Electrochemical Sensors Based on Organic Conjugated Polymers
Abstract
:1. Introduction
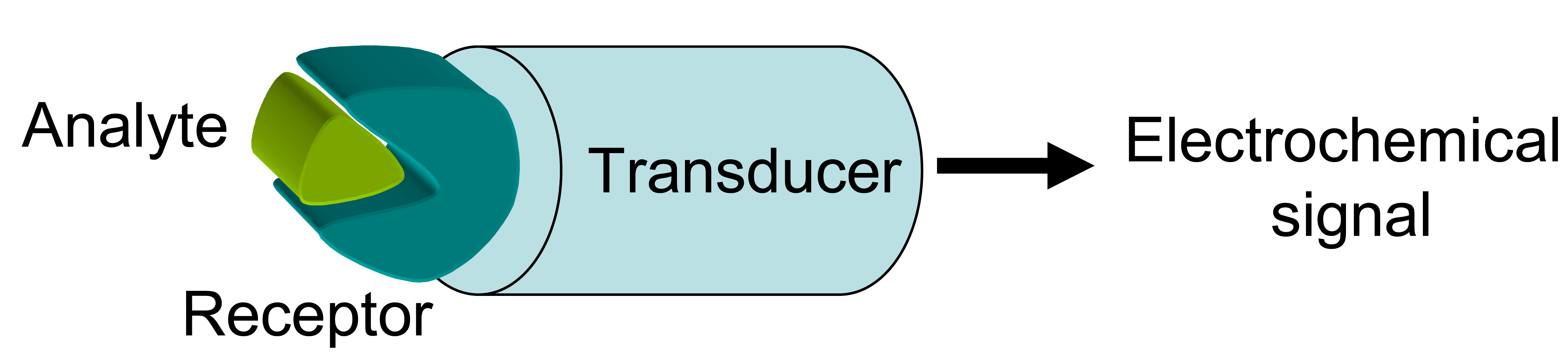
2. Electrochemical Sensors
2.1. Classification of electrochemical techniques
2.2. Conducting polymer-based electrochemical sensors
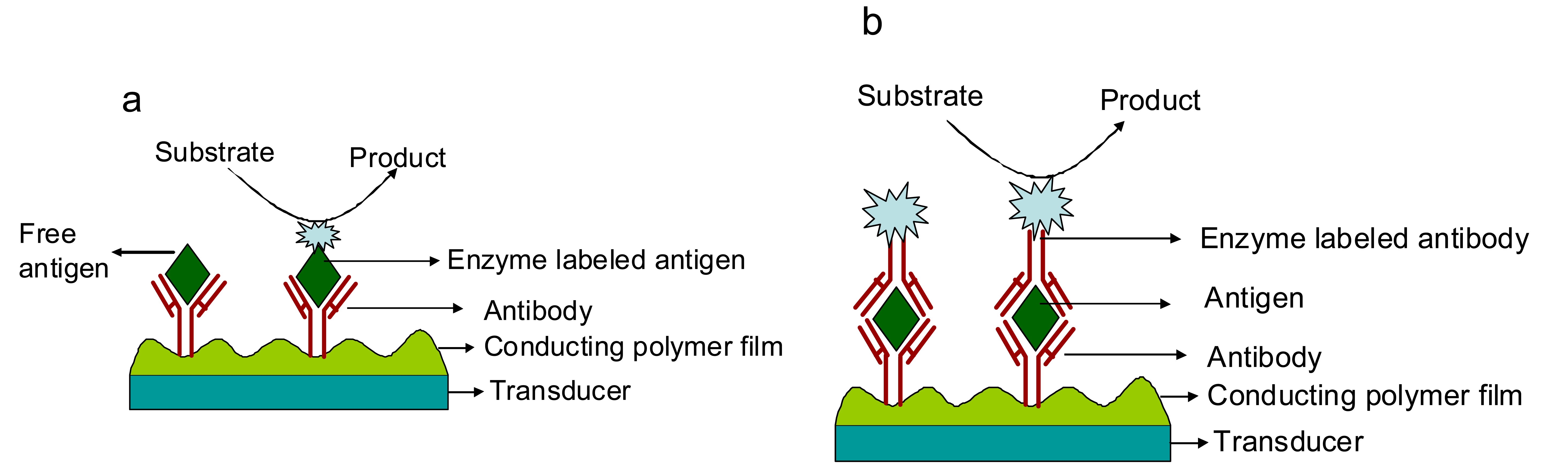
2.2. Conducting polymer-based electrochemical biosensors
3. Conclusions
Acknowledgments
References
- Park, S.-M. Electrochemistry of π-Conjugated Polymers. In Handbook of Organic Conductive Molecules and Polymers; Nalwa, H. S., Ed.; Chichester, Wiley, 1997; Vol. 3, pp. 429–469. [Google Scholar]
- Guiseppi-Elie, A.; Wallace, G. G.; Matsue, T. Handbook of Conducting Polymers, 2nd ed.; Skotheim, T. A., Elsenbaumer, R., Reynolds, J. R., Eds.; Marcel Dekker: New York, 1998; pp. 963–991. [Google Scholar]
- Arbizzani, C.; Mastragostino, M.; Scrosati, B. In: Handbook of Organic Conductive Molecules and Polymers; Nalwa, H. S., Ed.; Chichester, Wiley, 1997; Vol. 4, Chapter 11; pp. 595–619. [Google Scholar]
- Shim, Y.-B.; Won, M.-S.; Park, S.-M. Electrochemistry of conductive polymers VIII, In situ spectroelectrochemical studies of polyaniline growth mechanisms. J. Electrochem. Soc. 1990, 137, 538–544. [Google Scholar]
- Shim, Y.-B.; Park, S.-M. Electrochemistry of conductive polymers VII, Autocatalytic rate constant for polyaniline growth. Synth. Met. 1989, 29, E169–E174. [Google Scholar]
- Park, D.-S.; Shim, Y.-B.; Park, S.-M. Degradation kinetics of polypyrrole films. J. Elrctrochem. Soc. 1993, 140, 2749–2752. [Google Scholar]
- Park, D.-S.; Shim, Y.-B.; Park, S.-M. Degradation of electrochemically prepared polypyrrole in aqueous sulfuric acid. J. Elrctrochem. Soc. 1993, 140, 609–614. [Google Scholar]
- Shim, Y.-B.; Park, S.-M. Electrochemistry of conductive polymers XXII, Electrochemical and spectroelectrochemical studies of polyazulene growth and its characterization. J. Electrochem. Soc. 1997, 144, 3027–3033. [Google Scholar]
- Blanchard, P.; Leriche, P.; Frere, P.; Roncali, J. Advanced functional polythiophene based on tailored precursors. In Handbook of Conducting PolymersConjugated polymers, 3rd ed.; Skotheim, T. A., Reynolds, J. R., Eds.; CRC press: Bocaraton, Florida, 2006; Chapter 13; pp. 1–77. [Google Scholar]
- Jeffries-El, M.; Mccullough, D. Regioregular polythiophene, J. In Handbook of Conducting PolymersConjugated polymers, 3rd ed.; Skotheim, T. A., Reynolds, J. R., Eds.; CRC press: Bocaraton, Florida, 2006; Chapter 9; pp. 1–49. [Google Scholar]
- Lee, J.-W.; Park, D.-S.; Shim, Y.-B.; Park, S.-M. Electrochemical characterization of poly (1,8-diaminonaphthalene): a functionalized polymer. J. Electrochem. Soc. 1992, 139, 3507–3514. [Google Scholar]
- Paul, E.W.; Ricco, A.J.; Wrighton, M.S. Resistance of polyaniline films as a function of electrochemical potential and the fabrication of polyaniline-based microelectronic devices. J. Phys. Chem. 1985, 89, 1441–1447. [Google Scholar]
- Kim, D. H.; Kang, B. S.; lim, S. M.; Bark, K.-M.; Kim, B. G.; Shiro, M.; Shim, Y.-B.; Shin, S. C. Polyterthiophene-bearing pendant organomolybdenum complexes: electropolymerization of erythro-[Mo2(μ-C5H5)2(CO)4{μ-η2:η2-C(R)≡C[C4HS(C4H3S-2)2-2,5] } ]. J. Chem. Soc. Dalton Trans. 1998, 1893–1898. [Google Scholar]
- Jung, T.S.; Kim, J. H.; Jang, E. K.; Kim, D. H.; Shim, Y.-B.; Park, B.; Shin, S. C. Synthesis and physical properties of α, ω-bis [Co2(CO)6{μ-η2 :η2 -C(R) ≡C}[ oligothiophenes. J. Organomet. Chem. 2000, 599, 232–237. [Google Scholar]
- Kang, B.S.; Kim, D.H.; Jung, T. S.; Jang, E. K.; Pak, Y.; Shin, S. C.; Park, D. S.; Shim, Y.-B. Polytherthiophene appended by transition-metal cluster: electropolymerization of 3′- [CCo3(CO)9]-5,2′;5′,2″-terthiophene. Synth. Met. 1999, 105, 9–12. [Google Scholar]
- Kim, D.H.; Park, D. S.; Shim, Y.-B.; Shin, S. C. Polytherthiophene p-conjugated by organomolybdenum complex (II): electropolymerization of erythro-[ (μ5-C5H5)2 Mo2-(O)2(μ -O) {μ-η2:η2-C(Ph)≡C[C4HS(C4H3S-2)2-2,5] } ]. J. Organo. Met. 2000, 608, 133–138. [Google Scholar]
- Chaubey, A.; Malhotra, B. D. Mediated biosensors. Biosens. Bioelectron. 2002, 17, 441–456. [Google Scholar]
- Mulchandani, A.K.; Wang, C. L. Bienzyme sensors based on poly(anilinomethylferrocene)-modified electrodes. Electroanalysis 1996, 8, 414–419. [Google Scholar]
- Bartlett, P.N.; Whitaker, R. G. Strategies for the development of amperometric enzyme electrodes. Biosensors 1988, 3, 359–379. [Google Scholar]
- Cartwell, R. W. Chemical sensors; Oxford University Press, 1997. [Google Scholar]
- Shim, Y.-B.; Park, J.-H. Humidity sensor using chemically synthesized poly (1,5-diaminonaphthalene) doped with carbon. J. Eletrcochem. Soc. 2000, 147, 381–385. [Google Scholar]
- Boopathi, M.; Won, M.-S.; Kim, Y. H.; Shin, S. C.; Shim, Y.-B. Eletcrocatalytic reduction of molecular oxygen using a polyterthiophene carboxylic acid) appended by 1,5-diamiononaphthalene copper complex. J. Electrochem. Soc. 2002, 149, E265–E271. [Google Scholar]
- Lindfors, T.; Ivaska, A. Calcium-selective electrode based on polyaniline functionalized with bis[4-(1,1,3,3-tetramethylbutyl)phenyl]phosphate. Anal. Chim. Acta 2001, 437, 171–182. [Google Scholar]
- Zachara, J.E.; Owska, R. T.; Pokrop, R.; Zagorska, M.; Dybko, A.; Wroblewski, W. Miniaturised all-solid-state potentiometric ion sensors based on PVC-membranes containing conducting polymers. Sens. Actuats. B 2004, 101, 207–212. [Google Scholar]
- Kaden, H.; Jahn, H.; Berthold, M. Study of the glass/polypyrrole interface in an all-solid-state pH sensor. Solid State Ionics 2004, 169, 129–133. [Google Scholar]
- Faria, R.C.; Bulhões, L. O. S. Hydrogen ion selective electrode based on poly(1-aminoanthracene) film. Anal. Chim. Acta 1998, 377, 21–27. [Google Scholar]
- Mousavi, Z.; Bobacka, J.; Lewenstam, A.; Ivaska, A. Response mechanism of potentiometric Ag+ sensor based on poly(3,4-ethylenedioxythiophene) doped with silver hexabromocarborane. J. Electroanal. Chem. 2006, 593, 219–226. [Google Scholar]
- Bobacka, J.; Ivaska, A.; Lewenstam, A. Plasticizer-free all-solid-state potassium-selective electrode based on poly (3-octylthiophene) and valinomycin. Anal. Chim. Acta 1999, 385, 195–202. [Google Scholar]
- Si, P.; Chi, O.; Li, Z.; Ulstrup, J.; Muller, P.J.; Mortensen, J. Functional Polythiophene Nanoparticles: Size-Controlled Electropolymerization and Ion Selective Response. J. Am. Chem. Soc. 2007, 129, 3888–3896. [Google Scholar]
- Zanganeh, A.R.; Amini, M. K. A potentiometric and voltammetric sensor based on polypyrrole film with electrochemically induced recognition sites for detection of silver ion. Electrochim. Acta 2007, 52, 3822–3830. [Google Scholar]
- Ocypa, M.; Michalska, A.; Maksymiuk, K. Accumulation of Cu(II) cations in poly(3,4-ethylenedioxythiophene) films doped by hexacyanoferrate anions and its application in Cu2+-selective electrodes with PVC based membranes. Electrochim. Acta 2006, 51, 2298–2305. [Google Scholar]
- Zejli, H.; Sharrock, P.; Hidalgo-Hidalgo de Cisneros, J.L.; Naranjo-Rodriguez, I.; Temsamani, K.R. Voltammetric determination of trace mercury at a sonogel–carbon electrode modified with poly-3-methylthiophene. Talanta 2006, 68, 79–85. [Google Scholar]
- Seol, H.; Shin, S.C.; Shim, Y. B. Trace analysis of Al (III) ions based on the redox current of a conducting polymer. Electroanalysis 2004, 16, 2051–2057. [Google Scholar]
- Park, D.-S.; Shim, Y.-B.; Park, S.-M. Characterization of electrochemically prepared polyaminopyridines. Electroanalysis 1996, 8, 44–49. [Google Scholar]
- Rahman, M.A.; Won, M.-S.; Shim, Y.-B. Characterization of an EDTA bonded conducting polymer modified electrode: its application for the simultaneous determination of heavy metal ions. Anal. Chem. 2003, 75, 1123–1129. [Google Scholar]
- Rahman, M.A.; Park, D.-S.; Won, M.-S.; Park, S.-M.; Shim, Y.-B. Selective electrochemical analysis of various metal ions at an EDTA bonded conducting polymer modified electrode. Electroanalysis 2004, 16, 1366–1370. [Google Scholar]
- Kwon, N.H.; Won, M. S.; Park, D. S.; Shim, Y. B. Stability enhancement of all-solid-state H+ ISSEs with cross-linked silicon-urethane matrices. Electroanalysis 2005, 17, 641–647. [Google Scholar]
- Won, M.S.; Yoon, J. H.; Shim, Y. B. Determination of Selenium with a Poly(1,8-diamino-naphthalene)-Modified Electrode. Electroanalysis 2005, 17, 1952–1958. [Google Scholar]
- Konopka, A.; Sokalski, T.; Michalska, A.; Lewenstam, A.; Maj-Zurawska, M. Factors Affecting the Potentiometric Response of All-Solid-State Solvent Polymeric Membrane Calcium-Selective Electrode for Low-Level Measurements. Anal. Chem. 2004, 76, 6410–6418. [Google Scholar]
- Aquino-Binag, C.N.; Kumar, N.; Lamb, R. N.; Pigram, P. J. Fabrication and Characterization of a Hydroquinone-Functionalized Polypyrrole Thin-Film pH Sensor. Chem. Mater. 1996, 8, 2579–2585. [Google Scholar]
- Zotti, G.; Zecchin, S.; Schiavon, G.; Berlin, A.; Penso, M. Ionochromic and Potentiometric Properties of the Novel Polyconjugated Polymer from Anodic Coupling of 5,5′-Bis(3,4-(ethylenedioxy)thien-2-yl)-2,2′-bipyridine. Chem. Mater. 1999, 11, 3342–3351. [Google Scholar]
- Wu, X.; Kim, J.; Dordick, J.S. Enzymatically and Combinatorially Generated Array-Based Polyphenol Metal Ion Sensor. Biotechnol. Prog. 2000, 16, 513–516. [Google Scholar]
- Jovanovi, V. M.; Markievi, L.; Stankovi, S.; Stankovi, R.; Jovanovi, M.S. The behavior of polypyrrole doped with different anions as an ion-selective electrode. Electroanalysis 1995, 7, 574–578. [Google Scholar]
- Gonzalez-Bellavista, A.G.; Macanas, J.; Munoz, M.; Fabregas, E. Sulfonated poly(ether ether ketone), an ion conducting polymer, as alternative polymeric membrane for the construction of anion-selective electrodes. Sens. Actuat. B 2007, 125, 100–105. [Google Scholar]
- Sjoberg-Eerola, P.; Bobaccka, J.; Lewenstam, A.; Ivaska, A. All-solid-state chloride sensors based on electronically conducting, semiconducting and insulating polymer membranes. Sens. Actuat. B 2007. doi: 10.1016.j.snb.2007.05.004. [Google Scholar]
- Rahman, M.A.; Park, D. S.; Chang, S. C.; McNeil, C. J.; Shim, Y. B. The biosensor based on the pyruvate oxidase modified conducting polymer for phosphate ions determinations. Biosens. Bioelectron. 2006, 21, 1116–1124. [Google Scholar]
- Li, Y.; Wang, P.; Wang, L.; Lin, X. Overoxidized polypyrrole film directed single-walled carbon nanotubes immobilization on glassy carbon electrode and its sensing applications. Biosens. Bioelectron. 2007, 22, 3120–3125. [Google Scholar]
- Bendikov, T.A.; Harmon, T.C. Long-lived solid state perchlorate ion selective sensor based on doped poly(3,4-ethylenedioxythiophene) (PEDOT) films. Anal. Chim. Acta 2005, 551, 30–36. [Google Scholar]
- Badea, M.; Amine, A.; Palleschi, G.; Moscone, D.; Volpe, G.; Curulli, A. New electrochemical sensors for detection of nitrites and nitrates. J. Electroanal. Chem. 2001, 509, 66–72. [Google Scholar]
- Atta, N.F.; Galal, A.; Mark, H. B., Jr; Yu, T.; Bishop, P. L. Conducting polymer ion sensor electrodes-III. Potentiometric sulfide ion selective electrode. Talanta 1998, 47, 987–999. [Google Scholar]
- Fabre, B.; Burlet, S.; Cespuglio, R.; Bidan, G. Voltammetric detection of NO in the rat brain with an electronic conducting polymer and Nafion bilayer-coated carbon fibre electrode. J. Electroanal. Chem. 1997, 426, 75–83. [Google Scholar]
- Sjöberg-Eerola, P.; Bobacka, J.; Sokalski, T.; Mieczkowski, J.; Ivaska, A.; Lewenstam, A. All-Solid-State Chloride Sensors with Poly(3-Octylthiopene) Matrix and Trihexadecylmethylammonium Chlorides as an Ion Exchanger Salt. Electroanalysis 2004, 16, 379–385. [Google Scholar]
- Mousavi, Z.; Bobacka, J.; Ivaska, A. Potentiometric Ag+ Sensors Based on Conducting Polymers: A Comparison between Poly (3,4-ethylenedioxythiophene) and Polypyrrole Doped with Sulfonated Calixarenes. Electroanalysis 2005, 17, 1609–1615. [Google Scholar]
- Sutar, D.S.; Padma, N.; Aswal, D.K.; Deshpande, S.K.; Gupta, S.K.; Yakhmi, J.V. Preparation of nanofibrous polyaniline films and their application as ammonia gas sensor. Sens. Actuats. B 2007. doi:10:1016/j.snb.2007.06.015. [Google Scholar]
- Koull, S.; Chandra, R.; Dhawan, S.K. Conducting polyaniline composite: a reusable sensor material for aqueous ammonia. Sens. Actuats. B 2001, 75, 151–159. [Google Scholar]
- Kumar, S.A.; Chen, S. Electrochemically polymerized composites of conducting poly(p-ABSA) and flavins (FAD, FMN, RF) films and their use as electrochemical sensors: A new potent electroanalysis of NADH and NAD+. Sens. Actuats. B 2007, 123, 964–977. [Google Scholar]
- Agüí, L.; Peña-Farfal, C.; Yáñez-Sedeño, P.; Pingarrón, J. M. Poly-(3-methylthiophene)/carbon nanotubes hybrid composite-modified electrodes. Electrochim Acta 2007, 52, 7946–7952. [Google Scholar]
- Radhakrishnan, S.; Paul, S. Conducting polypyrrole modified with ferrocene for applications in carbon monoxide sensors. Sens. Actuats. B 2007, 125, 60–65. [Google Scholar]
- Vidotti, M.; Antonia, L.D.; Cintra, E. P.; Torresi, S. C. Reduction of interference signal of ascorbate and urate in poly(pyrrole)-based ammonia sensors in aqueous solutions. Electrochim. Acta 2004, 49, 3665–3670. [Google Scholar]
- Boopathi, M.; Won, M.S.; Shim, Y.-B. A sensor for acetaminophen in a blood medium using a Cu (II)-conducting polymer complex modified electrode. Anal. Chim. Acta 2004, 512, 191–197. [Google Scholar]
- Calbreath, D. F. The Scope of Clinical Chemistry. In Clinical Chemistry. A Fundamental Text book; Ozmar, S., Ed.; Philadelphia; W.B. Saunders Company, 1992; pp. 3–9. [Google Scholar]
- Turner, A. P. F.; Karube, I.; Wilson, G. S. Biosensors: Fundamentals, and Applications; Oxford; Oxford University Press, 1987. [Google Scholar]
- Schultz, J.S.; Taylor, R. F. Introduction to chemical and biological sensors. In Handbook of chemical and biological sensors; Taylor, R. F., Schultz, J.S., Eds.; London; IOP, 1996; pp. 1–10. [Google Scholar]
- Scheller, F. W.; Schubert, F.; Fedowitz, J. Present state and frontiers in biosensorics. In Frontiers in biosensorics I. Fundamental aspects; Scheller, F. W., Schubert, F., Fedowitz, J., Eds.; Basel; Birkhauser Verlag, 1997; pp. 1–12. [Google Scholar]
- Cass, A. E. G.; Cass, T.; Cooper, J. Biosensors: A Practical Approach; Oxford; Oxford University Press, 2004. [Google Scholar]
- Wang, J.; Musameh, M. Carbon nanotube/Teflon composite electrochemical sensors and biosensors. Anal. Chem. 2003, 75, 2075–2079. [Google Scholar]
- Bidan, G. Electroconducting conjugated polymers. New sensitive matrices to build up chemical or electrochemical sensors. A review. Sens. Actuat. B 1992, 6, 45–56. [Google Scholar]
- Bartlett, P.N.; Cooper, J. M. A review of the immobilization of enzymes in electropolymerized films. J. Electroanal. Chem. 1993, 362, 1–12. [Google Scholar]
- Cosnier, S. Biomolecule immobilization on electrode surfaces by entrapment or attachment to electrochemically polymerized films. A review. Biosens. Bioelectron. 1999, 14, 443–456. [Google Scholar]
- Gerard, M.; Chaubey, A.; Malhotra, B.D. Application of conducting polymers to biosensors. Biosens. Bioelectron. 2002, 17, 345–359. [Google Scholar]
- Umana, M.; Waller, J. Protein modified electrodes. The glucose oxidase / polypyrrole system. Anal. Chem. 1986, 64, 2979–2983. [Google Scholar]
- Foulds, N.C.; Lowe, C. R. Enzyme entrapment in electrically conducting polymers. Immobilisation of glucose oxidase in polypyrrole and its application in amperometric glucose sensors. J. Chem. Soc. Faraday Trans. I 1986, 82, 1259–1264. [Google Scholar]
- Clark, L.C., Jr.; Lyons, C. Electrode systems for continuous monitoring cardiovascular surgery. Ann. N. Y. Acad. Sci. 1962, 102, 29–45. [Google Scholar]
- Rahman, M. A.; Park, D.S.; Shim, Y.-B. A performance comparison of choline biosensors: anodic or cathodic detections of H2O2 generated by enzyme immobilized on a conducting polymer. Biosens. Bioelectron. 2004, 19, 1565–1571. [Google Scholar]
- Rahman, M. A.; Kwon, N.-H.; Won, M.-S.; Choe, E. S.; Shim, Y.-B. Functionalized conducting polymer as an enzyme-immobilizing substrate: an amperometric glutamate microbiosensor for in vivo measurements. Anal. Chem. 2005, 77, 4854–4860. [Google Scholar]
- Rahman, M. A.; Won, M.-S.; Shim, Y.-B. Xanthine sensors based on anodic and cathodic detection of enzymatically generated hydrogen peroxide. Electroanalysis 2007, 19, 631–637. [Google Scholar]
- Darain, F.; Park, J.-S.; Akutsu, H.; Shim, Y.-B. Superoxide radical sensing using a cytochrome c3 immobilized conducting polymer electrode. Biosens. Bioelectron. 2007, 23, 161–167. [Google Scholar]
- Kwon, N.-H.; Rahman, M. A.; Won, M.-S.; Shim, Y.-B. Lipid-bonded conducting polymer layers for a model biomembrane: application to superoxide biosensors. Anal. Chem. 2006, 78, 52–60. [Google Scholar]
- Rahman, M.A.; Won, M.-S.; Shim, Y.-B. The potential use of hydrazine as an alternative to peroxidase in a biosensor: comparison between hydrazine and HRP-based glucose sensors. Biosens. Bioelectron. 2005, 21, 257–265. [Google Scholar]
- Shiddiky, M.J. A.; Park, D.-S.; Shim, Y.-B. Detection of polymerase chain reaction fragments using a conducting polymer-modified screen-printed electrode in a microfluidic device. Electrophoresis 2005, 26, 4656–4663. [Google Scholar]
- Shiddiky, M. J. A.; Rahman, M. A.; Park, J.-S.; Shim, Y.-B. Analysis of polymerase chain reaction amplifications through phosphate detection using an enzyme-based microbiosensor in a microfluidic device. Electrophoresis 2006, 27, 2951–2959. [Google Scholar]
- Tian, F.; Xu, B.; Zhu, G.; Zhu, L. Hydrogen peroxide biosensor with enzyme entrapped within electrodeposited polypyrrole based on mediated sol–gel derived composite carbon electrode. Anal. Chim. Acta 2001, 443, 9–16. [Google Scholar]
- Uang, Y.M.; Chou, T. C. Fabrication of glucose oxidase/polypyrrole biosensor by galvanostatic method in various pH aqueous solutions. Biosens. Bioelectron. 2003, 19, 141–147. [Google Scholar]
- Borole, D. D.; Kapadi, U. R.; Mahulikar, P.P.; Hundiwale, D. G. Glucose oxidase electrode for polyaniline, poly(o-toluidine and their copolymers for advance technology. Polym. Adv. Technol. 2003, 15, 306–312. [Google Scholar]
- Seker, S.; Becerik, I. A Neural Network Model in the Calibration of Glucose Sensor Based on the Immobilization of Glucose Oxidase into Polypyrrole Matrix. Electroanalysis 2004, 16, 1542–1549. [Google Scholar]
- Ramanavicius, A.; Kausaite, A.; Ramanavicience, A.; Acaite, J.; Malinauskas, A. Redox enzyme – glucose oxidase – initiated synthesis of polypyrrole. Synth. Met. 2006, 156, 409–413. [Google Scholar]
- Curulli, A.; Valentini, F.; Orlanduci, S.; Terranova, M.L.; Palleschi, G. Pt based enzyme electrode probes assembled with Prussian Blue and conducting polymer nanostructures. Biosens. Bioelectron. 2004, 20, 1223–1232. [Google Scholar]
- Barton, A.C.; Collyer, S. D.; Davis, F.; Gornall, D.D.; Law, K. A.; Lawrence, E.C.D.; Mills, D. W.; Myler, S.; Prtichand, J.A.; Thomson, M.; Higson, S. P. J. Sonochemically fabricated microelectrode arrays for biosensors-part II: Modification with a polysiloxane coating. Biosens. Bioelectron. 2004, 20, 408–412. [Google Scholar]
- Arslan, A.; Kiralp, S.; Toppare, L.; Yagci, Y. Immobilization of tyrosinase in polysiloxane/polypyrrole copolymer matrices. Int. J. Biol. Macromole. 2005, 35, 163–167. [Google Scholar]
- Vedrine, C.; Fabiano, S.; Tran-Minh, C. Amperometric tyrosinase based biosensor using an electrogenerated polythiophene film as an entrapment support. Talanta 2003, 59, 535–544. [Google Scholar]
- Boyukbayram, A.E.; Kiralp, S.; Toppare, L.; Yagci, Y. Preparation of biosensors by immobilization of polyphenol oxidase in conducting copolymers and their use in determination of phenolic compounds in red wine. Bioelectrochemistry 2006, 69, 164–171. [Google Scholar]
- Jiang, L.; Jun, H.K.; . Hoh, Y.S.; Lim, J.O.; Lee, D.D.; Huh, J.S. Sensing characteristics of polypyrrole–poly(vinyl alcohol) methanol sensors prepared by in situ vapor state polymerization. Sens. Actuators B 2005, 105, 132–137. [Google Scholar]
- Kan, J.; Pan, X.; Chen, C. Polyaniline–uricase biosensor prepared with template process. Biosens. Bioelectron. 2004, 19, 1635–1640. [Google Scholar]
- Haccoun, J.; Piro, B.; Noel, V.; Pham, M.C. The development of a regentless lactate biosensor based on a novel conducting polymer. Bioelectrochemistry 2006, 68, 218–236. [Google Scholar]
- Bartlett, P. N.; Simon, E.; Toh, C.S. Modified electrodes for NADH oxidation and dehydrogenase-based biosensors. Bioelectrochemistry 2002, 56, 117–122. [Google Scholar]
- Chaubey, A.; Pande, K.K.; Singh, V.S.; Malhotra, B.D. Co-immobilization of lactate oxidase and lactate dehydrogenase on conducting polyaniline films. Anal. Chim. Acta 2000, 407, 97–103. [Google Scholar]
- Asberg, P.; Inganas, O. Hydrogels of a conducting conjugated polymer as 3-D enzyme electrode. Biosens. Bioelectron. 2003, 19, 199–207. [Google Scholar]
- Grennan, K.; Killard, A.J.; Smyth, M.R. Chemically Polymerized Polyaniline Films for the Mass-Production of Biosensor Devices. Electroanalysis 2005, 17, 1360–1369. [Google Scholar]
- Morrin, A.; Ngamna, O.; Kllard, A.J.; Moulton, S. E.; Smyth, M. E.; Wallace, G. G. An Amperometric Enzyme Biosensor Fabricated from Polyaniline Nanoparticles. Electroanalysis 2005, 17, 423–430. [Google Scholar]
- Jia, J.; Wang, B.; Wu, A.; Cheng, G.; Li, Z.; Dong, S. A method to construct third generation Horseradish peroxidase biosensor: Self assembling gold nanoparticles to three dimensional sol-gel network. Anal. Chem. 2002, 74, 2217–2223. [Google Scholar]
- Ngamna, O.; Morrin, A.; Moulton, S. E.; Killrad, A.J.; Smyth, M. R.; Wallace, G. G. An HRP based biosensor using sulphonated polyaniline. Synth. Met. 2005, 153, 185–188. [Google Scholar]
- Mathebe, N.G.R.; Morrin, A.; Iwuoha, E.I. Electrochemistry and scanning electron microscopy of polyaniline/peroxidase-based biosensor. Talanta 2005, 64, 115–120. [Google Scholar]
- Brahim, S.; Narinesinh, D.; Gueseppi-Elie, A. Polypyrrole-hydrogel composites for the construction of clinically important biosensors. Biosens. Bioelectron. 2002, 17, 53–59. [Google Scholar]
- Ivanov, A.N.; Evtugyn, G. A.; Lukachova, L. V.; Karyakina, E.E.; Budnikov, H. C.; Kiseleva, S. G.; Orlov, A. V.; Karpacheva, G. P.; Karyakin, A. A. New polyaniline based potentiometric biosensor for pesticides detection. IEEE Sens. J. 2003, 3, 333–340. [Google Scholar]
- Cosnier, S.; Pellec, A.L.; Marks, R.S.; Perie, K.; Lellouche, J. -P. A permselective biotinylated polydicarbazole film for the fabrication of amperometric enzyme electrodes. Electrochem. Commun. 2003, 5, 973–977. [Google Scholar]
- Langer, J.J.; Filipiak, M.; J. Kecinska, J.; Jasnowska, J.; Wlodareczak, J.; Buladowski, B. Polyaniline biosensor for choline determination. Surf. Sci. 2004, 573, 140–145. [Google Scholar]
- Qu, F.; Yang, M.; Jiang, J.; Shen, J.; Yu, R. Amperometric biosensor for choline based on layer-by-layer assembled functionalized carbon nanotube and polyaniline multilayer film. Anal. Biochem. 2005, 344, 108–114. [Google Scholar]
- E. Gizeli, E.; Lowe, C.R. Immunosensors. Curr. Opin. Biotechnol. 1996, 7, 66–71. [Google Scholar]
- McNeil, C. J.; Athey, D.; Renneberg, R. Immunosensors for Clinical Diagnosis. In Frontiers in Biosensorics II. Practical Applications; Scheller, F.W., Schubert, F., Fedowitz, J., Eds.; Basel; Birkhauser Verlag, 1997; pp. 17–25. [Google Scholar]
- Luppa, P.B.; Sokoll, L. J.; Chan, D. W. Immunosesnors-principles and applications to clinical chemistry. Clinica Chimica Acta 2001, 314, 1–26. [Google Scholar]
- Rogers, K.R. Principles of affinity-based biosensors. Mol. Biotechnol. 2000, 14, 109–129. [Google Scholar]
- Aizawa, M. Immunosensors for clinical analysis. Adv. Clin. Chem. 1994, 31, 247–275. [Google Scholar]
- Darain, F.; Park, S.-U.; Shim, Y.-B. Disposable amperometric immunosensor system for rabbit IgG using a conducting polymer modified screen-printed electrode. Biosens. Bioelectron. 2003, 18, 773–780. [Google Scholar]
- Darain, F.; Park, D.S.; Park, J.S.; Shim, Y.-B. Development of an immunosensor for the detection of vitellogenin using impedance spectroscopy. Biosnes. Bioelectron. 2004, 19, 1245–1252. [Google Scholar]
- Darain, F.; Park, D.-S.; Park, J.-S.; Chang, S.-C.; Shim, Y.-B. A separation-free amperometric immunosensor for vitellogenin based on screen-printed carbon arrays modified with a conductive polymer. Biosens. Bioelectron. 2005, 20, 1780–1787. [Google Scholar]
- Darain, F.; Ban, C.; Shim, Y. -B. Development of a new and simple method for the detection of histdine-tagged proteins. Biosens. Bioelectron. 2004, 20, 857–863. [Google Scholar]
- Rahman, M.A.; Shiddiky, M. J. A.; Park, J.-S.; Shim, Y.-B. An impedimetric immunosensor for the label-free detection of bisphenol A. Biosens. Bioelectron. 2007, 22, 2464–2470. [Google Scholar]
- Grant, S.; Dauris, F.; Law, K.A.; Berton, A.C.; Collyer, S.D.; Higson, S.P.J.; Gibson, T.D. Label- free and reversible immunosensor based upon an ac impedance interrogation protocol. Anal. Chim. Acta 2005, 537, 163–168. [Google Scholar]
- Ramanaviciene, A.; Ramanavicius, A. Application of polypyrrole for the creation of immunosensors. Crit. Rev. Anal. Chem. 2002, 32, 245–252. [Google Scholar]
- Gooding, J. J.; Wasiowych, C.; Barnett, D.; Hibbert, D.B.; Barisci, J. N.; Wallace, G.G. Electrochemical modulation of antigen–antibody binding. Biosens. Bioelectron. 2004, 20, 260–268. [Google Scholar]
- Zhang, L.; Yuan, R.; Huang, X.; Chai, Y.; Tang, D.; Cao, S. A new label-free amperometric immunosensor for rubella vaccine. Anal. Bioanal. Chem. 2005, 381, 1036–1040. [Google Scholar]
- Tahir, Z.M.; Alcocilza, E. C.; Grooms, D. L. Polyaniline synthesis and its biosensor application. Biosens. Bioelectron. 2005, 20, 1690–1695. [Google Scholar]
- Lillie, G.; Payne, P.; Vadgama, P. Electrochemical impedance spectroscopy as a platform for reagentless bioaffinity sensing. Sens. Actuat. B 2001, 78, 249–256. [Google Scholar]
- Farace, G.; Lillie, G.; Hianik, T.; Payne, P.; Vadgama, P. Reagentless biosensing using electrochemical impedance spectroscopy. Bioelectrochemistry 2002, 55, 1–3. [Google Scholar]
- Lee, Y.-T.; Shim, Y.-B. Direct DNA hybridization detection based on the oligonucleotide-functionalized conductive polymer. Anal. Chem. 2001, 73, 5629–5632. [Google Scholar]
- Ban, C.; Chung, S.; Park, D.-S.; Shim, Y.-B. Detection of protein-DNA interaction with a DNA probe: distinction between single-strand and double-strand DNA-protein interaction. Nucleic Acids Research 2004, 32, e110–e117. [Google Scholar]
- Shiddiky, M.J. A.; Rahman, M. A.; Shim, Y.-B. Hydrazine-Catalyzed Ultrasensitive Detection of DNA and Proteins. Anal. Chem. 2007, 79, 6886–6890. [Google Scholar]
- Gambhir, A.; Gerard, M.; Ain, S. K. J.; Malhotra, B. D. Characterization of DNA Immobilized on Electrochemically Prepared Conducting Polypyrrole-Polyvinyl Sulfonate Films. Appl. Biochem. Biotech. 2001, 96, 303–309. [Google Scholar]
- Cha, J.; Han, J.I.; Choi, Y.; Yoon, D.S.; Who, K.; Lim, G. DNA hybridization electrochemical sensor using conducting polymer. Biosens. Bioelectron. 2003, 18, 1241–1247. [Google Scholar]
- Youssoufi, H.K.; Makrouf, B. Electrochemical biosensing of DNA hybridization by ferrocenyl groups functionalized polypyrrole. Anal. Chim. Acta 2002, 469, 85–92. [Google Scholar]
- Shi, L.; Willner, I. Electrical contacting of glucose oxidase by DNA-templated polyaniline wires on surfaces. Electrochem. Commun. 2004, 6, 1057–1060. [Google Scholar]
- Wu, J.; Zhou, Y.; Li, X.; Liu, H.; Shen, G.; Yu, R. A biosensor monitoring DNA hybridization based on polyaniline intercalated graphite oxide nanocomposite. Sens. Actuat. B 2005, 104, 43–49. [Google Scholar]
- Zhu, N.; Chang, Z.; He, P.; Fang, Y. Electrochemically fabricated polyaniline nanowire-modified electrode for voltammetric detection of DNA hybridization. Electrochim. Acta 2006, 18, 3758–3762. [Google Scholar]
- Taira, S.; Yokoyama, K. Immobilization of single-stranded DNA by self-assembled polymer on gold substrate for a DNA chip. Biotechnol. Bioeng. 2005, 89, 835–838. [Google Scholar]
- Lori, J.; Morrin, A.; Killard, A.J.; Smyth, M.R. Development and Characterization of Nickel-NTA-Polyaniline Modified Electrodes. Electroanalysis 2006, 18, 77–81. [Google Scholar]
- Lassalle, N.; Mailley, P.; Vieil, E.; Livache, T.; Roget, A.; Correia, J.P.; . Abrantes, L.M. Electronically conductive polymer grafted with oligonucleotides as electrosensors of DNA: Preliminary study of real time monitoring by in situ techniques. J. Electroanal. Chem. 2001, 509, 48–57. [Google Scholar]
- Cai, H.; Xu, Y.; He, P.-G.; Fang, Y.-Z. Indicator Free DNA Hybridization Detection by Impedance Measurement Based on the DNA-Doped Conducting Polymer Film Formed on the Carbon Nanotube Modified Electrode. Electroanalysis 2003, 15, 1864–1870. [Google Scholar]


| Conducting Polymer | Structure | Conductivity (S/cm) |
|---|---|---|
| Polyacetylene | 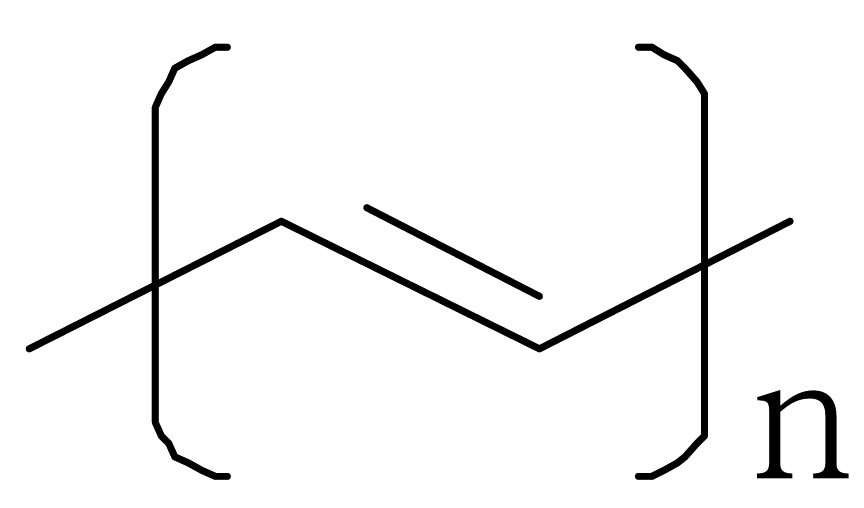 | 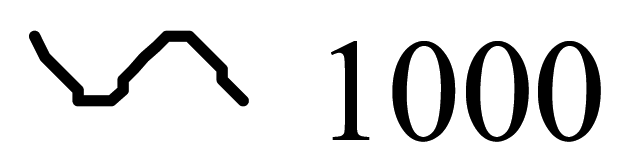 |
| Polyparaphenylene | 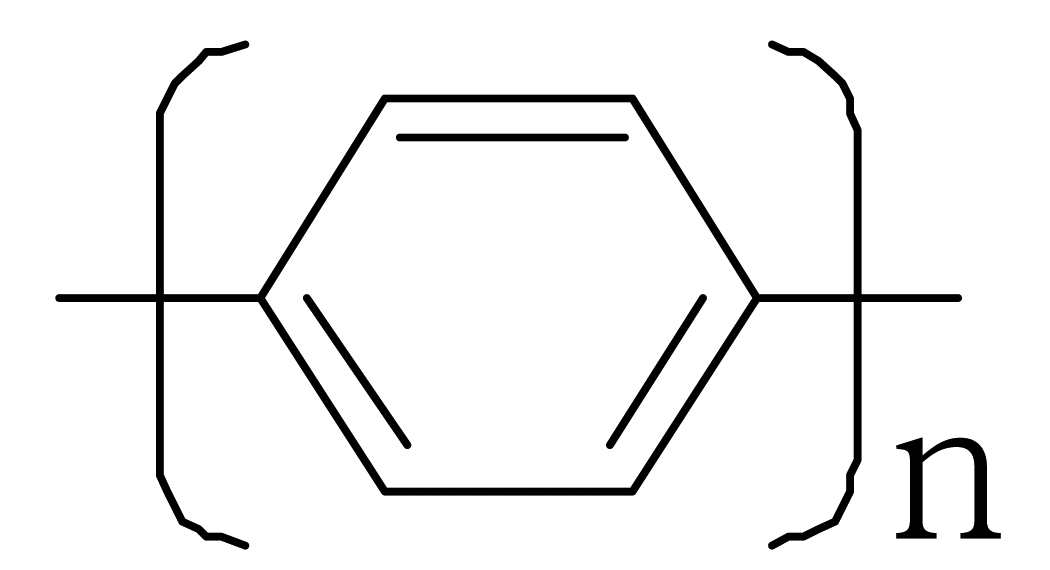 | 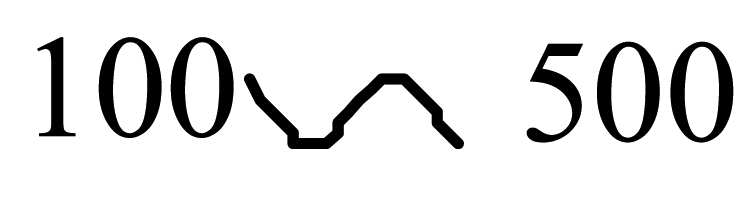 |
| Polyparaphenylene vinylene |  |  |
| Polyazulene | 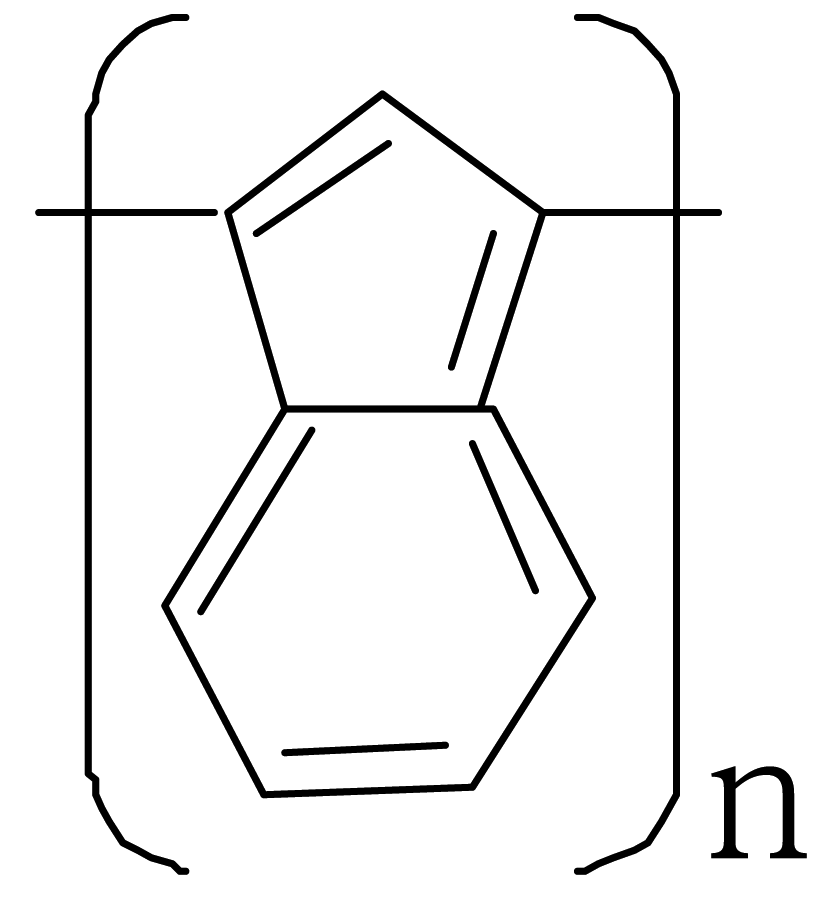 | 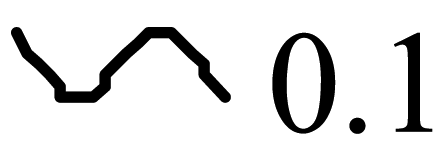 |
| Polyaniline | 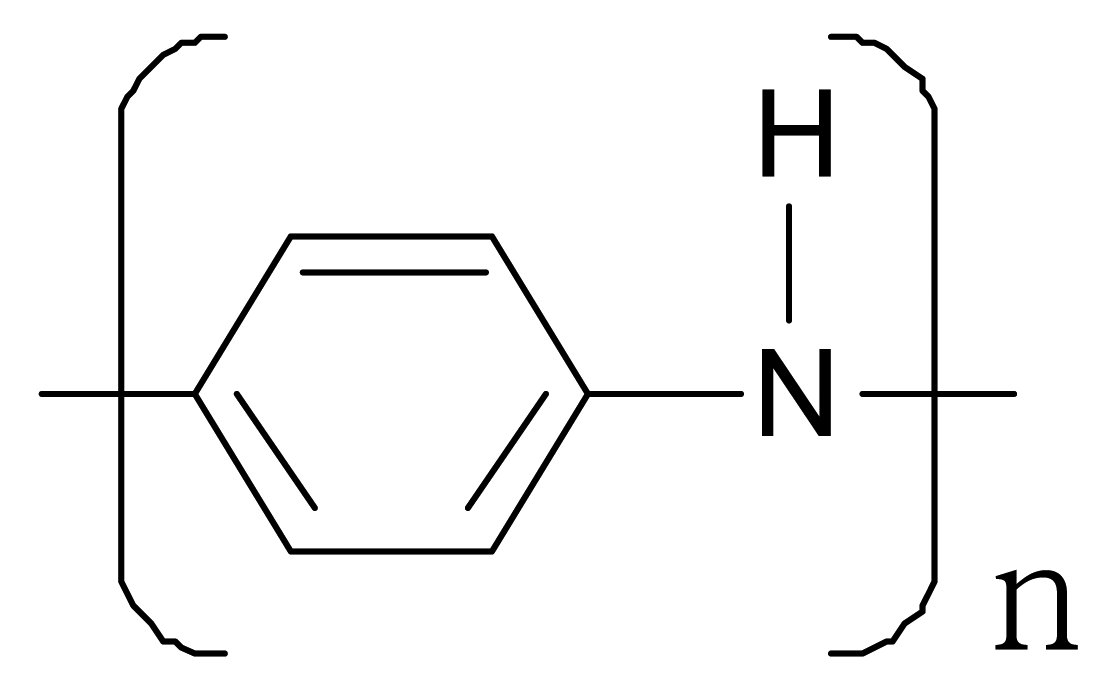 |  |
| Polyparaphenylene sulfide | 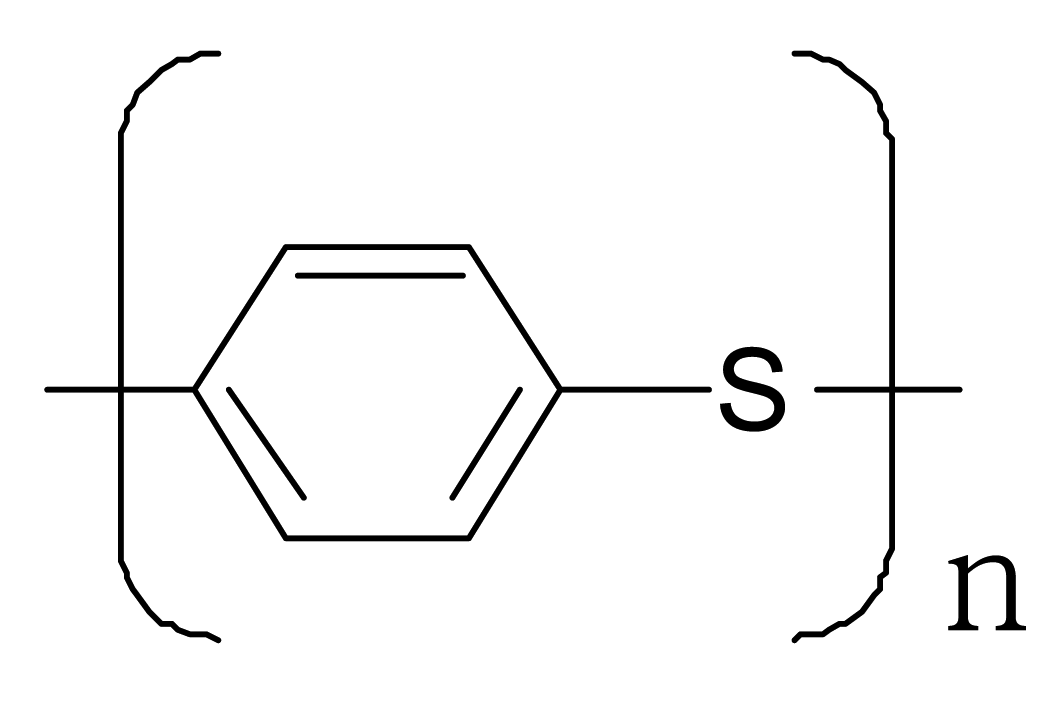 |  |
| Polypyrrole | 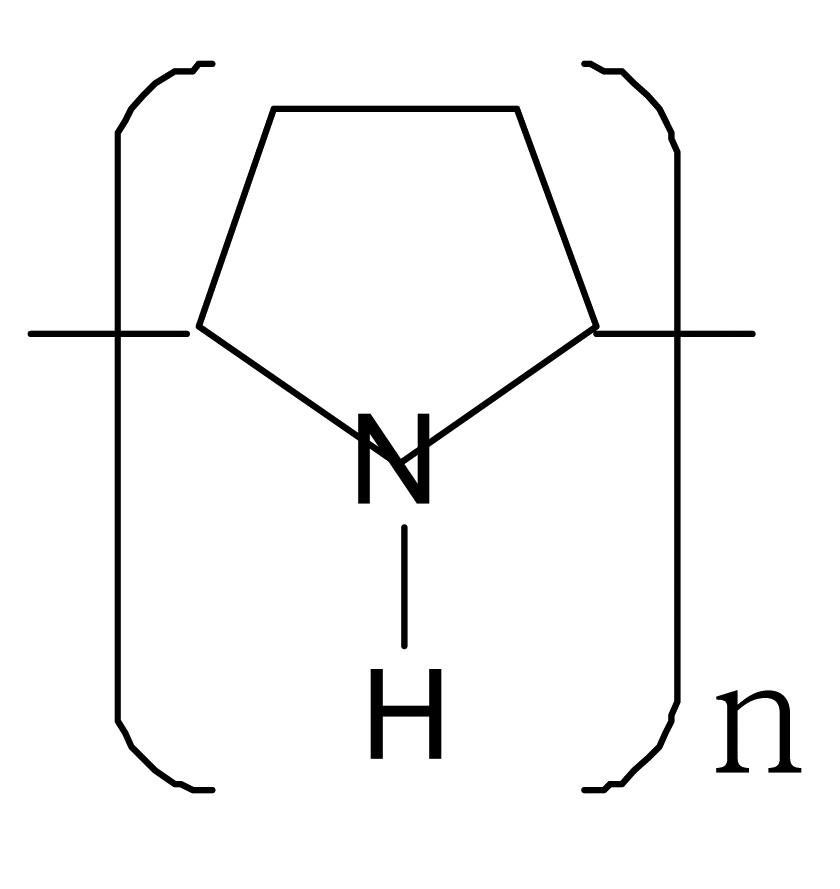 |  |
| Polythiophene |  |  |
| Polycarbazole | 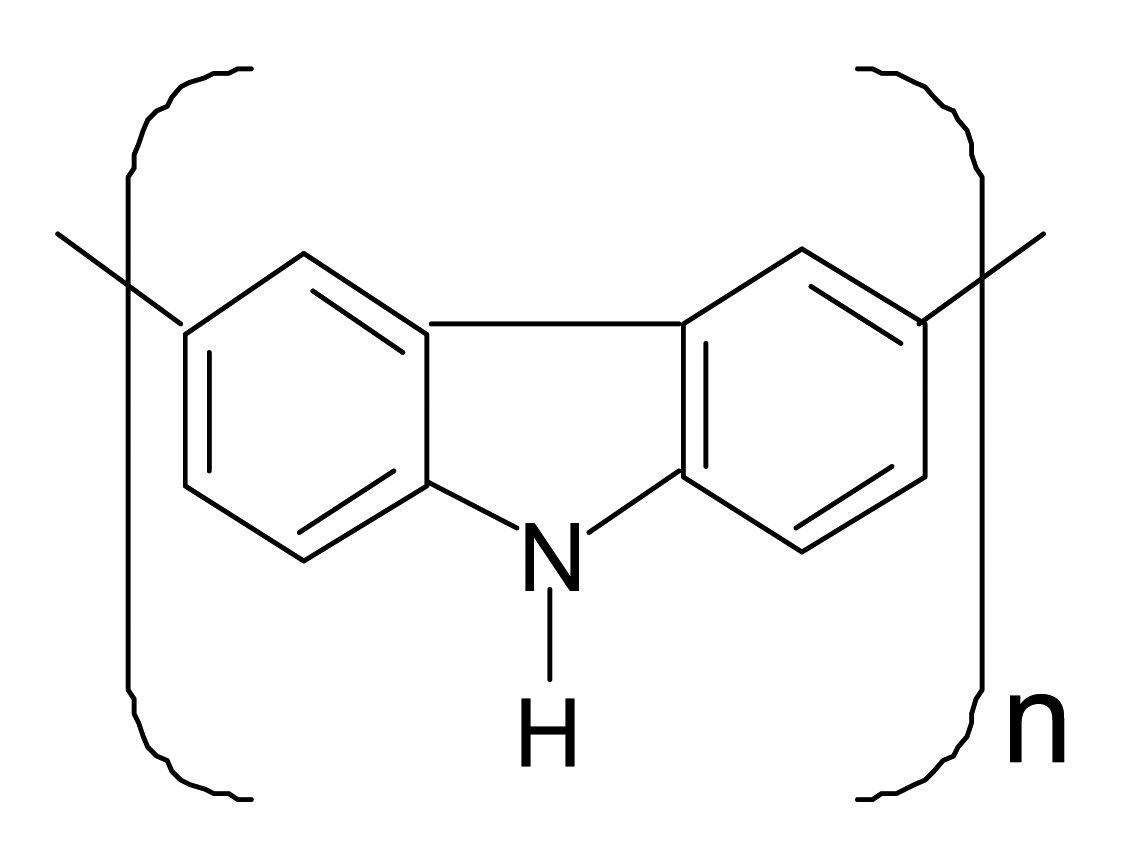 |  |
| Polydiaminonaphthalene | 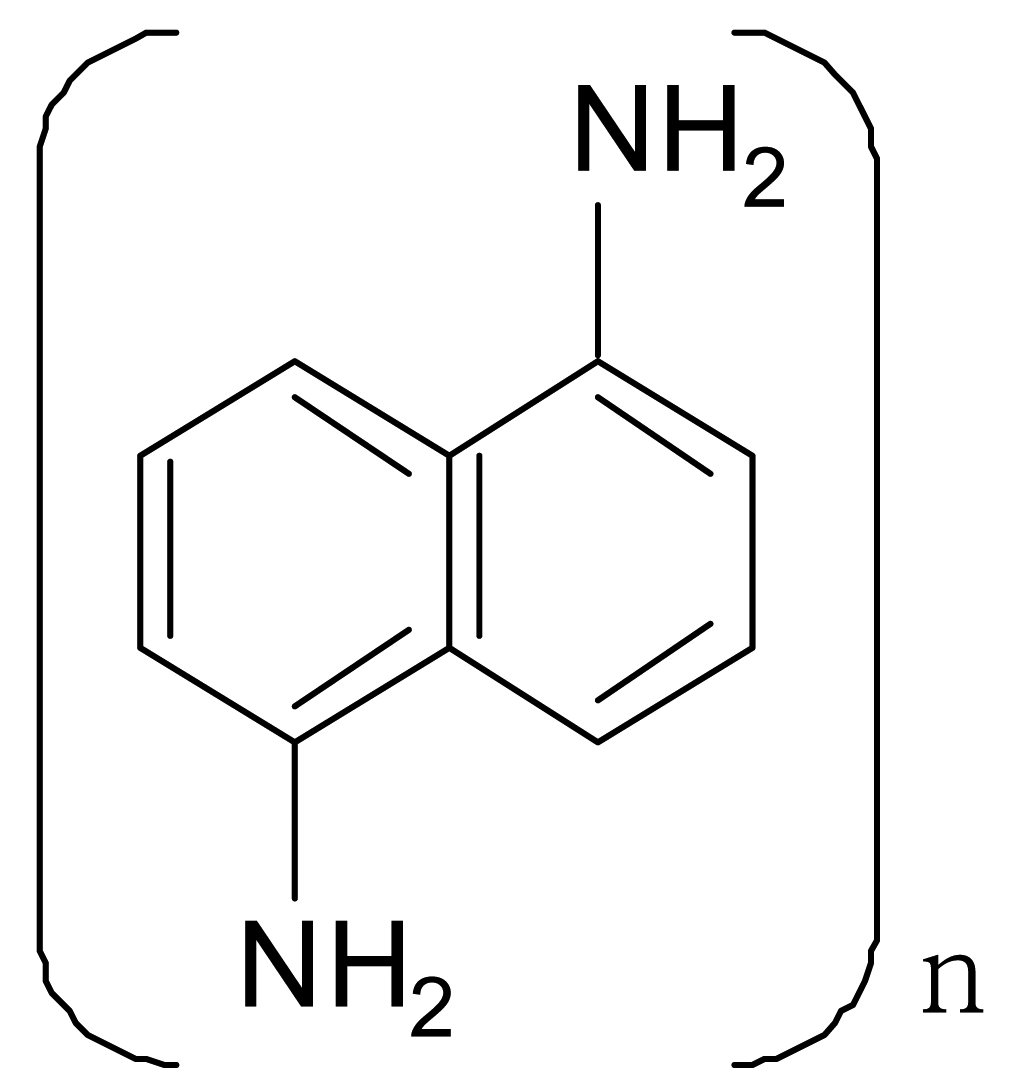 | 10−3 |
© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Rahman, M.A.; Kumar, P.; Park, D.-S.; Shim, Y.-B. Electrochemical Sensors Based on Organic Conjugated Polymers. Sensors 2008, 8, 118-141. https://doi.org/10.3390/s8010118
Rahman MA, Kumar P, Park D-S, Shim Y-B. Electrochemical Sensors Based on Organic Conjugated Polymers. Sensors. 2008; 8(1):118-141. https://doi.org/10.3390/s8010118
Chicago/Turabian StyleRahman, Md. Aminur, Pankaj Kumar, Deog-Su Park, and Yoon-Bo Shim. 2008. "Electrochemical Sensors Based on Organic Conjugated Polymers" Sensors 8, no. 1: 118-141. https://doi.org/10.3390/s8010118
APA StyleRahman, M. A., Kumar, P., Park, D.-S., & Shim, Y.-B. (2008). Electrochemical Sensors Based on Organic Conjugated Polymers. Sensors, 8(1), 118-141. https://doi.org/10.3390/s8010118





