Preparation of Silver Nanoparticle and Its Application to the Determination of ct-DNA
Abstract
:1. Introduction
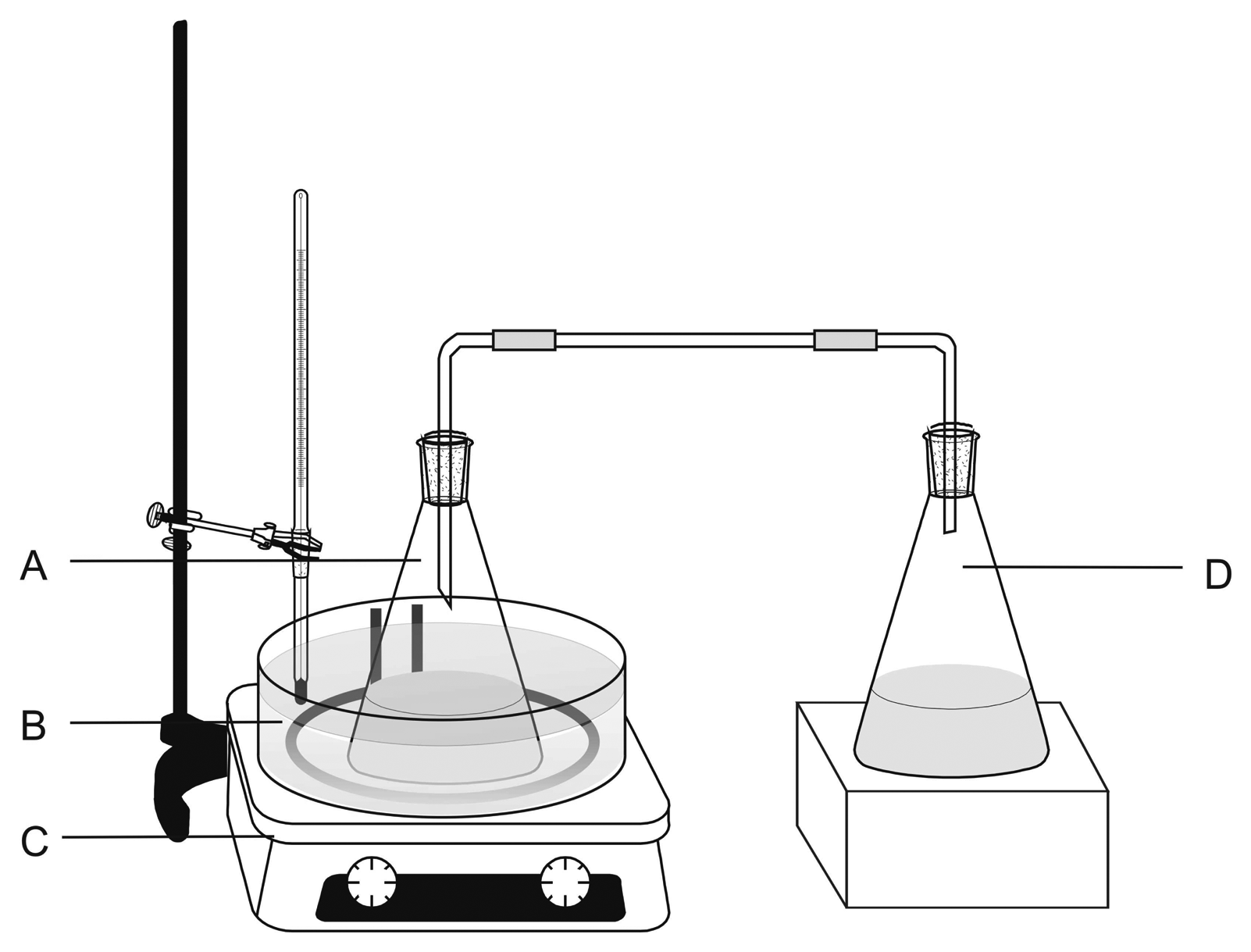
2. Experimental
3. Results and Discussion
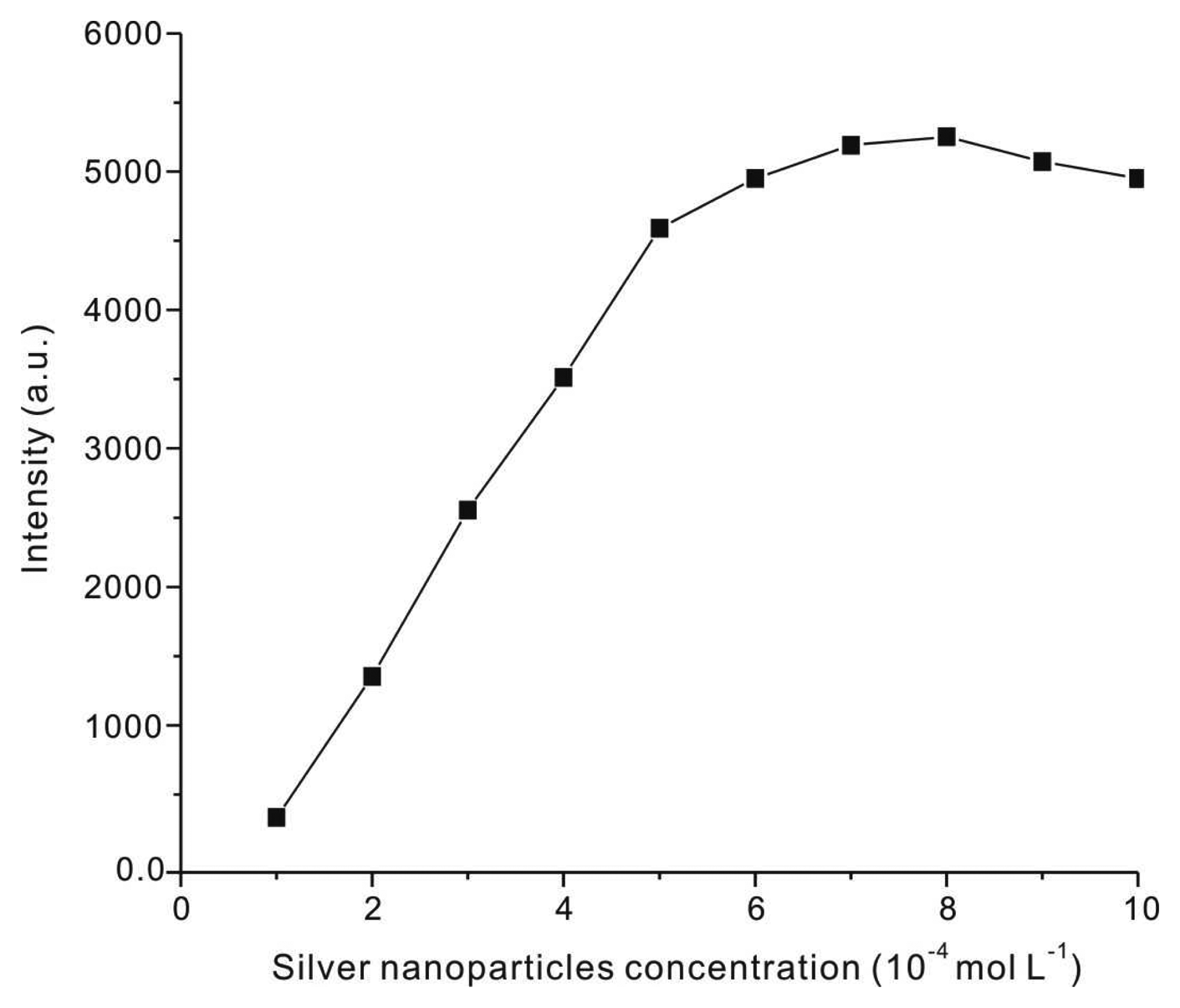
3.1. Optimization of General Procedure
3.1.1. Effect of Reactant Concentrations
3.1.2. Effect of Reaction Time
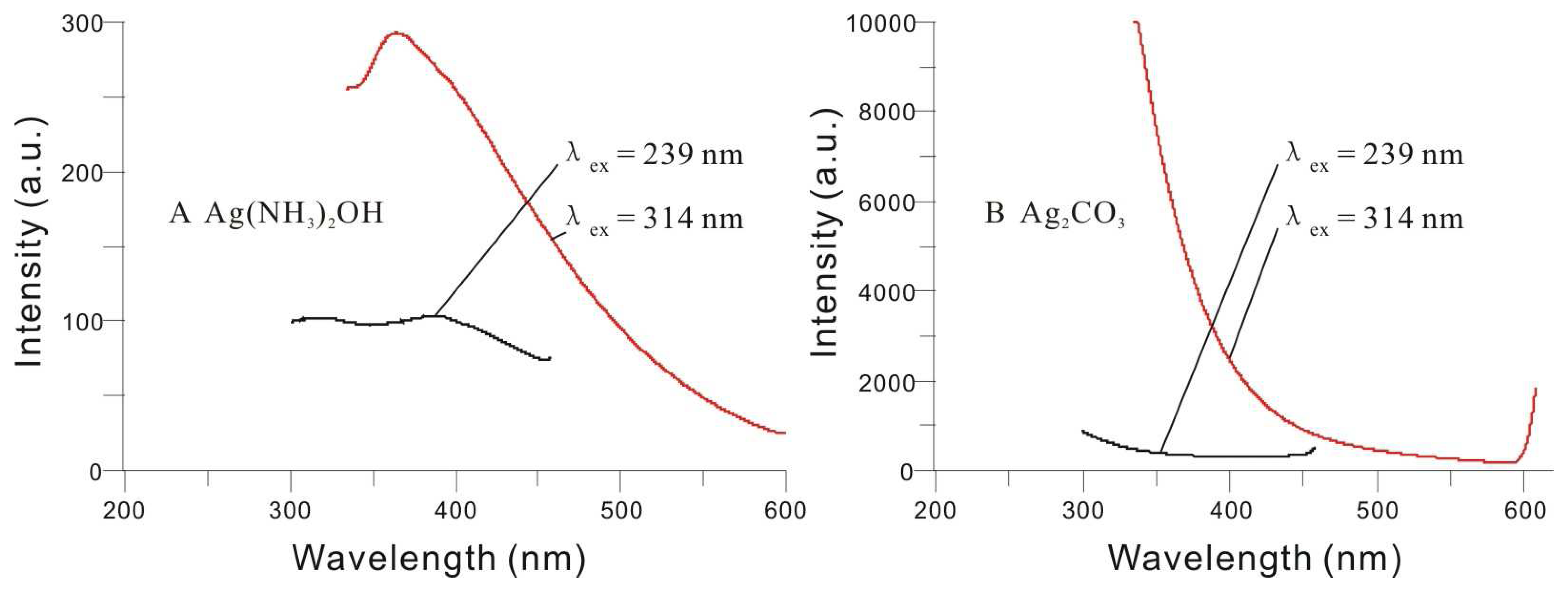
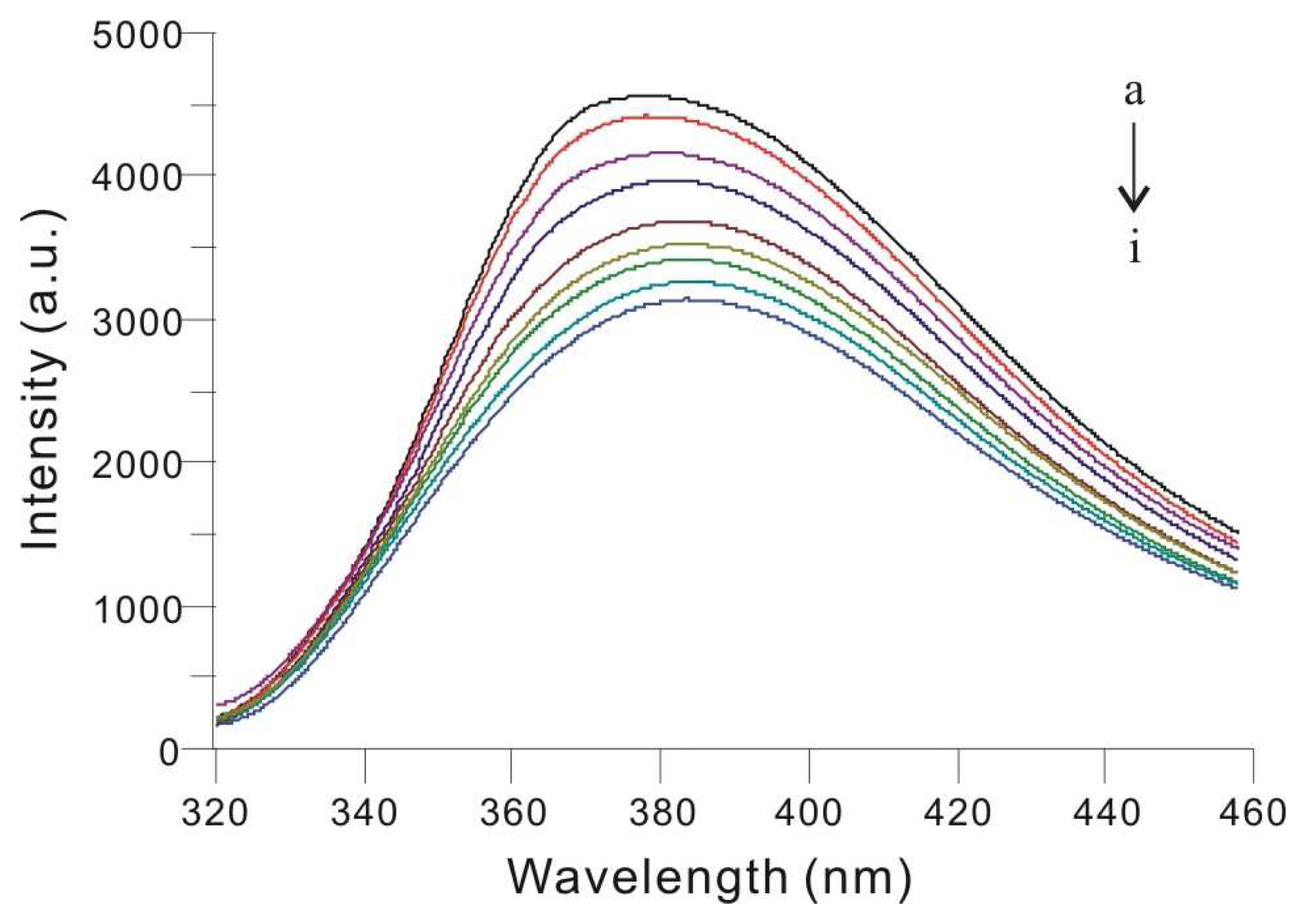
3.2. Fluorescence Spectral Characteristics of the Silver Nanoparticles
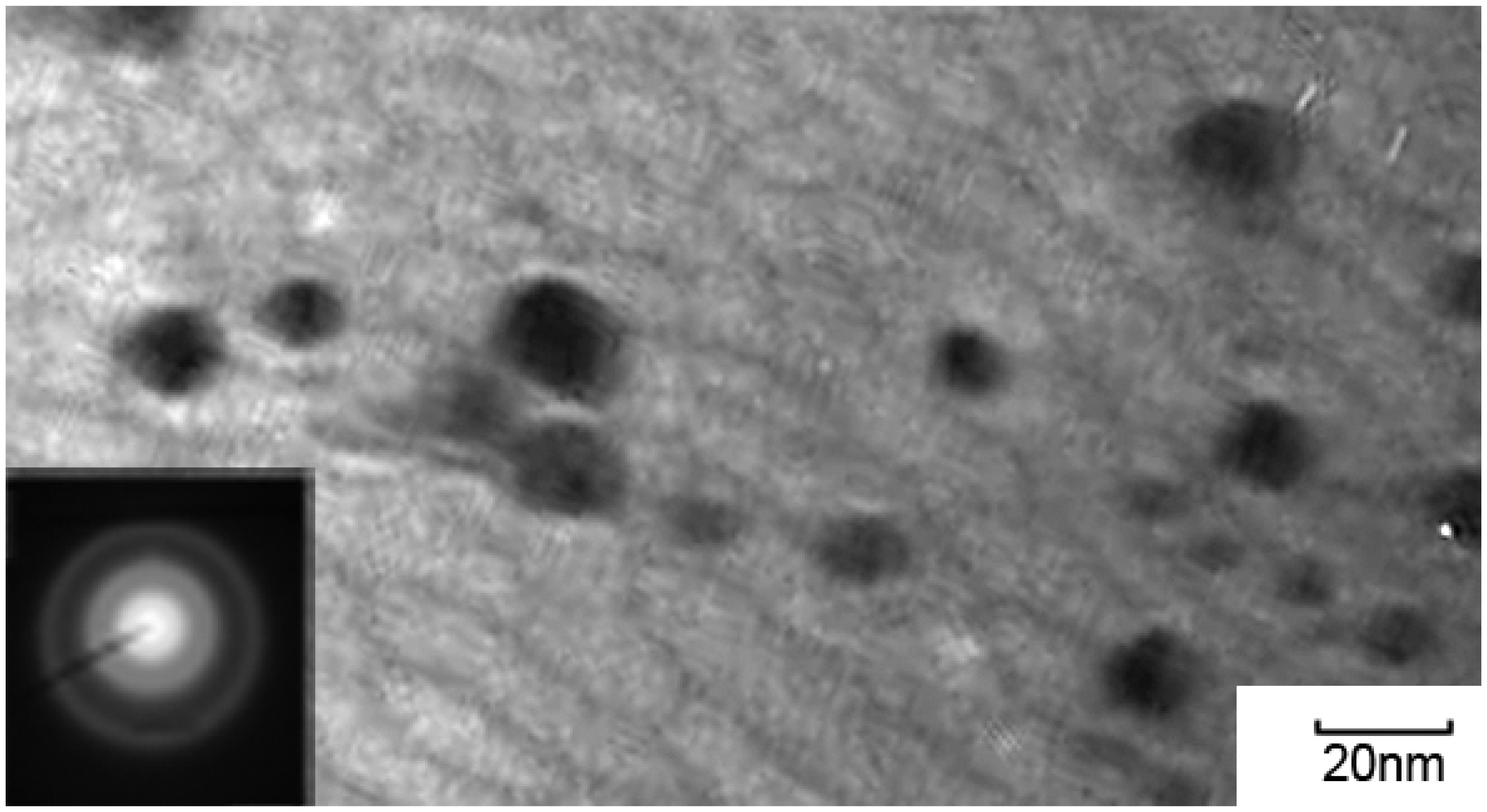
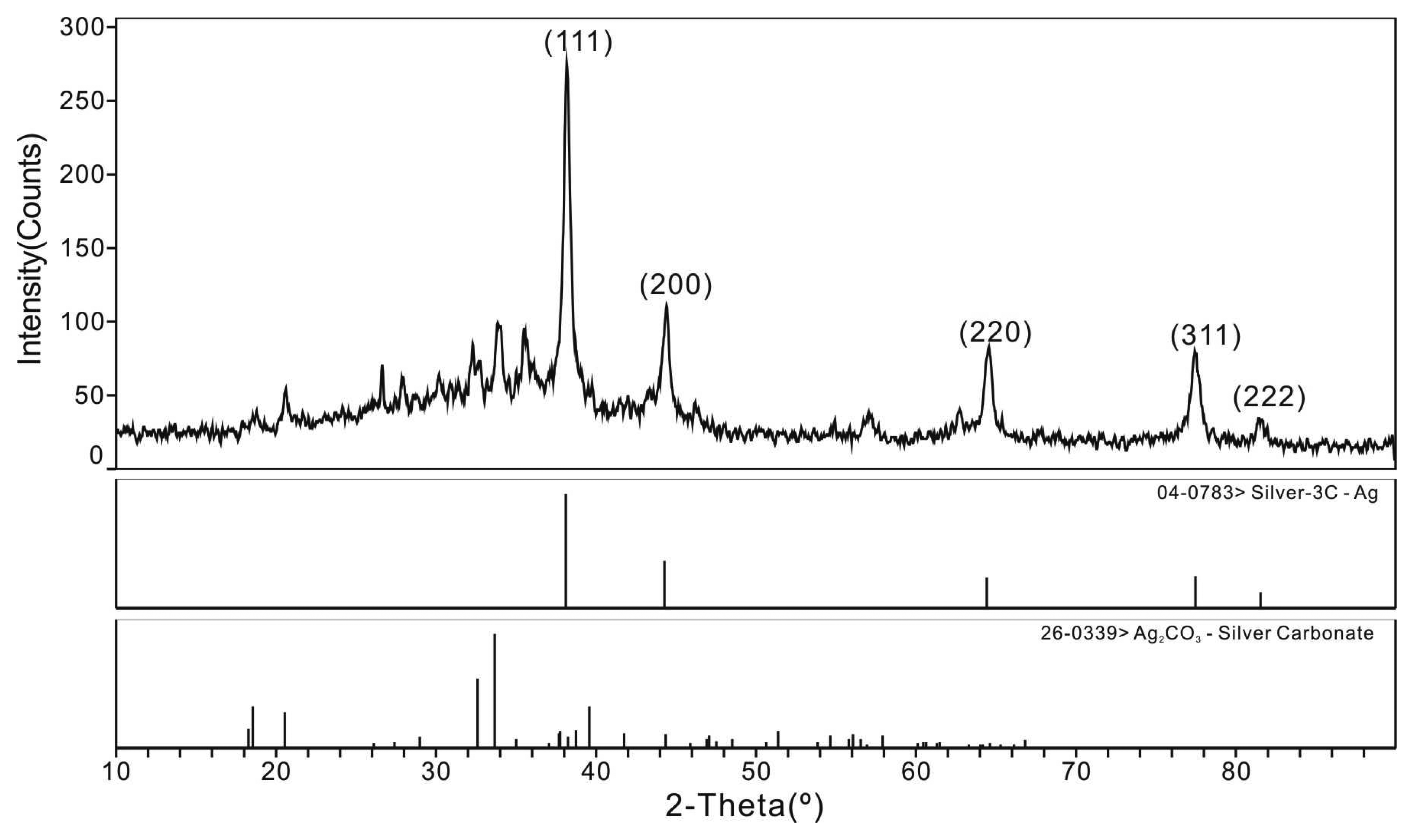
3.3. TEM, ED and XRD Analysis
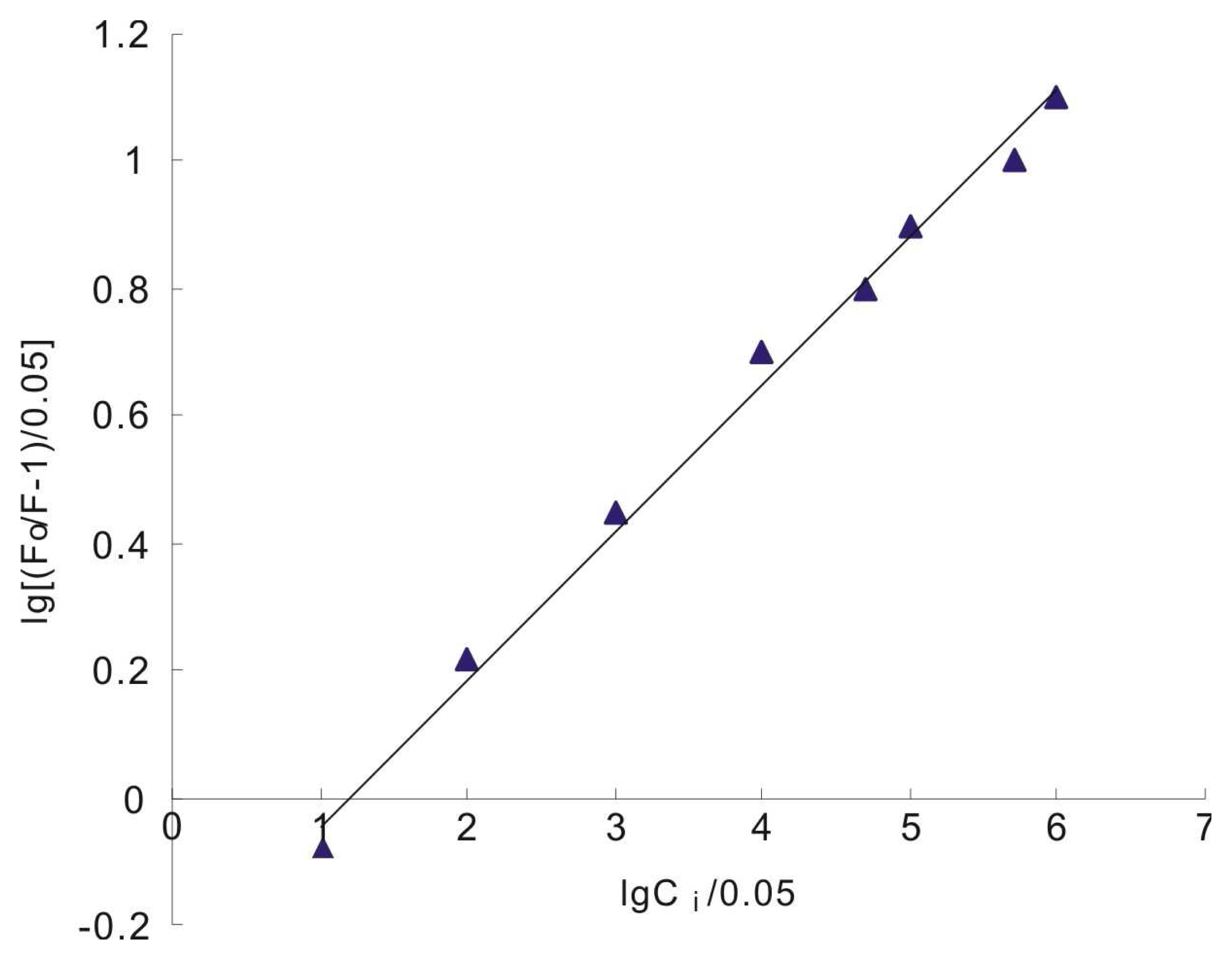
3.4. Determination of ct-DNA
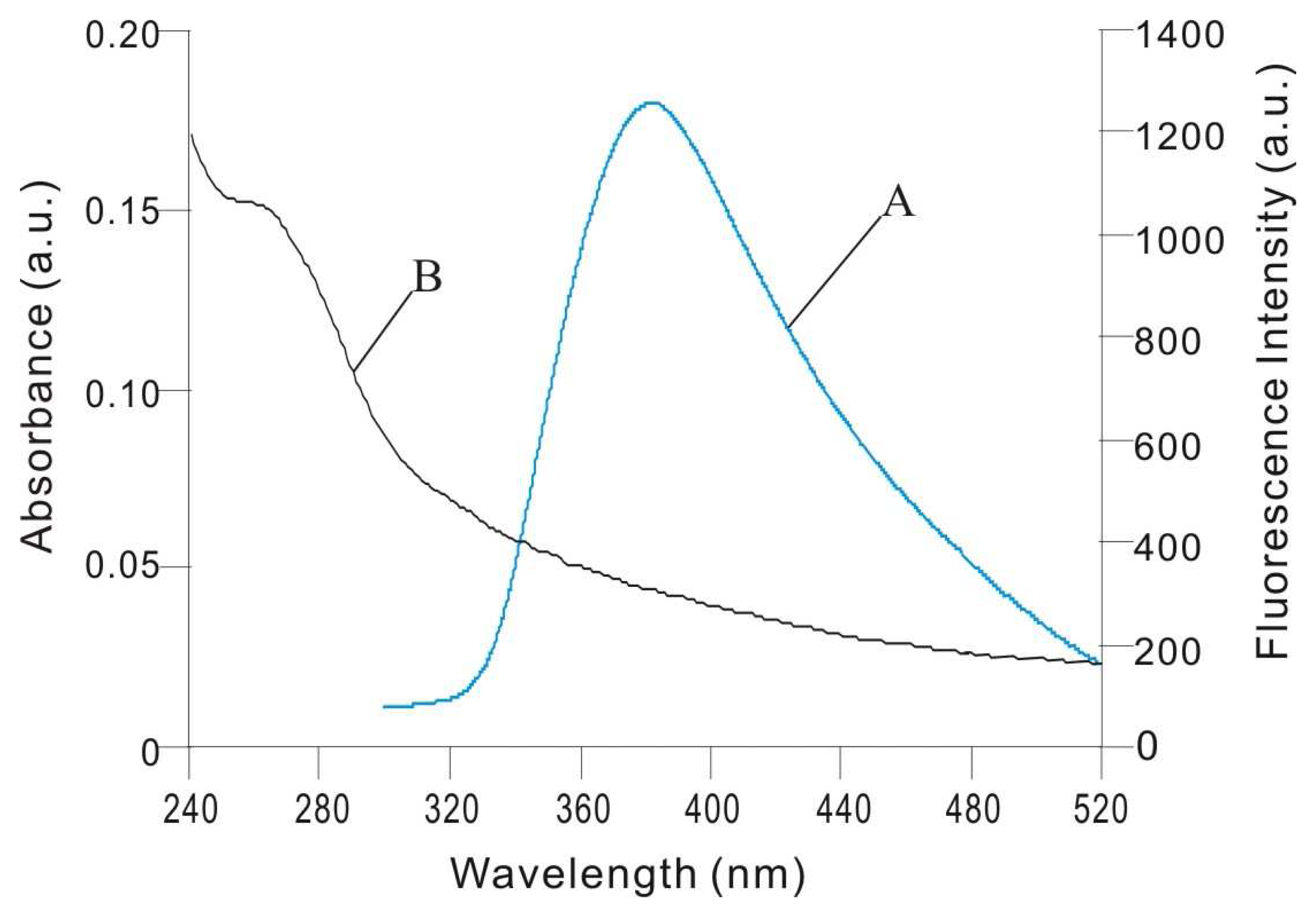
3.5. Energy Transfer between the Silver Nanoparticles and ct-DNA
3.6 Interference of the Coexisting Foreign Substances
4. Conclusions
Acknowledgments
References
- Yin, B.; Ma, H.; Wang, S.; Chen, S. Electrochemical synthesis of silver nanoparticles under protection of poly(N-vinylpyrrolidone). J. Phys. Chem. B. 2003, 107, 8898–8904. [Google Scholar]
- Penner, R.M. Mesoscopic metal particles and wires by electrodeposition. J. Phys. Chem. B. 2002, 106, 3339–3353. [Google Scholar]
- Raveendran, P.; Fu, J.; Wallen, S.L. Completely “green” synthesis and stabilization of metal nanoparticles. J. Am. Chem. Soc. 2003, 125, 13940–13941. [Google Scholar]
- Lin, X.Z.; Teng, X.; Yang, H. Direct synthesis of narrowly dispersed silver nanoparticles using a single-source precursor. Langmuir 2003, 19, 10081–10085. [Google Scholar]
- Carotenuto, G. Synthesis and characterization of poly(N-vinylpyrrolidone) filled by monodispersed silver clusters with controlled size. Appl. Organometal. Chem. 2001, 15, 344–351. [Google Scholar]
- Okitsu, K.; Yue, A.; Tanabe, S.; Matsumoto, H. Sonochemical preparation and catalytic behavior of highly dispersed palladium nanoparticles on alumina. Chem. Mater. 2000, 12, 3006–3011. [Google Scholar]
- Ghosh, K.; Maiti, S.N. Mechanical properties of silver-powder-filled polypropylene composites. J. Appl. Polym. Sci. 1996, 60, 323–331. [Google Scholar]
- Nersisyan, H.H.; Lee, J.H.; Son, H.T.; Won, C.W.; Maeng, D.Y. A new and effective chemical reduction method for preparation of nanosized silver powder and colloid dispersion. Mater. Res. Bull. 2003, 38, 949–956. [Google Scholar]
- Rabin, I.; Schulze, W.; Ertl, G.; Felix, C.; Sieber, C.; Harbich, W.; Buttet, J. Absorption and fluorescence spectra of Ar-matrix-isolated Ag3 clusters. J. Chem. Phys. Lett. 2000, 320, 59–64. [Google Scholar]
- Geddes, C.D.; Parfenov, A.; Gryczynski, I.; Lakowicz, J.R. Luminescent blinking from silver nanostructures. J. Phys. Chem. B. 2003, 107, 9989–9993. [Google Scholar]
- Jiang, Z.; Yuan, W.; Pan, H. Luminescence effect of silver nanoparticle in water phase. Spectrochim. Acta A. 2005, 61, 2488–2494. [Google Scholar]
- Maali, A.; Cardinal, T.; Tréguer-Delapierre, M. Intrinsic fluorescence from individual silver nanoparticles. Physica E. 2003, 17, 559–560. [Google Scholar]
- Evanoff, D.D., Jr.; Chumanov, G. Size-controlled synthesis of nanoparticles. 1. “silver-only” aqueous suspensions via hydrogen reduction. J. Phys. Chem. B. 2004, 108, 13948–13956. [Google Scholar]
- Hailstone, R.K. Computer simulation studies of silver cluster formation on AgBr microcrystals. J. Phys. Chem. 1995, 99, 4414–4428. [Google Scholar]
- Shiraishi, Y.; Toshima, N. Colloidal silver catalysts for oxidation of ethylene. J. Mol. Catal. A: Chem. 1999, 141, 187–192. [Google Scholar]
- Sclafani, A.; Mozzanega, M.; Pichat, P. Effect of silver deposits on the photocatalytic activity of titanium dioxide samples for the dehydrogenation or oxidation of 2-propanol. J. Photochem. Photobiol. A: Chem. 1991, 59, 181–189. [Google Scholar]
- Tada, H.; Teranishi, K.; Inubushi, Y.-i.; Ito, S. Ag nanocluster loading effect on TiO2 photocatalytic reduction of bis(2-dipyridyl)disulfide to 2-mercaptopyridine by H2O. Langmuir 2000, 16, 3304–3309. [Google Scholar]
- Shirtcliffe, N.; Nickel, U.; Schneider, S. Reproducible preparation of silver sols with small particle size using borohydride reduction: for use as nuclei for preparation of larger particles. J. Colloid. Interface. Sci. 1999, 211, 122–129. [Google Scholar]
- Bright, R.M.; Musick, M.D.; Natan, M.J. Preparation and characterization of Ag colloid monolayers. Langmuir 1998, 14, 5695–5701. [Google Scholar]
- Wang, X.M.; Li, Y.; Gong, S.J.; Fu, D.G. A spectroscopic study on the DNA binding behavior of the anticancer drug dacarbazine. Spectrosc. Lett. 2002, 35, 751–756. [Google Scholar]
- Park, S.J.; Taton, T. A.; Mirkin, C. A. Arry-based electrical detection of DNA with nanoparticle. Scienc. 2002, 295, 1503–1506. [Google Scholar]
- Li, J.; Xue, M.; Wang, H.; Cheng, L.; Gao, L.; Lu, Z.; Chan, M. Amplifying the electrical hybridization signals of DNA array by multilayer assembly of Au nanoparticles probes. Analyst 2003, 128, 917–923. [Google Scholar]
- Zhang, Y.; Kim, H.H.; Heller, A. Enzyme-amplified amperometric detection of 3000 copies of DNA in a 10 μL droplet. Anal. Chem. 2003, 75, 3267–3269. [Google Scholar]


| No. | Reagents | Concentration (mol L-1) | Phenomena | |
|---|---|---|---|---|
| Precipitation | Fluorescence | |||
| 1 | AgNO3 | 1.0 × 10-5 | No | No |
| NH3.H2O | 1.0 × 10-5 | |||
| 2 | AgNO3 | 1.0 × 10-5 | No | No |
| NH3.H2O | 1.0 | |||
| 3 | AgNO3 | 1.0 × 10-4 | No | No |
| NH3.H2O | 1.0 | |||
| 4 | AgNO3 | 1.0 × 10-3 | No | Yes |
| NH3.H2O | 1.0 | |||
| 5 | AgNO3 | 1.0 × 10-2 | Yes | Yes |
| NH3.H2O | 1.0 | |||
| 6 | AgNO3 | 1.0 | Yes | Yes |
| NH3.H2O | 1.0 | |||
| No. | Reaction time (hours) | Phenomena | |
|---|---|---|---|
| Precipitation | Fluorescence | ||
| 1 | 2 | No | No |
| 2 | 6 | No | No |
| 3 | 10 | No | No |
| 4 | 20 | No | No |
| 5 | 30 | No | Weak |
| 6 | 40 | No | Weak |
| 7 | 53 | No | Strong |
| 8 | 54 | No | Strong |
| 9 | 55 | No | Strong |
| 10 | 60 | Yes | Strong |
| Co-existing Substance | Co-existing concentration (μg mL-1) | Change in intensity of fluorescence (%) |
|---|---|---|
| Bovine Serum Albumin | 5.0 | +2.1 |
| Glucose | 20.0 | +2.0 |
| L-Lysine | 15.0 | -3.3 |
| L-Tyrosine | 10.0 | -4.0 |
| Glutamic acid | 16.0 | +1.5 |
| Glysine | 13.0 | +2.1 |
| L-Histidine | 5.0 | +1.2 |
| SO42- | 5.5 | +1.5 |
| PO43- | 2.0 | +4.0 |
| Cl- | 6.0 | +3.5 |
| I- | 5.5 | +3.7 |
| Zn2+ | 10.0 | +1.6 |
| Cu2+ | 2.0 | +4.1 |
| Mg2+ | 15.0 | +1.8 |
| Fe3+ | 6.0 | +3.5 |
| Ca2+ | 15.0 | +1.9 |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Liu, C.; Yang, X.; Yuan, H.; Zhou, Z.; Xiao, D. Preparation of Silver Nanoparticle and Its Application to the Determination of ct-DNA. Sensors 2007, 7, 708-718. https://doi.org/10.3390/s7050708
Liu C, Yang X, Yuan H, Zhou Z, Xiao D. Preparation of Silver Nanoparticle and Its Application to the Determination of ct-DNA. Sensors. 2007; 7(5):708-718. https://doi.org/10.3390/s7050708
Chicago/Turabian StyleLiu, Chunhua, Xiupei Yang, Hongyan Yuan, Zaide Zhou, and Dan Xiao. 2007. "Preparation of Silver Nanoparticle and Its Application to the Determination of ct-DNA" Sensors 7, no. 5: 708-718. https://doi.org/10.3390/s7050708




