UVB-Protective Effects of Isoflavone Extracts from Soybean Cake in Human Keratinocytes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Isoflavone extracts
2.3. Cell Culture
2.4. Drug treatment and UVB irradiation
2.5. Cell viability assay (MTT assay)
2.6. Flow cytometric analysis of intracellular H2O2
2.7. Cell lysate preparation and Western blot analysis of JNK, ERK1/2 and p38
2.8. Statistical analysis
3. Results and Discussion
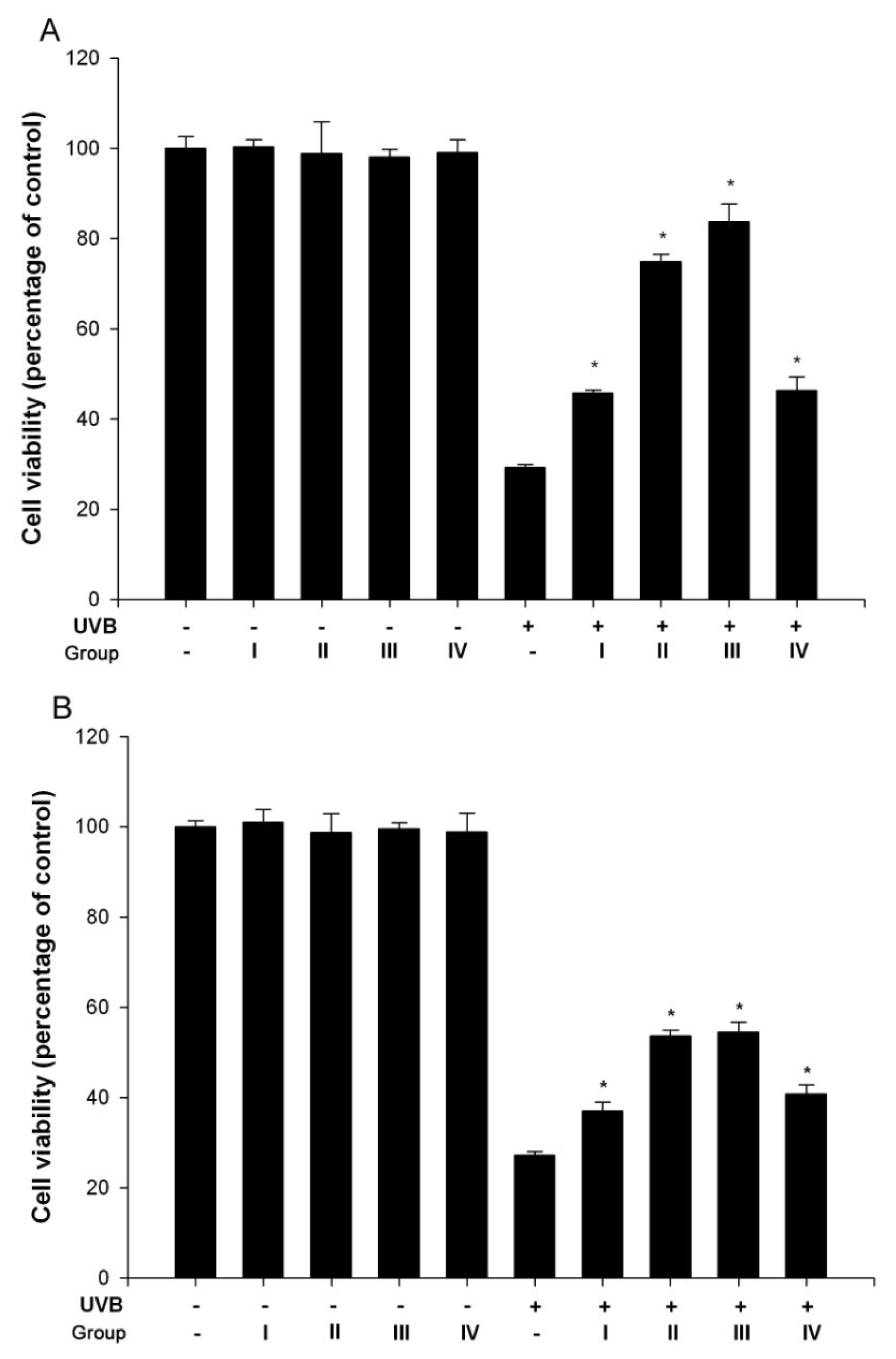
3.1. Isoflavone extracts inhibited UVB-induced cell death
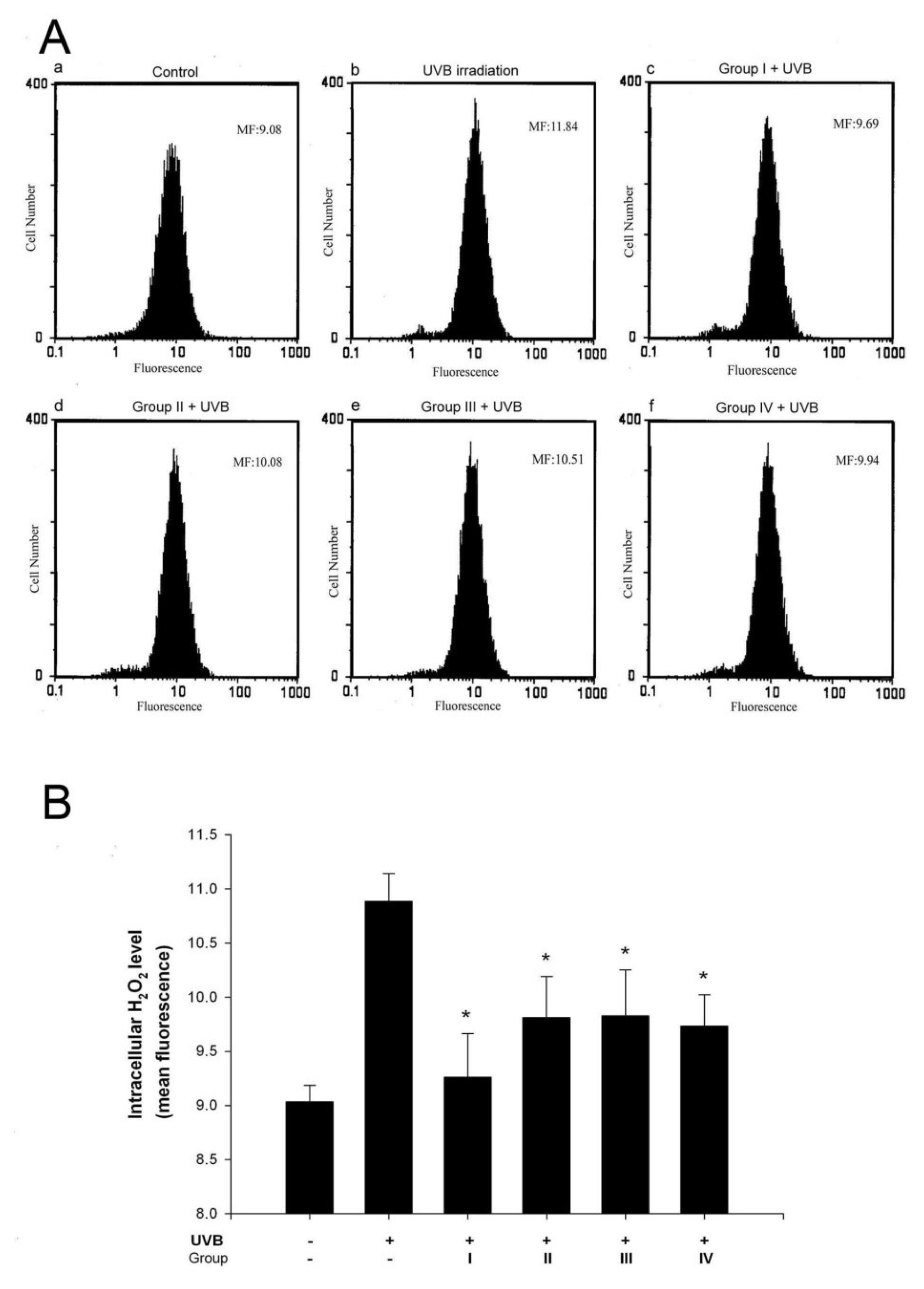
3.2. Isoflavone extracts inhibit UVB-induced H2O2 generation in keratinocytes
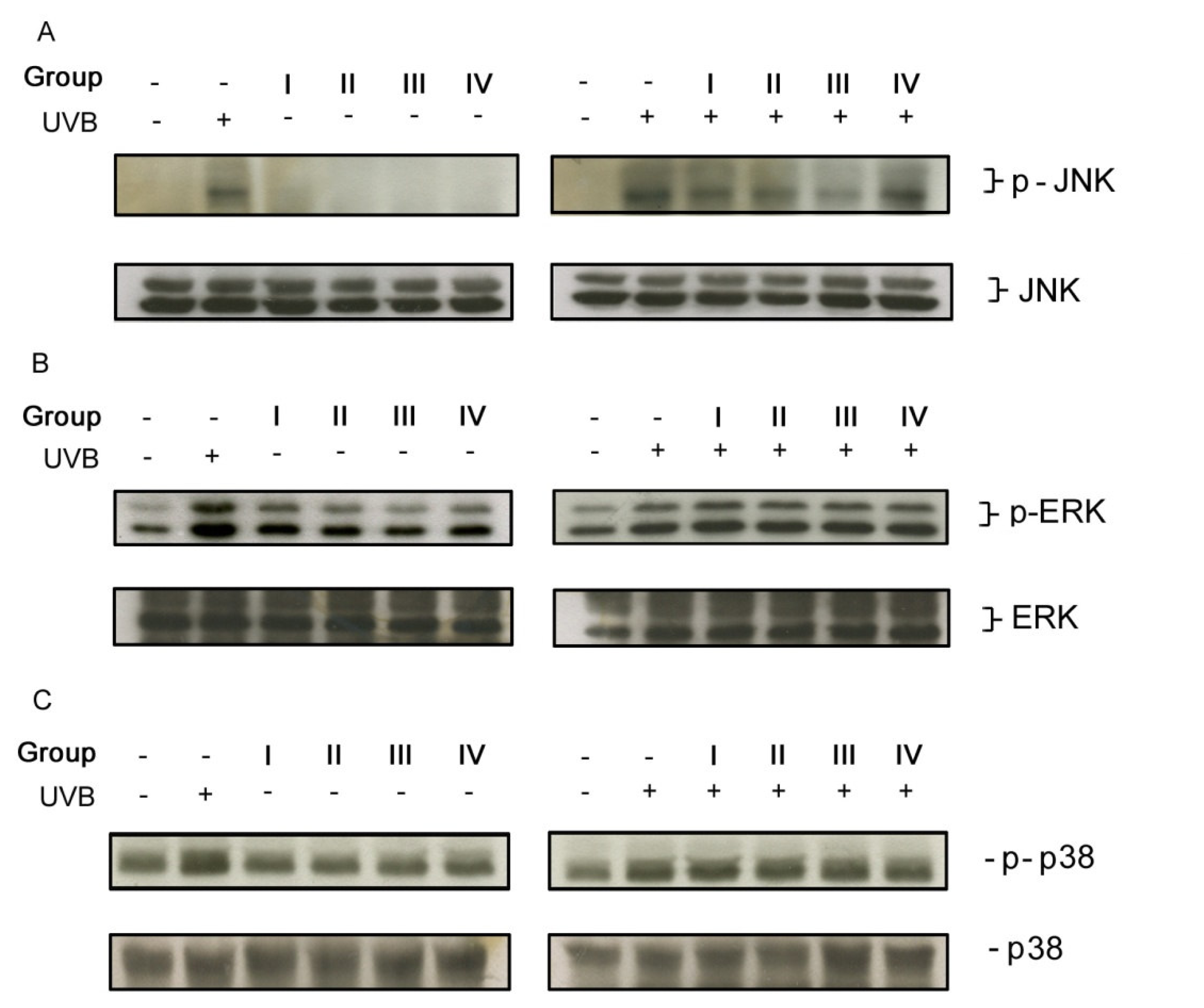
3.3. Isoflavone extracts differentially inhibited UVB-induced MAP kinase signaling pathway
Discussion
| Extracts | Isoflavone name | Concentration (μg/ml) |
|---|---|---|
| aglycone extract (Group I) | daidzein | 47.3 ± 0.5 |
| genistein | 36.4 ± 0.1 | |
| glycitein | 16.3 ± 0.2 | |
| glucoside extract (Group II) | daidzin | 25.3 ± 0.1 |
| genistin | 45.9 ± 1.6 | |
| glycitin | 29.2 ± 0.2 | |
| acetylglucoside extract (Group III) | acetyldaidzin | 19.7 ± 0.9 |
| acetylgenistin | 69.0 ± 1.6 | |
| acetylglycitin | 12.8 ± 0.7 | |
| malonylglucoside extract (Group IV) | malonyldaidzin | 195.2 ± 1.2 |
| malonylgenistin | 140.4 ± 4.8 | |
| malonylglycitin | 64.0 ± 0.8 |
Acknowledgements
References
- Armstrong, B.; Doll, R. Environmental factors and cancer incidence and mortality in different countries, with special reference to dietary practices. Int. J. Cancer 1975, 15, 617–631. [Google Scholar]
- Correa, P. Epidemiological correlations between diet and cancer frequency. Cancer Res 1981, 41, 3685–3690. [Google Scholar]
- Messina, M.; Barnes, S. The role of soy products in reducing risk of cancer. J. Natl. Cancer Inst 1991, 83, 541–546. [Google Scholar]
- Kao, T.H.; Chen, B.H. An improved method for determination of isoflavones in soybean powder by liquid chromatography. Chromatographia 2002, 56, 423–430. [Google Scholar]
- Kao, T.H.; Chen, B.H. Functional components in soybean cake and their effects on antioxidant activity. J. Agric. Food Chem 2006, 54, 7544–7555. [Google Scholar]
- Wei, H.; Saladi, R.; Lu, Y.; Wang, Y.; Palep, S.R.; Moore, J.; Phelps, R.; Shyong, E.; Lebwohl, M.G. Isoflavone genistein: photoprotection and clinical implications in dermatology. J. Nutr 2003, 133, 3811S–3819S. [Google Scholar]
- Russo, A.; Cardile, V.; Lombardo, L.; Vanella, L.; Acquaviva, R. Genistin inhibits UV light-induced plasmid DNA damage and cell growth in human melanoma cells. J. Nutr. Biochem 2006, 17, 103–108. [Google Scholar]
- Wei, H.; Spencer, J.; elfand, J.; Phelps, R.; Lebwohl, M. The isoflavone genistein: A new agent in dermatology? Cosmet. Dermatol 2001, 14, 13–19. [Google Scholar]
- Ji, S; Willis, GM; Frank, GR; Cornelius, SG; Spurlock, ME. Soybean isoflavones, genistein and genistin, inhibit rat myoblast proliferation, fusion and myotube protein synthesis. J. Nutr. 1999, 129, 1291–1297. [Google Scholar]
- Kao, T.H.; Lu, Y.F.; Chen, B.H. Preparative column chromatography of four groups of isoflavones from soybean cake. Eur. Food Res. Technol 2005, 221, 459–465. [Google Scholar]
- Huang, C.C.; Fang, J.Y.; Wu, W.B.; Chiang, H.S.; Wei, Y.J.; Hung, C.F. Protective effects of (−)- epicatechin-3-gallate on UVA-induced damage in HaCaT keratinocytes. Arch. Dermatol. Res. 2005, 296, 473–481. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar]
- Green, L.M.; Reade, J.L.; Ware, C.F. Rapid colorimetric assay for cell viability: application to the quantitation of cytotoxic and growth inhibitory lymphokines. J. Immunol. Methods 1984, 70, 257–268. [Google Scholar]
- Wu, W.B.; Chiang, H.S.; Fang, J.Y.; Chen, S.K.; Huang, C.C.; Hung, C.F. (+)-Catechin prevents ultraviolet B-induced human keratinocyte death via inhibition of JNK phosphorylation. Life Sci. 2006, 79, 801–807. [Google Scholar]
- Peus, D.; Meves, A.; Vasa, R.A.; Beyerle, A.; O’Brien, T.; Pittelkow, M.R. H2O2 is required for UVB-induced EGF receptor and downstream signaling pathway activation. Free Radic. Biol. Med 1999, 27, 1197–1202. [Google Scholar]
- Katiyar, S.K.; Afaq, F.; Azizuddin, K.; Mukhtar, H. Inhibition of UVB-induced oxidative stress-mediated phosphorylation of mitogen-activated protein kinase signaling pathways in cultured human epidermal keratinocytes by green tea polyphenol (-)-epigallocatechin-3-gallate. Toxicol. Appl. Pharmacol 2001, 176, 110–117. [Google Scholar]
- Peus, D.; Vasa, R.A.; Beyerle, A.; Meves, A.; Krautmacher, C.; Pittelkow, M.R. UVB activates ERK1/2 and p38 signaling pathways via reactive oxygen species in cultured keratinocytes. J. Invest. Dermatol 1999, 112, 751–756. [Google Scholar]
- Afaq, F.; Malik, A.; Syed, D.; Maes, D.; Matsui, M.S.; Mukhtar, H. Pomegranate fruit extract modulates UV-B-mediated phosphorylation of mitogen-activated protein kinases and activation of nuclear factor kappa B in normal human epidermal keratinocytes paragraph sign. Photochem. Photobiol 2005, 81, 38–45. [Google Scholar]
- Takahashi, H.; Suzuki, Y.; Miyauchi, Y.; Hashimoto, Y.; Ishida-Yamamoto, A.; Iizuka, H. Roxithromycin decreases ultraviolet B irradiation-induced reactive oxygen intermediates production and apoptosis of keratinocytes. J. Dermatol. Sci 2004, 34, 25–33. [Google Scholar]
- Devary, Y.; Gottlieb, R.A.; Smeal, T.; Karin, M. The mammalian ultraviolet response is triggered by activation of Src tyrosine kinases. Cell 1992, 71, 1081–1091. [Google Scholar]
- Bender, K.; Blattner, C.; Knebel, A.; Iordanov, M.; Herrlich, P.; Rahmsdorf, H.J. UV-induced signal transduction. J. Photochem. Photobiol. B 1997, 37, 1–17. [Google Scholar]
- Su, B.; Karin, M. Mitogen-activated protein kinase cascades and regulation of gene expression. Curr. Opin. Immunol 1996, 8, 402–411. [Google Scholar]
- Peus, D.; Vasa, R.A.; Meves, A.; Beyerle, A.; Pittelkow, M.R. UVB-induced epidermal growth factor receptor phosphorylation is critical for downstream signaling and keratinocyte survival. Photochem. Photobiol 2000, 72, 135–140. [Google Scholar]
- Assefa, Z.; Van Laethem, A.; Garmyn, M.; Agostinis, P. Ultraviolet radiation-induced apoptosis in keratinocytes: on the role of cytosolic factors. Biochim. Biophys. Acta 2005, 1755, 90–106. [Google Scholar]
- Assefa, Z.; Vantieghem, A.; Garmyn, M.; Declercq, W.; Vandenabeele, P.; Vandenheede, J.R.; Bouillon, R.; Merlevede, W.; Agostinis, P. p38 mitogen-activated protein kinase regulates a novel, caspase-independent pathway for the mitochondrial cytochrome c release in ultraviolet B radiation-induced apoptosis. J. Biol. Chem 2000, 275, 21416–21421. [Google Scholar]
- Lei, K.; Davis, R.J. JNK phosphorylation of Bim-related members of the Bcl2 family induces Bax-dependent apoptosis. Proc. Natl. Acad. Sci. U S A 2003, 100, 2432–2437. [Google Scholar]
- Takasawa, R.; Nakamura, H.; Mori, T.; Tanuma, S. Differential apoptotic pathways in human keratinocyte HaCaT cells exposed to UVB and UVC. Apoptosis 2005, 10, 1121–30. [Google Scholar]
- Courdavault, S.; Baudouin, C.; Charveron, M.; Canguilhem, B.; Favier, A.; Cadet, J.; Douki, T. Repair of the three main types of bipyrimidine DNA photoproducts in human keratinocytes exposed to UVB and UVA radiations. DNA Repair (Amsterdam) 2005, 4, 836–44. [Google Scholar]
© 2007 by MDPI ( http://www.mdpi.org) Reproduction is permitted for noncommercial purposes.
Share and Cite
Chiang, H.-S.; Wu, W.-B.; Fang, J.-Y.; Chen, B.-H.; Kao, T.-H.; Chen, Y.-T.; Huang, C.-C.; Hung, C.-F. UVB-Protective Effects of Isoflavone Extracts from Soybean Cake in Human Keratinocytes. Int. J. Mol. Sci. 2007, 8, 651-661. https://doi.org/10.3390/i8070651
Chiang H-S, Wu W-B, Fang J-Y, Chen B-H, Kao T-H, Chen Y-T, Huang C-C, Hung C-F. UVB-Protective Effects of Isoflavone Extracts from Soybean Cake in Human Keratinocytes. International Journal of Molecular Sciences. 2007; 8(7):651-661. https://doi.org/10.3390/i8070651
Chicago/Turabian StyleChiang, Han-Sun, Wen-Bin Wu, Jia-You Fang, Bing-Huei Chen, Tsai-Hua Kao, Ying-Ting Chen, Chieh-Chen Huang, and Chi-Feng Hung. 2007. "UVB-Protective Effects of Isoflavone Extracts from Soybean Cake in Human Keratinocytes" International Journal of Molecular Sciences 8, no. 7: 651-661. https://doi.org/10.3390/i8070651




