Consideration of the Factors Influencing the Specific Rates of Solvolysis of p-Methoxyphenyl Chloroformate
Abstract
:1. Introduction
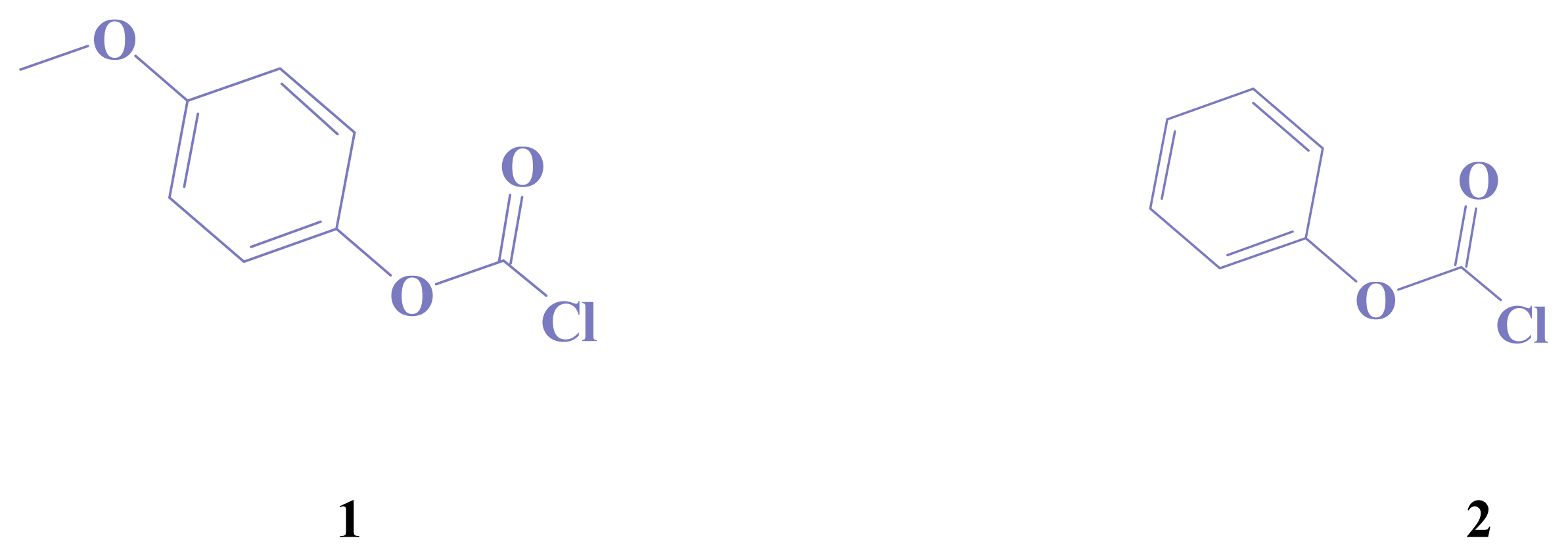
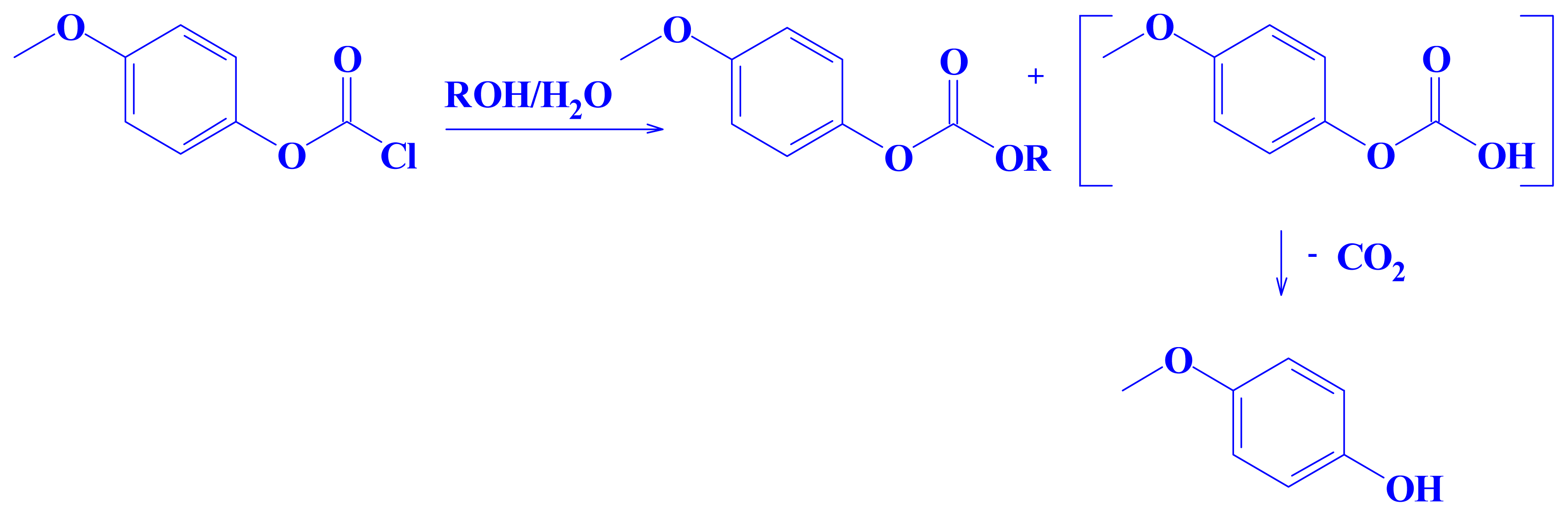
2. Results and Discussion
3. Conclusions
4. Experimental Section
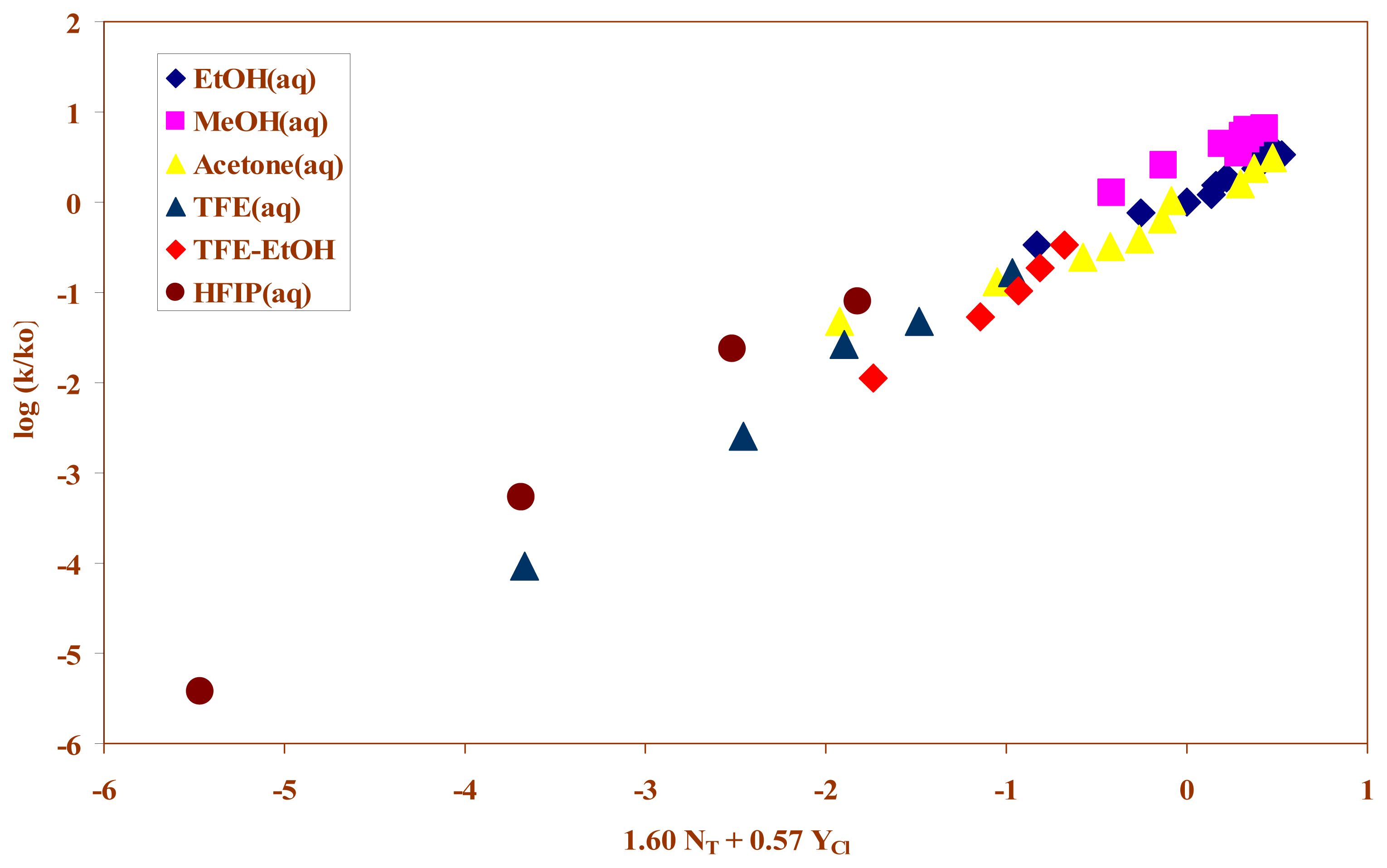
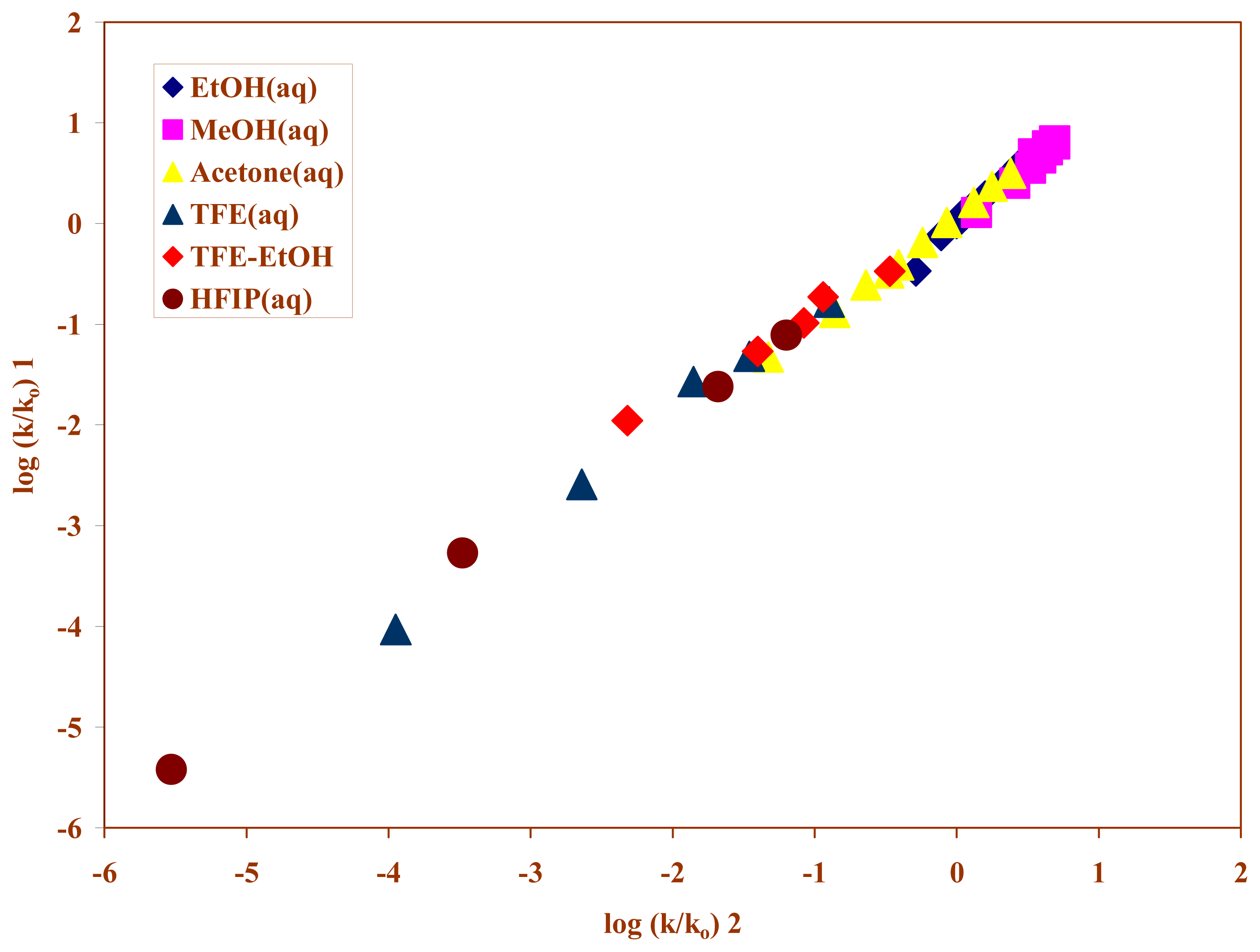

| Solvent (%)a | 105k(s−1)b | NTc | YCld | Ie |
|---|---|---|---|---|
| 90% Acetone (v/v) | 15.0±0.6 | −0.35 | −2.22 | −0.17 |
| 97% TFE (w/w) | 0.0300±0.0013 | −3.30 | 2.83 | 0.49 |
| 90% TFE (w/w) | 0.825±0.032 | −2.55 | 2.85 | 0.47 |
| 80% TFE (w/w) | 8.63±0.24 | −2.22 | 2.90 | 0.28 |
| 70% TFE (w/w) | 15.2±0.6 | −1.98 | 2.96 | 0.25 |
| 50% TFE (w/w) | 52.6±2.8 | −1.73 | 3.16 | 0.09 |
| 80T-20E (v/v) | 3.52±0.13 | −1.76 | 1.89 | 0.52 |
| 60T-40E (v/v) | 17.0±0.5 | −0.94 | 0.63 | 0.59 |
| 50T-50E (v/v) | 32.7±1.7 | −0.64 | 0.16 | 0.51 |
| 40T-60E (v/v) | 59.2±2.3 | −0.34 | −0.48 | 0.43 |
| 20T-80E (v/v) | 107±3 | 0.08 | −1.42 | 0.31 |
| 97%HFIP (w/w) | 1.20(±0.13)x10−3 | −5.26 | 5.17 | 0.73 |
| 70%HFIP (w/w) | 7.58±0.22 | −2.94 | 3.83 | 0.69 |
| Substrate | nb | ℓc | mc | hc | cc | Rd | Fe |
|---|---|---|---|---|---|---|---|
| 1 | 44f | 1.60±0.05 | 0.57±0.05 | 0.18±0.06 | 0.981 | 517 | |
| 1.70±0.08 | 0.61±0.04 | 0.29±0.18 (0.114)g | 0.19±0.06 | 0.982 | 359 | ||
| 31h | 1.46±0.08 | 0.53±0.03 | 0.18±0.06 | 0.964 | 182 | ||
| 1.75±0.07 | 0.66±0.03 | 0.85±0.15 | 0.22±0.04 | 0.984 | 274 | ||
| 2 | 49h | 1.66±0.05 | 0.56±0.03 | 0.15±0.07 | 0.980 | 568 | |
| 1.77±0.08 | 0.61±0.04 | 0.35±0.19 (0.068)g | 0.16±0.06 | 0.982 | 400 | ||
| 44i | 1.60±0.05 | 0.54±0.03 | 0.15±0.06 | 0.979 | 468 | ||
| 1.67±0.08 | 0.57±0.04 | 0.19±0.20 (0.332)g | 0.15±0.06 | 0.979 | 312 |
Acknowledgement
References and Notes
- Grunwald, E.; Winstein, S. The Correlation of Solvolysis Rates. J. Am. Chem. Soc 1948, 70, 846–854. [Google Scholar]
- Bentley, T.W.; Llewellyn, G. Yx Scales of Solvent Ionizing Power. Prog. Phys. Org. Chem 1990, 17, 121–158. [Google Scholar]
- Winstein, S.; Grunwald, E.; Jones, H.W. The Correlation of Solvolyses Rates and the Classification of Solvolysis Reactions into Mechanistic Categories. J. Am. Chem. Soc 1951, 73, 2700–2707. [Google Scholar]
- Wells, P. R. Linear Free Energy Relationships; Academic Press: New York, 1968; pp. 67–68. [Google Scholar]
- Schadt, F.L.; Bentley, T.W.; Schleyer, P.v.R. The SN2-SN1 Spectrum. 2. Quantitative Treatments of Nucleophilic Solvent Assistance. A Scale of Solvent Nucleophilicities. J. Am. Chem. Soc 1976, 98, 7667–7674. [Google Scholar]
- Kevill, D.N.; Anderson, S.W. An Improved Scale of Solvent Nucleophilicity Based on the Solvolysis of the S-Methyldibenzothiophenium Ion. J. Org. Chem 1991, 56, 1845–1850. [Google Scholar]
- Kevill, D.N. Development and Uses of Scales of Solvent Nucleophilicity. In Advances in Quantitative Structure-Property Relationships; Volume 1, Charton, M, Ed.; JAI Press: Greenwich, CT, 1996; pp. 81–115. [Google Scholar]
- Bentley, T.W.; Koo, I.S.; Norman, S.J. Distinguishing Between Solvation Effects and Mechanistic Changes. Effects Due to Differences in Solvation of Aromatic Rings and Alkyl Groups. J. Org. Chem 1991, 56, 1604–1609. [Google Scholar]
- Liu, K.-T; Sheu, H.-C. Solvolysis of 2-Aryl-2-chloroadamantanes. A New Y Scale for Benzylic Chlorides. J. Org. Chem 1991, 56, 3021–3025. [Google Scholar]
- Liu, K.-T; Lin, Y.-S.; Duann, Y.-F. Solvent Effects on the Solvolysis of Some Secondary Tosylates. Applications of YBnOTs and YxBnOTs Scales to Mechanistic Studies. J. Phys. Org. Chem 2002, 15, 750–757. [Google Scholar]
- Kevill, D.N.; Koyoshi, F.; D’Souza, M.J. Correlation of the Specific Rates of Solvolysis of Aromatic Carbamoyl Chlorides, Chloroformates, Chlorothionoformates, and Chlorodithioformates Revisited. Int. J. Mol. Sci 2007, 8, 346–352. [Google Scholar]
- Liu, K.–T.; Chen, H.–I.; Lin, Y.–S.; Jin, B.-Y. Solvolysis of N, N-Diphenylcarbamoyl Chloride Revisted. Extended Positive Charge Delocalization on Phenyl Rings in the Transition State and Possible Contribution of Non-Canonical Resonance Structure. J. Phys. Org. Chem 2000, 13, 322–329. [Google Scholar]
- Kaspi, J.; Rappoport, Z. Nucleophilicity and Ionizing Power in Binary Solvent Mixtures. Tetrahedron Lett 1977, 2035–2038. [Google Scholar]
- Kevill, D.N.; Park, B.-C.; Park, K.-H.; D’Souza, M.J.; Yaakoubd, L.; Mlynarski, S.L.; Kyong, J.B. Rate and Product Studies in the Solvolyses of N, N-Dimethylsulfamoyl and 2-Propanesulfonyl 2-Propanesulfonyl Chlorides. Org. Biomol. Chem 2006, 4, 1580–1586, , and references therein. [Google Scholar]
- Kevill, D.N.; Ryu, Z.H. Additional Solvent Ionizing Power Values for Binary Water – 1,1,1,3,3,3,-Hexafluoro-2-propanol Solvents. Int. J. Mol. Sci 2006, 7, 451–455. [Google Scholar]
- Bentley, T.W.; Carter, G.E. The SN2-SN1 Spectrum. 4. Mechanism for Solvolyses of tert-Butyl Chloride: A Revised Y Scale of Solvent Ionizing Power based on Solvolyses of 1-Adamantyl Chloride. J. Am. Chem. Soc 1982, 104, 5741–5747. [Google Scholar]
- Kevill, D.N.; D’Souza, M.J. Additional YClValues and Correlation of the Specific Rates of Solvolysis of tert-Butyl Chloride in Terms of NTand YClScales. J. Chem. Res. Synop 1993, 174–175. [Google Scholar]
- Kevill, D.N.; Ismail, N.HJ.; D’Souza, M.J. Solvolysis of the (p- Methoxybenzyl)dimethylsulfonium Ion. Development and Use of a Scale to Correct for Dispersion in Grunwald-Winstein Plots. J. Org. Chem 1994, 59, 6303–6312. [Google Scholar]
- Kevill, D.N.; D’Souza, M.J. Correlation of the Rates of Solvolysis of Phenyl Chloroformate. J. Chem. Soc., Perkin Trans. 2 1997, 1721–1724. [Google Scholar]
- Kevill, D.N.; D’Souza, M.J. Correlation of the Rates of Solvolysis of Benzoyl Chloride and Derivatives Using Extended Forms of the Grunwald-Winstein Equation. J. Phys. Org. Chem 2002, 15, 881–888. [Google Scholar]
- Frost, A.A.; Pearson, R.G. Kinetics and Mechanism-a Study of Homogeneous Chemical Reactions, 2nd Ed ed; Wiley: New York, 1961; pp. 49–50. [Google Scholar]
- Kevill, D.N.; Abduljaber, M.H. Correlation of the Rates of Solvolysis of Cyclopropylcarbinyl and Cyclobutyl Bromides Using the Extended Grunwald-Winstein Equation. J. Org. Chem 2000, 65, 2458–2554. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
D’Souza, M.J.; Reed, D.; Koyoshi, F.; Kevill, D.N. Consideration of the Factors Influencing the Specific Rates of Solvolysis of p-Methoxyphenyl Chloroformate. Int. J. Mol. Sci. 2007, 8, 788-796. https://doi.org/10.3390/i8080788
D’Souza MJ, Reed D, Koyoshi F, Kevill DN. Consideration of the Factors Influencing the Specific Rates of Solvolysis of p-Methoxyphenyl Chloroformate. International Journal of Molecular Sciences. 2007; 8(8):788-796. https://doi.org/10.3390/i8080788
Chicago/Turabian StyleD’Souza, Malcolm J., Darneisha Reed, Fumie Koyoshi, and Dennis N. Kevill. 2007. "Consideration of the Factors Influencing the Specific Rates of Solvolysis of p-Methoxyphenyl Chloroformate" International Journal of Molecular Sciences 8, no. 8: 788-796. https://doi.org/10.3390/i8080788




