PVC Membrane Sensors for Potentiometric Determination of Acebutolol
Abstract
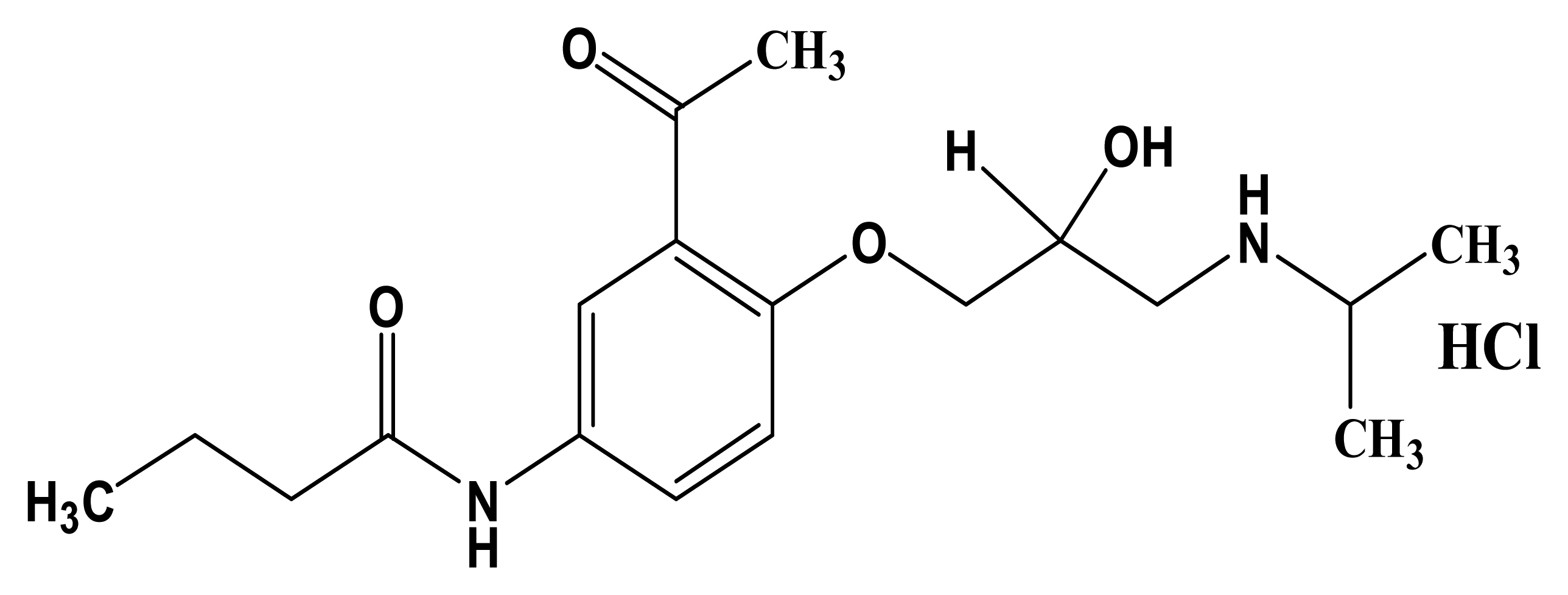
:1. Introduction
2. Results and Discussion
2.1. Sensor characteristics
2.2. Response mechanism of the proposed sensor
2.3. Effect of pH
2.4. Response time
2.5. Effect of plasticizer type on the characteristic performance of the sensor
2.6. Effect of divers ions
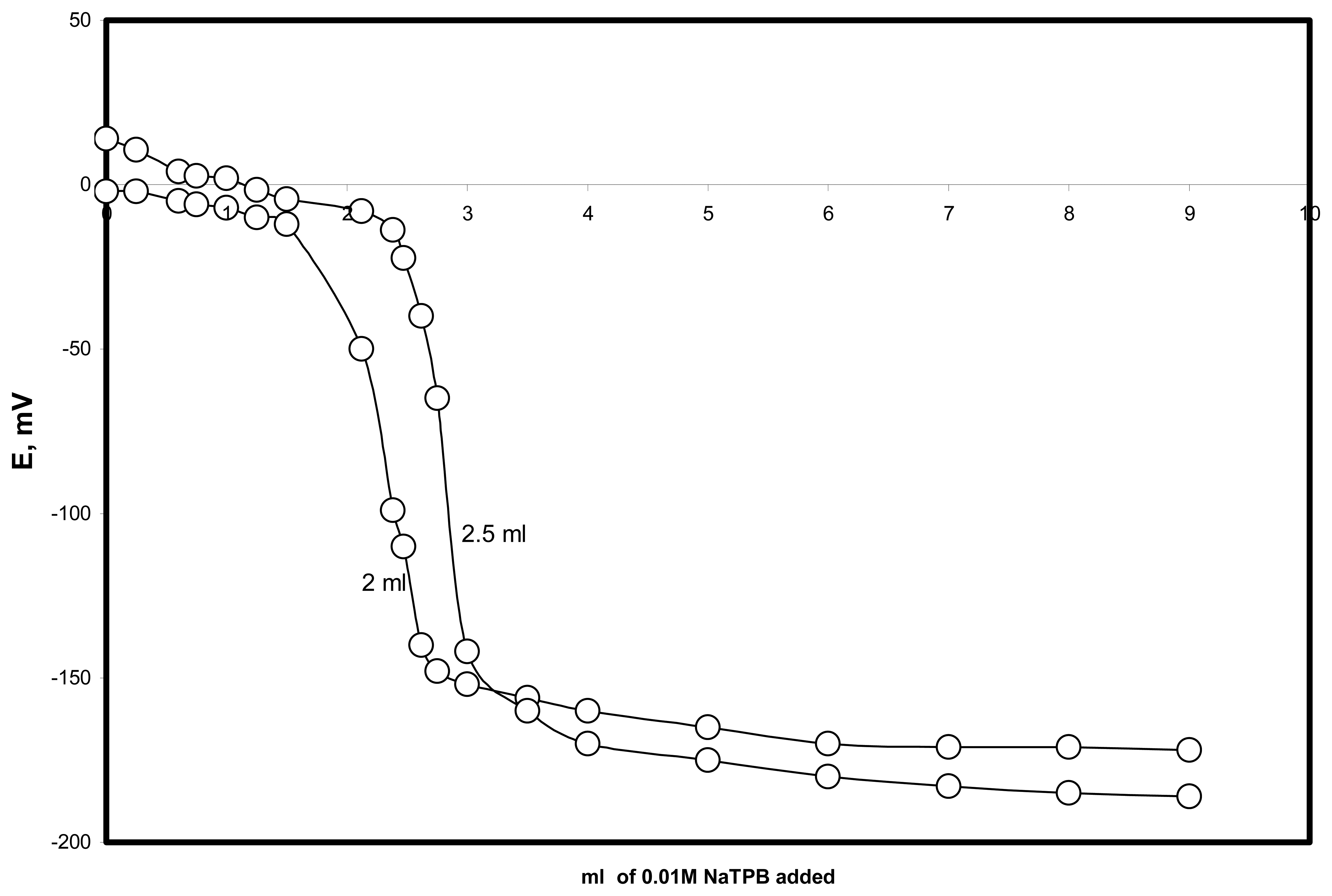
2.7. Validity of the proposed method
2.7.1. Limit of quantification and limit of detection
2.7.2. Precision and Accuracy of the method
2.7.3. Ruggedness
2.7.4. Robustness
2.8. Analytical applications
2.8.1. Determination of AC in Tablets
2.8.2. Application of AC –PVC electrodes as indicator electrode
3. Experiment
3.1. Apparatus
2.3. Reagents and materials
3.3. Sensor preparation
3.4. Procedure
3.5. Determination of AC in Sectral Tablets
4. Conclusion
References and Notes
- Martindal. The Complete Drug Reference, 33rd Ed. ed; Pharmaceutical Press: London, 2002; p. 569. [Google Scholar]
- Delamoye, M.; Duverneuil, C.; Paraire, F.; de Mazancourt, P.; Alvarez, J.C. Simultaneous determination of thirteen beta-blockers and one metabolite by gradient high-performance liquid chromatography with photodiode-array UV detection. Forensic Science International 2004, 141, 23–31. [Google Scholar]
- Saarinen, M.T.; Sirèn, H.; Riekkola, M.-L. Screening and determination of B-blockers, narcotic analgesics and stimulants in urine by high-performance liquid chromatography with column switching. J. Chromatography B.: Biomedical Science and applications 1995, 664, 341–346. [Google Scholar]
- Pelander, A.; Ojanpera, I.; Laks, S.; Rasanen, I.; Vuori, E. Toxicological screening with formula-based metabolite identification by liquid chromatography/time-of-flight Mass Spectrometer. Anal. Chem. 2003, 75, 5710–5718. [Google Scholar]
- Brunelli, C.; Bicchi, C.; Di Stilo, A.; Salomone, A.; Vincenti, M. High-speed gas chromatography in doping control: fast-GC and fast-GC/MS determination of beta -adrenoceptor ligands and diuretics. J. Sep. Sci. 2006, 29, 2765–2771. [Google Scholar]
- Bai, X.; You, T.Y.; Sun, H.; Yang, X.; Wang, E. Determination of three-β-blockers by capillary electrophoresis with end column electrochemical detection. Electroanalysis 2000, 12, 1379–1382. [Google Scholar]
- Sastry, C.S.P.; Rao, S.G.; Naidu, P.Y.; Srinivas, K.R. New spectrophotometric method for the determination of some drugs with iodine and wool fast blue BL. Talanta 1998, 45, 1227–1234. [Google Scholar]
- Abdelatef, H.E.; El-Sayed, M.M.; Ayad, H.M. Spectrophotometric and spectrofluorimetric method for acyclovir and acebutolol hydrochloride. Spectrochim. Acta A. Mol. Biomol. Spectrosc. 2006, 65, 996–998. [Google Scholar]
- El-Gindy, A.; Ashour, A.; Abdel-Fattah, L.; Shabana, M.M. First derivative spectrophotometric, TLC-densitometric and HPLC determination of acebutolol HCl in presence of its acid-induced degradation product. J. Pharm. Biomed. Anal. 2001, 24, 527–534. [Google Scholar]
- El-Walily, A.F.M. Analysis of nifedipine-acebutolol hydrochloride binary combination in tablets using UV-derivative spectroscopy, capillary gas chromatography and high-performance liquid chromatography. J. Pharm. Biomed. Anal. 1997, 16, 21–30. [Google Scholar]
- Nasierowska, Z.; Deres, J.; Suffczynski, J.; Stanczak, W. Polarographic determination of acebutolol. Farm. Pol. 1984, 40, 87–89. [Google Scholar]
- Sanbe, H.; Haginaka, J. Restricted access media-molecularly imprinted polymer for propranolol and its application to direct injection analysis of β-blockers in biological fluids. Analyst 2003, 118, 593–597. [Google Scholar]
- Pospìšilová, M.; Kavalìrová, A.; Polášek, M. Assay of acebutolol in pharmaceutical by analytical capillary isotachophoresis. J. Chromatogr. A. 2005, 1081, 72–76. [Google Scholar]
- Mostafa, G.A.E.; Ghazy, S.E. Potentiometric PVC membrane sensors for selective determination of pyridoxine hydrochloride (vitamin B6) in some pharmaceutical formulations. Annali di Chimica 2003, 93, 691–699. [Google Scholar]
- Yuan, J.; Yao, S. A. Sulpha-drug sensitive sensor based on ion-pair complex modified PQC resonator. Talanta 2002, 58, 641–648. [Google Scholar]
- Riahi, S.; Mousavi, M.F.; Bathaie, S.Z.; Shmsipur, M. A novel potentiometric sensor for selective determination of theophylline: Theoretical and practical investigation. Anal. Chim. Acat. 2005, 548, 192–198. [Google Scholar]
- Shamsipur, M.; Jalali, F.; Ershad, S. Preparation of a diclofenac potentiometric sensor and its application to pharmaceutical analysis and to drug recovery from biological fluids. J. Pharm. Biomed. Analysis 2005, 37, 943–947. [Google Scholar]
- Cunningham, L.; Freiser, H. Ion-selective electrodes for some β-adeneric and calcium blockers. Anal. Chim. Acta. 1984, 157, 157–162. [Google Scholar]
- IUPAC Analytical Chemistry Division. Recommendation for Nomenclature of Ion Selective Electrode. Pure Appl. Chem. 1994, 66, 2527–2536. [Google Scholar]
- Anthony, C.M.; M David, O.; Brian, W. Clarke's analysis of drugs and poisons, 3rd Ed. ed; Pharmaceutical Press: Great Britain, 2004; Vol. 2, p. 569. [Google Scholar]
- Ma, T.S.; Hassan, S.S.M. Organic analysis using ion selective electrodes; Academic press: London, 1982; Vol. 1&2. [Google Scholar]
- IUPAC Analytical Chemistry Division. Potentiometric selectivity coefficients of ion selective electrodes. Pure Appl. Chem. 2000, 72, 1851–185. [Google Scholar]
- British Pharmacopoeia; The Stationery Office: London, 2003.
- Miller, J.C.; Miller, J.N. Statistics for analytical chemistry, 1st Ed. ed; Ellis Harwood Limited: England, 1986. [Google Scholar]
- British Pharmacopoeia; The Stationery Office: London, 1980; volume II, appendix 1B; p. A45.
- Hassan, S.S.M.; Marzouk, S.A.M. A novel ferroin membrane sensor for potentiometric determination of iron. Talanta 1994, 41, 891–899. [Google Scholar]
- Carggs, A.; Moody, G.J.; Tomas, J.D.R. PVC matrix membrane ion-selective electrodes. J. Chem. Educ. 1974, 51, 541–544. [Google Scholar]




| Parameter | Value* | |
|---|---|---|
| AC-TPB | AC-PM | |
| Slope, (mV/ decade) | 51.5 ± 0.5 | 53.0 ± 0.5 |
| Intercept, mV | 134.0 ± 0.5 | 164.0 ± 0.5 |
| Correlation Coefficient, (r) | 0.998 | 0.999 |
| Detection limit, M | 6×10-6 | 4×10-6 |
| Response time for1×10-3 M solution, s | 25 ±0.5 | 20± 0.6 |
| Working pH range | 2 - 6.0 | 2- 6.0 |
| Parameter | AC(100μg/ml)* Within–day | AC(100μg/ml)* Between-days | ||
|---|---|---|---|---|
| AC-TPB | AC-PM | AC-TPB | AC-PM | |
| R, % | 98.0 | 98.7 | 97.7 | 98.5 |
| R.S. D, % | 1.5 | 1.5 | 1.6 | 1.5 |
| Slope | 51.5 ± 0.5 | 53.0 ± 0.5 | 51.5± 0.6 | 53.0± 0.6 |
| Correlation coefficient | 0.998 | 0.999 | 0.998 | 0.998 |
| Interferent, J | ||
|---|---|---|
| AC-TPB | AC-PM | |
| Na+ | 2 × 10-3 | 2 × 10-3 |
| K+ | 1 × 10-3 | 1 × 10-3 |
| Ca2+ | 3 × 10-3 | 3 × 10-3 |
| Mg2+ | 3.5 × 10-3 | 3.4×10-3 |
| Fe3+ | 3 × 10-3 | 2.0×10-3 |
| Glucose | 2 × 10-4 | 2 × 10-4 |
| Sucrose | 2 × 10-4 | 2×10-4 |
| Lactose | 2 × 10-4 | 2×10-4 |
| Starch | 3 × 10-4 | 3 × 10-4 |
| Avisil | 1 × 10-4 | 1.5 × 10-4 |
| gelatin | 1 × 10-4 | 1 × 10-4 |
| Caffeine | 3 × 10-3 | 3 ×10-3 |
| Tryptophan | 2 × 10-3 | 2 ×10-3 |
| Urea | 1 × 10-3 | 1 ×10-3 |
| Added (μg/ml) | Found (μg/ml) | Recovery, %* | ||
|---|---|---|---|---|
| AC-TPB | AC-PM | AC-TPB | AC-PM | |
| 3.0 | 2.93 | 2.93 | 97.6 ± 1.9 | 97.6 ± 1.8 |
| 6.0 | 5.87 | 5.88 | 97.8 ±1.8 | 98.0 ± 1.8 |
| 10.0 | 9.8 | 9.8 | 98.0 ±1.7 | 99.0 ± 1.7 |
| 50.0 | 49.0 | 49.5 | 98.0 ±1.6 | 99.0 ±1.6 |
| 100.0 | 99.4 | 99.5 | 99.4 ±1.5 | 99.5 ± 1.5 |
| 200.0 | 198.0 | 199.0 | 99.0 ±1.4 | 99.5± 1.5 |
| 300.0 | 298.0 | 298.0 | 99.3 ± 1.4 | 99.3 ± 1.4 |
| 370.0 | 369.0 | 369.0 | 99.7 ± 1.4 | 99.7 ± 1.4 |
| Drug (trade name) | Nominal value of AC, (mg/tablet) | AC, content ±RSD (%) (mg/tablet) (n=5) | Official, ** method content ±RSD(%) (mg/tablet) (n=5) | |t|2 AC- TPB(PM) | F AC- TPB(PM) | |
|---|---|---|---|---|---|---|
| AC-TPB | AC-PM | |||||
| Sectral tablets | 200mg | 197.0 ± 1.4 | 198.0 ± 1.4 | 197.0 ± 1.5 | 1.3 (1.2) | 1.5 (1.4) |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Mostafa, G.A.-H.; Hefnawy, M.M.; Al-Majed, A. PVC Membrane Sensors for Potentiometric Determination of Acebutolol. Sensors 2007, 7, 3272-3286. https://doi.org/10.3390/s7123272
Mostafa GA-H, Hefnawy MM, Al-Majed A. PVC Membrane Sensors for Potentiometric Determination of Acebutolol. Sensors. 2007; 7(12):3272-3286. https://doi.org/10.3390/s7123272
Chicago/Turabian StyleMostafa, Gamal Abdel-Hafiz, Mohamed Mahmoud Hefnawy, and Abdulrahman Al-Majed. 2007. "PVC Membrane Sensors for Potentiometric Determination of Acebutolol" Sensors 7, no. 12: 3272-3286. https://doi.org/10.3390/s7123272




