A Novel Type of Tri-Colour Light-Emitting-Diode-Based Spectrometric Detector for Low-Budget Flow-Injection Analysis
Abstract
:1. Introduction
2. Results and Discussion
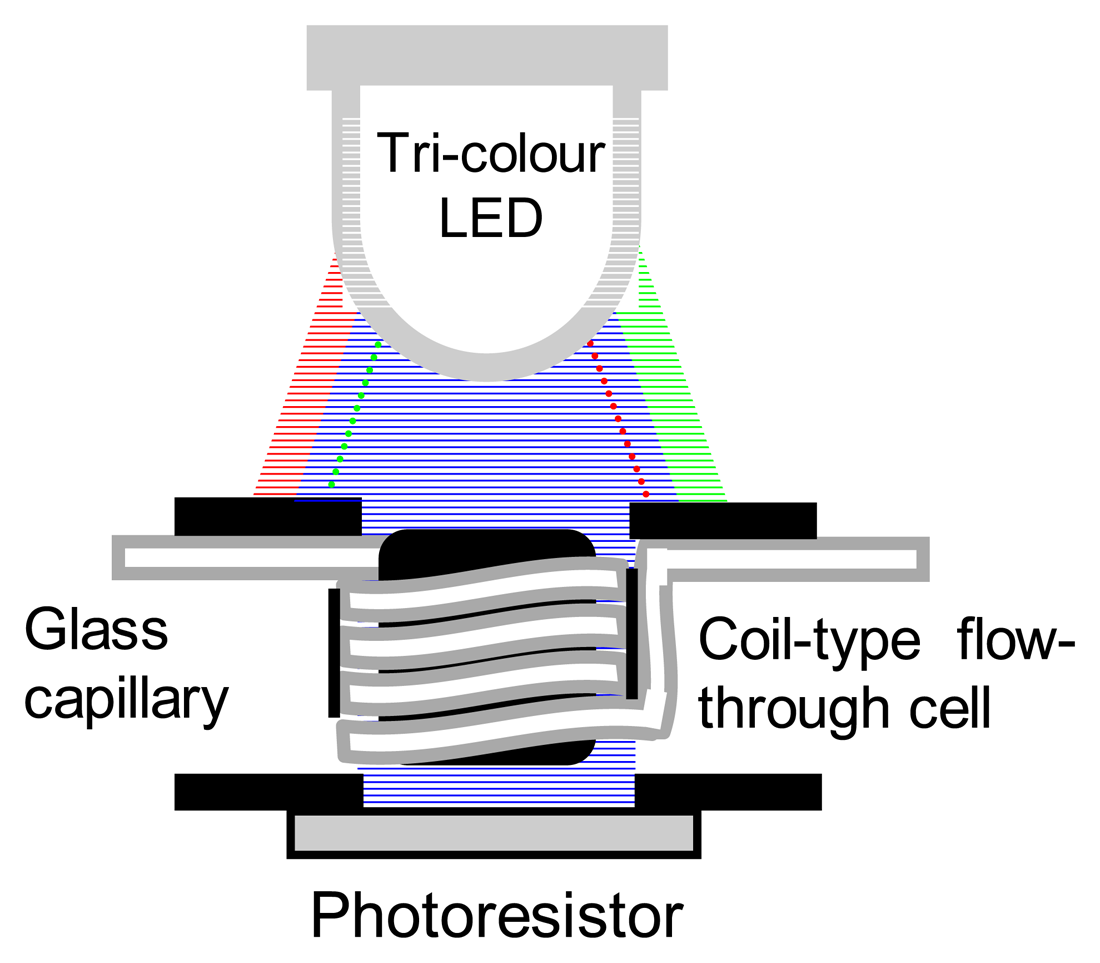
2.1. Optical geometry of the tri-colour LED-based spectrometric detector
2.2 Performance of the low-cost FIA with the tri-colour LED-based spectrometric detector
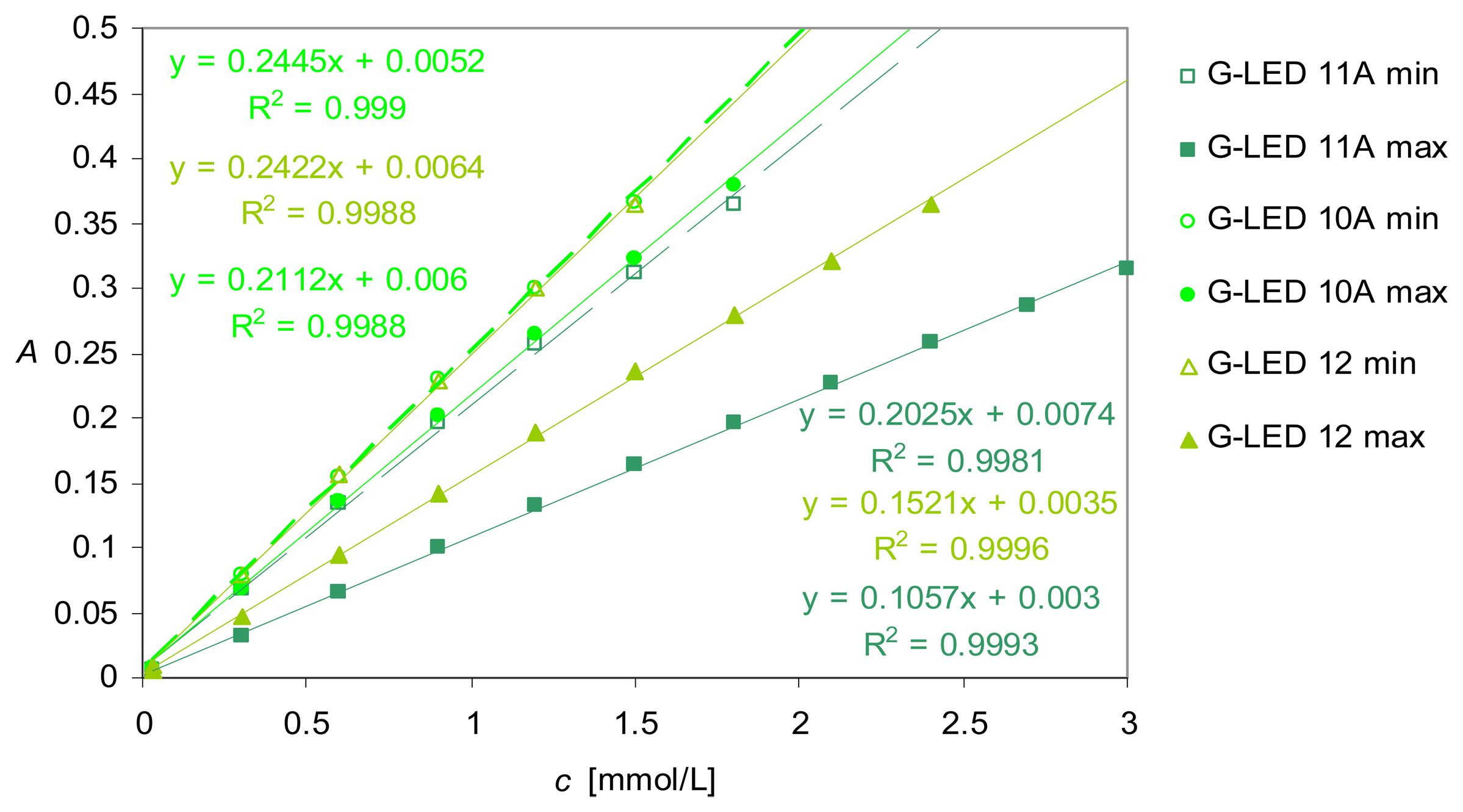
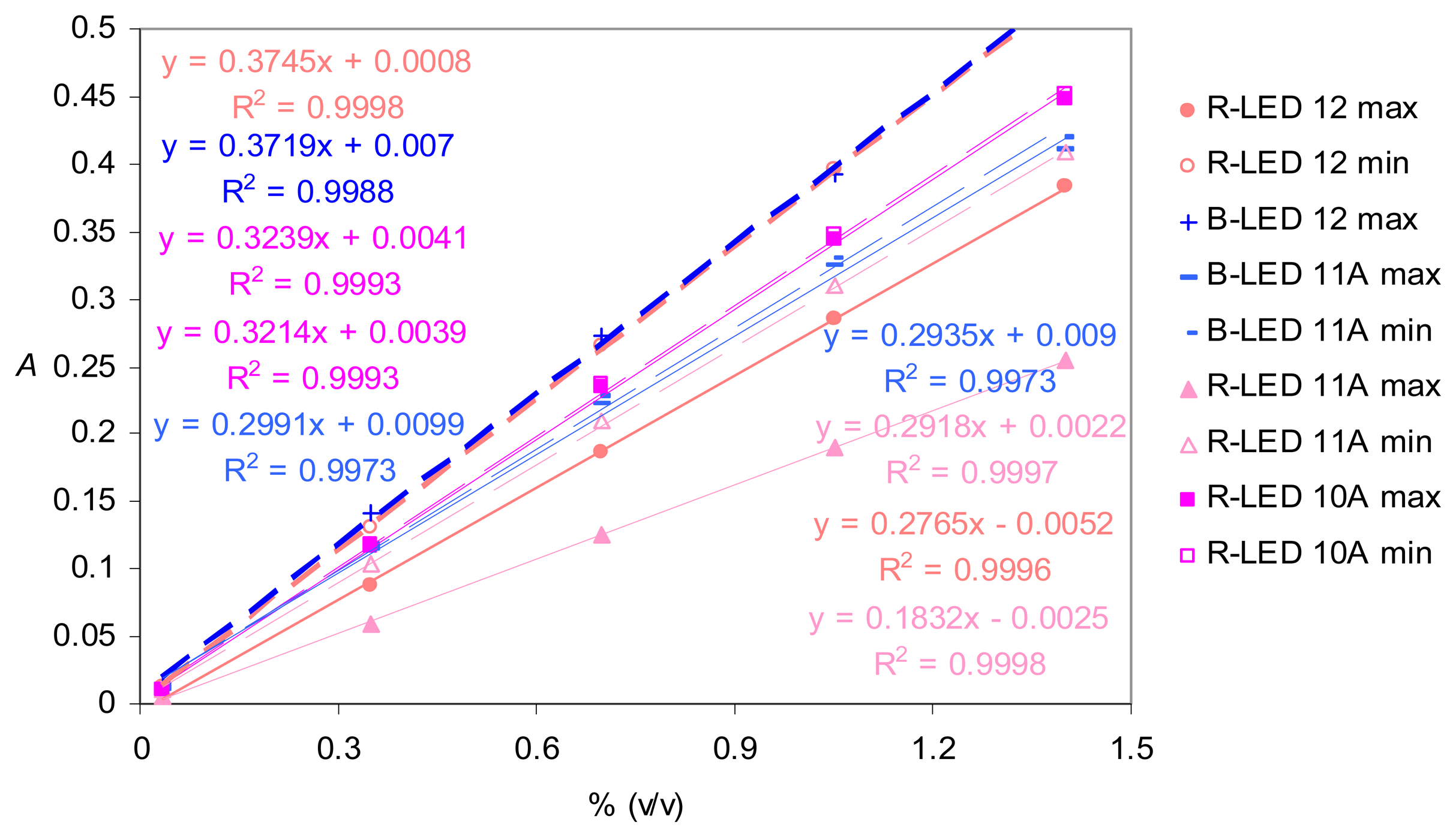
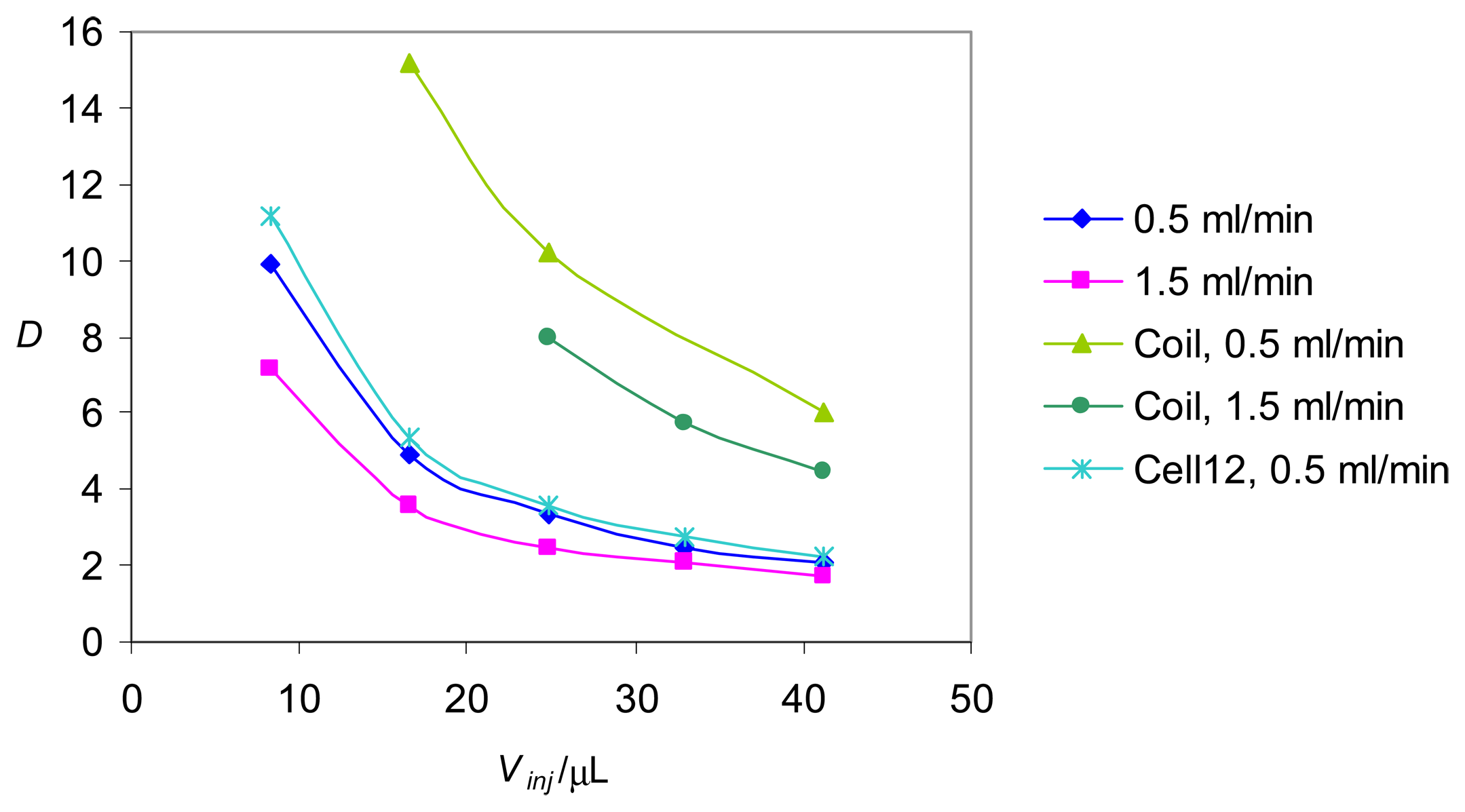
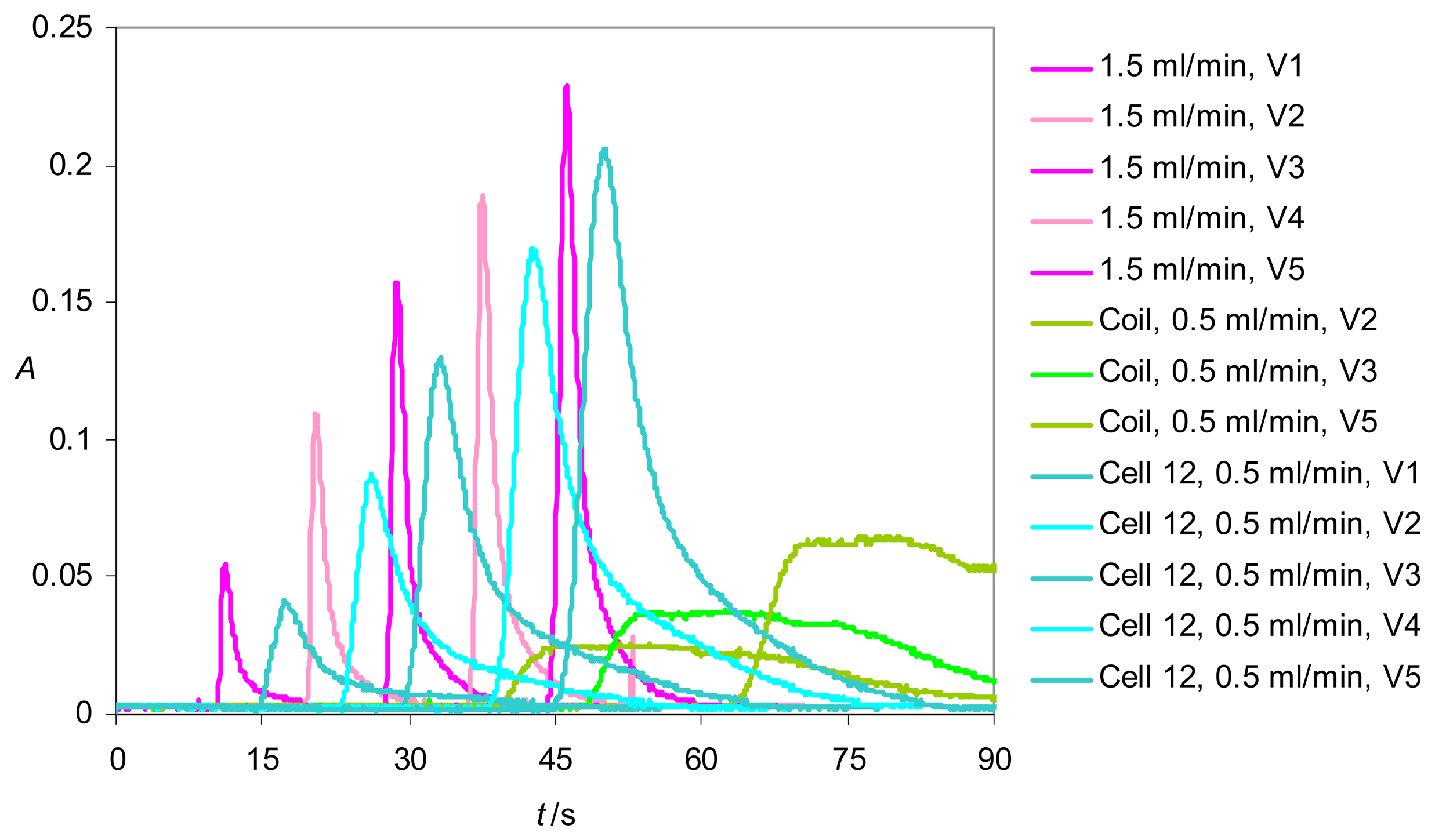
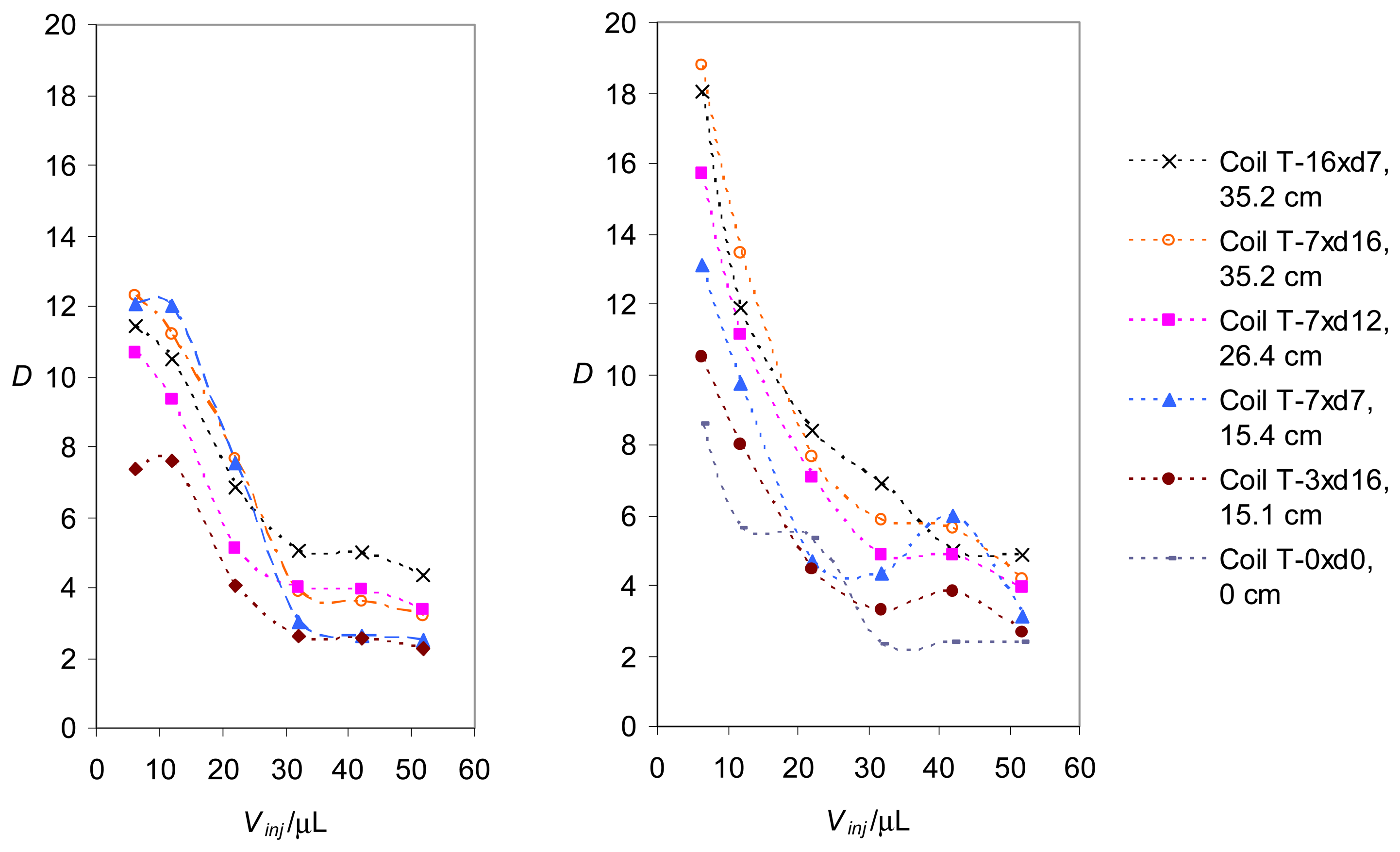
Determination of the dispersion coefficients
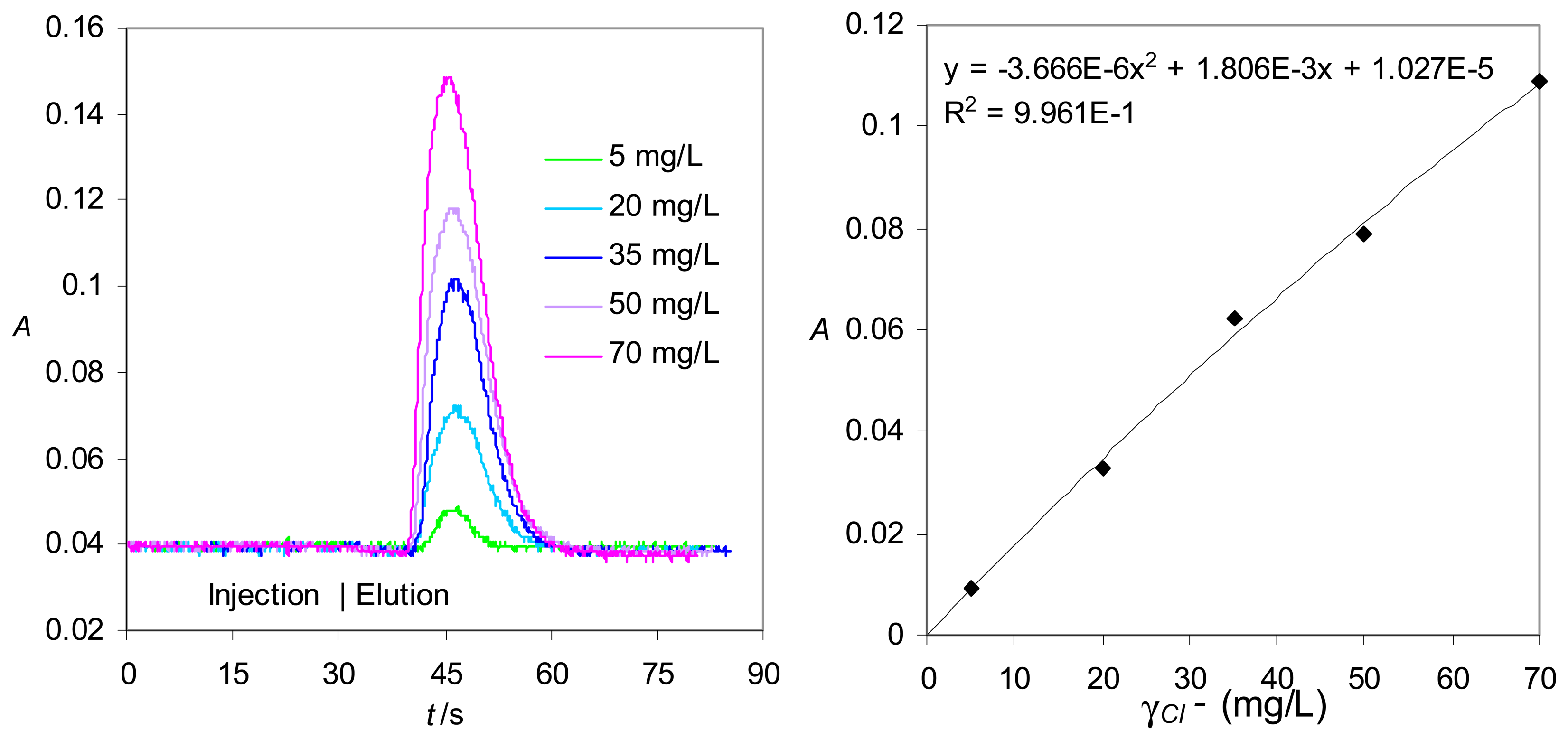
Real-life application in the blue light range - Chloride determination
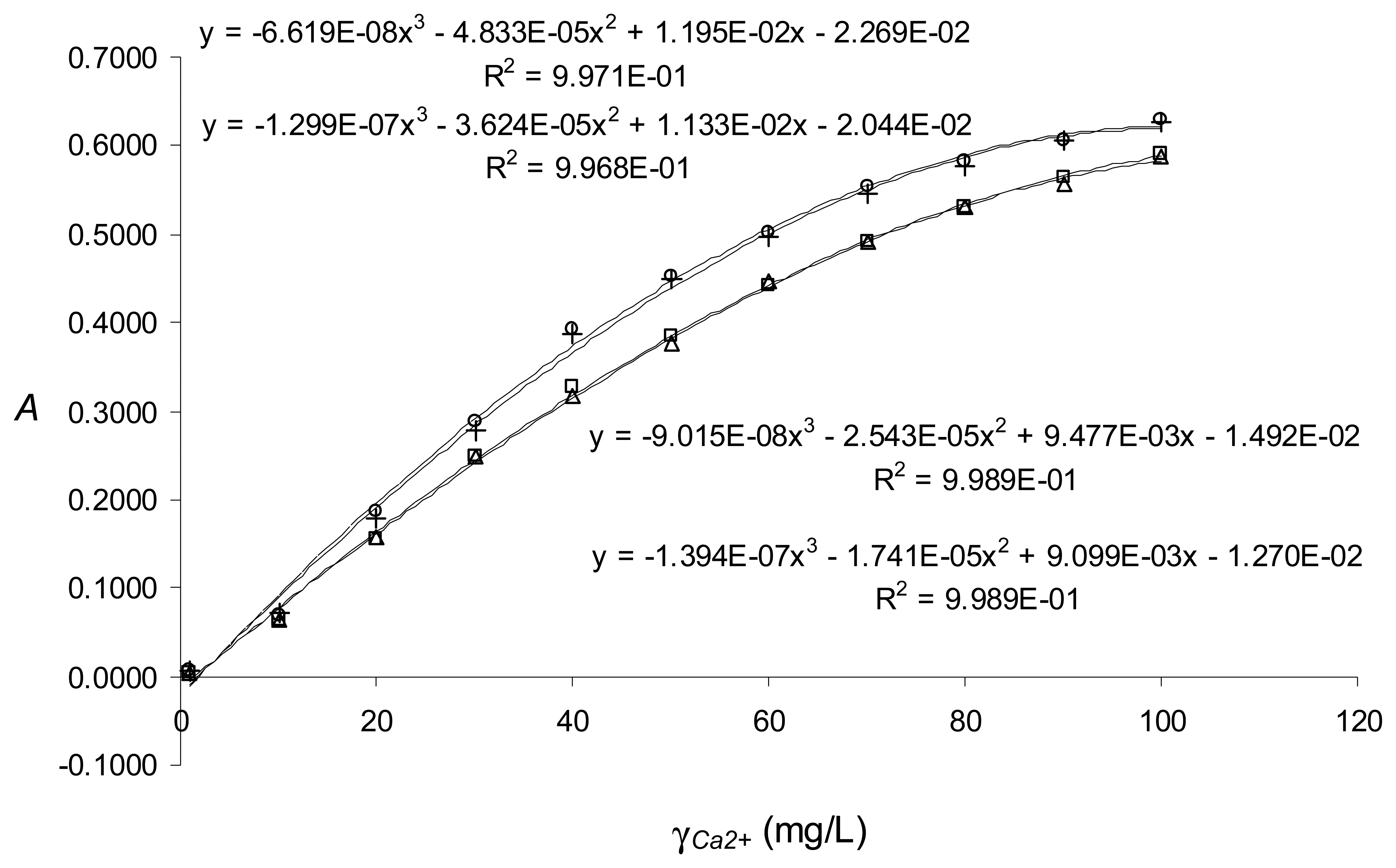
Real-life application in the green light range - Calcium determination with a data-point sampling rate limited to one measurement per second
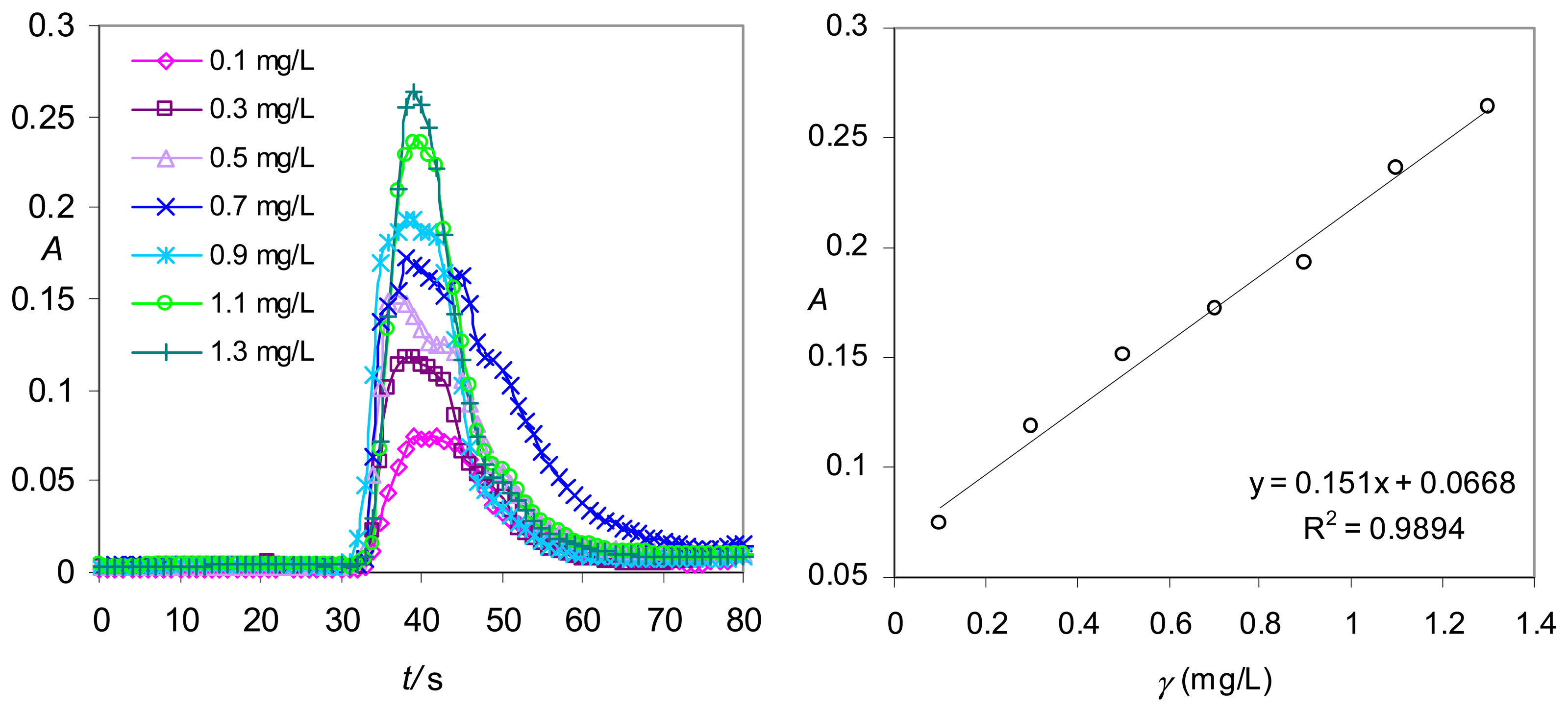
Real-life application in the red light range – Hydrogen phosphate determination with a data-point sampling rate limited to one measurement per second
3. Experimental Section
3.1. Prototyping of a tri-colour LED-based detector
Optical geometry
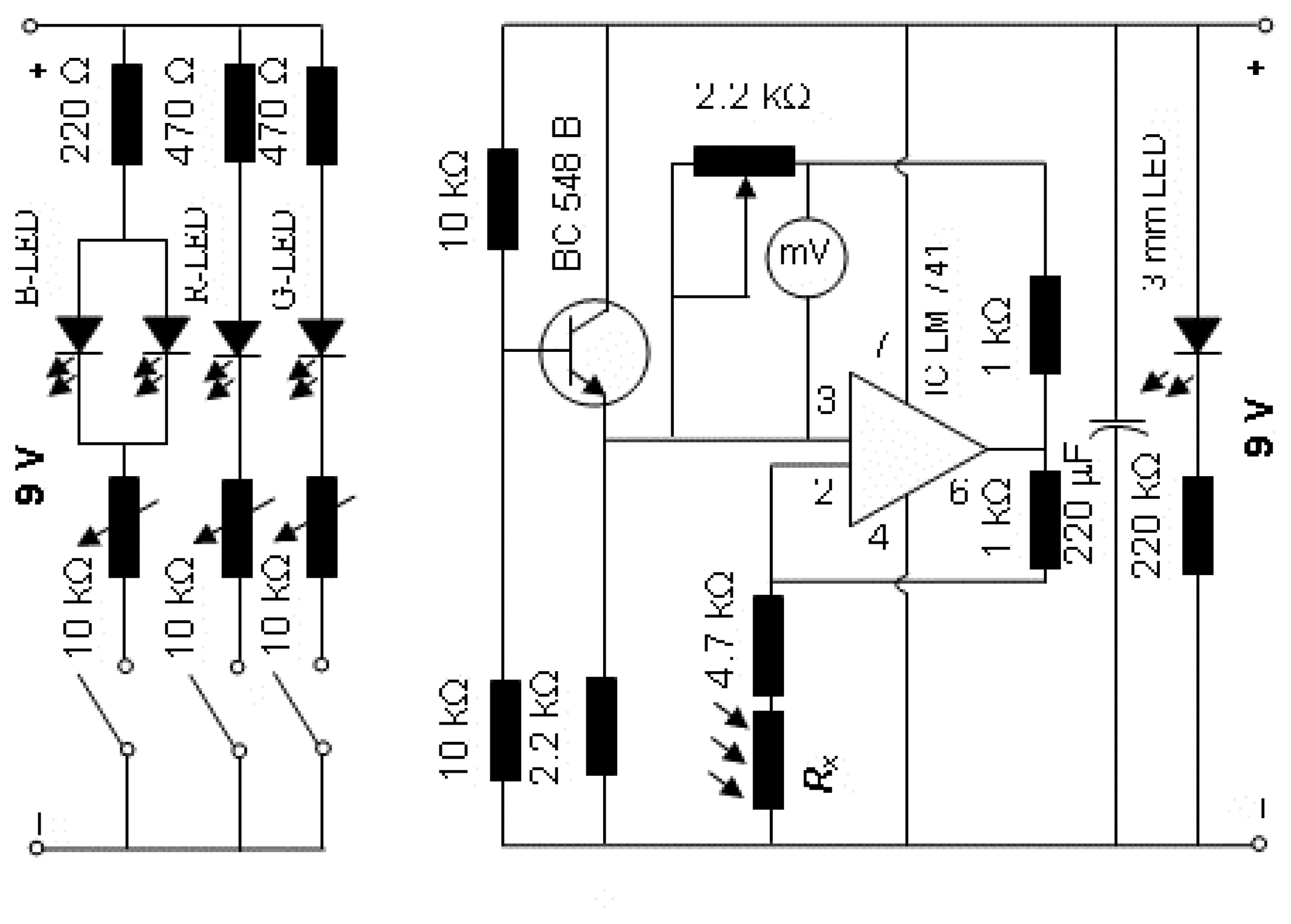
Electronic circuitry
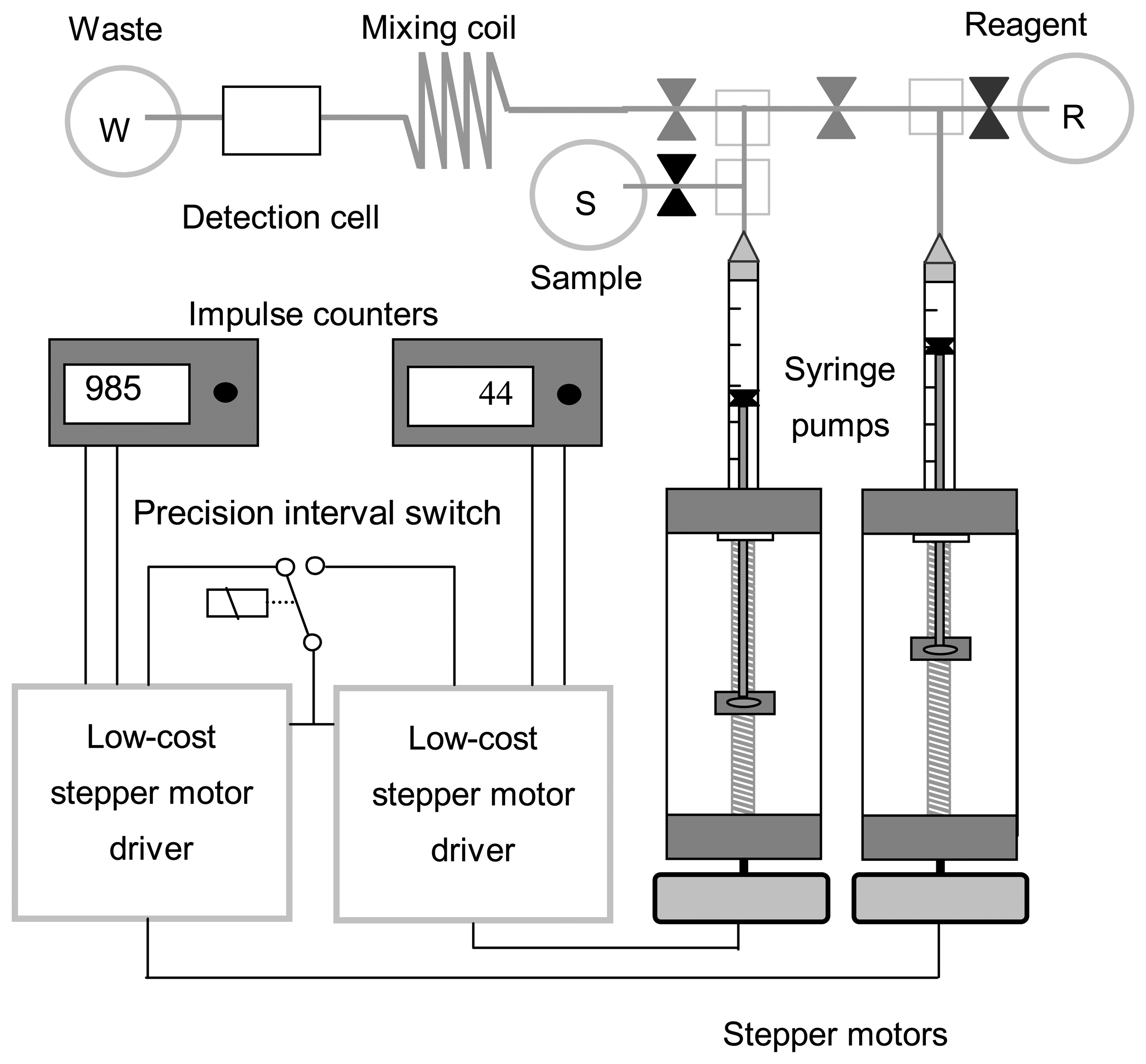
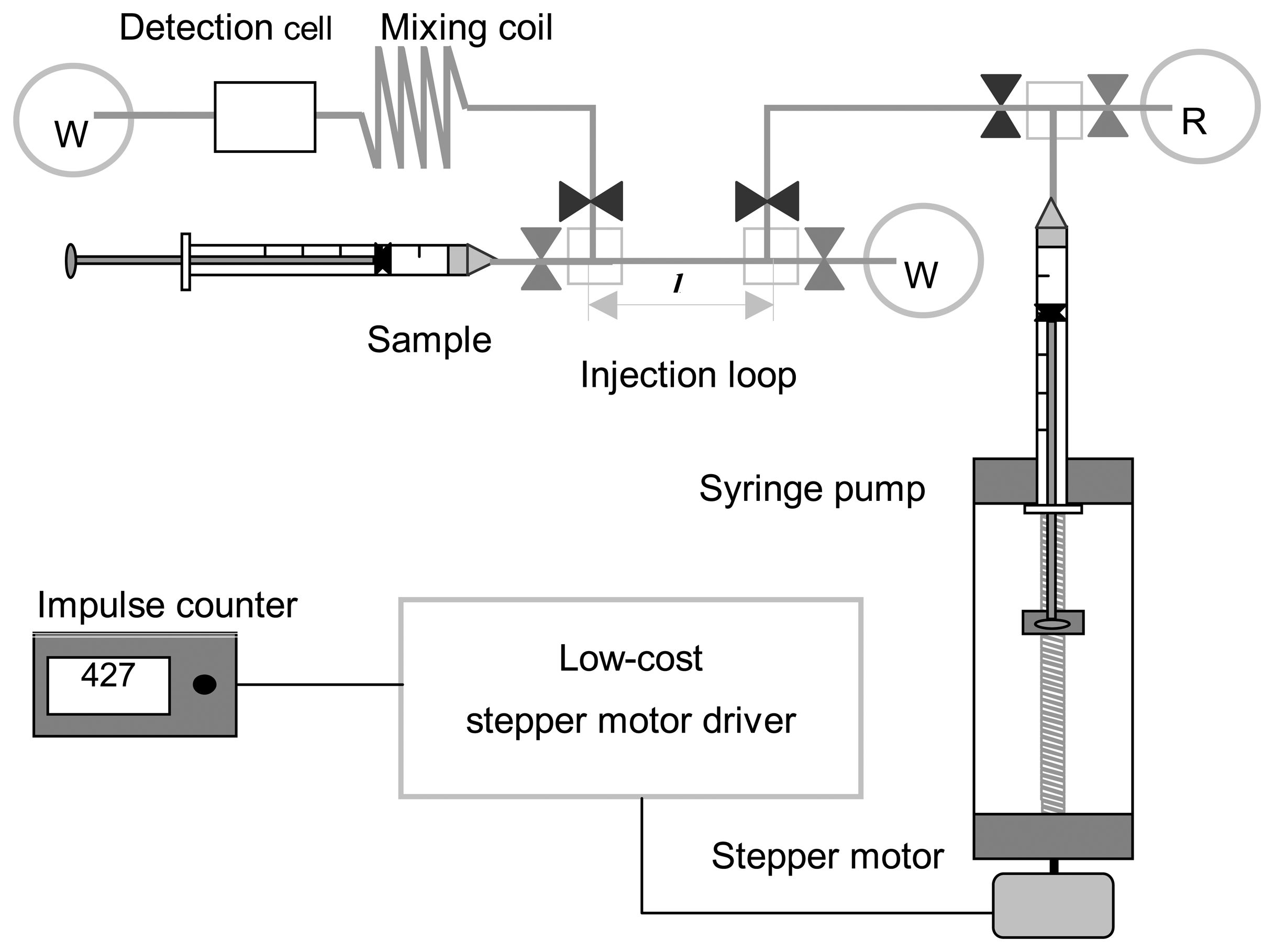
3.2. Prototyping of a low-cost FIA with time-based injection
3.3 Performance tests
Conclusions
Acknowledgments
References
- Flaschka, H.; McKeithan, C.; Barnes, R. Light emitting diodes and phototransistors in photometric modules. Anal. Lett. 1973, 6, 585–594. [Google Scholar]
- Betteridge, D.; Dagless, E.L.; Fields, B.; Graves, N.F. Highly sensitive flow-through phototransducer for unsegmented continuous-flow analysis demonstrating high-speed spectrophotometry at parts per 109 level and a new method of refractometric determinations. Analyst 1978, 103, 897–908. [Google Scholar]
- Betteridge, D. Flow-injection analysis in the teaching laboratory. Fresenius Z. Anal. Chem. 1982, 312, 441–443. [Google Scholar]
- Worsfold, P.J.; Clinch, J.R.; Casey, H. Spectrophotometric field-monitor for water-quality parameters - the determination of phosphate. Anal. Chim. Acta 1987, 197, 43–50. [Google Scholar]
- Schmidt, G. J.; Scott, R.P.W. Simple and sensitive ion chromatograph for trace-metal determination. Analyst 1984, 109, 997–1002. [Google Scholar]
- Liu, H.; Dasgupta, P.K. A liquid drop: A windowless optical cell and a reactor without walls for flow injection analysis. Anal. Chim. Acta 1996, 326, 13–22. [Google Scholar]
- Jambunathan, S.; Dasgupta, P.K.; Wolcott, D.K.; Marshall, G.D.; Olson, D.C. Optical fiber coupled light emitting diode based absorbance detector with a reflective flow cell. Talanta 1999, 50, 481–490. [Google Scholar]
- Trojanowicz, M.; Szpunar-Lobinska, J. Simultaneous flow-injection determination of aluminium and zinc using LED photometric detection. Anal. Chim. Acta 1990, 230, 125–130. [Google Scholar]
- Huang, J.L.; Liu, H.H.; Tan, A.M.; Xu, J.H.; Zhao, X.N. A dual-wavelength light-emitting diode based detector for flow-injection analysis process analyzers. Talanta 1992, 39, 589–592. [Google Scholar]
- Liu, H.; Dasgupta, P.K. Dual-wavelength photometry with light-emitting-diodes - compensation of refractive-index and turbidity effects in flow-injection analysis. Anal. Chim. Acta 1994, 289, 347–353. [Google Scholar]
- Hauser, P.C.; Rupasinghe, T.W.T.; Cates, N.E. A multiwavelength photometer based on light-emitting diodes. Talanta 1995, 42, 605–612. [Google Scholar]
- Ruzicka, J.; Hansen, E.H. Flow Injection Analysis; Wiley: New York, 1981. [Google Scholar]
- http://home.cogeco.ca/∼rpaisley4/Stepper.html (9 August 2005).
- Gros, N. Spectrometer with microreaction chamber and tri-colour light emitting diode as a light source. Talanta 2004, 62, 143–150. [Google Scholar]
- Yoshimura, K.; Nawata, S.; Kura, G. Gel-phase absorptiometry of phosphate with molybdate and Malachite Green and its Application to Flow Analysis. Analyst 1990, 115, 843–848. [Google Scholar]
| 10a cell | 11a cell | 12 cell | |
| Coil's outer diameter | 4 mm | 5 mm | 6.5 mm |
| Coil's internal volume | 20 μL | 27 μL | 41 μL |
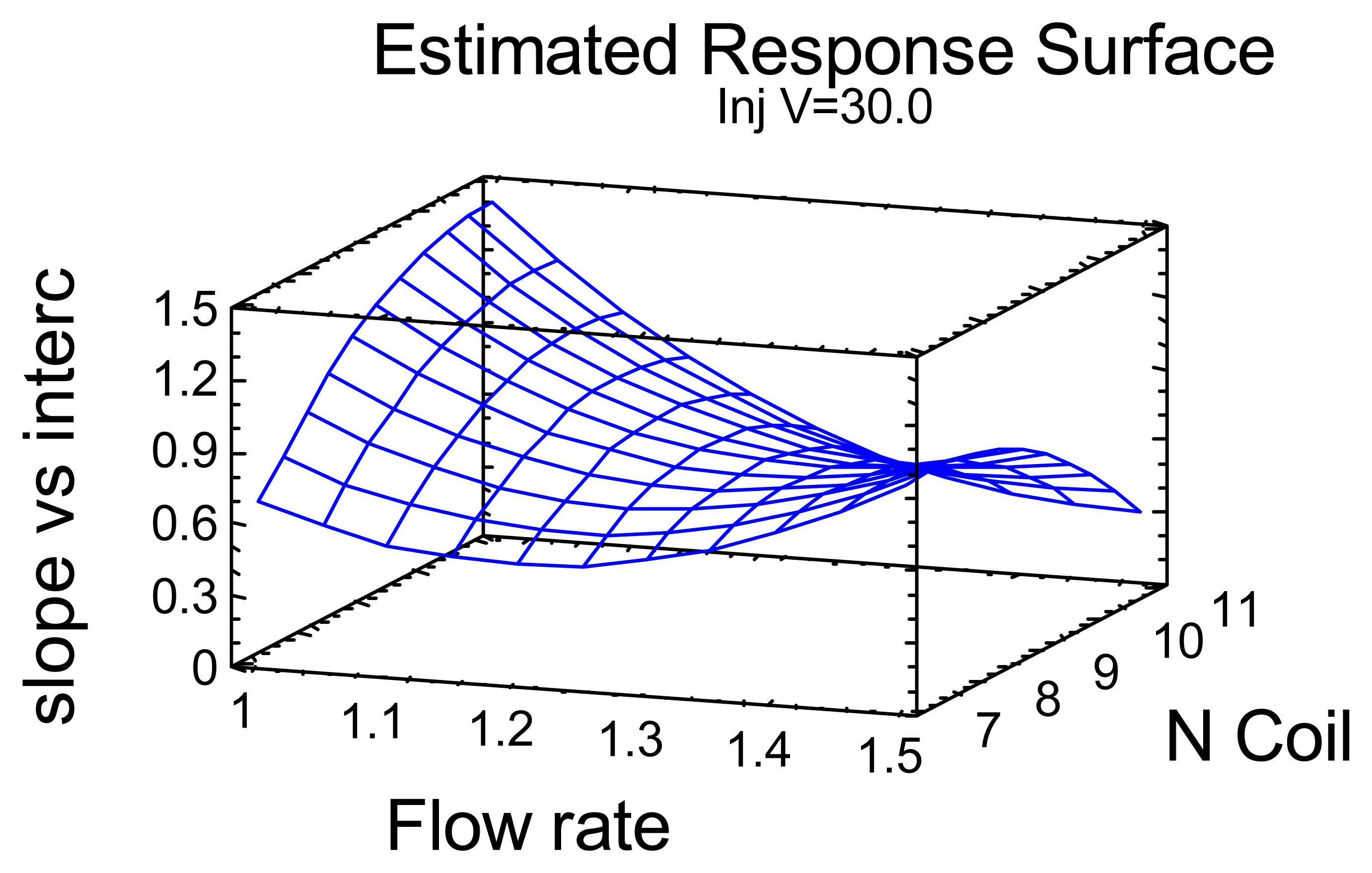
| Experiment | Flow rate (mL/min) | Number of turns of a mixing coil | Injection volume (μL) | Response variables | ||
|---|---|---|---|---|---|---|
| Slope b | Intercept a | b/a | ||||
| 1 | 1.5 | 9 | 25 | 0.1197 | 0.0823 | 1.4534 |
| 2 | 1.25 | 11 | 35 | 0.1183 | 0.1445 | 0.8191 |
| 3 | 1 | 9 | 25 | 0.1532 | 0.0955 | 1.6033 |
| 4 | 1.25 | 9 | 30 | 0.1043 | 0.1368 | 0.7629 |
| 5 | 1.25 | 7 | 35 | 0.1253 | 0.1343 | 0.9335 |
| 6 | 1.25 | 9 | 30 | 0.1268 | 0.1424 | 0.8909 |
| 7 | 1.25 | 7 | 25 | 0.0303 | 0.2389 | 0.1270 |
| 8 | 1 | 9 | 35 | 0.1592 | 0.1524 | 1.0442 |
| 9 | 1 | 11 | 30 | 0.1618 | 0.1197 | 1.3524 |
| 10 | 1.5 | 11 | 30 | 0.0387 | 0.1956 | 0.1976 |
| 11 | 1.25 | 9 | 30 | 0.0880 | 0.1600 | 0.5500 |
| 12 | 1.25 | 11 | 25 | 0.0847 | 0.1173 | 0.7216 |
| 13 | 1.5 | 7 | 30 | 0.1272 | 0.1269 | 1.0018 |
| 14 | 1 | 7 | 30 | 0.1102 | 0.1370 | 0.8039 |
| 15 | 1.5 | 9 | 35 | 0.0860 | 0.1760 | 0.4886 |
| Mixing coil's mark | T-16xd7 | T-7xd16 | T-7xd12 | T-7xd7 | T-3xd16 | T-0xd0 |
|---|---|---|---|---|---|---|
| Number of turns | 16 | 7 | 7 | 7 | 3 | - |
| Coil support diameter | 7 mm | 16 mm | 12 mm | 7 mm | 16 mm | - |
| Coil tube length* | 35.2 cm | 35.2 cm | 26.4 cm | 15.4 | 15.1 cm | - |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Gros, N. A Novel Type of Tri-Colour Light-Emitting-Diode-Based Spectrometric Detector for Low-Budget Flow-Injection Analysis. Sensors 2007, 7, 166-184. https://doi.org/10.3390/s7020166
Gros N. A Novel Type of Tri-Colour Light-Emitting-Diode-Based Spectrometric Detector for Low-Budget Flow-Injection Analysis. Sensors. 2007; 7(2):166-184. https://doi.org/10.3390/s7020166
Chicago/Turabian StyleGros, Nataša. 2007. "A Novel Type of Tri-Colour Light-Emitting-Diode-Based Spectrometric Detector for Low-Budget Flow-Injection Analysis" Sensors 7, no. 2: 166-184. https://doi.org/10.3390/s7020166




