydrogels useful for microactuators. Sens. Actuat. A 1999, 77(2), 139–144. [Google Scholar]
- Kuckling, D.; Adler, H.J.P.; Arndt, K.F.; Wolff, T.; Hoffman, J.; Fischer, W.J. Photocrosslinking of thin polymer films - Materials for sensors and actuators. Macromol. Symp. 1999, 142, 111–120. [Google Scholar]
- Kuckling, D.; Adler, H.J.P.; Arndt, K.F.; Hoffmann, J.; Plötner, M.; Wolff, T. Photocrosslinking of thin films of temperature-sensitive polymers. Polym. Adv. Technol. 1999, 10(6), 345–352. [Google Scholar]
- Beebe, D.J.; Moore, J.S.; Bauer, J.M.; Yu, Q.; Liu, R.H.; Devadoss, C.; Jo, B.H. Functional hydrogel structures for autonomous flow control inside microfluidic channels. Nature 2000, 404, 588–590. [Google Scholar]
- Arndt, K.-F.; Kuckling, D.; Richter, A. Application of sensitive hydrogels in flow control. Polym. Adv. Techn. 2000, 11(8-12), 496–505. [Google Scholar]
- Richter, A.; Kuckling, D.; Howitz, S.; Gehring, T.; Arndt, K.F. Electronically controllable microvalves based on smart hydrogels: magnitudes and potential applications. J. Microelectromech. Syst. 2003, 12(5), 748–753. [Google Scholar]
- Richter, A.; Howitz, S.; Kuckling, D.; Kretschmer, K.; Arndt, K.F. Automatically and electronically controllable hydrogel based valves and microvalves – design and operating performance. Macromol. Symp. 2004, 210, 447–456. [Google Scholar]
- Yu, C.; Mutlu, S.; Selvaganapathy, P.; Mastrangelo, C.H.; Svec, F.; Fréchet, J.M.J. Flow control valves for analytical microfluidic chips without mechanical parts based on thermally responsive monolithic polymers. Anal. Chem. 2003, 75(8), 1958–1961. [Google Scholar]
- Wang, J.; Chen, Z.; Mauk, M.; Hong, K.S.; Li, M.; Yang, S.; Bau1, H.H. Self-actuated, thermo-responsive hydrogel valves for lab on a chip. Biomed. Microdev. 2005, 7(4), 313–322. [Google Scholar]
- Richter, A.; Arndt, K.F. Design and properties of a chemomechanical valve. In 5th Internat. Symp. on Polym. Adv. Technol.; Tokyo, 1999; pp. 31.8.–5.9.1999.156. [Google Scholar]
- Baldi, A.; Gu, Y.; Loftness, P.E.; Siegel, R.A.; Ziaie, B. A hydrogel-actuated environmentally sensitive microvalve for active flow control. J. Microelectromech. Syst. 2003, 12(5), 613–621. [Google Scholar]
- Kuckling, D.; Arndt, K.F.; Richter, A. Temperature and pH dependent swelling behavior of poly(N-isopropylacrylamide)-copolymer and their use in flow control. Macromol. Mater. Eng. 2003, 288, 144–151. [Google Scholar]
- Harmon, M.E.; Tang, M.; Frank, C.W. A microfluidic actuator based on thermoresponsive hydrogels. Polymer 2003, 44, 4547–4556. [Google Scholar]
- Suzuki, H.; Tokuda, T.; Kobayashi, K. A disposable “intelligent mosquito” with a reversible sampling mechanism using the volume-phase transition of a gel. Sens. Actuat. B 2002, 83(1-3), 53–59. [Google Scholar]
- Eddington, D.T.; Beebe, D.J. A valved responsive hydrogel microdispensing device with integrated pressure source. J. Microelectromech. Syst. 2004, 13(4), 586–593. [Google Scholar]
- Richter, A.; Klenke, C.; Arndt, K.F. Adjustable low dynamic pumps based on hydrogels. Macromol. Symp. 2004, 210, 377–384. [Google Scholar]
- Richter, A.; Türke, A.; Pich, A. Controlled double-sensitivity of microgels applied to electronically adjustable chemostats. Adv. Mater. 2007, 19, 1109–1112. [Google Scholar]
- Richter, A.; Wenzel, J.; Kretschmer, K. Mechanically adjustable chemostats based on stimuli-responsive polymers. Sens. Actuat. B 2007, 125, 569–573. [Google Scholar]
- Hydrogel Valves – Dead-Volume-Free Microfluidic Switches. Data sheet 2007. http://www.gesim.de/upload/PDFs/Hydrogel_en.pdf downloaded at 10/22/2007.
- van der Linden, H.J.; Herber, S.; Olthuis, W.; Bergveld, P. Stimulus-sensitive hydrogels and their applications in chemical (micro)analysis. Analyst 2003, 128, 325–331. [Google Scholar]
- Arndt, K.F.; Schmidt, T.; Richter, A.; Kuckling, D. High response smart gels: synthesis and application. Macromol. Symp. 2004, 207, 257–268. [Google Scholar]
- Flory, P.J.; Rehner, J. Statistical mechanics of cross-linked polymer networks I. Rubber elasticity. J. Chem. Phys. 1943, 11, 512–520. [Google Scholar]
- Flory, P.J.; Rehner, J. Statistical mechanics of cross-linked polymer networks II. Swelling. J. Chem. Phys. 1943, 11, 521–526. [Google Scholar]
- Huggins, M.L. Solutions of long chain compounds. J. Chem. Phys. 1941, 9(5), 440. [Google Scholar]
- Flory, P.J. Thermodynamics of high polymer solutions. J. Chem. Phys. 1941, 9(8), 660–661. [Google Scholar]
- Siegel, R.A.; Firestone, B.A. pH-dependent equilibrium swelling properties of hydrophobic polyelectrolyte copolymer gels. Macromolecules 1988, 21(11), 3254–3259. [Google Scholar]
- English, A.E.; Tanaka, T.; Edelman, E.R. Equilibrium and non-equilibrium phase transitions in copolymer polyelectrolyte hydrogels. J. Chem. Phys. 1997, 107(5), 1645–1654. [Google Scholar]
- English, A.E.; Edelman, E.R.; Tanaka, T. Polymer hydrogel phase transitions. In Experimental Methods in Polymer Science; Tanaka, T., Ed.; Academic Press Cambridge Burlington: London, 2000; p. 547. [Google Scholar]
- Tanaka, T.; Hocker, L.; Benedek, G.B. Spectrum of light scattered from a viscoelastic gel. J. Chem. Phys. 1973, 59, 5151–5159. [Google Scholar]
- Tanaka, T.; Fillmore, D.J. Kinetics of swelling of gels. J. Chem. Phys. 1979, 70(3), 1214–1218. [Google Scholar]
- Li, Y.; Tanaka, T. Kinetics of swelling and shrinking of gels. J. Chem. Phys. 1990, 92(2), 1365–1371. [Google Scholar]
- Lesho, M.J.; Sheppard, N.F. A method for studying swelling kinetics based on measurement of electrical conductivity. Polym. Gels Networks 1997, 5, 503–523. [Google Scholar]
- Shakhsher, Z.M.; Odeh, I.; Jabr, S.; Seitz, W.R. An optical chemical sensor based on swellable dicarboxylate functionalized polymer microspheres for pH copper and calcium determination. Microchim. Acta 2004, 144, 147–153. [Google Scholar]
- Oktar, O.; Caglar, P.; Seitz, W.R. Chemical modulation of thermosensitive poly(N-isopropylacryl-amide) microsphere swelling: a new strategy for chemical sensing. Sens. Actuat. B 2005, 104, 179–185. [Google Scholar]
- Dübendorfer, J.; Kunz, R.E.; Jobst, G.; Moser, I.; Urban, G. Integrated optical pH sensor using replicated chirped grating coupler sensor chips. Sens. Actuat. B 1998, 50(3), 210–219. [Google Scholar]
- Rooney, M.T.V.; Seitz, W.R. An optically sensitive membrane for pH based on swellable polymer microspheres in a hydrogel. Anal. Comm. 1999, 36, 267–270. [Google Scholar]
- Seitz, W.R.; Rooney, M.T.V.; Miele, E.W.; Wang, H.; Kaval, N.; Zhang, L.; Doherty, S.; Milde, S.; Lenda, J. Derivatized, swellable polymer microspheres for chemical transduction. Anal. Chim. Acta 1999, 400, 55–64. [Google Scholar]
- Shakhsher, Z.; Seitz, W.R.; Legg, K.D. Single fiber-optic pH sensor based on changes in reflection accompanying polymer swelling. Anal. Chem. 1994, 66, 1731–1735. [Google Scholar]
- Zhang, L.; Langmuir, M.E.; Bai, M.; Seitz, W.R. A sensor for pH based on an optical reflective device coupled to the swelling of an aminated polystyrene membrane. Talanta 1997, 44, 1691–1698. [Google Scholar]
- Zhang, Z.; Shakhser, Z.; Seitz, W.R. Aminated polystyrene membranes for a fiber optic pH sensor based on reflectance changes accompanying polymer swelling. Microchim. Acta 1995, 121, 41–50. [Google Scholar]
- Schalkhammer, T.; Lobmaier, C.; Pittner, F.; Aussenegg, A.F.R. The use of metal-island-coated pH-sensitive swelling polymers for biosensor applications. Sens. Actuat B 1995, 24-25, 166–172. [Google Scholar]
- Aussenegg, F.R.; Brunner, H.; Leitner, A.; Lobmaier, C.; Schalkhammer, T. Metal island coated polymer swelling over mirror system (MICSPOMS): a new principle for measure ionic strength. Sens. Actuat. B 1995, 29, 204–209. [Google Scholar]
- Holtz, J.H.; Asher, S.A. Polymerized colloidal crystal hydrogel films as intelligent chemical sensing materials. Nature 1997, 389, 829–832. [Google Scholar]
- Lee, K.; Asher, A.S. Photonic crystal chemical sensors: pH and ionic strength. J. Am. Chem. Soc. 2000, 122, 9534–9537. [Google Scholar]
- Yan, F.; Asher, S.A. Cation identity dependence of crown ether photonic crystal Pb2+ sensing. Anal. Bioanal. Chem. 2007, 387, 2121–2130. [Google Scholar]
- Ben-Moshe, M.; Alexeev, V.L.; Asher, S.A. Fast responsive crystalline colloidal array photonic crystal glucose sensors. Anal. Chem. 2006, 78, 5149–5157. [Google Scholar]
- Marshall, A.J.; Blyth, J.; Davidson, C.A.B.; Lowe, C.R. PH-sensitive holographic sensors. Anal. Chem. 2003, 75(17), 4423–4431. [Google Scholar]
- Sartain, F.K.; Yang, X.; Lowe, C.R. Holographic lactate sensor. Anal. Chem. 2006, 78, 5664–5670. [Google Scholar]
- McCurley, M.F. An optical biosensor using a fluorescent, swelling sensing element. Biosens. Bioelectronics 1994, 9, 527–533. [Google Scholar]
- Saunders, B.R.; Vincent, B. Osmotic de-swelling of polystyrene microgel particles. Colloid Polym. Sci. 1997, 275, 9–17. [Google Scholar]
- Sheppard, N.F.; Tucker, R.C.; Salehi-Had, S. Design of a conductimetric pH microsensor swelling hydrogels. Sens. Actuat. B 1993, 10, 73–77. [Google Scholar]
- Sheppard, N.F.; Lesho, M.J.; McNally, P.; Francomacaro, S. Microfabricated conductimetric pH sensor. Sens. Actuat. B 1995, 28, 95–102. [Google Scholar]
- Richter, A.; Bund, A.; Keller, M.; Arndt, K.F. Characterization of a microgravimetric sensor based on pH sensitive hydrogels. Sens. Actuat. B 2004, 99(2-3), 579–585. [Google Scholar]
- Stojanov, P.G.; Doherty, S.A.; Grimes, C.A.; Seitz, W.R. A remotely interogatable sensor for chemical monitoring. IEEE Transactions on Magnetics 1998, 34(4), 1315–1317. [Google Scholar]
- Grimes, C.A.; Stojanov, P.G.; Liu, Y.; Tong, C.; Ong, K.G.; Loiselle, K.; Shaw, M.; Doherty, S.A; Seitz, W.R. A magnetostatic-coupling basedremote query sensor for environmental monitoring. J. Phys. D 1999, 32, 1329–1335. [Google Scholar]
- Ruana, C.; Zenga, K.; Grimes, C.A. A mass-sensitive pH sensor based on a stimuli-responsive polymer. Anal. Chimica Acta 2003, 497, 123–131. [Google Scholar]
- Seitz, W.R. New directions in fiber optic chemical sensors: sensors based on polymer swelling. J. Mol. Structure 1993, 292, 105–114. [Google Scholar]
- McCurley, M.F.; Seitz, W.R. Swelling of a polymer membrane for use in a glucose biosensor. 1992, 487, 301–309. [Google Scholar]
- Cong, J.; Zhang, X.; Chen, K.; Xu, J. Fiber optic Bragg grating sensor based on hydrogels for measuring salinity. Sens. Actuat. B 2002, 87, 487–490. [Google Scholar]
- Liu, X.; Zhang, X.; Cong, J.; Xu, J.; Chen, K. Demonstration of etched cladding fiber Bragg grating-based sensors with hydrogel coating. Sens. Actuat. B 2003, 96, 468–472. [Google Scholar]
- Carrascosa, L.G.; Moreno, M.; Àlvarez, M.; Lechuga, L.M. Nanomechanical biosensors: a new sensing tool. Trends in Analytical Chemistry 2006, 25(3), 196–206. [Google Scholar]
- Bashir, R.; Hilt, J.Z.; Elibol, O.; Gupta, A.M.; Peppas, N.A. Micromechanical cantilever as an ultrasensitive pH microsensor. Appl. Phys. Lett. 2002, 81, 3091–3093. [Google Scholar]
- Hilt, J.Z; Gupta, A.M.; Bashir, R.; Peppas, N.A. Ultrasensitive biomems sensors based on microcantilevers patterned with environmentally responsive hydrogels. Biomed. Microdev. 2003, 5(3), 177–184. [Google Scholar]
- Zhang, Y.; Ji, H.F.; Snow, D.; Sterling, R.; Brown, G.M. A pH sensor based on a microcantilever coated with intelligent hydrogel. Instrumentation Sci. Technol. 2004, 32(4), 361–369. [Google Scholar]
- Han, I.S.; Han, M.H.; Kim, J.; Lew, S.; Lee, Y.J.; Horkay, F.; Magda, J.J. Constant-volume hydrogel osmometer: a new device concept for miniature biosensors. Biomacromol. 2002, 3, 1271–1275. [Google Scholar]
- Gerlach, G.; Günther, M.; Suchaneck, G.; Sorber, J.; Arndt, K.F.; Richter, A. Application of sensitive hydrogels in chemical and pH sensors. Macromol. Symp. 2004, 210, 403–410. [Google Scholar]
- Gerlach, G.; Guenther, M.; Sorber, J.; Suchaneck, G.; Arndt, K.-F.; Richter, A. Chemical and pH sensors based on the swelling behavior of hydrogels. Sens. Actuat. B 2005, 111-112, 555–561. [Google Scholar]
- Trinh, Q.T.; Gerlach, G.; Sorber, J.; Arndt, K.-F. Hydrogel-based piezoresistive pH sensors: design, simulation and output characteristics. Sens. Actuat. B 2006, 117, 17–26. [Google Scholar]
- Herber, S.; Olthuis, W.; Bergveld, P. A swelling hydrogel-based PCO2 sensor. Sens. Actuat. B 2003, 91, 378–382. [Google Scholar]
- Herber, S.; Eijkel, J.; Olthuis, W.; Bergveld, P.; van den Berg, A. Study of chemically induced pressure generation of hydrogels under isochoric conditions using a microfabricated device. J Chem. Phys. 2004, 121(6), 2746–2751. [Google Scholar]
- Strong, Z.A.; Wang, A.W.; Conaghy, C.F. Hydrogel-actuated capacitive transducer for wireless biosensors. Biomed. Microdev. 2002, 4(2), 97–103. [Google Scholar]
- Arndt, K.F.; Richter, A.; Ludwig, S.; Zimmermann, J.; Kressler, J.; Kuckling, D.; Adler, H.J. Poly(vinyl alcohol)/(poly(acrylic acid) hydrogels: FT-IR spectroscopic characterization of crosslinking reaction and work at transition point. Acta Polym. 1999, 50, 383–390. [Google Scholar]
- Kretschmer, K.; Kuckling, D.; Richter, A. SPR investigations of pH dependent swelling of thin poly(vinyl alcohol)/poly(acrylic acid) layers. unpublished.
- Suzuki, A.; Suzuki, H. Hysteretic behavior and irreversibility of polymer gels by pH change. J. Chem. Phys. 1995, 103(11), 4706–4710. [Google Scholar]
- Harmon, M.E.; Kuckling, D.; Frank, C.W. Photo-cross-linkable PNIPAAm copolymers. 2. Effects of constraint on temperature and pH-responsive hydrogel layers. Macromolecules 2003, 36, 162–172. [Google Scholar]
- Harmon, M.E.; Kuckling, D.; Pareek, P.; Frank, C.W. Photo-cross-linkable PNIPAAm copolymers. 4. Effects of copolymerization and cross-linking on the volume-phase transition in constrained hydrogel layers. Langmuir 2003, 19, 10947–10956. [Google Scholar]
- Richter, A.; Howitz, S.; Kuckling, D.; Arndt, K.F. Influence of phenomena of volume phase transition at the behavior of hydrogel based valves. Sens. Actuat. B 2004, 99(2-3), 451–458. [Google Scholar]
- Kuckling, D.; Hoffmann, J.; Plötner, M.; Ferse, D.; Kretschmer, K.; Adler, H.J.P.; Arndt, K.F.; Reichelt, R. Photo cross-linkable poly(N-isopropylacrylamide) copolymers III: micro-fabricated temperature responsive hydrogels. Polymer 2003, 44(16), 4455–4462. [Google Scholar]
- Bhattacharya, S.; Eckert, F.; Boyko, V.; Pich, A. Temperature-, pH-, and magnetic-field-sensitive hybrid microgels. Small 2007, 4, 650–657. [Google Scholar]

 ) are ionised by deprotonation in basic solutions, which have an excess of hydroxyl groups. Basic hydrogels (
) are ionised by deprotonation in basic solutions, which have an excess of hydroxyl groups. Basic hydrogels (
 ) swell in acidic solutions due to the ionisation of their basic groups by protonation. Amphiphilic hydrogels (
) swell in acidic solutions due to the ionisation of their basic groups by protonation. Amphiphilic hydrogels (
 ) contain both acidic and basic groups. Therefore they show two phase transitions.
) contain both acidic and basic groups. Therefore they show two phase transitions.
 ) are ionised by deprotonation in basic solutions, which have an excess of hydroxyl groups. Basic hydrogels (
) are ionised by deprotonation in basic solutions, which have an excess of hydroxyl groups. Basic hydrogels (
 ) swell in acidic solutions due to the ionisation of their basic groups by protonation. Amphiphilic hydrogels (
) swell in acidic solutions due to the ionisation of their basic groups by protonation. Amphiphilic hydrogels (
 ) contain both acidic and basic groups. Therefore they show two phase transitions.
) contain both acidic and basic groups. Therefore they show two phase transitions.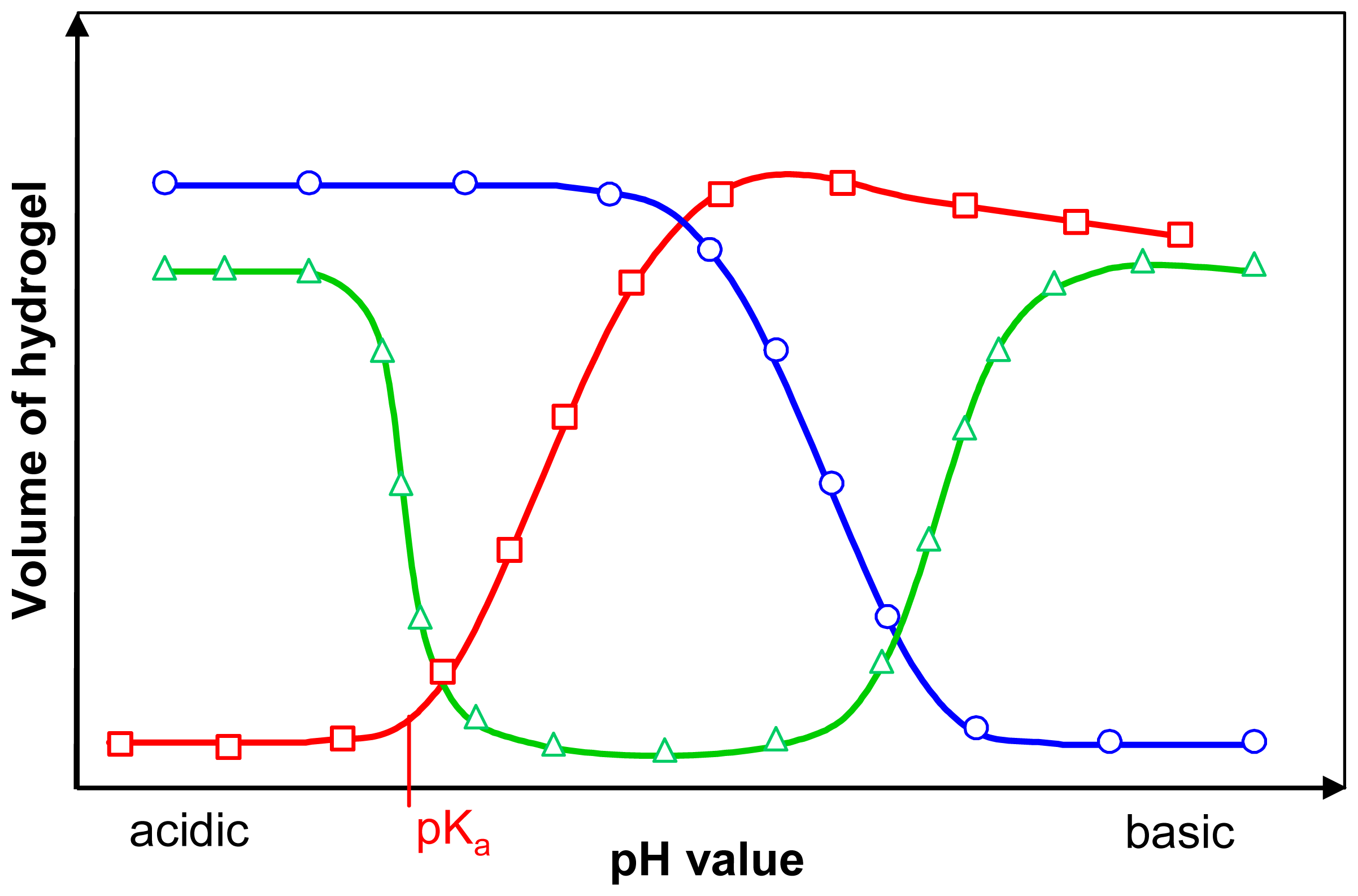

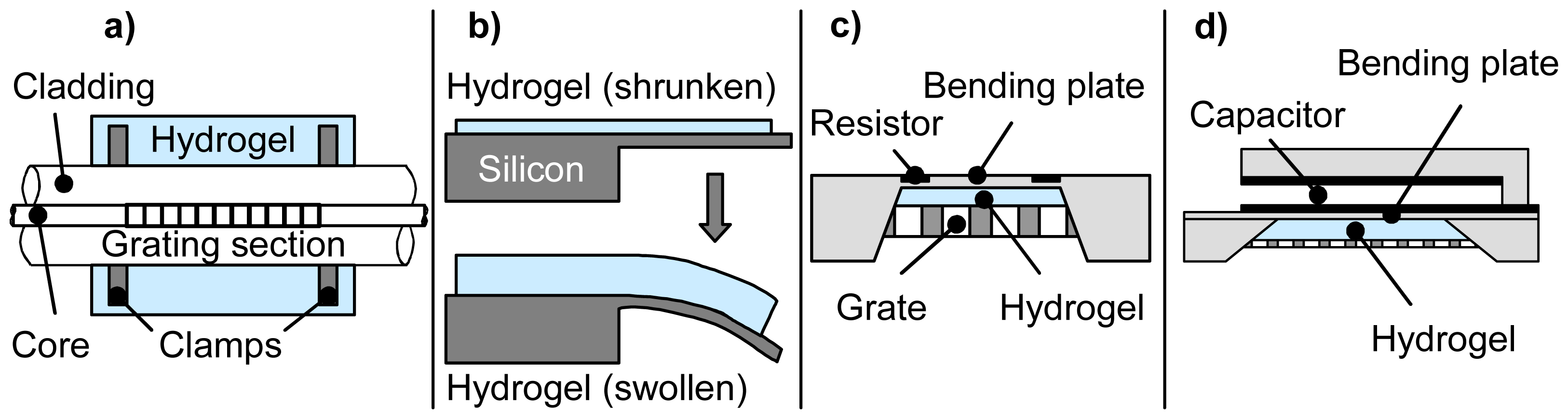
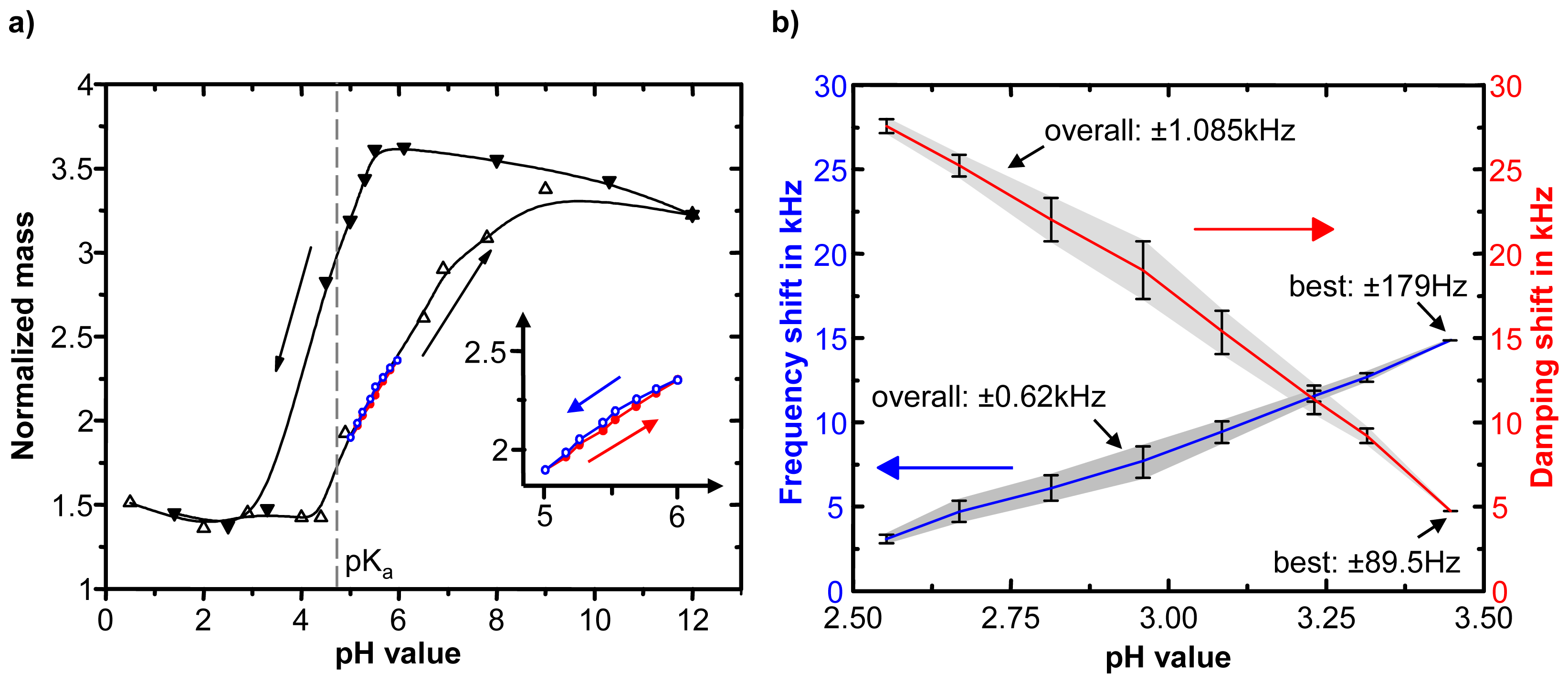
| Sensor type | Data channel | Transducer resolution | Sensitivity per pH unit | Accuracy of measurement | pH working range | Hydrogel | Minimal response time | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| overall | best point | ||||||||||
| Transducer signal | pH unit | Transducer signal | pH unit | Material | Thickness | ||||||
| Refractometric sensor [46] | refractive index | 2.5x10−4 10−5/4x10−2 | 3.11x10−2 | ± 3.75x10- * | ± 1.64x10-3 * | ± 1.1x10-4 | ± 4.8x10-4 | 7-7.75 | HEMA-DMAEMA | 300 nm (dry) | ∼ 80 s |
| Holographic sensor [58] | wavelength | 6x10−3 (1nm/165nm) | 165 nm | ± 5.3 nm * | ± 0.032 * | ± 3.1 nm * | ± 0.019 * | 5 - 7 | PHEMA-co-MAA | 10 μm | ∼ 250 s |
| CCA sensor[55] (Braggdiffraction) | wavelength | 3,3x10−3 (1nm/300nm) | 74.2 nm | - | - | - | - | 4.3-8.5 | PAAm-PCCA | 125 μm | 10 min |
| Conductimetric sensor [63] | resistance | - | 100 Ω | - | - | ± 5.2Ω | ± 0.052 | 7 - 8 | HEMA-DMAEMA | 8 μm | 350 s |
| Quartz crystalmicro balance[64] | frequency damping | 3.3x10−6 (0.1Hz/30kHz) | 13.2 kHz 25.467 kHz | ± 0.62 kHz ± 1.085 kHz | ± 0.047 ± 0.042 | ± 179 Hz ± 89.5 Hz | ± 0.013 ± 3.5x10-3 | 2.55 – 3.45 | PVA-PAA | 390 nm (dry) | 500 ms |
| Magnetoelastic sensor [67] | frequency | 2,4x10−4 0.4Hz/1.7kHz | 506 Hz | ± 66 Hz | ± 0.13 | - | - | 4,4 – 8,5 | poly(AA-co-IOA) | 1.4 μm | 120 s |
| Microcantilever [74] | deflexion | 5x105 (1nm/20μm) | 20.3 μm | ± 1.15 μm * | ± 0.057 * | ± 0.3 μm * | ± 0.015 * | 6 - 6,8 | PMAA-PEGDMA | 2.2 μm | few minutes |
| Microcantilever [75] | deflexion | 2,9x105 (1nm/35μm) | 1000 nm | ± 5 % | ± 0.05 | - | - | 6 - 9 | AAm-DMAEMA | 15 μm | 15 min |
| Bending plate sensor [79] | resistance | 1,67x10-5 (1μV/60mV) | 14.55 mV * | ± 3.5 mV * | ± 0.24 * | ± 1.9 mV * | ± 0.13 * | 5.5 - 11 | PVA-PAA | 40 μm | ∼ 12 min |
© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Richter, A.; Paschew, G.; Klatt, S.; Lienig, J.; Arndt, K.-F.; Adler, H.-J.P. Review on Hydrogel-based pH Sensors and Microsensors. Sensors 2008, 8, 561-581. https://doi.org/10.3390/s8010561
Richter A, Paschew G, Klatt S, Lienig J, Arndt K-F, Adler H-JP. Review on Hydrogel-based pH Sensors and Microsensors. Sensors. 2008; 8(1):561-581. https://doi.org/10.3390/s8010561
Chicago/Turabian StyleRichter, Andreas, Georgi Paschew, Stephan Klatt, Jens Lienig, Karl-Friedrich Arndt, and Hans-Jürgen P. Adler. 2008. "Review on Hydrogel-based pH Sensors and Microsensors" Sensors 8, no. 1: 561-581. https://doi.org/10.3390/s8010561
APA StyleRichter, A., Paschew, G., Klatt, S., Lienig, J., Arndt, K.-F., & Adler, H.-J. P. (2008). Review on Hydrogel-based pH Sensors and Microsensors. Sensors, 8(1), 561-581. https://doi.org/10.3390/s8010561




