Improved Classification of Orthosiphon stamineus by Data Fusion of Electronic Nose and Tongue Sensors
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Selection and Preparation
2.2. Electronic Tongue Setup and Measurement
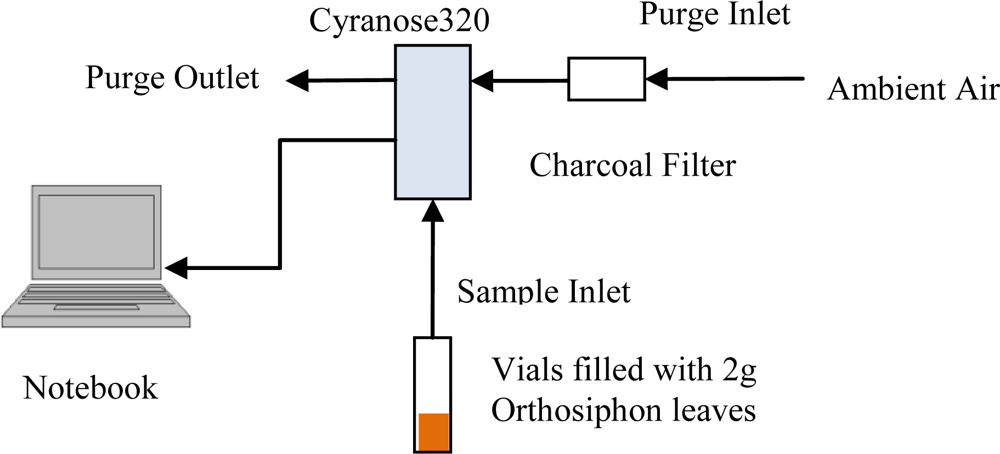
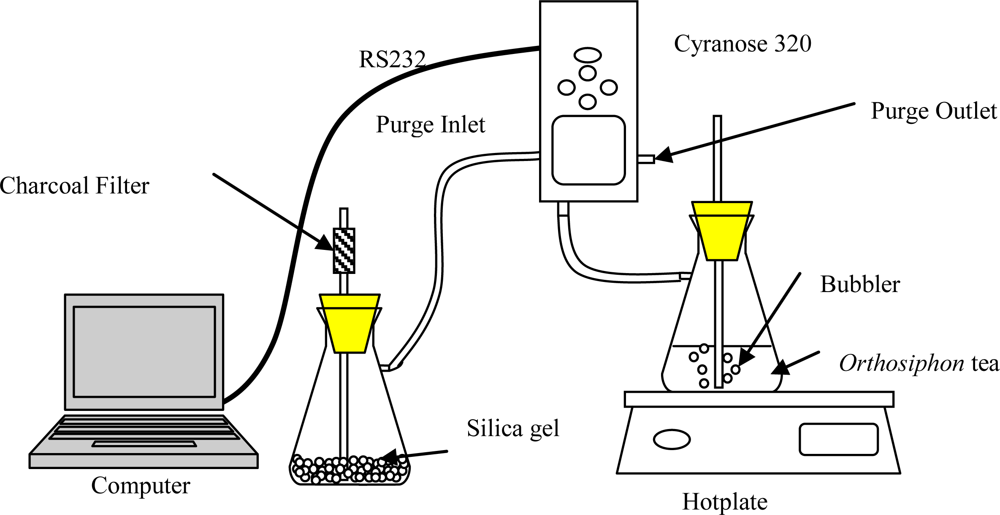
2.3. Electronic Nose Setup and Measurement
2.4. Data Analysis
2.5. Data Fusion
3. Results and Discussion
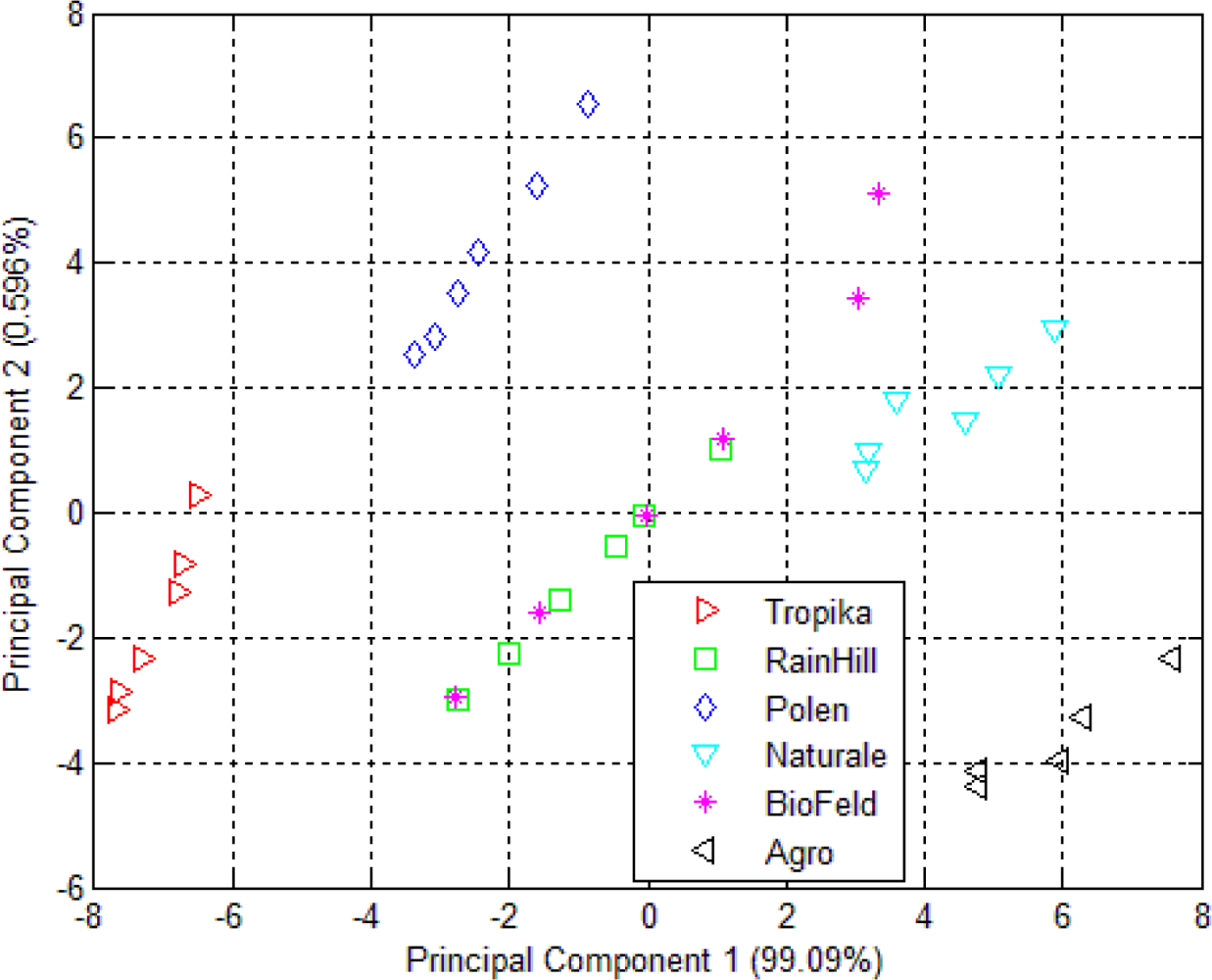
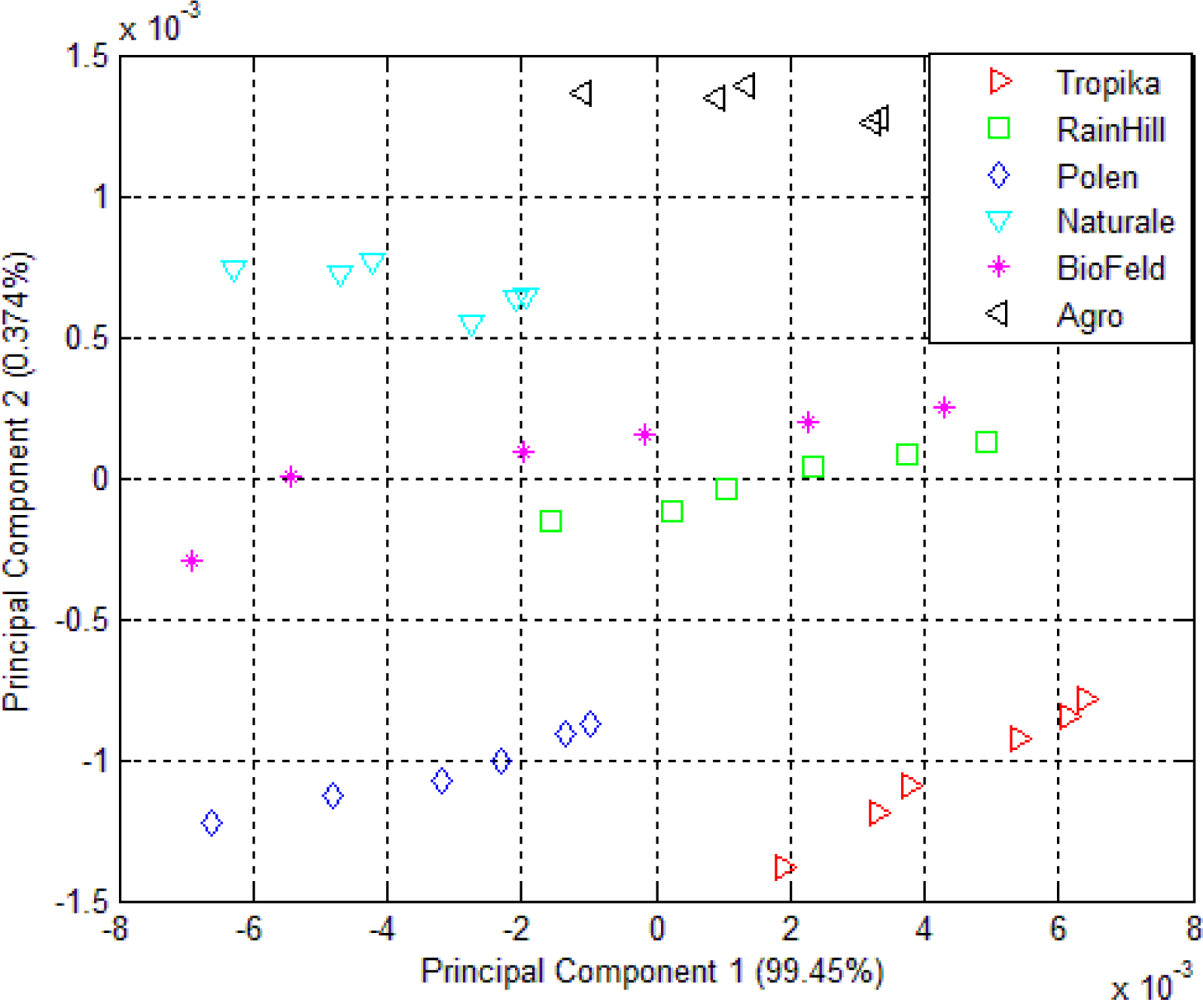
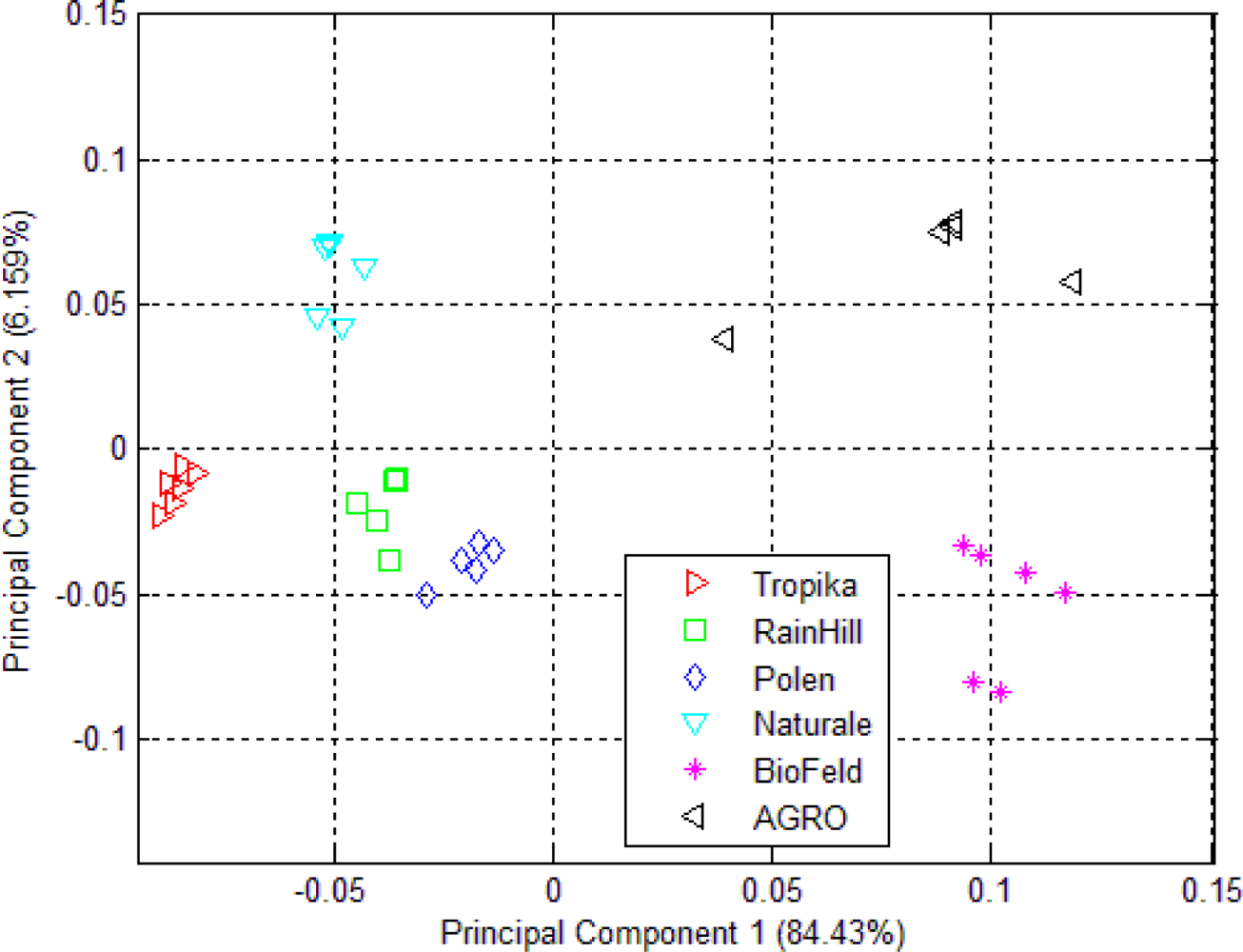
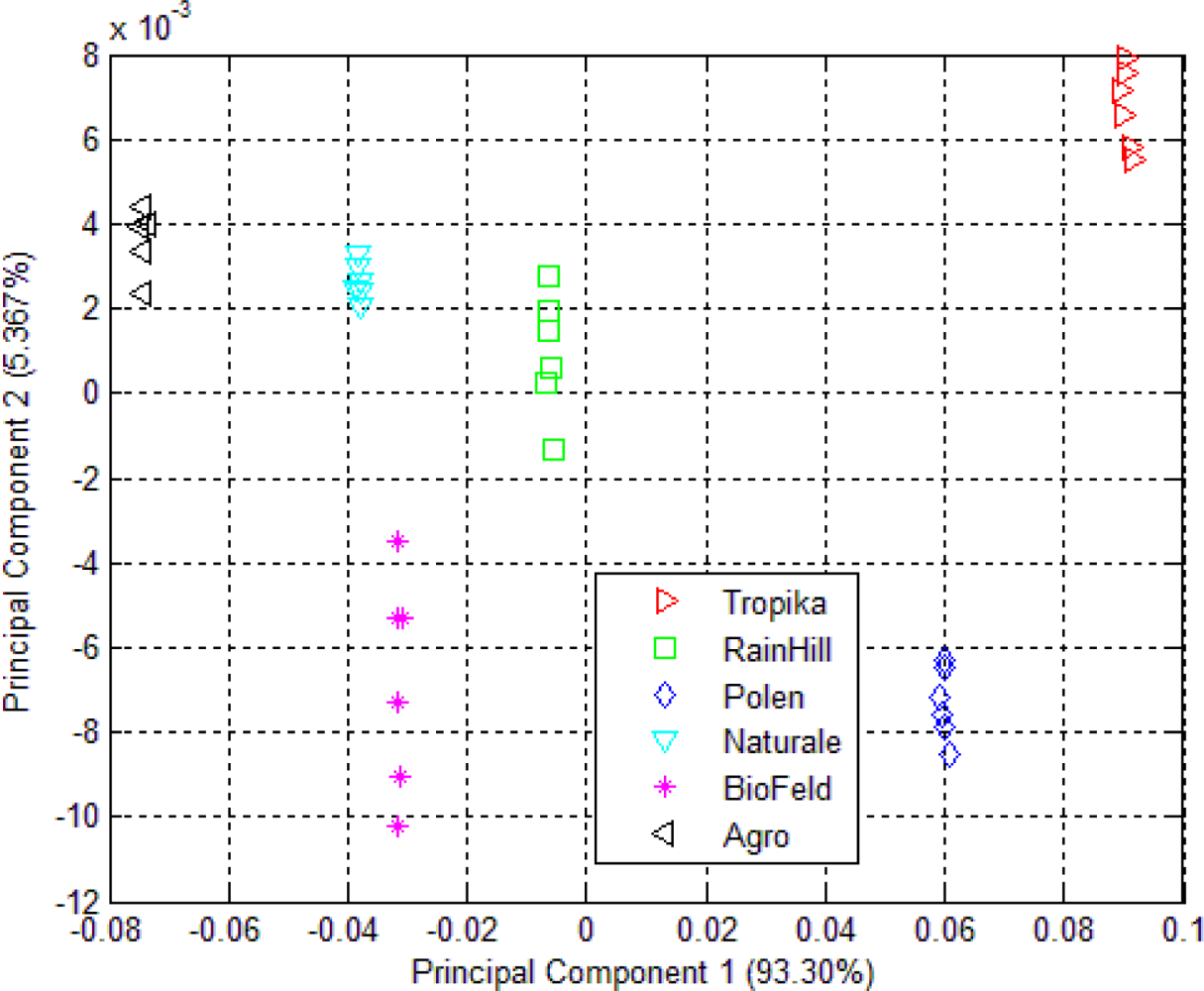
3.1. Principal Component Analysis (PCA)
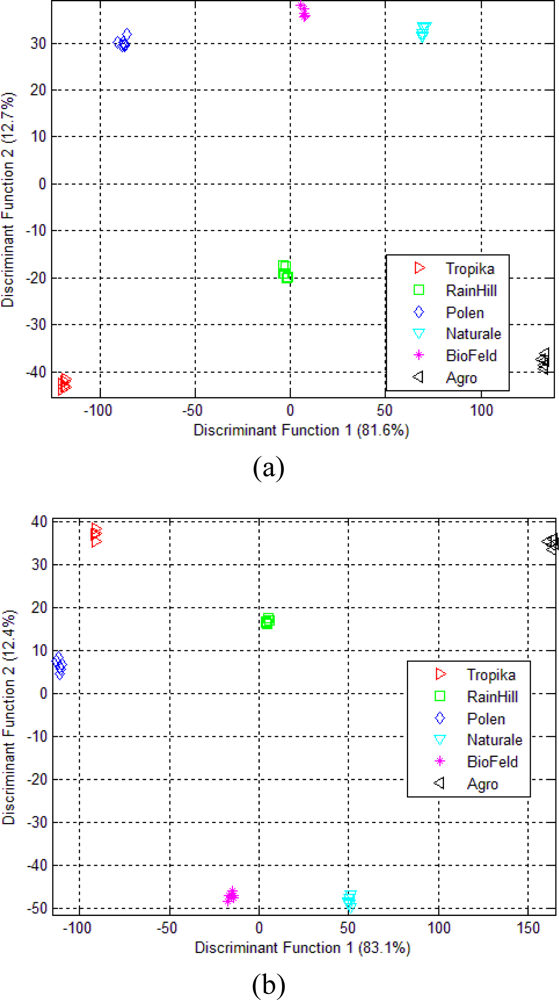
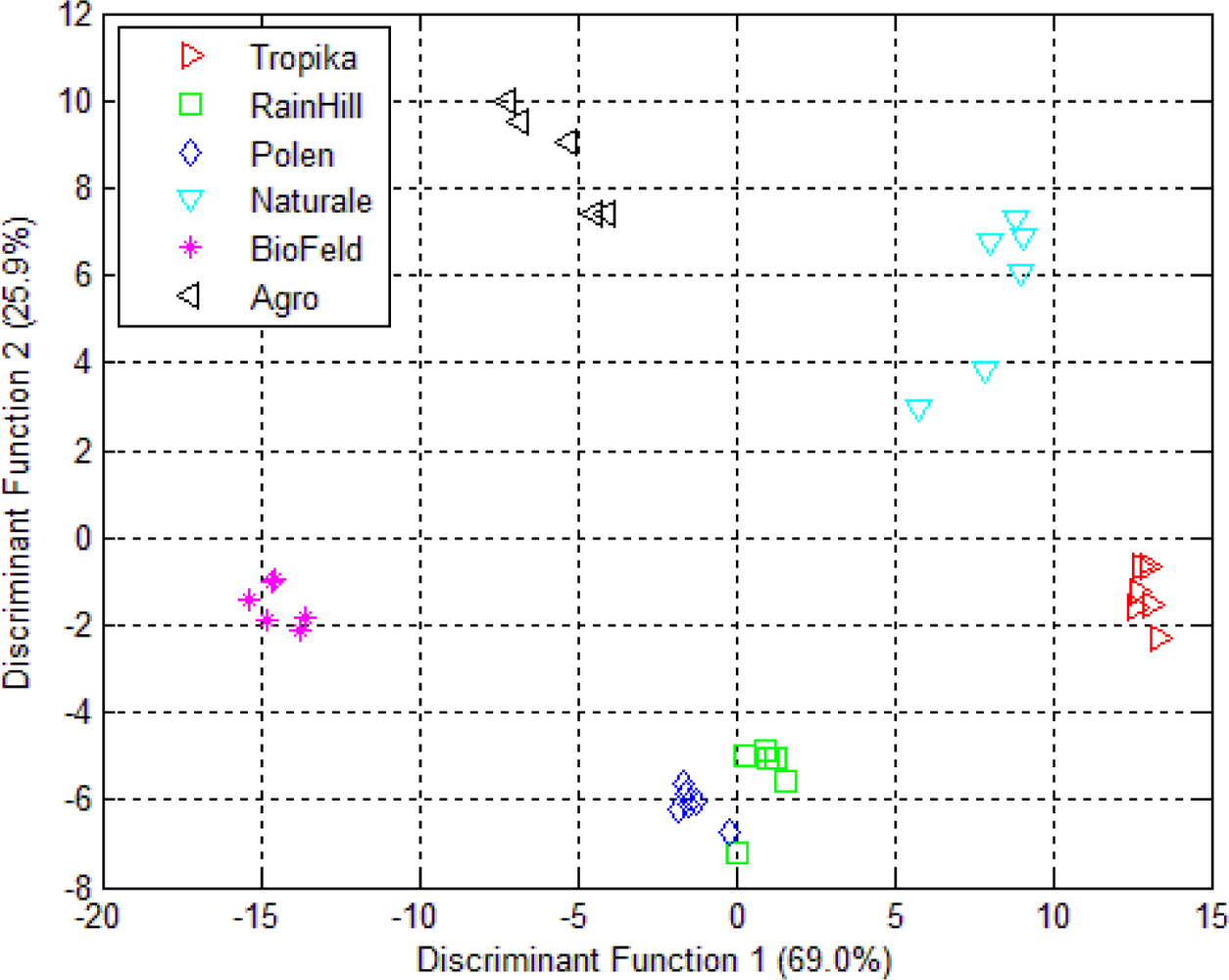
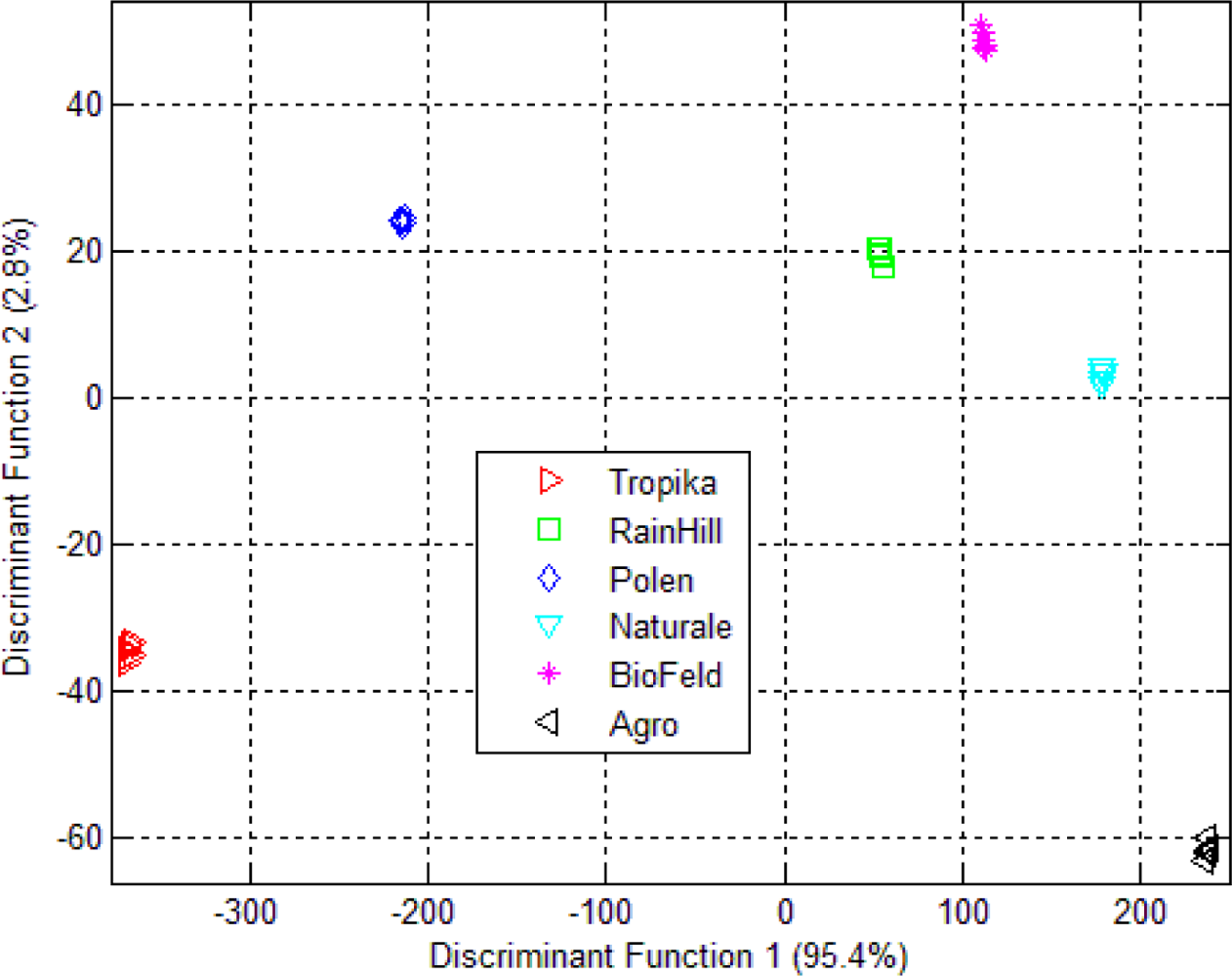
3.2. Linear Discriminant Analysis (LDA)
4. Conclusions
Acknowledgments
References
- Mendez, MLR; Arrieta, AA; Parra, V; Bernal, A; Vegas, A; Villanueva, S; Osuna, RG; de Saja, JA. Fusion of three sensory modalities for the multimodal characterization of red wines. IEEE Sens. J. Vol 2004, 4, 342–347. [Google Scholar]
- Guadarrama, A; Fernández, JA; Íñiguez, M; Souto, J; de Saja, JA. Discrimination of wine aroma using an array of conducting polymers sensors in conjunction with solid-phase micro-extraction (SPME) technique. Sens. Actuat. B: Chem 2001, 77, 401–408. [Google Scholar]
- Schaller, E; Zenhausern, S; Zesiger, T; Bosset, JO; Escher, F. Use of preconcentration techniques applied to a MS-based “electronic nose”. Analysis 2000, 28, 743–749. [Google Scholar]
- Gardner, JW; Bartlett, PN. Electronic Noses: Principles and Applications; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Lvova, L; Legin, A; Vlasov, Y; Cha, GS; Nam, H. Multicomponent analysis of Korean green tea by means of disposable all-solid-state potentiometric electronic tongue microsystem. Sens. Actuat. B:Chem 2003, 95, 391–399. [Google Scholar]
- Hidayat, W; Shakaff, AYM; Ahmad, MN; Adom, AH. Classification of agarwood oil using an electronic nose. Sensors 2010, 10, 4675–4685. [Google Scholar]
- Bhattacharya, N; Tudu, B; Jana, A; Ghosh, D; Bandhopadhyaya, R; Bhuyan, M. Preemptive identification of optimum fermentation time for black tea using electronic nose. Sens. Actuat. B: Chem 2008, 131, 110–116. [Google Scholar]
- Dutta, R; Kashwan, KR; Bhuyan, M; Hines, EL; Gardner, JW. Electronic nose based tea quality standardization. Neural Netw 2003, 16, 847–853. [Google Scholar]
- Legin, AM; Rudnitskaya, A; Yu, G; Vlasov, AV; Di Natale, C; D'Amico, A. The features of the electronic tongue in comparison with the characteristics of the discrete ion-selective sensors. Sens. Actuat. B: Chem 1999, 58, 464–468. [Google Scholar]
- Vlasov, Y; Legin, A; Rudnitskaya, A; D’Amico, A; Di Natale, C. Electronic tongue-new analytical tool for liquid analysis on the basis of non-specific sensors and methods of pattern recognition. Sens. Actuat. B: Chem 2000, 65, 235–236. [Google Scholar]
- Toko, K. Taste sensor. Sens. Actuat. B: Chem 2000, 64, 205–215. [Google Scholar]
- Persaud, K; Dodd, G. Analysis of discrimination mechanisms in the mammalian olfactory system using a model nose. Nature 1982, 299, 352–355. [Google Scholar]
- Delwiche, JF. The impact of perceptual interactions on perceived flavor. Food Qual. Prefer 2004, 15, 137–146. [Google Scholar]
- Winquist, F; Lundstrom, I; Wide, P. The combination of an electronic tongue and an electronic nose. Sens. Actuat. B: Chem 1999, 58, 512–517. [Google Scholar]
- Natale, CD; Paolesse, R; Macagnano, A; Mantini, A; D'Amico, A; Legin, A; Lvova, L; Rudnitskaya, A; Vlasov, Y. Electronic nose and electronic tongue integration for improved classification of clinical and food samples. Sens. Actuat. B: Chem 2000, 64, 15–21. [Google Scholar]
- Boilot, P; Hines, EL; Gongora, MA; Folland, RS. Electronic Noses inter-comparison, data fusion and sensor selection in discrimination of standard fruit solutions. Sens. Actuat. B: Chem 2002, 88, 80–88. [Google Scholar]
- Winquist, F; Holmin, S; Krantz-Rulcker, C; Wide, P; Lundstrom, I. A hybrid electronic tongue. Anal. Chim. Acta 2000, 406, 147–157. [Google Scholar]
- Sim, CO; Ahmad, MN; Ismail, Z; Othman, AR; Noor, NA; Zaihidee, EM. Chemometric Classification of Herb—Orthosiphon stamineus according to its geographical origin using virtual chemical sensor based upon fast GC. Sensors 2003, 3, 458–471. [Google Scholar]
- Legin, A; Rudnitskaya, A. Chalcogenide Glass Electrodes. Available online: http://www.electronictongue.com/price01.html (accessed on 15 March 2009).
- National Instruments. NI USB-6008. Available online: http://sine.ni.com/nips/cds/view/p/lang/en/nid/14604 (accessed on 15 March 2009).
- Gardner, JW; Boilot, P; Hines, EL. Enhancing electronic nose performance by sensor selection using a new integer-based genetic algorithm. Sens. Actuat. B: Chem 2005, 106, 114–121. [Google Scholar]
- Steinmetz, V; Sevila, F; Bellon-Maurel, V. A Methodology for sensor fusion design: Application to fruit quality assessment. J. Agric. Engng. Res 1999, 74, 21–31. [Google Scholar]
- Berrueta, LA; Alonso-Slaces, RM; Heberger, K. Supervised pattern recognition in food analysis. J. Chrom: A 2007, 1158, 196–214. [Google Scholar]
- Tabachnick, BG; Fidell, LS. Using Multivariate Statistics; Pearson Education: Upper Saddle River, NJ, USA, 2007. [Google Scholar]
| Brands | Number of Samples | Number of replicated e-nose measurements* | Number of replicated e-tongue measurements | Colour of tea infusion |
|---|---|---|---|---|
| Tropika | 6 | 10 | 3 | Light yellow |
| RainHill | 6 | 10 | 3 | Dark yellow |
| Polen | 6 | 10 | 3 | Very light yellow |
| Naturale | 6 | 10 | 3 | Light yellow |
| BioFeld | 6 | 10 | 3 | Very light yellow |
| Agro | 6 | 10 | 3 | Greenish |
| Sensor Label | Description |
|---|---|
| Fe3+ | Ion-selective sensor for Iron ions |
| Cd2+ | Ion-selective sensor for Cadmium ions |
| Cu2+ | Ion-selective sensor for Copper ions |
| Hg2+ | Ion-selective sensor for Mercury ions |
| Ti+ | Ion-selective sensor for Titanium ions |
| S2− | Ion-selective sensor for Sulfur ions |
| Cr (VI) | Ion-selective sensor for Chromium ions |
| HI 5311 | Reference probe using Ag/AgCl electrode |
| Sampling setting | Dried leaves | Tea infusion | ||
|---|---|---|---|---|
| Cycle | Time(s) | Pump Speed | Time(s) | Pump Speed |
| Baseline Purge | 15 | 60 mL/min | 10 | 120 mL/min |
| Sample Draw | 20 | 60 mL/min | 30 | 120 mL/min |
| Idle Time | 3 | - | 3 | - |
| Air Intake Purge | 50 | 160/min | 80 | 160 mL/min |
| Experiment | PC1 | PC2 | PC3 | PC4 | PC5 |
|---|---|---|---|---|---|
| Dried Orthosiphon leaves using e-nose | 99.09 | 0.596 | 0.142 | 0.088 | 0.024 |
| Orthosiphon tea infusions using e-nose | 99.46 | 0.374 | 0.086 | 0.024 | 0.017 |
| Orthosiphon tea infusions using e-tongue | 85.12 | 6.095 | 4.220 | 1.906 | 1.526 |
| Before Fusion | After Fusion | ||||
|---|---|---|---|---|---|
| Modality | Sensor Label | Wilks' Lambda | F | Wilks' Lambda | F |
| E-NOSE | SENSOR01 | .209 | 22.672 | .005 | 1,128.765 |
| SENSOR02 | .200 | 23.988 | .003 | 1,853.475 | |
| SENSOR03 | .290 | 14.707 | .004 | 1,392.132 | |
| SENSOR04 | .161 | 31.174 | .002 | 2,488.416 | |
| SENSOR05 | .366 | 10.393 | .274 | 15.884 | |
| SENSOR06 | .211 | 22.449 | .229 | 20.196 | |
| SENSOR07 | .045 | 128.200 | .002 | 2,668.615 | |
| SENSOR08 | .180 | 27.277 | .002 | 3,404.481 | |
| SENSOR09 | .022 | 268.335 | .005 | 1,250.789 | |
| SENSOR10 | .015 | 391.406 | .001 | 5,290.785 | |
| SENSOR11 | .036 | 159.070 | .002 | 2,686.906 | |
| SENSOR12 | .169 | 29.496 | .003 | 2,131.702 | |
| SENSOR13 | .037 | 154.903 | .002 | 2,614.114 | |
| SENSOR14 | .038 | 150.154 | .002 | 2,843.676 | |
| SENSOR15 | .028 | 209.879 | .001 | 6,947.781 | |
| SENSOR16 | .024 | 239.119 | .001 | 4,037.560 | |
| SENSOR17 | .020 | 286.910 | .001 | 5,061.946 | |
| SENSOR18 | .120 | 44.176 | .002 | 3,124.103 | |
| SENSOR19 | .239 | 19.096 | .005 | 1,170.584 | |
| SENSOR20 | .266 | 16.516 | .002 | 2,996.106 | |
| SENSOR21 | .031 | 186.642 | .002 | 2,892.857 | |
| SENSOR22 | .019 | 307.061 | .002 | 3,767.506 | |
| SENSOR23 | .298 | 14.143 | .199 | 24.082 | |
| SENSOR24 | .163 | 30.855 | .003 | 1,821.131 | |
| SENSOR25 | .054 | 105.628 | .002 | 3,826.430 | |
| SENSOR26 | .243 | 18.692 | .012 | 492.505 | |
| SENSOR27 | .522 | 5.499 | .005 | 1,145.081 | |
| SENSOR28 | .228 | 20.373 | .010 | 602.827 | |
| SENSOR29 | .031 | 188.591 | .008 | 722.956 | |
| SENSOR30 | .045 | 126.357 | .002 | 3,636.019 | |
| SENSOR31 | .390 | 9.402 | .421 | 8.246 | |
| SENSOR32 | .408 | 8.693 | .004 | 1,540.924 | |
| E-TONGUE | SENSOR01 | .060 | 93.797 | .000 | 16,946.764 |
| SENSOR02 | .687 | 2.734 | .520 | 5.547 | |
| SENSOR03 | .051 | 110.770 | .194 | 25.003 | |
| SENSOR04 | .070 | 80.328 | .000 | 30,371.417 | |
| SENSOR05 | .817 | 1.345 | .771 | 1.782 | |
| SENSOR06 | .020 | 296.335 | .000 | 57,563.900 | |
| SENSOR07 | .802 | 1.477 | .645 | 3.305 | |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zakaria, A.; Shakaff, A.Y.M.; Adom, A.H.; Ahmad, M.; Masnan, M.J.; Aziz, A.H.A.; Fikri, N.A.; Abdullah, A.H.; Kamarudin, L.M. Improved Classification of Orthosiphon stamineus by Data Fusion of Electronic Nose and Tongue Sensors. Sensors 2010, 10, 8782-8796. https://doi.org/10.3390/s101008782
Zakaria A, Shakaff AYM, Adom AH, Ahmad M, Masnan MJ, Aziz AHA, Fikri NA, Abdullah AH, Kamarudin LM. Improved Classification of Orthosiphon stamineus by Data Fusion of Electronic Nose and Tongue Sensors. Sensors. 2010; 10(10):8782-8796. https://doi.org/10.3390/s101008782
Chicago/Turabian StyleZakaria, Ammar, Ali Yeon Md. Shakaff, Abdul Hamid Adom, MohdNoor Ahmad, Maz Jamilah Masnan, Abdul Hallis Abdul Aziz, Nazifah Ahmad Fikri, Abu Hassan Abdullah, and Latifah Munirah Kamarudin. 2010. "Improved Classification of Orthosiphon stamineus by Data Fusion of Electronic Nose and Tongue Sensors" Sensors 10, no. 10: 8782-8796. https://doi.org/10.3390/s101008782
APA StyleZakaria, A., Shakaff, A. Y. M., Adom, A. H., Ahmad, M., Masnan, M. J., Aziz, A. H. A., Fikri, N. A., Abdullah, A. H., & Kamarudin, L. M. (2010). Improved Classification of Orthosiphon stamineus by Data Fusion of Electronic Nose and Tongue Sensors. Sensors, 10(10), 8782-8796. https://doi.org/10.3390/s101008782





