Detection of a High-Density Brachiolaria-Stage Larval Population of Crown-of-Thorns Sea Star (Acanthaster planci) in Sekisei Lagoon (Okinawa, Japan)
Abstract
:1. Introduction
2. Materials and Methods
2.1. General Background Information and A. Planci Outbreak in the Yaeyama Islands
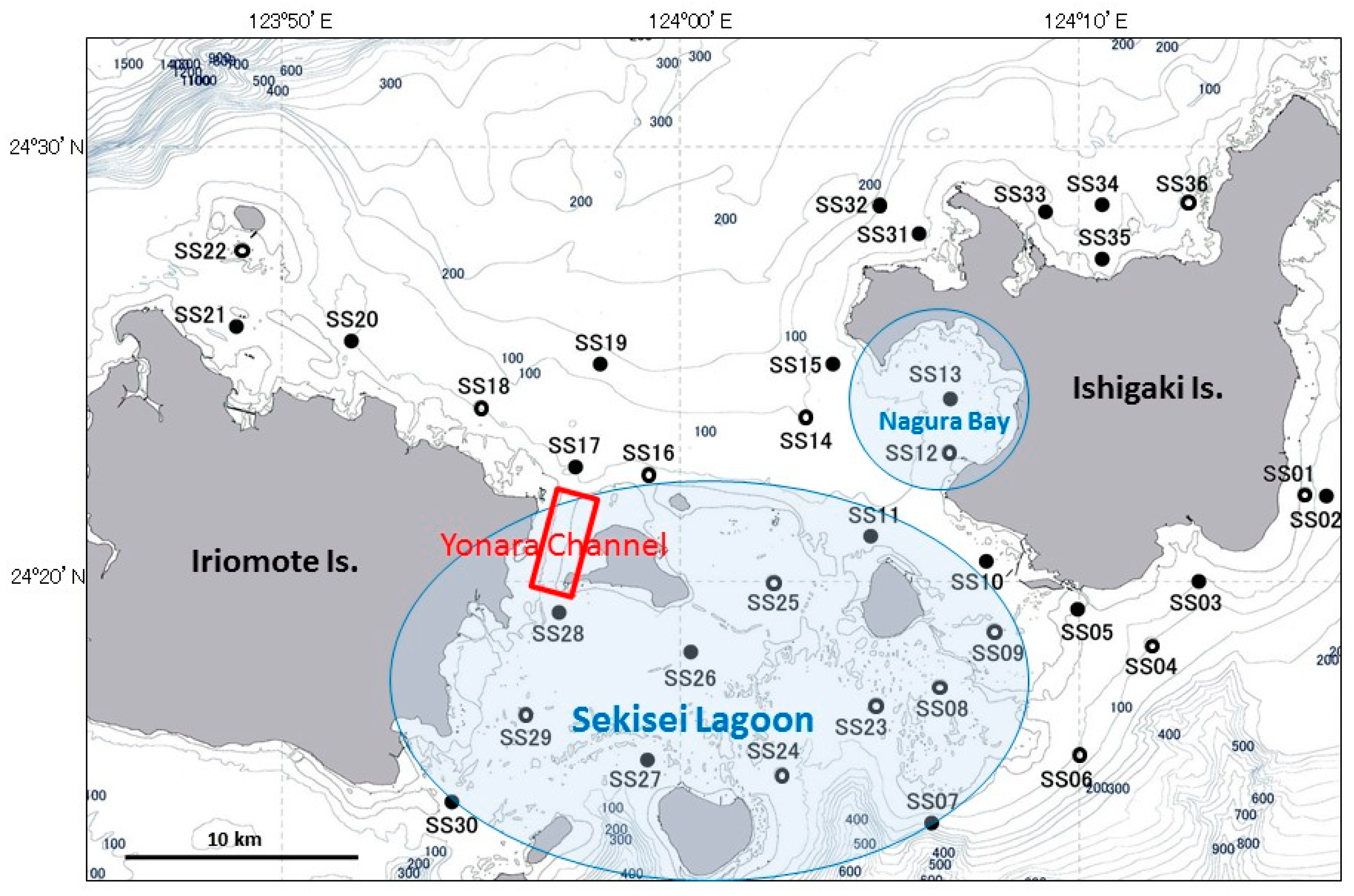
2.2. A. Planci Larvae and Seawater Sampling
2.3. Identification of A. Planci Larvae
2.4. Estimating Nutrient and Chl-a Concentrations
2.5. Statistical Analyses
- (1)
- High–High: + at target site, + at neighboring site (larval density was significantly higher at the target and neighboring sites)
- (2)
- High–Low: + at target site, − at neighboring site (larval density was significantly higher at the target site than that at the neighboring site)
- (3)
- Low–High: − at target site, + at neighboring site (larval density was significantly lower at the target site than that at the neighboring site)
- (4)
- Low–Low: − at target site, − at neighboring site (larval density was significantly lower at the target and neighboring sites)
3. Results
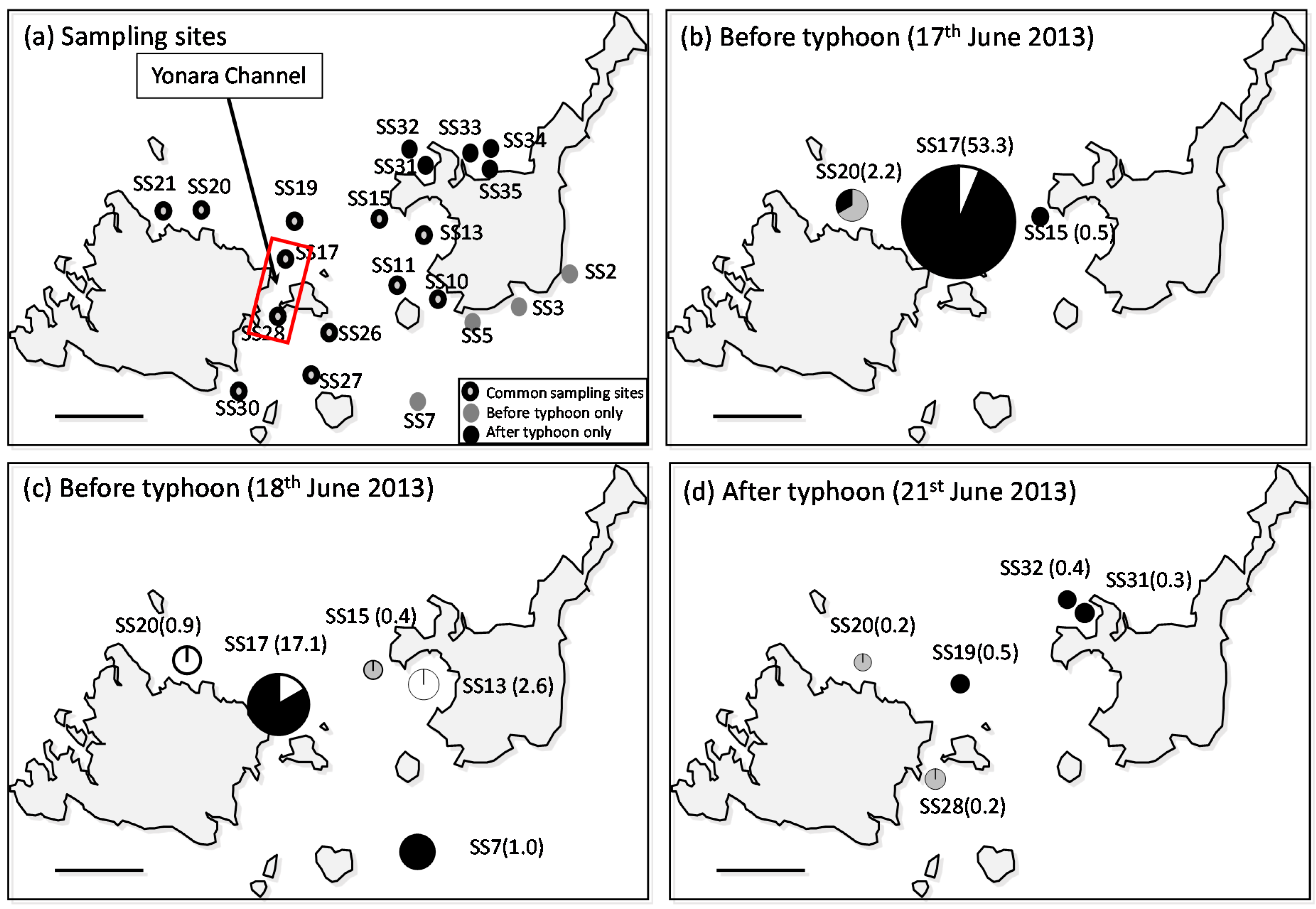
3.1. Larval Distribution
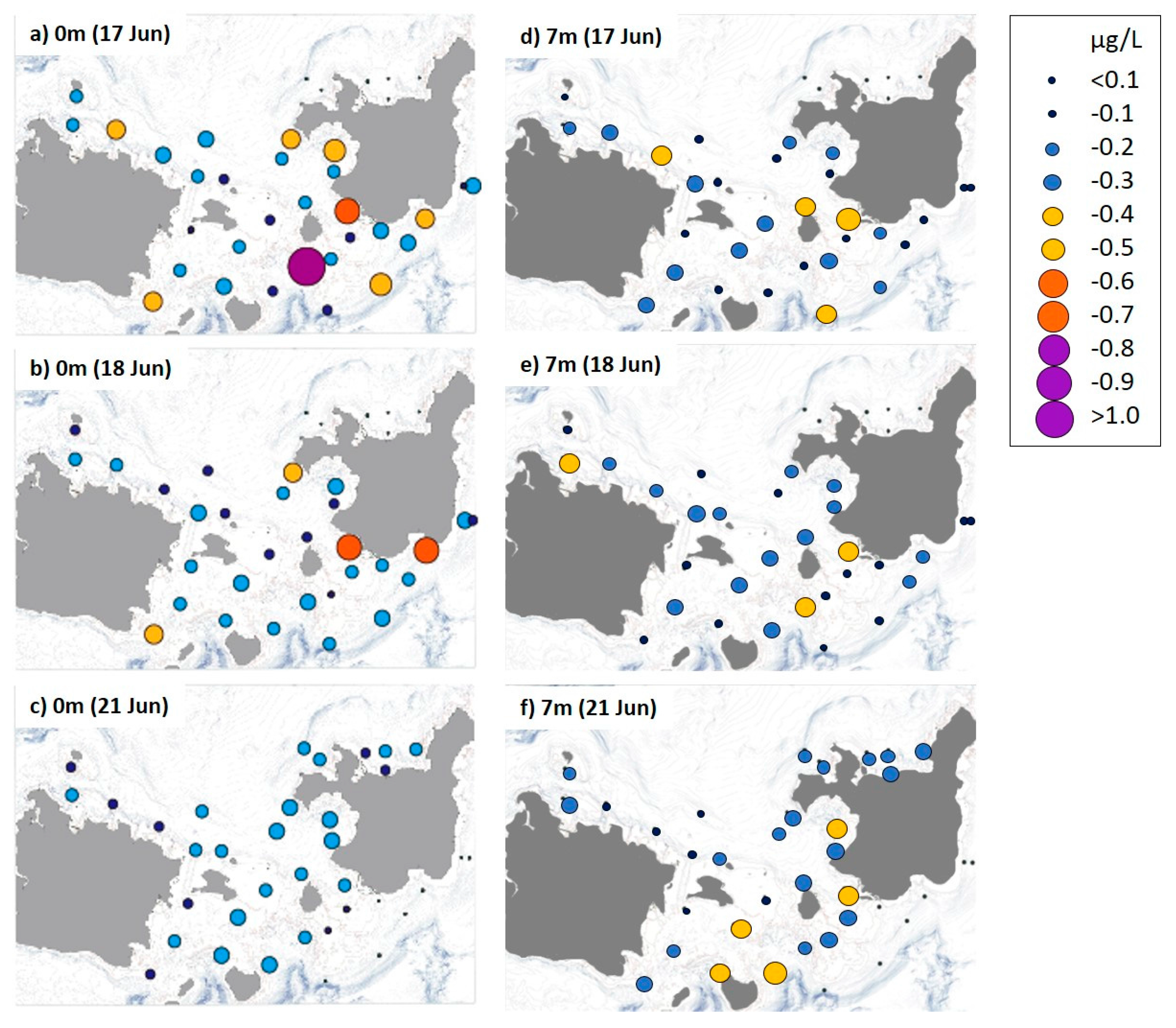
3.2. Distribution of Nutrient and Chl-a Concentrations
4. Discussion
4.1. High-Density A. Planci Larval Population before the Typhoon
4.2. A. Planci Larval Distribution and Density in Association with Food Availability
4.3. Possible A. Planci Larval Dispersal inside and outside Sekisei Lagoon
4.4. Screening Method Used in This Study
4.5. Future Perspectives; Prediction of Population Outbreaks
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pratchett, M.S.; Caballes, C.F.; Rivera-Posada, J.A.; Sweatman, H.P.A. Limits to understanding and managing outbreaks of crown-of-thorns starfish (Acanthaster spp.). In Oceanography and Marine Biology: An Annual Review; Hughes, R.N., Hughes, D.J., Smith, I.P., Eds.; CRC Press: Boca Raton, FL, USA, 2014; Volume 52, pp. 133–200. [Google Scholar]
- Pratchett, M. Dynamics of an outbreak population of Acanthaster planci at Lizard Island, northern Great Barrier Reef (1995–1999). Coral Reefs 2005, 24, 453–462. [Google Scholar] [CrossRef]
- Endean, R. Acanthaster planci infestations on reefs of the Great Barrier Reef. In Proceedings of the Third International Coral Reef Symposium, Miami, FL, USA, May 1977; Volume 1, pp. 185–191. Available online: http://www.reefbase.org/resource_center/publication/icrs.aspx?icrs=ICRS3 (accessed on 31 March 2016).
- Moran, P.; De’ath, G.; Baker, V.; Bass, D.; Christie, C.; Miller, I.; Miller-Smith, B.; Thompson, A. Pattern of outbreaks of crown-of-thorns starfish (Acanthaster planci L.) along the Great Barrier Reef since 1966. Mar. Freshw. Res. 1992, 43, 555–567. [Google Scholar] [CrossRef]
- Yamaguchi, M. Acanthaster planci infestations of reefs and coral assemblages in Japan: A retrospective analysis of control efforts. Coral Reefs 1986, 5, 23–30. [Google Scholar] [CrossRef]
- Suzuki, G.; Kai, S.; Yamashita, H. Mass stranding of crown-of-thorns starfish. Coral Reefs 2012, 31, 821. [Google Scholar] [CrossRef]
- Yasuda, N. Implications from the records of population outbreaks of crown-of-thorns Starfish Acanthaster planci in Japan over 100 years (1912–2015). In Coral Reef Studies in Japan; Iguchi, A., Hongo, C., Eds.; Chapter 10; Springer: Tokyo, Japan, 2016; under review. [Google Scholar]
- Fabricius, K.E.; Okaji, K.; De’ath, G. Three lines of evidence to link outbreaks of the crown-of-thorns seastar Acanthaster planci to the release of larval food limitation. Coral Reefs 2010, 29, 593–605. [Google Scholar] [CrossRef]
- Birkeland, C.; Lucas, J.S. Acanthaster Planci: Major Management Problem of Coral Reefs; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Yamaguchi, M. Larval behavior and geographic distribution of coral reef asteroids in the Indo-West Pacific. Micronesia 1977, 13, 283–296. [Google Scholar]
- Keesing, J.; Halford, A. Importance of postsettlement processes for the population dynamics of Acanthaster planci (L.). Mar. Freshw. Res. 1992, 43, 635–651. [Google Scholar] [CrossRef]
- Birkeland, C. Terrestrial runoff as a cause of outbreaks of Acanthaster planci (Echinodermata: Asteroidea). Mar. Biol. 1982, 69, 175–185. [Google Scholar] [CrossRef]
- Brodie, J. Enhancement of larval and juvenile survival and recruitment in Acanthatser planci from the effects of terrestrial runoff: A review. Mar. Freshw. Res. 1992, 43, 539–553. [Google Scholar] [CrossRef]
- Brodie, J.; Fabricius, K.; De’ath, G.; Okaji, K. Are increased nutrient inputs responsible for more outbreaks of crown-of-thorns starfish? An appraisal of the evidence. Mar. Pollut. Bull. 2005, 51, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, K.; Graba-Landry, A.; Dworjanyn, S.A.; Byrne, M. Larval starvation to satiation: Influence of nutrient regime on the success of Acanthaster planci. PLoS ONE 2015, 10, e0122010. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, K.; Graba-Landry, A.; Dworjanyn, S.A.; Byrne, M. Larval phenotypic plasticity in the boom-and-bust crown-of-thorns seastar, Acanthaster planci. Mar. Ecol. Prog. Ser. 2015, 539, 179–189. [Google Scholar] [CrossRef]
- Keesing, J.; Halford, A. Field measurements of survival rates of juvenile Acanthaster planci: Techniques and preliminary results. Mar. Ecol. Prog. Ser. 1992, 85, 107–114. [Google Scholar] [CrossRef]
- Sweatman, H. No-take reserves protect coral reefs from predatory starfish. Curr. Biol. 2008, 18, R598–R599. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, N.; Kajiwara, K.; Nagai, S.; Ikehara, K.; Nadaoka, K. The first report of field sampling and identification of the crown-of-thorns starfish larvae. Galaxea 2015, 17, 1–2. [Google Scholar] [CrossRef]
- Uthicke, S.; Doyle, J.; Duggan, S.; Yasuda, N.; McKinnon, A.D. Outbreak of coral-eating Crown-of-Thorns creates continuous cloud of larvae over 320 km of the Great Barrier Reef. Sci. Rep. 2015, 5, 16885. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.F.; Miyazawa, Y.; Cui, W.; Nadaoka, K. Numerical study of surface water circulation around Sekisei Lagoon, southwest Japan. Ocean Dyn. 2010, 60, 359–375. [Google Scholar] [CrossRef]
- Nadaoka, K.; Mitsui, J.; Hamasaki, K.; Harii, S.; Tamura, J.; Suzuki, Y. Field measurements of currents and turbidity, freshwater and thermal transport characteristics in Sekisei lagoon, Okinawa. Proc. Coast. Eng. JSCE 2003, 50, 1036–1040. (In Japanese) [Google Scholar]
- Hasegawa, H.; Yamano, H. Status of coral reefs around the country. In Coral Reefs of Japan; Ministry of the Environment, Japanese Coral Reef Society: Tokyo, Japan, 2004; pp. 153–286. [Google Scholar]
- Yasuda, N.; Ogasawara, K.; Kajiwara, K.; Ueno, M.; Oki, K.; Taniguchi, H.; Kakuma, S.; Okaji, K.; Nadaoka, K. Latitudinal differentiation of crown-of-thorns starfish (Acanthaster planci) in the reproduction patterns through the Ryukyu Island archipelago. Plankton Benthos Res. 2010, 5, 156–164. [Google Scholar] [CrossRef]
- Ministry of the Environment BCiJ. Annual Report of Coral Reef Monitoring around Sekisei Lagoon National Park and Its Surrounding Areas (H26, May 2014). Available online: http://www.biodic.go.jp/moni1000/findings/reports/index.html (accessed on 31 March 2016).
- Motoda, S. North Pacific standard plankton net. Inf. Bull. Planktology Japan 1957, 4, 13–15. [Google Scholar]
- Vogler, C.; Benzie, J.A.H.; Tenggardjaja, K.; Ambariyanto-Barber, P.H.; Wörheide, G. Phylogeography of the crown-of-thorns starfish: Genetic structure within the Pacific species. Coral Reefs 2013, 32, 515–525. [Google Scholar] [CrossRef]
- Timmers, M.; Bird, C.; Skillings, D.; Smouse, P.; Toonen, R. There’s no place like home: Crown-of-thorns outbreaks in the Central Pacific are regionally derived and independent events. PLoS ONE 2012, 7, e31159. [Google Scholar] [CrossRef] [PubMed]
- Timmers, M.A.; Andrews, K.R.; Bird, C.E.; de Maintenton, M.J.; Brainard, R.E.; Toonen, R.J. Widespread dispersal of the crown-of-thorns sea star, Acanthaster planci, across the Hawaiian archipelago and Johnston atoll. J. Mar. Biol. 2011, 2011, 10. [Google Scholar] [CrossRef]
- Yasuda, N.; Taquet, C.; Nagai, S.; Yoshida, T.; Adjeroud, M. Genetic connectivity of the coral-eating sea star Acanthaster planci during the severe outbreak of 2006–2009 in the Society Islands, French Polynesia. Mar. Ecol. 2015, 36, 668–678. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. In Nucleic Acids Symposium Series; Oxford University Press: Oxford, UK, 1999; Volume 41, pp. 95–98. [Google Scholar]
- Meeker, N.D.; Hutchinson, S.A.; Ho, L.; Trede, N.S. Method for isolation of PCR-ready genomic DNA from zebrafish tissues. Biotechniques 2007, 43, 610, 612 and 614. [Google Scholar] [CrossRef] [PubMed]
- The General Environmental Techinos co., LTD. Available online: http://www.kanso.co.jp/eng/index.html (accessed on 10 May 2016).
- Suzuki, R.; Ishimaru, T. An improved method for the determination of phytoplankton chlorophyll using N, N-dimethylformamide. J. Oceanogr. Soc. Japan 1990, 46, 190–194. [Google Scholar] [CrossRef]
- Holm-Hansen, O.; Lorenzen, C.J.; Holmes, R.W.; Strickland, J.D.H. Fluorometric determination of chlorophyll. J. Cons. 1965, 30, 3–15. [Google Scholar] [CrossRef]
- Moran, P.A.P. Notes on continuous stochastic phenomena. Biometrika 1950, 37, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Anselin, L. Local indicators of spatial association—LISA. Geogr. Anal. 1995, 27, 93–115. [Google Scholar] [CrossRef]
- Nanami, A.; Ohta, I.; Sato, T. Estimation of spawning migration distance of the white-streaked grouper (Epinephelus ongus) in an Okinawan coral reef system using conventional tag-and-release. Environ. Biol. Fishes 2014, 98, 1387–1397. [Google Scholar] [CrossRef]
- Nakamura, M.; Okaji, K.; Higa, Y.; Yamakawa, E.; Mitarai, S. Spatial and temporal population dynamics of the crown-of-thorns starfish, Acanthaster planci, over a 24-year period along the central west coast of Okinawa Island, Japan. Mar. Biol. 2014, 161, 2521–2530. [Google Scholar] [CrossRef]
- Yamaguchi, M.; (Luminous Himuka Research Institute, Miyazaki, Japan). personal communication, 18 August 2012.
- Lucas, J.S. Quantitative studies of feeding and nutrition during larval development of the coral reef asteroid Acanthaster planci (L.). J. Exp. Mar. Biol. Ecol. 1982, 65, 173–193. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O. Uptake of dissolved organic matter by larval stage of the crown-of-thorns starfish Acanthaster planci. Mar. Biol. 1994, 120, 55–63. [Google Scholar]
- Johnson, L.G.; Cartwright, C.M. Thyroxine-accelerated larval development in the crown-of-thorns starfish, Acanthaster planci. Biol. Bull. 1996, 190, 299–301. [Google Scholar] [CrossRef]
- Basch, L.V. Effects of algal and larval densities on development and survival of asteroid larvae. Mar. Biol. 1996, 126, 693–701. [Google Scholar] [CrossRef]
- Roche, R.C.; Pratchett, M.S.; Carr, P.; Turner, J.R.; Wagner, D.; Head, C.; Sheppard, C.R. Localized outbreaks of Acanthaster planci at an isolated and unpopulated reef atoll in the Chagos Archipelago. Mar. Biol. 2015, 162, 1695–1704. [Google Scholar] [CrossRef]
- Dight, I.; James, M.; Bode, L. Modelling the larval dispersal of Acanthaster planci II. Patterns of reef connectivity. Coral Reefs 1990, 9, 125–134. [Google Scholar] [CrossRef]
- Nash, W.J.; Goddard, M.; Lucas, J.S. Population genetic studies of the crown-of-thorns starfish, Acanthaster planci (L.), in the Great Barrier Reef region. Coral Reefs 1988, 7, 11–18. [Google Scholar] [CrossRef]
- Benzie, J. Review of the genetics, dispersal and recruitment of crown-of-thorns starfish (Acanthaster planci). Mar. Freshw. Res. 1992, 43, 597–610. [Google Scholar] [CrossRef]
- Yasuda, N.; Nagai, S.; Hamaguchi, M.; Okaji, K.; Gérard, K.; Nadaoka, K. Gene flow of Acanthaster planci (L.) in relation to ocean currents revealed by microsatellite analysis. Mol. Ecol. 2009, 18, 1574–1590. [Google Scholar] [CrossRef] [PubMed]
- Houk, P.; Bograd, S.; van Woesik, R. The transition zone chlorophyll front can trigger Acanthaster planci outbreaks in the Pacific Ocean: Historical confirmation. J. Oceanogr. 2007, 63, 149–154. [Google Scholar] [CrossRef]
- Johnson, C. Settlement and recruitment of Acanthatser planci on the Great Barrier Reef: Questions of process and scale. Mar. Freshw. Res. 1992, 43, 611–627. [Google Scholar] [CrossRef]
- Johnson, C.; Sutton, D.; Olson, R.; Giddins, R. Settlement of crown-of-thorns starfish: Role of bacteria on surfaces of coralline algae and a hypothesis for deepwater recruitment. Mar. Ecol. Prog. Ser. 1991, 71, 143–162. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, G.; Yasuda, N.; Ikehara, K.; Fukuoka, K.; Kameda, T.; Kai, S.; Nagai, S.; Watanabe, A.; Nakamura, T.; Kitazawa, S.; et al. Detection of a High-Density Brachiolaria-Stage Larval Population of Crown-of-Thorns Sea Star (Acanthaster planci) in Sekisei Lagoon (Okinawa, Japan). Diversity 2016, 8, 9. https://doi.org/10.3390/d8020009
Suzuki G, Yasuda N, Ikehara K, Fukuoka K, Kameda T, Kai S, Nagai S, Watanabe A, Nakamura T, Kitazawa S, et al. Detection of a High-Density Brachiolaria-Stage Larval Population of Crown-of-Thorns Sea Star (Acanthaster planci) in Sekisei Lagoon (Okinawa, Japan). Diversity. 2016; 8(2):9. https://doi.org/10.3390/d8020009
Chicago/Turabian StyleSuzuki, Go, Nina Yasuda, Kohta Ikehara, Kouki Fukuoka, Takahiko Kameda, Sayaka Kai, Satoshi Nagai, Atushi Watanabe, Takashi Nakamura, Shunsuke Kitazawa, and et al. 2016. "Detection of a High-Density Brachiolaria-Stage Larval Population of Crown-of-Thorns Sea Star (Acanthaster planci) in Sekisei Lagoon (Okinawa, Japan)" Diversity 8, no. 2: 9. https://doi.org/10.3390/d8020009




