1. Introduction
Reactive nitrogen and oxygen species (RNS, ROS) have been intensively studied in recent years with regard to their relevant physiological and pathological importance connected also with oxidative stress. Antioxidant compounds are able to neutralize the excess of ROS or RNS and, as a consequence of this activity, it has been suggested that they play an important role in prevention of many diseases, e.g. atherosclerosis, cardiovascular and neurological diseases and cancer [
1–
3]. The interest in searching for antioxidants that can be consumed in the habitual diet has increased considerably in the last years. Polyphenolic compounds have shown strong antioxidant effects [
4–
6] and so it has been suggested that they are responsible for reducing the risk of diseases associated with oxidative stress. Flavonoids, a family of polyphenolic compounds, are an important group of antioxidants present in vegetables and fruits and other products made from these, such as wine, beer or juice. Catechins, anthocyanins and procyanidins are an interesting class of flavonoids ubiquitously found in food. Catechins and procyanidins (
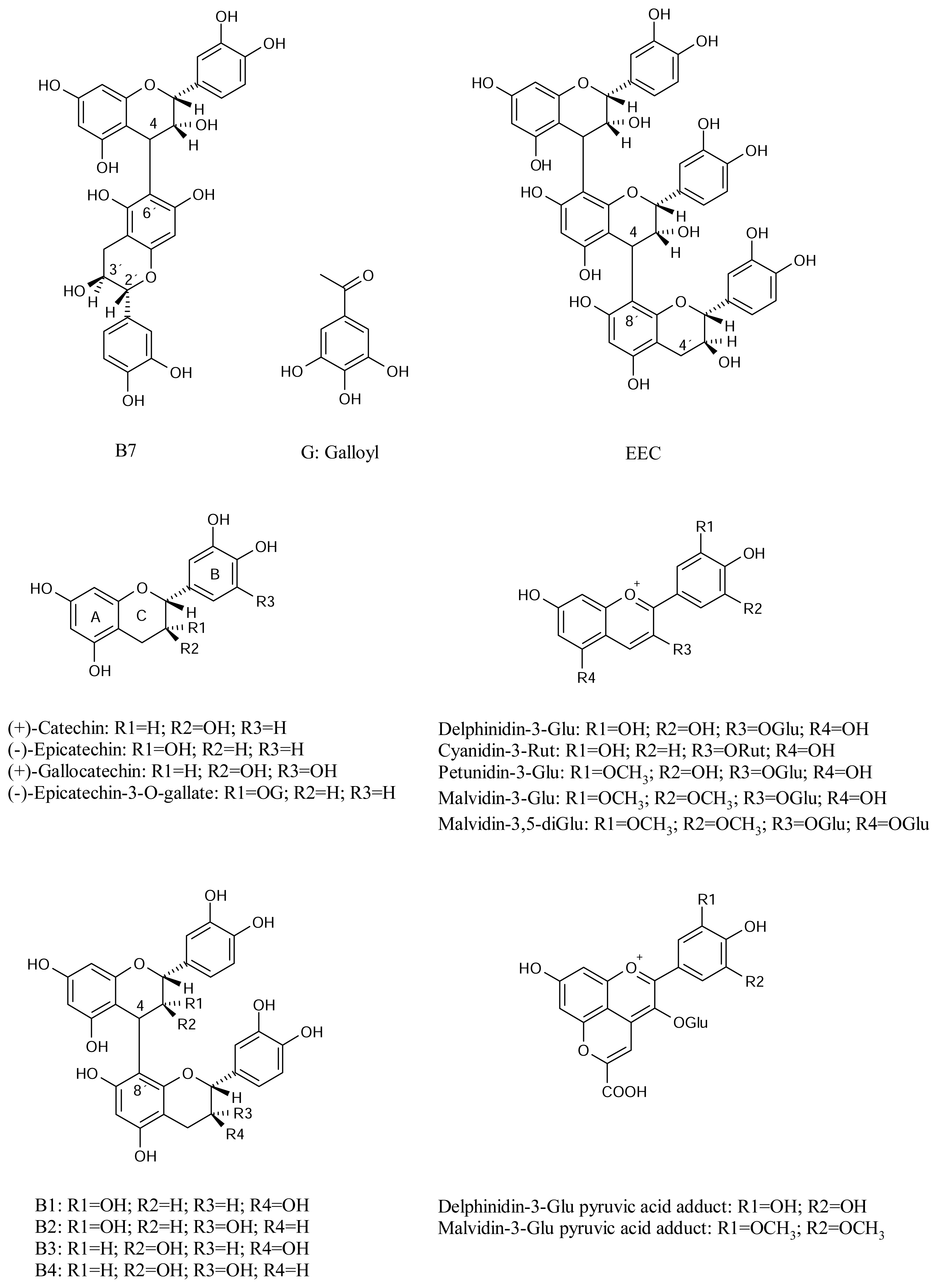
Figure 1), found in some fruits, such as plum and apple, as well as in tea and red wine [
7], have versatile biological effects such as anticancer, antiallergy and antioxidant activities [
8]. The main dietary sources of anthocyanin (
Figure 1), pigments responsible for the red-blue color, include red-colored fruits, vegetables and red wine. They have shown ability to prevent lipid oxidation and scavenging activity against free radicals [
9]. During red wine ageing, there is a loss of anthocyanins and, it appears, other pigments, anthocyanin-derived pigments, which are denominated pyranoanthocyanins. Some of these pigments are formed through the interaction of the original anthocyanins with pyruvic acid, and their structures (
Figure 1) are based on the anthocyanin-3-glucosides with additional C
3O
2 between position C4 and the 5-hydroxyl group of the molecule [
10]. The biological properties of these compounds have been little studied.
It has been proposed that regular consumption of red wine in moderate amounts reduces the risk of coronary heart disease via protection of LDL against oxidative damage and via inhibition of platelet aggregation [
11].
There are different methods to evaluate the
in vitro antioxidant capacity of isolated compounds, mixtures of compounds, biological fluids and tissues which involve different mechanisms of determination of antioxidant activity, for example: chemical methods based on scavenging of ROS or RNS such as peroxynitrite [
12], the hydroxyl radical and superoxide [
13]. Other methods measure the disappearance of free radicals using spectrophotometry, such as ABTS
•+ (2,2′-azinobis-(3-ethyl-benzothiazoline- 6-sulphonate) cation radical) [
14] or DPPH (2,2-diphenyl-1-picrylhydrazyl) [
15]. Other assays to determine the total antioxidant power include techniques such as the ferric reducing/antioxidant power method [
16] or use the in situ electrochemically generated bromine [
17]. The results in the measurement of antioxidant capacity depend on the method used. This is because a single method can not give a comprehensive prediction of antioxidant efficacy of the different compounds [
18].
The purpose of the present study was 2-fold: 1) to evaluate the in vitro antioxidant activity of catechins, procyanidins, anthocyanins and pyranoanthocyanins from wine by four methods based on different mechanisms 2) to investigate statistically the inter-relationship between the in vitro methods used.
2. Results and Discussion
Four in vitro methods based on different mechanisms of determination of the antioxidant capacity were used to test polyphenolic compounds. The inter-relationships between these methods were examined for all the tested compounds. The first method, peroxynitrite scavenging activity, was based on automated measurement of the inhibition of peroxynitrite mediated tyrosine nitration by the tested natural compounds. The next, the TEAC assay, was used for measuring the capacity of tested compounds to scavenge the stable cation radical ABTS•+ compared to Trolox C, a water soluble analogue of vitamin E. The FRAP assay, based on the reduction of a ferric-tripyridyltriazine complex to its ferrous, colored form was used to assess the total reducing power of antioxidants. The last, TBARS, was based on determination of the inhibition of peroxidation of the phosphatidylcholine substrate induced by the ascorbate/iron complex in lipid phase system.
The remarkable antioxidant activities of catechins, procyanidins, anthocyanins and pyranoanthocyanins in these different systems were analyzed and the results are presented in
Table 1. TBARS data for anthocyanins and pyranoanthocyanins are not presented in this table. IC50’s could not be determined since they present a maximum of absorbance around 520 nm, very close to the wavelength fixed for the measurement in this antioxidant method. It was, therefore, necessary to find a blank for these color compounds to avoid the interference caused by the color of the tested compounds. The blank should represent 100% inhibition of the sample in the assay, defined as baseline peroxidation of phosphatidylcholine without added iron/ascorbate. In these cases, due to the chemical properties of the studied pigments, it was impossible to find an adequate blank. These compounds in the acid mixture of the reaction, without Fe
III and ascorbate, present a red-pink color. Nevertheless, when Fe
III was added it resulted in unstable products which presented even less color than the blank. It is known that anthocyanins in acid medium are in a flavylium form and develop red-blue color and that in the presence of metal ions (such as Fe
III) they are able to form chelates and this could be the cause of the change in color.
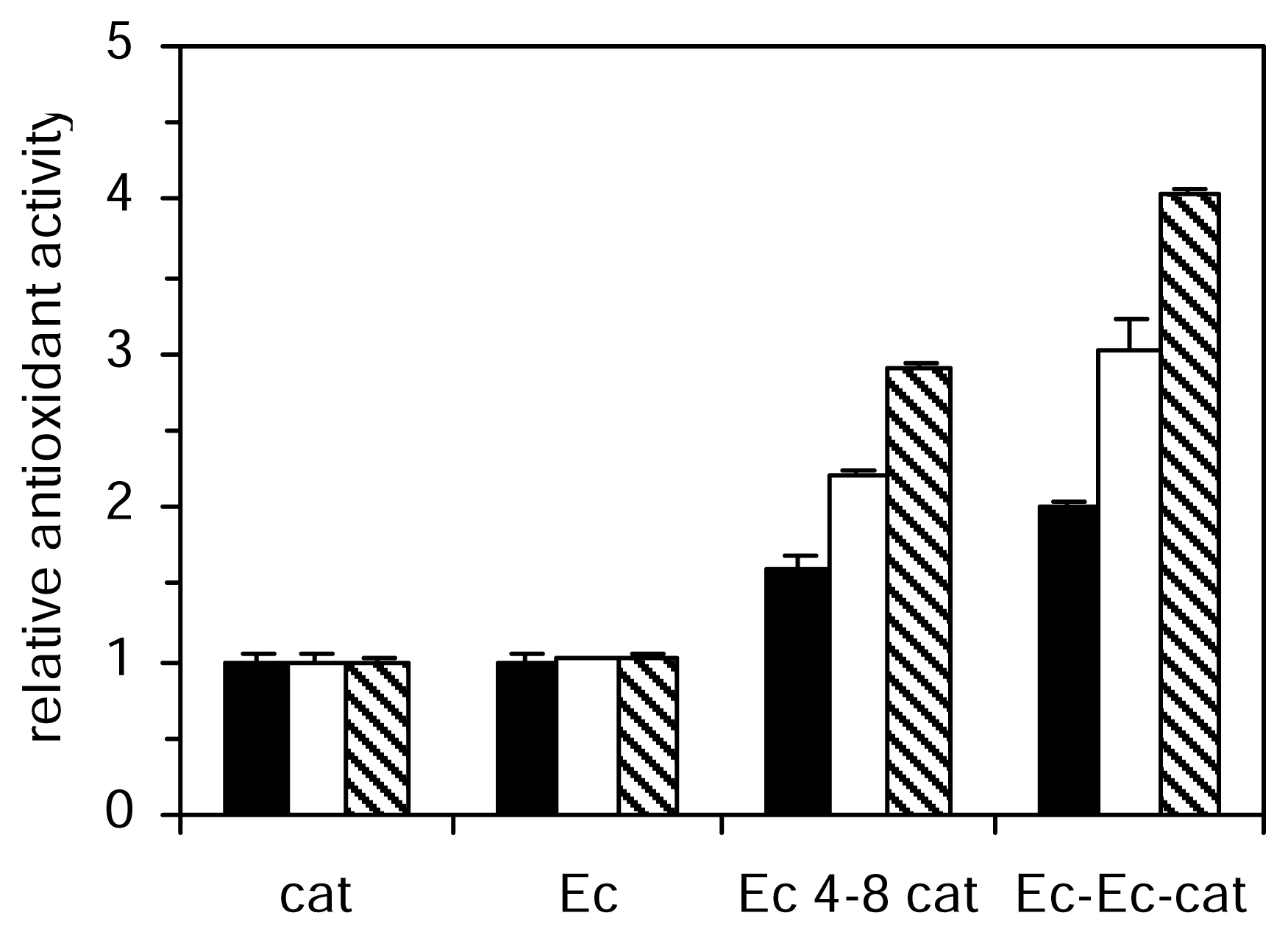
The aqueous phase antioxidant activity of the studied catechins and procyanidins increased from monomer to trimer (
Figure 2). The most active of all the compounds tested in the aqueous phase systems were the trimer (Ec-Ec-cat). The TEAC value of catechin was approximately doubled in dimer and threefold in trimer. Procyanidin dimers differ in their catechin moieties and the kind of interflavan linkages between them. We analyzed five different dimers, all of them belonging to the B series, linked 4–8 or 4–6. The antioxidant activities of catechin and epicatechin were not significantly different in the three aqueous methods however the activity of dimer Ec-Ec was significantly higher than the activity of dimer cat-cat. A similar effect was observed for cat-Ec and Ec-cat dimers, which showed higher activity than dimer cat-cat in these three aqueous methods. The linkage between positions 4 and 8 in procyanidins (Ec 4–8 cat) significantly increase antioxidant efficiency in aqueous phase assays with respect to 4 and 6 linkage (Ec 4–6 cat). In the lipid phase system the trend of decreasing antioxidant activity with polymerization was found in contrast to the antioxidant activity studied in aqueous phase, which increased from monomer to trimer. Galloylation reduced the ability to prevent peroxidation of phosphatidylcholine vesicles.
Nonetheless, in the aqueous phase methods, galloylation resulted in a significant increase of antioxidant activity. Hydroxylation of catechin to gallocatechin did not show a considerable effect in the inhibition of tyrosine nitration, however, it presented a significant increase in antioxidant effectiveness in the TEAC and FRAP assays, in contrast to the lipid phase where the antiradical activity was significantly reduced. The antioxidant activity of the tested series of polyphenolic compounds correlated with the number of aromatic hydroxyl groups in the aqueous phase assays. Different results were found in the lipid phase system where the trend of a decrease in the antioxidant efficiency with the number of aromatic hydroxyl groups was observed. These differences between aqueous and lipid phase methods are in agreement with previously published data [
19], where the results between the aqueous (TEAC method) and lipid phase system (TBARS method) for catechins and procyanidins were compared. Nevertheless, the present work provides more information about these features, since two more antioxidant methods (FRAP and peroxynitrite scavenging activity) were used to analyze the antioxidant capacity of these compounds. According to Auroma [
20], the use of more than one method is recommended in the study of antioxidant capacity, because it is clear that no single method can give a comprehensive prediction of antioxidant efficacy.
The activities of anthocyanin monoglycosides tested in TEAC and FRAP assays decreased in the order: delphinidin-3-monoglucoside > cyanidin-3-rutinoside > malvidin-3-monoglucoside > petunidin-3-monoglucoside. For the structure activity relationship study we did not have cyanidin-3- monoglucoside and for this reason we decided to test cyanidin-3-rutinoside. The completely conjugated structure of anthocyanins, which allows electron delocalization, results in very stable radical products, which is favorable for antioxidative capacity. The degree and position of hydroxylation and methoxylation in the B ring affect stability and reactivity and, thereby, also antioxidant actions. The TEAC and FRAP assays results for antioxidant activity of anthocyanins are in accordance with the previous observations of the effect of hydroxylation and methoxylation in ring B studied by DPPH radical scavenging capacity [
9]. The anthocyanins malvidin-3-glucoside and petunidin-3-glucoside showed lower efficiency compared to cyanidin-3-rutinoside and delphinidin-3-glucoside. The third hydroxyl group in the B ring enhanced the activity, as delphinidin-3-monoglucoside with hydroxyl groups in the 3′-, 4′-, and 5′-positions was significantly more effective than cyanidin-3-rutinoside with hydroxyl groups in 3′- and 4′-positions. Moreover, in this study, different glycosylation patterns may have modified the antioxidant and antiradical activities of the anthocyanins. Kähkönen et al., showed that delphinidin and cyanidin-3-rutinoside are less active in the DPPH scavenging activity than the corresponding monoglucosides, although this effect is very much dependent on the method used [
9]. For the FRAP and TEAC assays the methoxylation of hydroxyl groups in 5′ (petunidin-3-monoglucoside) or 3′ and 5′ positions (malvidin-3-monoglucoside) significantly reduced the antioxidant activity. However, the antioxidant activity showed by malvidin-3-monoglucoside in peroxynitrite mediated tyrosine nitration was the same as that of delphinidin-3-monoglucoside and cyanidin-3-rutinoside activities. The activity of anthocyanins in preventing tyrosine nitration decreased in the following order: cyanidin-3-rutinoside > malvidin-3-monoglucoside ≈ delphinidin-3-monoglucoside > petunidin-3-monoglucoside. Significant differences were not found between delphinidin-3-monoglucoside, cyanidin-3-rutinoside and malvidin-3-monoglucoside, but significant differences were found between the above mentioned compounds and petunidin-3-monoglucoside. Once more, these results show the importance of the substitution groups in the B ring of the anthocyanin molecules. Tsuda et al. [
21] demonstrated the mechanisms which pelargonidin, anthocyanidin with one hydroxyl group on the B ring, scavenge the ONOO
−. First, pelargonidin is broken by the radical and
p-hydroxybenzoic acid is then formed. Later, this acid reacts with ONOO
−, which results in the formation of 4-hydroxy-3-nitrobenzoic acid. It is probable that the different results among the glucosides of anthocyanidins are due, on the one hand, to different mechanisms from pelargonidin to protect against the peroxynitrite-mediated nitration of tyrosine [
21] and on the other hand that the different acids formed by the reaction between the anthocyanins and the radical present different affinities to ONOO
−.
Anthocyanins may exist in a variety of protonated, deprotonated, hydrated, and isomeric forms and the relative proportion of these molecules is strongly dependent on pH. These forms may play an important role in the antioxidant activity. Moreover, the peroxynitrite anion (ONOO
−) and its conjugate acid (HOONO) could have different reactivities [
22] and the relative proportion is too strongly dependent on pH (pK
a = 6.8). Inhibitions of tyrosine nitration were measured at pH 6.0 (≈ 15% of peroxynitrite was in the anionic form) (
Table 1) and a comparative study at physiological pH (7.4) was performed to be more representative of biological conditions. The peroxynitrite scavenging activity of anthocyanins at pH 7.4 (≈ 80% of peroxynitrite was in the anionic form) decreased in the following order: cyanidin-3-rutinoside > malvidin-3-monoglucoside ≈ delphinidin-3-monoglucoside > petunidin-3-monoglucoside (data not shown).
The glycosylation of malvidin-3-monoglucoside to malvidin-3,5-diglucoside caused significant reduction of the antioxidant power in the TEAC assay, but had no significant effect on the inhibition of tyrosine nitration, though the FRAP value for malvidin-3,5-diglucoside was higher than for malvidin-3-monoglucoside (
Table 1).
As it can see in
table 1, the incorporation of pyruvic acid into delphinidin-3-monoglucoside and malvidin-3-monoglucoside caused a significant decrease in antioxidant activity in aqueous phase assays. These results are in agreement with previously published data [
23,
24].
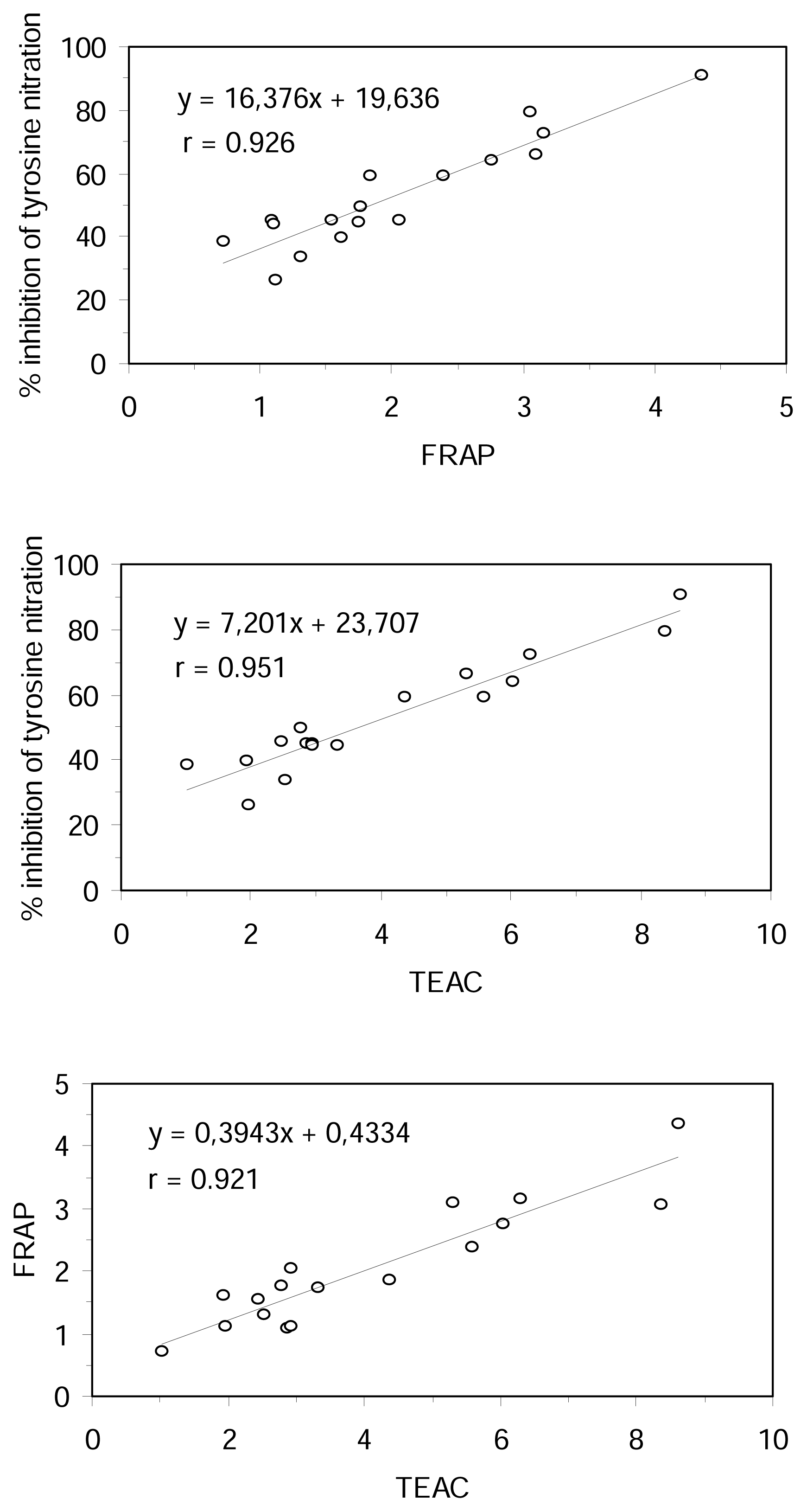
The correlation coefficients between the results obtained for inhibition of tyrosine nitration, FRAP, TEAC and TBARS assays were examined for all the tested compounds. On the basis of simple regression testing, there was a statistically significant relationship between the methods in aqueous phase (r > 0.92 and r
2 > 0.84, p < 0.0001, n = 17) (
Figure 3). However, the TBARS method results did not correlate with the results of the other methods. There was no statistically significant relationship between TBARS and aqueous phase methods at the 95% confidence level (|r| < 0.15, n = 10). These results could be explained, apart from the different hydrophilic or lipophilic properties of the substratum used in the different methods, by the different mechanism of antioxidant action measured for them.
It is evident that the expression of the results of antioxidant power of catechins, procyanidins, anthocyanins and pyranoanthocyanins, as well as the effect of diglycosylation of anthocyanins, depends on the assay used and, therefore, also the mechanisms of these methods used to evaluate the antioxidant activity. The TEAC, FRAP and inhibition tyrosine nitration methods can be recommended to evaluate the antioxidant capacities of wine polyphenols (procyanidins, anthocyanins and pyranoanthocyanins). Nonetheless, the TBARS assay is not recommended for anthocyanins and their adducts. In general, it could be concluded that procyanidins were, among the tested groups, the ones which showed more antioxidant capacity in the distinct aqueous methods used, followed by anthocyanins and pyranoanthocyanins.
3. Experimental Section
3.1 Chemicals and reagents
(+)-catechin (cat), (−)-epicatechin (Ec), (−)-epicatechin-3-O-gallate, gallocatechin and malvidin-3,5-diglucoside were obtained from Sigma-Aldrich (Steinhein, Germany). Butylated hydroxytoluene (BHT) was purchased from Aldrich (Milwaukee, USA), 2,2′-azinobis-(3-ethyl-benzo-thiazoline-6-sulphonate) (ABTS), (±)-6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox C), 2,4,6-tri(2-pyridyl)-1,3,5-pyridyltriazine (TPTZ) and trichloracetic acid (TCA) were obtained from Fluka Chemie (Buchs, Switzerland), thiobarbituric acid (TBA), phosphatidylcholine and myoglobin were obtained from Sigma (St. Louis, USA) and FeCl3 was obtained from Panreac Química SA (Barcelona, Spain). All other chemicals used were of analytical grade.
3.2 Isolation and purification of procyanidins
Procyanidin dimers B2, B3 and B4 were isolated from grape seeds; procyanidins of the B1 series (B1, B7 and trimer Ec-Ec-cat) were from almond fruit flesh. The procyanidins were isolated as previously described by Plumb et al., [
25]. Purities of dimers and trimers used in this work, which were tested by HPLC in the photodiode apparatus, selecting 280 nm as the preferred wavelength, were > 90%.
3.3 Isolation and purification of anthocyanins
Anthocyanins (delphinidin-3-monoglucoside, malvidin-3-monoglucoside, petunidin-3-monoglucoside and cyanidin-3-rutinoside) were isolated from a methanol-acid red grape skin extract by semipreparative HPLC using a Waters 600 chromatograph. The column was Ultracarb ODS (5 μm, 250 × 10 mm) (Phenomenex). The solvents were 5% acetic acid (A) and methanol (B) applied with the following gradient: from 10% to 15% B for 15 min, isocratic 15% B for 5 min, from 15% to 30% B for 30 min, from 30% to 45% B for 10 min, from 45% to 10% B for 10 min at a flow rate of 3 ml/min. Detection was carried out at 520 nm.
3.4 Pyranoanthocyanins syntheses
Syntheses of dephinidin-3-monoglucoside pyruvic acid adduct and malvidin-3-monoglucoside pyruvic acid adduct were performed as previously described by Romero et al., [
26]. Pyruvic acid was added to pure anthocyanin, dissolved in potassium hydrogen tartrate buffer containing 10% ethanol in a molar ratio of pyruvic acid to pure anthocyanins of 300:1. The pH was adjusted to 3.7 by addition of Na
2CO
3 and the solution was incubated at 32 °C in the dark in the presence of air.
The purity of anthocyanins and pyranoanthocyanins was tested by HPLC using the method of de Pascual-Teresa et al., [
27] with minor modifications. Briefly, a Hewlett-Packard 1100 HP system equipped with a quaternary pump and photodiode array detector was used. The column was an Aqua® C18, 5 μm (150 × 4.6 mm) (Phenomenex), thermostatted at 35 °C. The solvents were 0.1% trifluoroacetic acid (A) and acetonitrile (B) applied with the following gradient program: isocratic 10% B for 5 min, from 10% to 15% for 15 min, isocratic 15% B for 5 min, from 15% to 18% B for 5 min and from 18% to 35% B for 20 min at a flow rate of 0.5 ml/min. The measurement was carried out using the photodiode detection at 520 nm as the preferred wavelength. The purities of all the anthocaynins and pyranoanthocyanins used in this work, were in every case higher than 94%.
The identity of the anthocyanins and pyranoanthocyanins was confirmed by HPLC using a dual online detection by diode array detector and mass spectrometry (HPLC-DAD-MS). MS spectrometry was performed using a Finnigan LCQ equipped with API source, using an electrospray ionization (ESI) interface. Both the auxiliary and the sheath gas were a mixture of nitrogen and helium at flow rates of 1.2, and 6 L/min respectively. The capillary temperature was 195 °C and the capillary voltage was 4 V. The MS detector was programmed to perform a series of three consecutive scans: a full scan from 120 to 1500 amu, an MS2 scan of the most abundant ion in the full mass and MS3 of the most abundant ion in the MS2. The normalized energy of collision was 45%. Spectra were recorded in the positive mode.
3.5 Inhibition of tyrosine nitration
Peroxynitrite solution was prepared in accordance with the published method [
28]. A volume of 8 μl of 2.5 mM peroxynitrite solution in 0.05 M NaOH was drawn and mixed rapidly in the injector of the HPLC autosampler with 42 μl 1.0 mM tyrosine solution in 0.11 M KH
2PO
4-Na
2HPO
4 buffer (pH 6.0) containing 40 μM of tested compound. The reaction mixture was injected directly into the HPLC column (Aqua® C18, 5 μm, 150 × 4.6 mm, Phenomenex), the mobile phase, in isocratic conditions, consisted of 90% 40 mM HCOOH and 10% CH
3CN (v/v), at a flow rate of 1 ml/min. The chromatograms were recorded at 276 nm. The activity of the tested compounds was calculated as the percentage of tyrosine nitration relative to the measured peak area of 3-nitrotyrosine of the control.
3.6 Ferric reducing/antioxidant power (FRAP)
The FRAP assay was performed as described previously [
16]. FRAP reagent was freshly prepared each day by mixing 10 mM TPTZ, 20 mM FeCl
3 and 0.3 M acetate buffer pH 3.6 in the ratio 1:1:10 (v/v). The absorbance of the test components was read at 593 nm (Perkin Elmer UV/VIS Lambda Bio 20) 6 minutes after mixing at room temperature against a blank (FRAP and distilled water). For anthocyanins and pyranoanthocyanins an extra blank was needed. It was prepared by addition of a tested compound in acetate buffer (1:30, v/v). Data were expressed relative to values obtained for Trolox (200 μM) and expressed as Trolox equivalents.
3.7 Trolox equivalent antioxidant capacity (TEAC)
The assay was based on the relative ability of antioxidants to scavenge the cation radical ABTS
•+[
14]. The radical was generated by the interaction of ABTS (0.15 mM) with the ferrylmyoglobin radical, generated by the activation of metmyoglobin (2.5 μM) with H
2O
2 (0.1 mM). The extent of quenching of the ABTS
•+ was measured spectrophotometrically at 734 nm and compared to standard amounts of Trolox C. Results are expressed as TEAC value.
3.8 Lipid phase antioxidant activity (TBARS method)
Phospholipid liposomes (final concentration 1 mg/mL) were suspended in 150 mM KCl containing 0.2 mM FeCl
3 and the tested compound at a range of concentrations (0–25 μM). Peroxidation was started as described previously [
29] with ascorbate addition (final concentration 0.05 mM) in a final volume adjusted to 0.4 mL. Samples were incubated at 37 °C for 40 min and then the reactions were terminated by the addition of 0.8 ml of 20% (w/v) trichloroacetic acid (TCA)/0.4% (w/v) thiobarbituric acid (TBA)/0.25 N HCl and 0.01 ml of butylated hydroxytoluene dissolved in ethanol. The production of thiobarbituric acid reactive substances (TBARS) was measured spectrophotometrically at 535 nm during the 20 min of incubation at 80 °C and expressed as concentration causing 50% inhibition (IC
50).
3.9 Statistical analysis
Data are presented as means ± standard deviations (STDEV) of the three independent experiments. The results were processed by using one-way variance analysis (ANOVA). Differences at p < 0.05 were considered significant. In addition, simple regression analysis was performed to seek relationships between the different tests.
 ). Values are expressed relative to catechin activity. Each value represents the mean and standard deviation of three determinations.
). Values are expressed relative to catechin activity. Each value represents the mean and standard deviation of three determinations.
 ). Values are expressed relative to catechin activity. Each value represents the mean and standard deviation of three determinations.
). Values are expressed relative to catechin activity. Each value represents the mean and standard deviation of three determinations.