Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes
Abstract
:1. Introduction
2. Experimental
2.1 Chemicals
2.2 Immobilization of enzymes and proteins on the surface of CNT
2.3 Apparatus
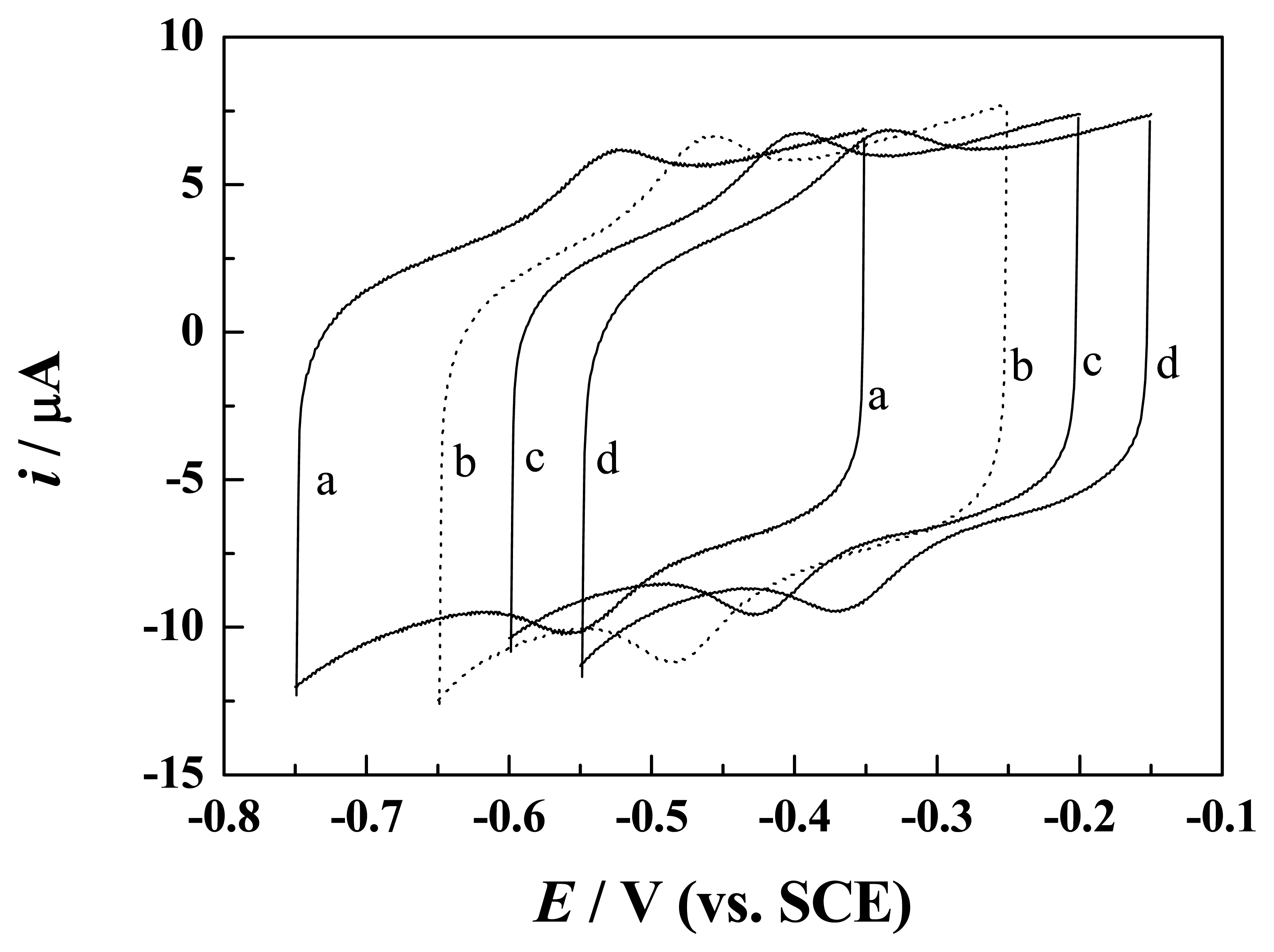
3. Results and discussion
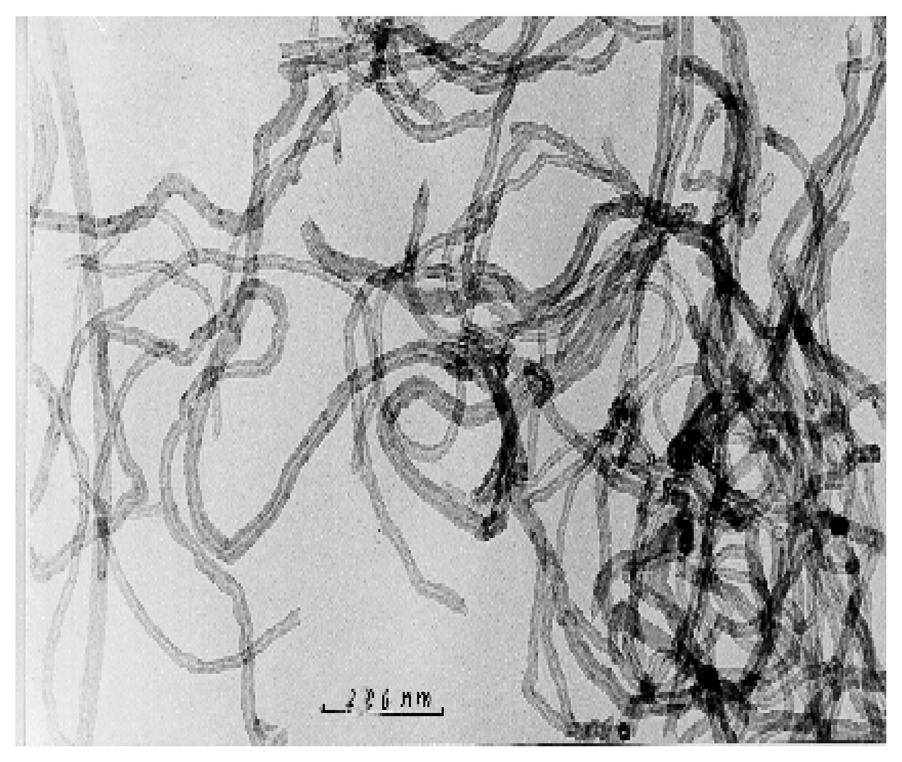
3.1 Physical characterization
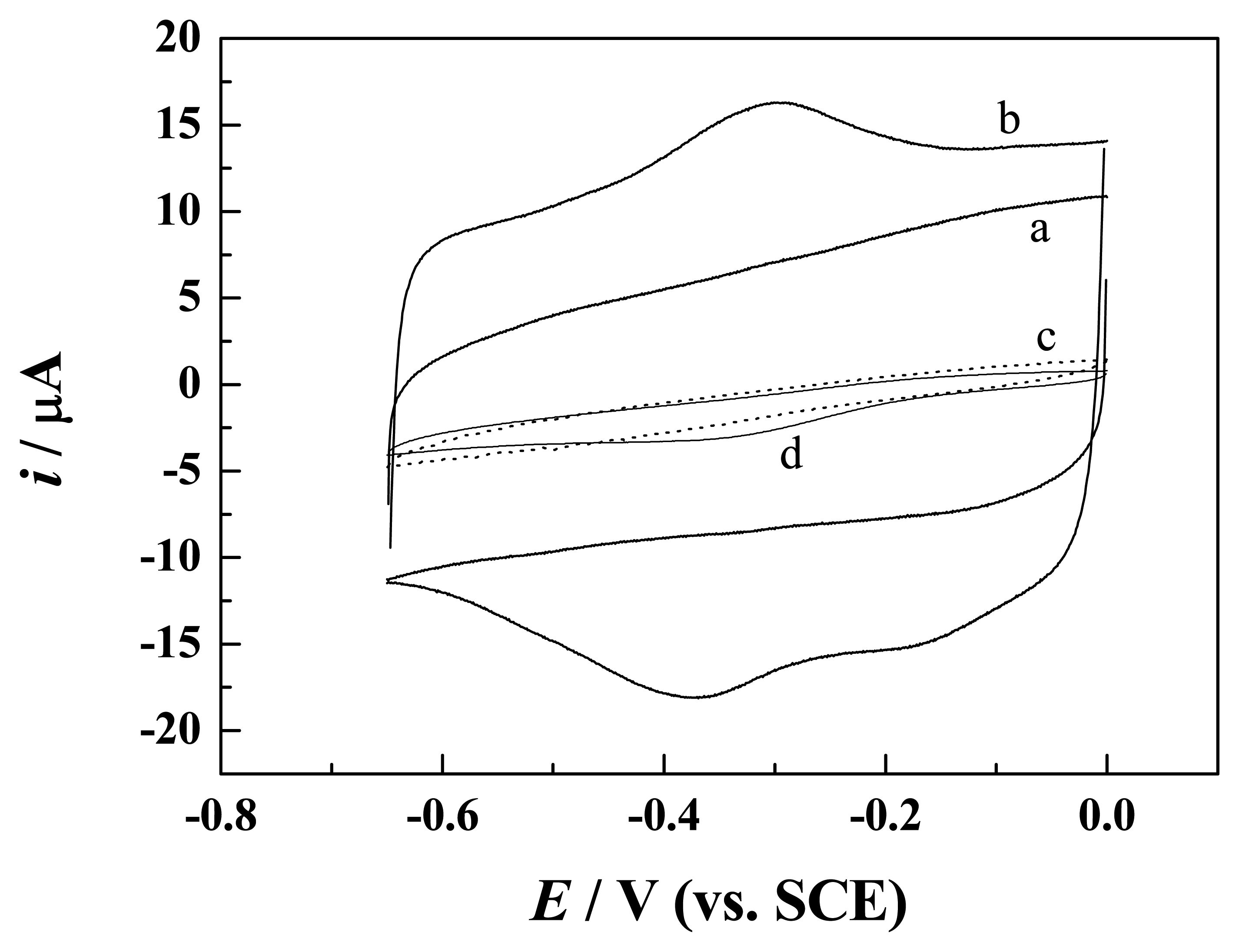
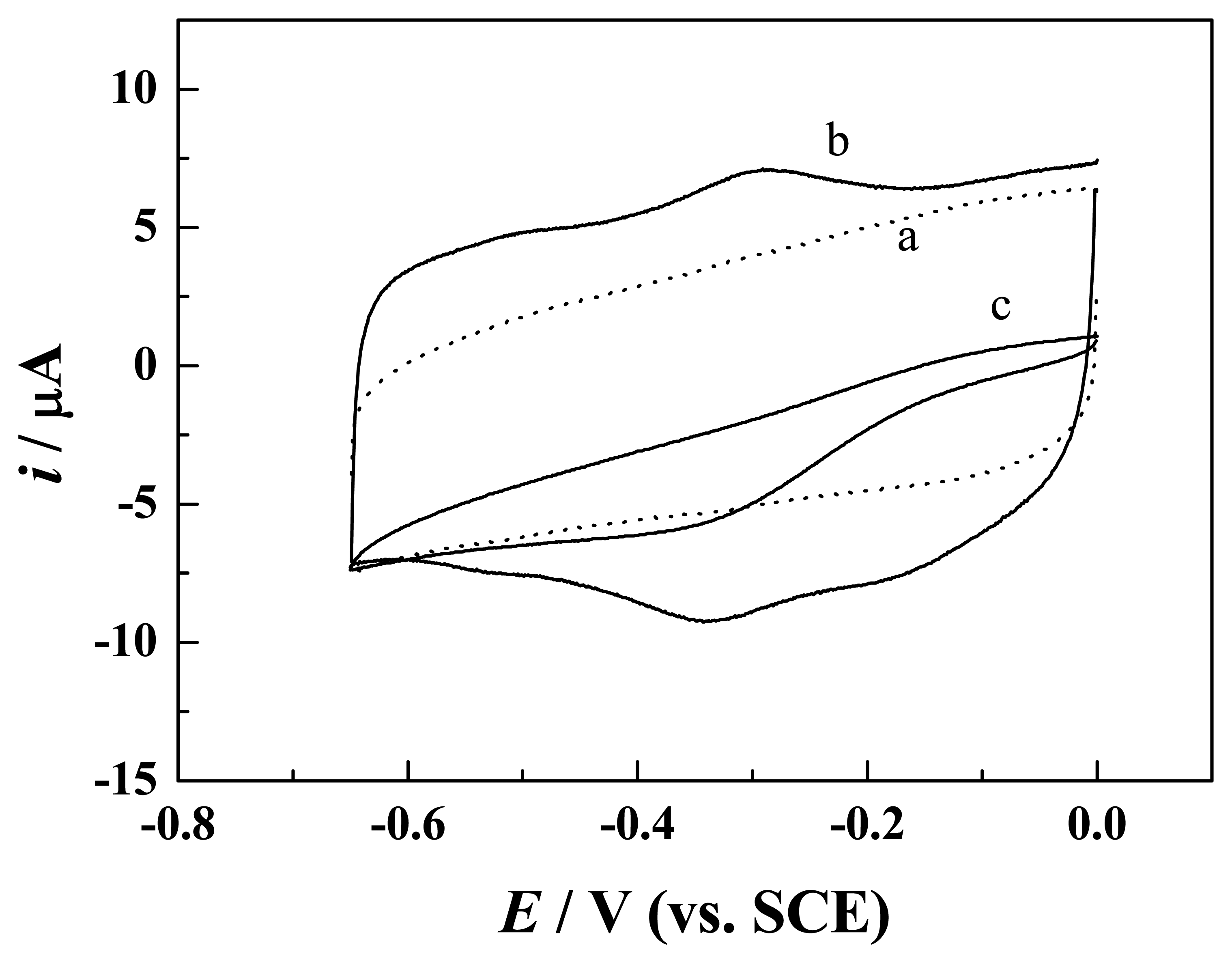
3.2 Direct electron transfer of Hb and HRP
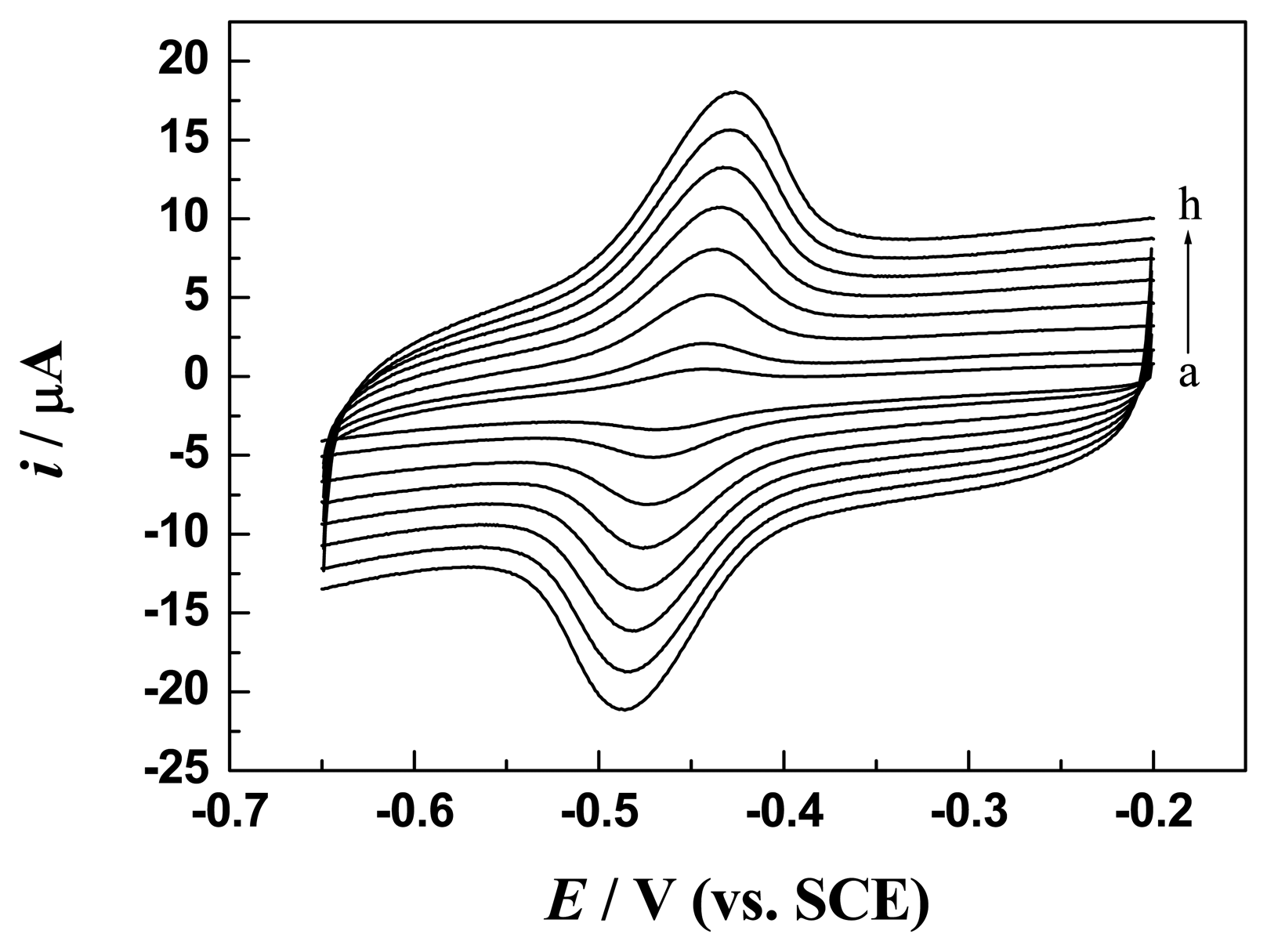
3.3 Direct electron transfer of GOx
3.4 Effect of solution pH
4. Conclusions
Acknowledgments
References and Notes
- Frew, J. E.; Hill, H. A. O. Direct and Indirect Electron Transfer between Electrodes and Redox Proteins. Eur. J. Biochem. 1988, 172, 261–269. [Google Scholar]
- Santucci, R.; Picciau, A.; Campanella, L.; Brunori, M. Electrochemistry of Metalloproteins. Curr. Top. Electrochem. 1994, 3, 313–328. [Google Scholar]
- Hess, C. R.; Juda, G. A.; Dooley, D. M.; Amii, R. N.; Hill, M. G.; Winkler, J. R.; Gray, H. B. Gold Electrodes Wired for Coupling with the Deeply Buried Active Site of Arthrobacter globiformis Amine Oxidase. J. Am. Chem. Soc. 2003, 125, 7156–7157. [Google Scholar]
- Elliott, S. J.; McElhaney, A. E.; Feng, C.; Enemark, J. H.; Armstrong, F. A. A Voltammetric Study of Interdomain Electron Transfer within Sulfite Oxidase. J. Am. Chem. Soc. 2002, 124, 11612–11613. [Google Scholar]
- Armstrong, F. A.; Wilson, G. S. Recent Developments in Faradaic Bioelectrochemistry. Electrochim. Acta 2000, 45, 2623–2645. [Google Scholar]
- Aguey-Zinsou, K.-F.; Bernhardt, P. V.; De Voss, J. J.; Slessor, K. E. Electrochemistry of P450cin: New Insights into P450 Electron Transfer. Chem. Commun. 2003, 418–419. [Google Scholar]
- Aguey-Zinsou, K.-F.; Bernhardt, P. V.; Kappler, U.; McEwan, A. G. Direct Electrochemistry of a Bacterial Sulfite Dehydrogenase. J. Am. Chem. Soc. 2003, 125, 530–535. [Google Scholar]
- Jhaveri, S. D.; Mauro, J. M.; Goldston, H. M.; Schauer, C. L.; Tender, L. M.; Trammell, S. A. A Reagentless Electrochemical Biosensor Based on a Protein Scaffold. Chem. Commun. 2003, 338–339.
- Xiao, Y.; Patolsky, F.; Katz, E.; Hainfeld, J. F.; Willner, I. “Plugging into Enzymes”: Nanowiring of Redox Enzymes by a Gold Nanoparticle. Science 2003, 299, 1877–1881. [Google Scholar]
- Iijima, S. Helical Microtubules of Graphitic Carbon. Nature 1991, 354, 56–58. [Google Scholar]
- Ebbesen, T. W.; Ajayan, P. M. Large-Scale Synthesis of Carbon Nanotubes. Nature 1992, 358, 220–222. [Google Scholar]
- Ajayan, P. M. Nanotubes from Carbon. Chem. Rev. 1999, 99, 1787–1799. [Google Scholar]
- Treacy, M. M. J.; Ebbesen, T. W.; Gibson, J. M. Exceptionally High Young's Modulus Observed for Individual Carbon Nanotubes. Nature 1996, 381, 678–680. [Google Scholar]
- Schlittler, R. R.; Seo, J. W.; Gimzewski, J. K.; Durkan, C.; Saifullah, M. S. M.; Welland, M. E. Single Crystals of Single-Walled Carbon Nanotubes Formed by Self-Assembly. Science 2001, 292, 1136–1139. [Google Scholar]
- White, C. T.; Todorov, T. N. Carbon Nanotubes as Long Ballistic Conductors. Nature 1998, 393, 240–242. [Google Scholar]
- Poncharal, Ph.; Wang, Z. L.; Ugarte, D.; de Heer, W. A. Electrostatic Deflections and Electromechanical Resonances of Carbon Nanotubes. Science 1999, 283, 1513–1516. [Google Scholar]
- Kong, J.; Franklin, N. R.; Zhou, C.; Chapline, M. G.; Peng, S.; Cho, K.; Dai, H. Nanotube Molecular Wires as Chemical Sensors. Science 2000, 287, 622–625. [Google Scholar]
- Collins, P. G.; Bradley, K.; Ishigami, M.; Zettl, A. Extreme Oxygen Sensitivity of Electronic Properties of Carbon Nanotubes. Science 2000, 287, 1801–1804. [Google Scholar]
- Vieira, R.; Pham-Huu, C.; Keller, N.; Ledoux, M. J. New Carbon Nanofiber/Graphire Felt Composie for Use as a Catalyst Support for Hydrazine Catalytic Decomposition. Chem. Commun. 2002, 954–955. [Google Scholar]
- Jin, Y. X.; Liu, Z. J.; Chen, W. X.; Xu, Z. D. Hydrogenation of Chlorophyll over Carbon Nanotubes-Supported Nickel Catalyst at Normal Temperature and Pressure. Acta Phys.–Chim. Sin. 2002, 18, 459–462. [Google Scholar]
- Fan, S.; Chapline, M. G.; Franklin, N. R.; Tombler, T. W.; Cassell, A. M.; Dai, H. Self-Oriented Regular Arrays of Carbon Nanotubes and Their Field Emission Properties. Science 1999, 283, 512–514. [Google Scholar]
- Kim, P.; Lieber, C. M. Nanotube Nanotweezers. Science 1999, 286, 2148–2150. [Google Scholar]
- Dai, H.; Hafner, J. H.; Rinzler, A. G.; Colbert, D. T.; Smalley, R. E. Nanotubes as Nanoprobes in Scanning Probe Microscopy. Nature 1996, 384, 147–150. [Google Scholar]
- Wildöer, J.W. G.; Venema, L. C.; Rinzler, A. G.; Smalley, R. E.; Dekker, C. Electronic Structure of Atomically Resolved Carbon Nanotubes. Nature 1998, 391, 59–62. [Google Scholar]
- Odom, T.W.; Huang, J.-L.; Kim, P.; Lieber, C. M. Atomic Structure and Electronic Properties of Single-Walled Carbon Nanotubes. Nature 1998, 391, 62–64. [Google Scholar]
- Wang, J.; Musameh, M. Carbon Nanotube/Teflon Composite Electrochemical Sensors and Biosensors. Anal. Chem. 2003, 75, 2075–2079. [Google Scholar]
- Musameh, M.; Wang, J.; Merkoci, A.; Lin, Y. Low-Potential Stable NADH Detection at Carbon-Nanotube-Modified Glassy Carbon Electrodes. Electrochem. Commun. 2002, 4, 743–746. [Google Scholar]
- Wang, J.; Li, M.; Shi, Z.; Li, N.; Gu, Z. Direct electrochemistry of Cytochrome c at a Glassy Carbon Electrode Modified with Single-Wall Carbon Nanotubes. Anal. Chem. 2002, 74, 1993–1997. [Google Scholar]
- Yu, X.; Chattopadhyay, D.; Galeska, I.; Papadimitrakopoulos, F.; Rusling, J.F. Peroxidase Activity of Enzymes Bound to the Ends of Single-Wall Carbon Nanotube Forest Electrodes. Electrochem. Commun. 2003, 5, 408–411. [Google Scholar]
- Gooding, J.J.; Wibowo, R.; Liu, J.; Yang, W.; Losic, D.; Orbons, S.; Mearns, F.J.; Shapter, J. G.; Hibbert, D. B. Protein Electrochemistry Using Aligned Carbon Nanotube Arrays. J. Am. Chem. Soc. 2003, 125, 9006–9007. [Google Scholar]
- Wang, J.; Deo, R.P.; Poulin, P.; Mangey, M. Carbon Nanotube Fiber Microelectrodes. J. Am. Chem. Soc. 2003, 125, 14706–14707. [Google Scholar]
- Chen, J.; Bao, J.-C.; Cai, C.-X.; Lu, T.-H. Direct Electrochemical Oxidation of Dihydronicotiamide Adenine Dinucleotide (NADH) at an Ordered Carbon Nanotubes Electrode. Chin. Chem. Lett. 2003, 14, 1171–1174. [Google Scholar]
- Cai, C.-X.; Chen, J. Direct Electron Transfer and Bioelectrocatalysis of Hemoglobin at a Carbon Nanotube Electrode. Anal. Biochem. 2004, 325, 285–292. [Google Scholar]
- Chen, J.; Cai, C.-X. Direct Electrochemical Oxidation of NADPH at a Low Potential on the Carbon Nanotube Modified Glassy Carbon Electrode. Chin. J. Chem. 2004, 22, 167–171. [Google Scholar]
- Encyclopedia of Chemical Technology (in Chinese); Vol. 1, Chemical Engineering Press: Beijing, 1990; p. p. 705.
- Bard, A. J.; Faulkner, L. R. Electrochemical Methods, Fundamental and Applications, 2nd Edition ed; John Wiley & Sons, Inc.: New York, 2001. [Google Scholar]
- Martin, C. R.; Dollard, K. A. Effect of Hydrophobic Interactions on the Rates of Ionic Diffusion in Nafion Films at Electrode Surfaces. J. Electroanal. Chem. 1983, 159, 127–135. [Google Scholar]
- Journet, C.; Maser, W. K.; Loiseau, A.; de la Chapelle, M. L.; Lefrant, S.; Deniard, P.; Lee, R.; Fischer, J. E. Large Scale Production of Single-Walled Carbon Nanotubes by the Electric-Arc Technique. Nature 1997, 388, 756–758. [Google Scholar]
- Star, A.; Stoddart, J. F.; Streuerman, D.; Diehl, M.; Boukai, A.; Wong, E. W.; Yang, X.; Chung, S.-W.; Chio, H.; Heath, J. R. Preparation and Properties of Polymer-Wrapped Single-Walled Carbon Nanotubes. Angew. Chem., Int. Ed. 2001, 40, 1721–1725. [Google Scholar]
- Riggs, J.E.; Guo, Z.; Caroll, D.L.; Sun, Y.P. Strong Luminescence of Solubilized Carbon Nanotubes. J. Am. Chem. Soc. 2000, 122, 5879–5880. [Google Scholar]
- Wang, J.; Musameh, M.; Lin, Y. Solubilization of Carbon Nanotubes by Nafion toward the Preparation of Amperometric Biosensors. J. Am. Chem. Soc. 2003, 125, 2408–2409. [Google Scholar]
- Richard, C.; Balavoine, F.; Schultz, P.; Ebbesen, T. W.; Mioskowski, C. Supramolecular Self-Assembly of Lipid Derivatives on Carbon Nanotubes. Science 2003, 300, 775–778. [Google Scholar]
- O'Connell, M.J.; Bachilo, S.M.; Huffman, C.B.; Moore, V.C.; Strano, M.S.; Haroz, E.H.; Roialon, K.L.; Boul, P.J.; Noon, W.H.; Kottrell, C.; Ma, J.; Hauge, R.H.; Weisman, R. B.; Smalley, R.E. Gap Fluorescence from Individual Single-Walled Carbon Nanotubes. Science 2002, 297, 593–596. [Google Scholar]
- O'Connell, M.J.; Poul, P.; Ericson, L.; Huffman, C.; Wang, Y.; Haroz, E.; Kuper, C.; Tour, J.; Ausman, D.; Smalley, R. E. Reversible Water-Solubilization of Single-Walled Carbon Nanotubes by Polymer Wrapping. Chem. Phys. Lett. 2001, 342, 265–271. [Google Scholar]
- Dieckmann, G.R.; Dalton, A. B.; Johnson, P. A.; Razal, J.; Chen, J.; Giordano, G. M.; Muñoz, E.; Musselman, I.H.; Baughman, R.H.; Drape, R.K. Controlled Assembly of Carbon Nanotubes by Designed Amphiphilic Peptide Helices. J. Am. Chem. Soc. 2003, 125, 1770–1777. [Google Scholar]
- Rao, A.M.; Richter, E.; Bandow, S.; Chase, B.; Eklund, P.C.; Williams, K.A.; Fang, S.; Subbaswamy, K. R.; Menon, M.; Thess, A.; Smalley, R. E.; Dresselhaus, G.; Dresselhaus, M.S. Diameter-Selective Raman Scattering from Vibrational Modes in Carbon Nanotubes. Science 1997, 275, 187–191. [Google Scholar]
- Wang, L.; Hu, N. Direct Electrochemistry of Hemoglobin in Layer-by-Layer Films with Poly(vinyl sulfonate) Grown on Pyrolytic Graphite Electrodes. Bioelectrochem. 2001, 53, 205–212. [Google Scholar]
- Yang, J.; Hu, N.; Rusling, J. F. Enhanced Electron Transfer for Hemoglobin in Poly(ester sulfonic acid) Films on Pyrolytic Graphite Electrodes. J. Electroanal. Chem. 1999, 463, 53–62. [Google Scholar]
- Fan, C.; Wagner, G.; Li, G. Effect of Dimethyl Sulfoxide on the Electron Transfer Reactivity of Hemoglobin. Bioelectrochem. 2001, 54, 49–51. [Google Scholar]
- Ma, H.; Hu, N.; Rusling, J. F. Electroactive Myoglobin Films Grown Layer-by-Layer with Poly(styrenesulfonate) on Pyrolytic Graphite Electrodes. Langmuir 1999, 16, 4969–4975. [Google Scholar]
- Sun, D.-M.; Cai, C.-X.; Li, X.-G.; Xing, W.; Lu, T.-H. Direct Electrochemistry and Bioelectrocatalysis of Horseradish Peroxidase Immobilized on Active Carbon. J. Electroanal. Chem. 2004, 566, 415–421. [Google Scholar]
- Zhang, Z.; Nassar, A.-E. F.; Lu, Z.; Schenkman, J. B.; Rusling, J. F. Direct Electron Injection from Electrodes to Cytochrome P450cam in Biomembrane-like Films. J. Chem. Soc., Faraday Trans. 1997, 93, 1769–1774. [Google Scholar]
- Laviron, E. General Expression of the Linear Potential Sweep Voltammogram in the case of Diffusionless Electrochemical Systems. J. Electroanal. Chem. 1979, 101, 19–28. [Google Scholar]
- Liu, H.; Hu, N. Heme Protein-Gluten Films: Voltammetric Studies and Their Electrocatalytic Properties. Anal. Chim. Acta 2003, 481, 91–99. [Google Scholar]
- Yang, J.; Hu, N. Direct Electron Transfer for Hemoglobin in Biomembrane-like Dimyristoyl Phosphatidylcholine Films on Pyrolytic Graphite Electrodes. Bioeletrochem. Bioenerg. 1999, 48, 117–127. [Google Scholar]
- Ferri, T.; Poscia, A.; Santucci, R. Direct Electrochemistry of Membrane-Entrapped Horseradish Peroxidase. Part I. A Voltammetric and Spectroscopic Study. Bioelectrochem. Bioenerg. 1998, 44, 177–181. [Google Scholar]
- Ferri, T.; Poscia, A.; Santucci, R. Direct Electrochemistry of Membrane-entrapped Horseradish Peroxidase. Part II. Amperometric Detection of Hydrogen Peroxide. Bioelectrochem. Bioenerg. 1998, 45, 221–226. [Google Scholar]
- Huang, R.; Hu, N. Direct Electrochemistry and Electrocatalysis with Horseradish Peroxidase in Eastman AQ Films. Bioelectrochem. 2001, 54, 75–81. [Google Scholar]
- Rusling, J. F. Enzyme Bioelectrochemistry in Cast Biomembrane-like Films. Acc. Chem. Res. 1998, 31, 363–369. [Google Scholar]
- Wang, Z.; Liu, J.; Liang, Q.; Wang, Y.; Luo, G. Canbon Nanotube-intercalated Graphitr Electrodes for Simultaneous Determination of Dopamine and Serotonin in the Presence of Ascorbic Acid. J. Electroanal. Chem. 2003, 540, 129–134. [Google Scholar]
- Jiang, L.; McNeil, C. J.; Cooper, J. M. Direct Electron Transfer Reactions of Glucose Oxidase Immobilized at a Self-Assembled Monolayer. J. Chem. Soc., Chem. Commun. 1995, 1293–1295. [Google Scholar]
- Ianniello, R. M.; Lindsay, T. J.; Yacynych, A. M. Differential Pulse Voltammetric Study of Direct Electron Transfer in Glucose Oxidase Chemically Modified Graphite Electrodes. Anal. Chem. 1982, 54, 1098–1101. [Google Scholar]
- Scheller, F.; Strand, G.; Neumann, B.; Kühn, M.; Ostrowski, W. Polarographic Reduction of the Prosthetic in Flavoproteins. Bioelectrochem. Bioenerg. 1979, 6, 117–122. [Google Scholar]
- Lei, C.; Wollenberger, U.; Bistolas, N.; Guiseppi-Elis, A.; Scheller, F. W. Electron Transfer of Hemoglobin at Electrodes Modified with Colloidal Clay Nanoparticles. Anal. Bioanal. Chem. 2002, 372, 235–239. [Google Scholar]
- Hecht, H. J.; Kalisz, H. M.; Hendle, J.; Schmid, R. D.; Schomburg, D. Crystal Structure of Glucose Oxidase from Aspergillus niger Refined at 2.3 Å Reslution. J. Mol. Biol. 1993, 229, 153–172. [Google Scholar]



© 2005 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Yin, Y.; Lü, Y.; Wu, P.; Cai, C. Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes. Sensors 2005, 5, 220-234. https://doi.org/10.3390/s5040220
Yin Y, Lü Y, Wu P, Cai C. Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes. Sensors. 2005; 5(4):220-234. https://doi.org/10.3390/s5040220
Chicago/Turabian StyleYin, Yajing, Yafen Lü, Ping Wu, and Chenxin Cai. 2005. "Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes" Sensors 5, no. 4: 220-234. https://doi.org/10.3390/s5040220
APA StyleYin, Y., Lü, Y., Wu, P., & Cai, C. (2005). Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes. Sensors, 5(4), 220-234. https://doi.org/10.3390/s5040220



