Electrochemical Investigation of Oligonucleotide-DNA Hybridization on Poly(4-Methoxyphenethylamine)
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Apparatus
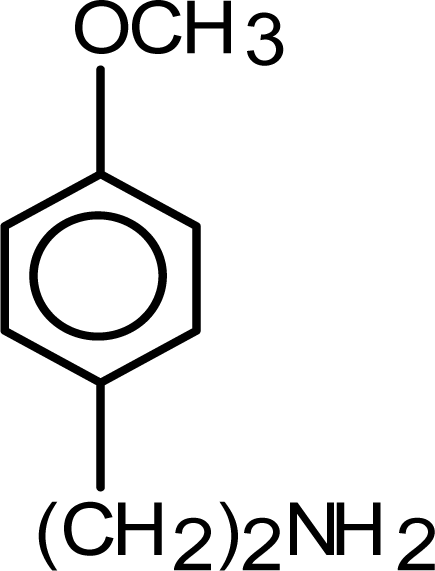
2.3. Production of poly(4-methoxyphenethylamine) films
2.4. Nitrogenated bases and oligonucleotide probe immobilization on graphite electrode/poly(4-methoxyphenethylamine)
2.5. Hybridization investigation of oligonucleotide immobilized onto poly(4-methoxyphenethylamine), using methylene blue as redox indicator or guanine and adenine monitoring
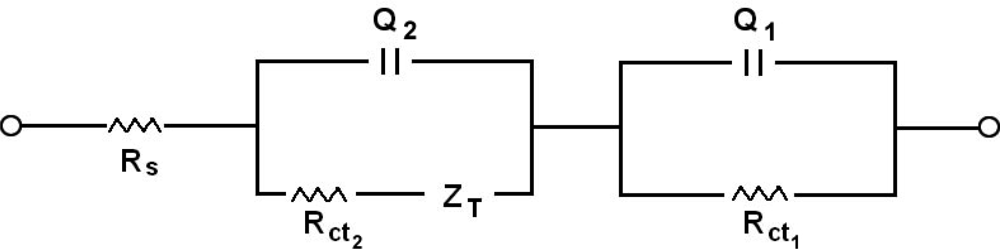
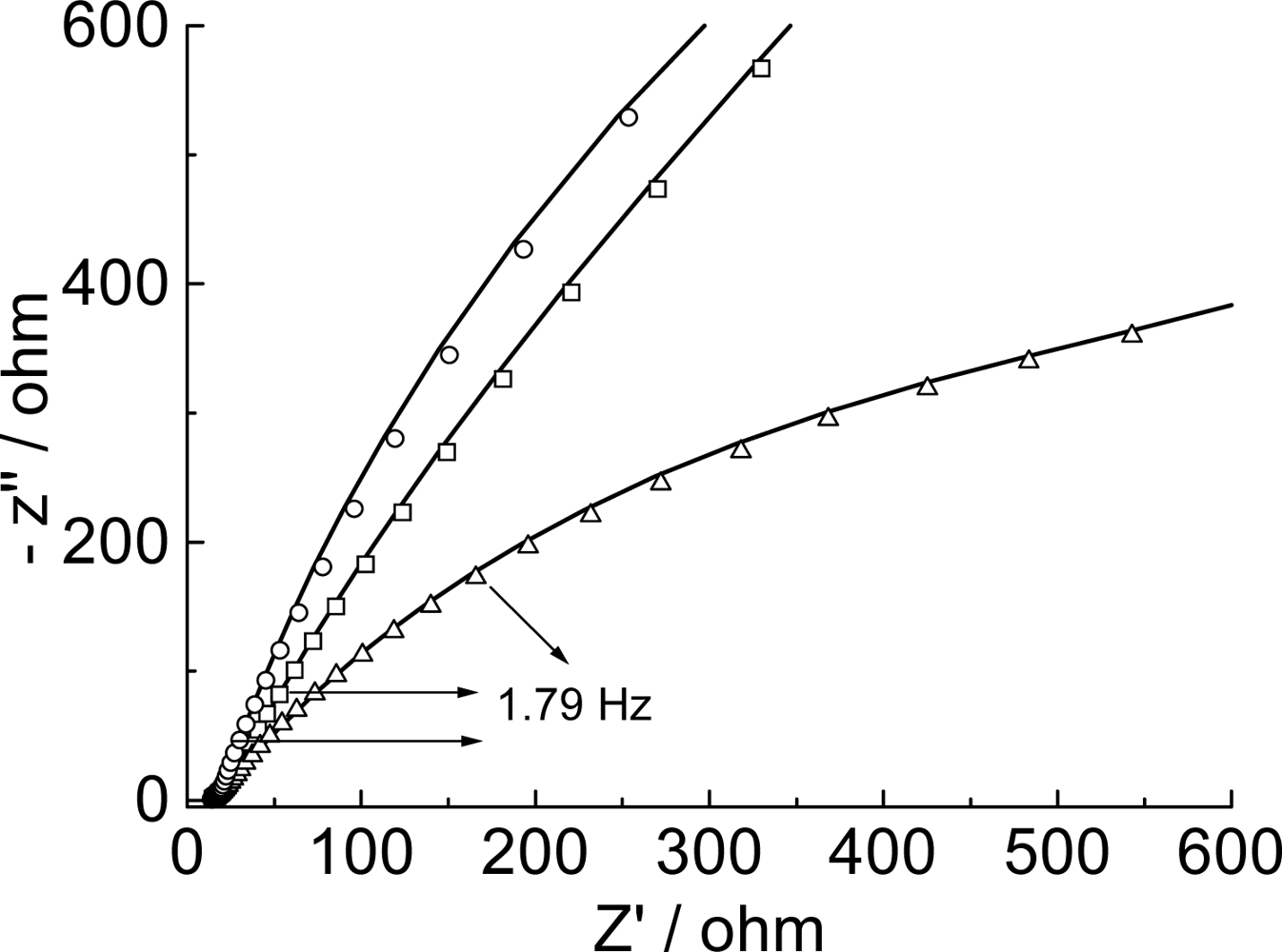
2.6. Impedance measurements
3. Results and discussion
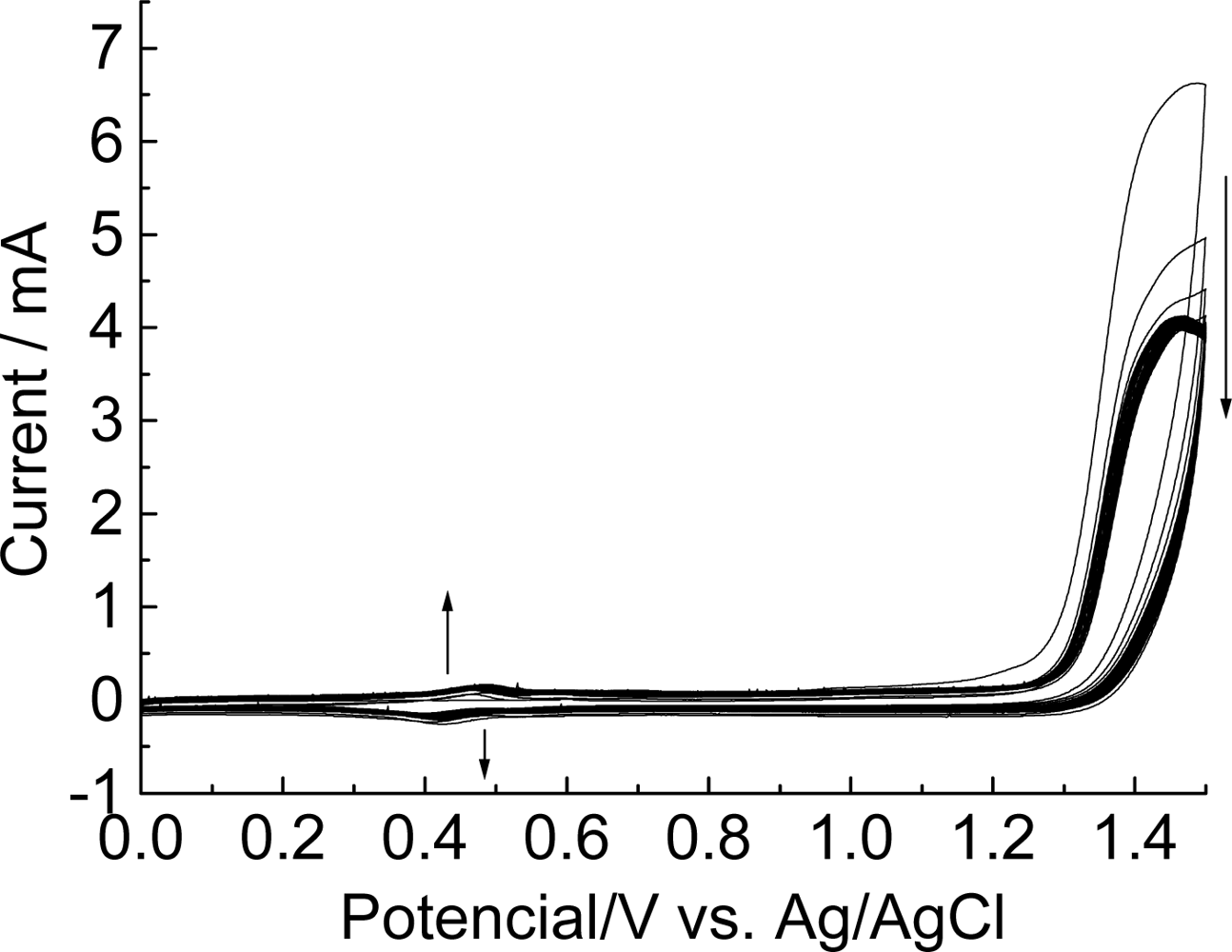
3.1. Electrochemical behavior of methoxyphenethylamine
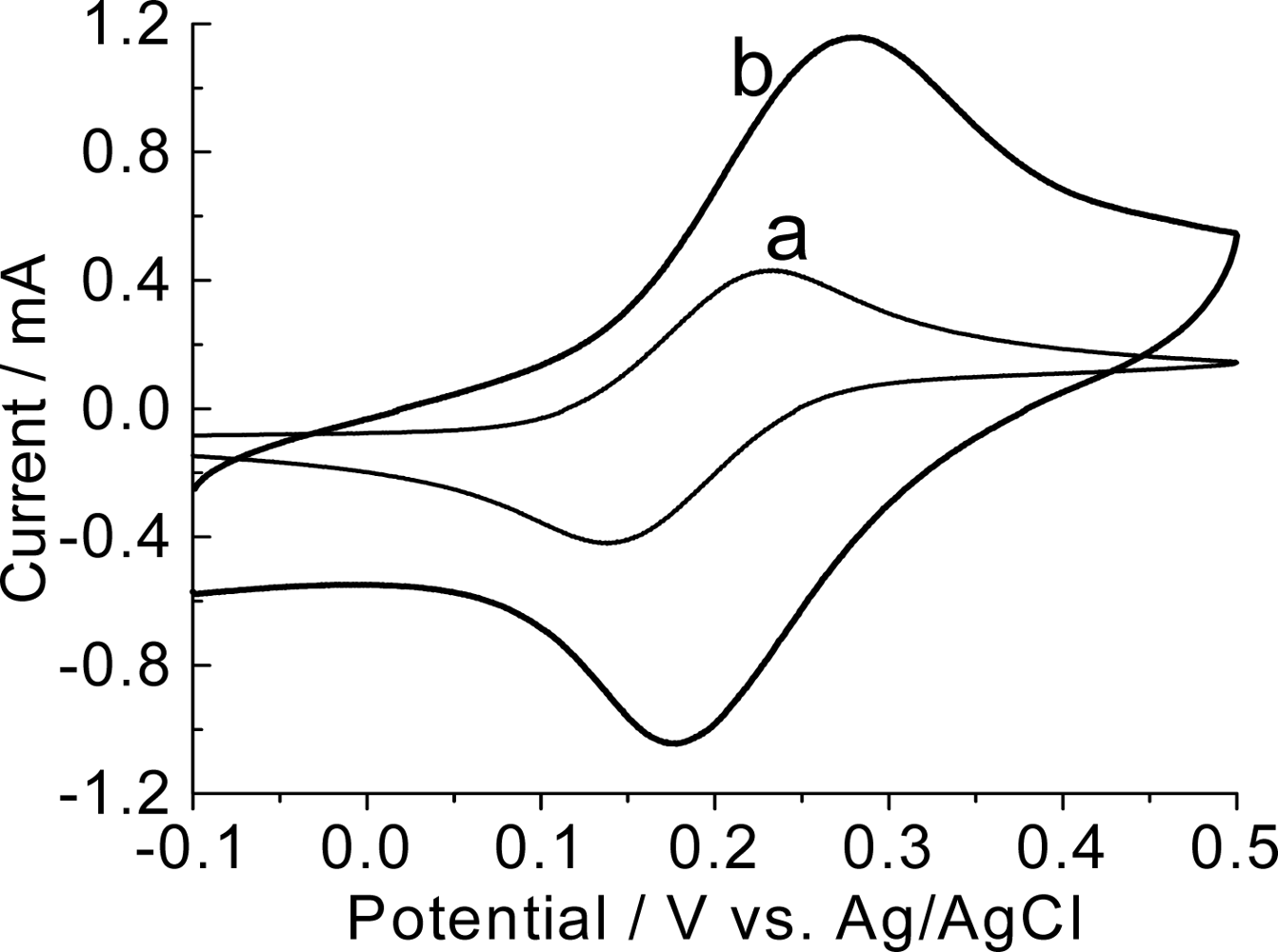
3.2. Electrochemical characterization of the modified electrodes
3.3. Immobilization of purines and pyrimidines
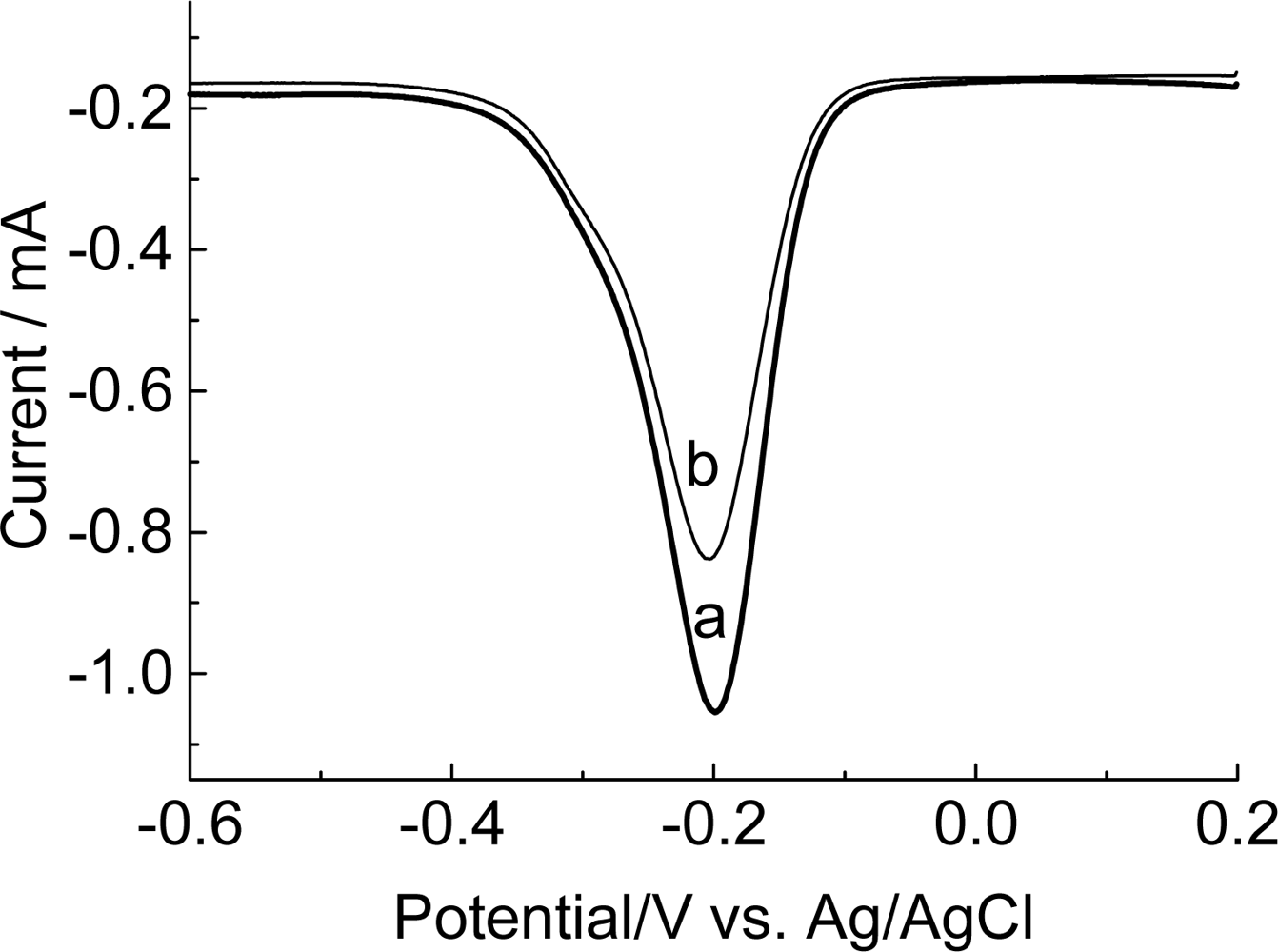
3.4. Investigation of the oligonucleotide immobilization
4. Conclusions
Acknowledgments
References
- Banks, CE; Davies, TJ; Wildgoose, GG; Compton, RG. Electrocatalysis at Graphite and Carbon Nanotube Modified Electrodes: Edge-Plane Sites and Tube Ends Are the Reactive Sites. Chem Commun 2005, 21, 829–841. [Google Scholar]
- Blanco-Lopez, MC; Lobo-Castanon, MJ; Miranda-Ordieres, AJ; Tunon-Blanco, P. Voltammetric Sensor for Vanillylmandelic Acid Based on Molecularly Imprinted Polymer-Modified Electrodes. Biosens Bioelectron 2003, 18, 353–362. [Google Scholar]
- Kutner, W; Wang, J; L'her, M; Buck, R. Analytical Aspects of Chemically Modified Electrodes: Classification, Critical Evaluation and Recommendations. Pure Appl Chem 1998, 70, 1301–1318. [Google Scholar]
- Chidsey, CED; Murray, RW. Electroactive Polymers and Macromolecular Electronics. Science 1986, 231, 25–31. [Google Scholar]
- Bardavid, Y; Kotlyar, AB; Yitzchaik, S. Conducting Polymers Coated DNA. Macromol Symp 2006, 240, 102–106. [Google Scholar]
- Kulkarni, MV; Viswanath, AK; Khanna, PK. Synthesis and Characterization of Poly(N-Methyl Aniline) Doped with Sulphonic Acids: Their Application as Humidity Sensors. J Appl Polym Sci 2006, 99, 812–820. [Google Scholar]
- Menezes, HA; Maia, G. Films Formed by the Electrooxidation of p-Aminophenol (p-Aph) in Aqueous Medium: What Do They Look Like? J Electroanal Chem 2006, 586, 39–48. [Google Scholar]
- Topcu-Sulak, M; Gokdogan, O; Gulce, A; Gulce, H. Amperometric Glucose Biosensor Based on Gold-Deposited Polyvinylferrocene Film on Pt Electrode. Biosens Bioelectron 2006, 21, 1719–1726. [Google Scholar]
- Ortega, JM. Electrodeposition of Copper on Poly (o-Aminophenol) Modified Platinum Electrode. Thin Solid Films 2000, 260, 159–165. [Google Scholar]
- Lofrano, RCZ; Madurro, JM; Abrantes, LM; Romero, JR. Electrocatalytic Hydrogenation of Carbonylic Compounds Using an Electrode with Platinum Particles Dispersed in Films of Poly-[Allyl Ether P-(2-Aminoethyl) Phenol]. J Mol Catal A: Chem 2004, 218, 73–79. [Google Scholar]
- Lofrano, RCZ; Madurro, JM; Romero, JR. Preparation and Properties of an Electrode Coated with a Cerium Poly(Allyl Ether p-Benzenesulfonate) Film for Electrorganic Reactions. J Mol Catal A: Chem 2000, 153, 237–242. [Google Scholar]
- Castro, CM; Vieira, SN; Goncalves, RA; Madurro, AGB; Madurro, JM. Electrochemical and Morphologic Studies of Nickel Incorporation on Graphite Electrodes Modified with Polytyramine. J Mater Sci 2008, 43, 475–482. [Google Scholar]
- Franco, DL; Afonso, AS; Vieira, SN; Ferreira, LF; Goncalves, RA; Madurro, AGB; Madurro, JM. Electropolymerization of 3-Aminophenol on Carbon Graphite Surface: Electric and Morphologic Properties. Mater Chem Phys 2008, 107, 404–409. [Google Scholar]
- Vieira, SN; Ferreira, LF; Franco, DL; Afonso, AS; Goncalves, RA; Madurro, AGB; Madurro, JM. Electrochemical Modification of Graphite Electrodes with Poly(4-Aminophenol). Macromol Symp 2006, 245, 236–242. [Google Scholar]
- Rho, S; Jahng, D; Lim, JH; Choi, J; Chang, JH; Lee, SC; Kim, KJ. Electrochemical DNA Biosensors Based on Thin Gold Films Sputtered on Capacitive Nanoporous Niobium Oxide. Biosens Bioelectron 2008, 23, 852–856. [Google Scholar]
- Ferreira, LF; Boodts, JFC; Madurro, AGB; Madurro, JM. Gold Electrodes Modified with Poly(4-Aminophenol): Incorporation of Nitrogenated Bases and an Oligonucleotide. Polym Int 2008, 57, 644–650. [Google Scholar]
- Madurro, AGB; Ferreira, LF; Vieira, SN; Goulart, LR; Madurro, JM. Immobilization of Purine Bases in Poly-4-Aminophenol Matrix. J Mater Sci 2007, 42, 3238–3243. [Google Scholar]
- Popovich, ND; Eckhardt, AE; Mikulecky, JC; Napier, ME; Thomas, RS. Electrochemical Sensor for Detection of Unmodified Nucleic Acids. Talanta 2002, 56, 821–828. [Google Scholar]
- Wang, J; Palecek, E; Nielsen, PE; Rivas, G; Cai, X; Shiraishi, H; Dontha, N; Luo, D; Farias, PAM. Peptide Nucleic Acid Probes for Sequence-Specific DNA Biosensors. J Am Chem Soc 1996, 118, 7667–7670. [Google Scholar]
- Xu, H; Liu, X; Cui, D; Li, M; Jiang, M. A Novel Method for Improving the Performance of ZnO Gas Sensors. Sens Actuators B 2006, 114, 301–307. [Google Scholar]
- Palecek, E. From Polarography of DNA to Microanalysis with Nucleic Acid-Modified Electrodes. Electroanalysis 1996, 8, 7–14. [Google Scholar]
- Mikkelson, SR. Electrochemical Biosensors for DNA Sequence Detection. Electroanalysis 1996, 8, 15–19. [Google Scholar]
- Palecek, E; Postbieglova, I. Adsorptive Stripping Voltammetry of Biomacromolecules with Transfer of the Adsorbed Layer. J Electroanal Chem 1986, 214, 359–371. [Google Scholar]
- Palecek, E. Oscillographic Polarography of Highly Polymerized Deoxyribonucleic Acid. Nature 1960, 188, 656–657. [Google Scholar]
- Lin, H; Xu, DK; Chen, HY. Simultaneous Determination of Purine Bases, Ribonucleosides and Ribonucleotides by Capillary Electrophoresis-Electrochemistry with a Copper Electrode. J Chromatogr 1997, 760, 227–233. [Google Scholar]
- Kafil, JB; Cheng, HY; Last, T. Quantitation of Nucleic Acids at the Picogram Level Using HPLC with Electrochemical Detection. Anal Chem 1986, 58, 285–289. [Google Scholar]
- Oliveira-Brett, AM; Piedade, JA; Silva, LA; Diculescu, VC. Voltammetric Determination of all DNA Nucleotides. Anal Biochem 2004, 332, 321–329. [Google Scholar]
- Ozkan, D; Erdem, A; Kara, P; Kerman, K; Meric, B; Hassmann, J; Ozsoz, M. Allele-Specific Genotype Detection of Factor V Leiden Mutation from Polymerase Chain Reaction Amplicons Based on Label-Free Electrochemical Genosensor. Anal Chem 2002, 74, 5931–5936. [Google Scholar]
- Lassalle, N; Vieil, E; Correia, JP; Abrantes, LM. Study of DNA Hybridization on Polypyrrole Grafted with Oligonucleotides by Photocurrent Spectroscopy. Biosens Bioelectron 2001, 16, 295–303. [Google Scholar]
- Franco, DL; Afonso, AS; Ferreira, LF; Goncalves, RA; Boodts, JFC; Madurro, AGB; Madurro, JM. Electrodes Modified with Polyaminophenols: Immobilization of Purines and Pyrimidines. Pol Eng Sci 2008, in press. [Google Scholar]
- Stempkowska, I; Ligaj, M; Jasnowska, J; Langer, J; Filipiak, M. Electrochemical Response of Oligonucleotides on Carbon Paste Electrode. Bioelectrochemistry 2007, 70, 488–494. [Google Scholar]
- Pham, MC; Piro, B; Tran, LD. Direct Electrochemical Detection of Oligonucleotide Hybridization on Poly(5-Hydroxy-1,4-Naphthoquinone-Co-5-Hydroxy-3-Thioacetic Acid-1,4-Naphthoquinone) Film. Anal Chem 2003, 75, 6748–6752. [Google Scholar]
- Okutucu, B; Telefoncu, A. Covalent Attachment of Oligonucleotides to Cellulose Acetate Membranes. Artif Cells Blood Substit Immobil Biotechnol 2004, 32, 599–608. [Google Scholar]
- Yang, M; McGovern, ME; Thompson, M. Genosensor Technology and the Detection of Interfacial Nucleic Acid Chemistry. Anal Chim Acta 1997, 346, 259–275. [Google Scholar]
- La-Scalea, MA; Serrano, SHP; Gutz, IGR. Eletrodos Modificados com DNA: uma Nova Alternativa em Eletroanálise. Quím Nova 1999, 22, 417–424. [Google Scholar]
- Jin, Y; Yao, X; Liu, Q; Li, J. Hairpin DNA Probe Based Electrochemical Biosensor Using Methylene Blue as Hybridization Indicator. Biosens Bioelectron 2007, 22, 1126–1130. [Google Scholar]
- Yan, F; Erdem, A; Meric, B; Kerman, K; Ozsoz, M. Electrochemical DNA Biosensor for the Detection of Specific Gene Related to Microcystis Species. Electrochem Commum 2001, 3, 224–228. [Google Scholar]
- Rohs, R; Sklenar, H; Lavery, R; Roder, B. Methylene Blue Binding to DNA with Alternating GC Base Sequence: a Modeling Study. J Am Chem Soc 2000, 122, 2860–2866. [Google Scholar]
- Enescu, M; Bernard, L; Gheorghe, V. Molecular Dynamics Simulation of Methylene Blue – Guanine Complex in Water: the Role of Solvent in Stacking. J Phys Chem 2000, 104, 1073–1077. [Google Scholar]
- Kara, P; Kermam, K; Ozkan, D; Meric, B; Erdem, A; Ozkan, Z; Ozsoz, M. Electrochemical Genosensor for the Detection of Interaction Between Methylene Blue and DNA. Electrochem Commun 2002, 4, 705–709. [Google Scholar]
- Bonanni, A; Esplandiu, MJ; Pividori, MI; Alegret, S; del Valle, M. Impedimetric Genosensors for the Detection of DNA Hybridization. Anal Bioanal Chem 2006, 385, 1195–1201. [Google Scholar]

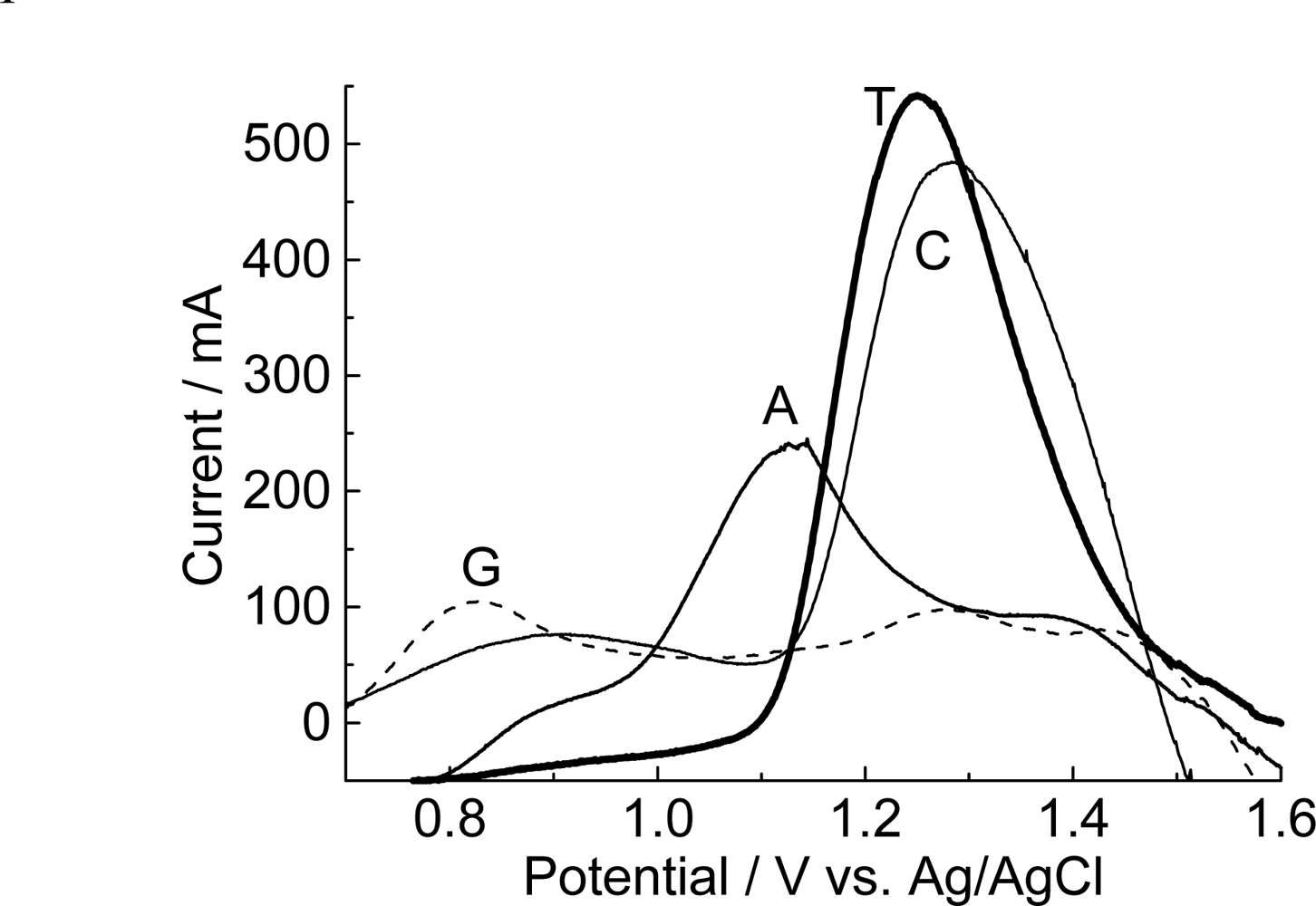




| Guanine | Adenine | Thymine | Cytosine | ||||
|---|---|---|---|---|---|---|---|
| Potential
(V) | Current
(mA) | Potential
(V) | Current
(mA) | Potential
(V) | Current
(mA) | Potential
(V) | Current
(mA) |
| 0.829 | 103 | 1.132 | 236 | 1.250 | 541 | 1.277 | 482 |
| RS(′Ω) | Q1 | R1ct(ohm) | Q2 | R2(ohm) | ZT | ||||
|---|---|---|---|---|---|---|---|---|---|
| Y0(μF) | n1 | Y0(mF) | n2 | Y0 | B | ||||
| Film | 15.7 | 13.4 | 0.62 | 875 | 0.1250 | 0.736 | 326 | 0.0200 | 0.201 |
| Film/Poly(GA) | 13.3 | 36.8 | 0.46 | 5.55 | 0.5401 | 0.848 | 3330 | - | - |
| Filme/Poly(GA): Poly(CT) | 14.0 | 32.0 | 0.54 | 5.16 | 0.1080 | 0.762 | 4400 | 0.003564 | 0.177 |
Share and Cite
Silva, F.B.; Vieira, S.N.; Goulart, L.R., Filho; Boodts, J.F.C.; Brito-Madurro, A.G.; Madurro, J.M. Electrochemical Investigation of Oligonucleotide-DNA Hybridization on Poly(4-Methoxyphenethylamine). Int. J. Mol. Sci. 2008, 9, 1173-1187. https://doi.org/10.3390/ijms9071173
Silva FB, Vieira SN, Goulart LR Filho, Boodts JFC, Brito-Madurro AG, Madurro JM. Electrochemical Investigation of Oligonucleotide-DNA Hybridization on Poly(4-Methoxyphenethylamine). International Journal of Molecular Sciences. 2008; 9(7):1173-1187. https://doi.org/10.3390/ijms9071173
Chicago/Turabian StyleSilva, Francielle B., Sabrina N. Vieira, Luiz R. Goulart, Filho, Julien F. C. Boodts, Ana G. Brito-Madurro, and João M. Madurro. 2008. "Electrochemical Investigation of Oligonucleotide-DNA Hybridization on Poly(4-Methoxyphenethylamine)" International Journal of Molecular Sciences 9, no. 7: 1173-1187. https://doi.org/10.3390/ijms9071173




